Introduction
The landscape of clinical research is rapidly evolving, with paid clinical trials emerging as a crucial element in the development of innovative treatments. Participants not only play a pivotal role in advancing medical breakthroughs but also gain access to cutting-edge therapies and financial compensation.
Nevertheless, the decision to partake in these trials is often accompanied by a multitude of questions concerning:
- Safety
- Ethical considerations
- Personal motivations
What compels individuals to navigate these complexities? Understanding their experiences can significantly enhance future research endeavors.
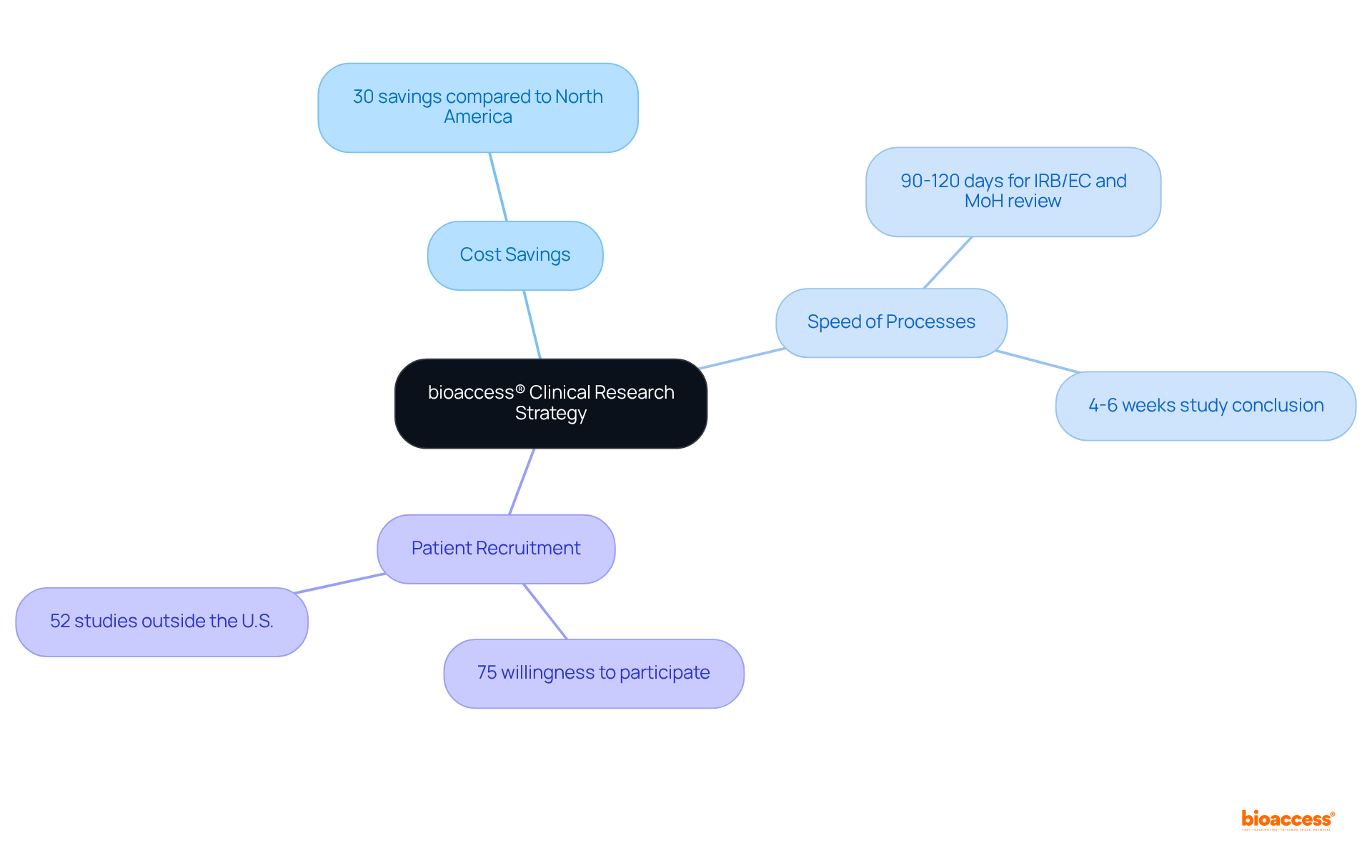
bioaccess®: Accelerating Paid Clinical Research Trials with Global Agility
bioaccess® leverages its extensive knowledge and strategically located sites across Latin America, particularly in Colombia, to expedite ethical approvals and . for , offering over 30% and Western Europe, coupled with a where the total IRB/EC and MoH (INVIMA) review is completed in just 90-120 days.
By tapping into the region’s , bioaccess® can conclude research studies in as little as 4-6 weeks—remarkably quicker than traditional markets. This exceptional agility not only streamlines the research process but also ensures that participants gain and therapies.
Notably, recent findings indicate that 75% of patients who were previously unaware of research studies expressed a willingness to participate if informed, underscoring the critical role of in accelerating enrollment.
Furthermore, with , bioaccess® stands at the forefront of this evolving landscape, driving innovation and improving patient access to essential medical advancements.
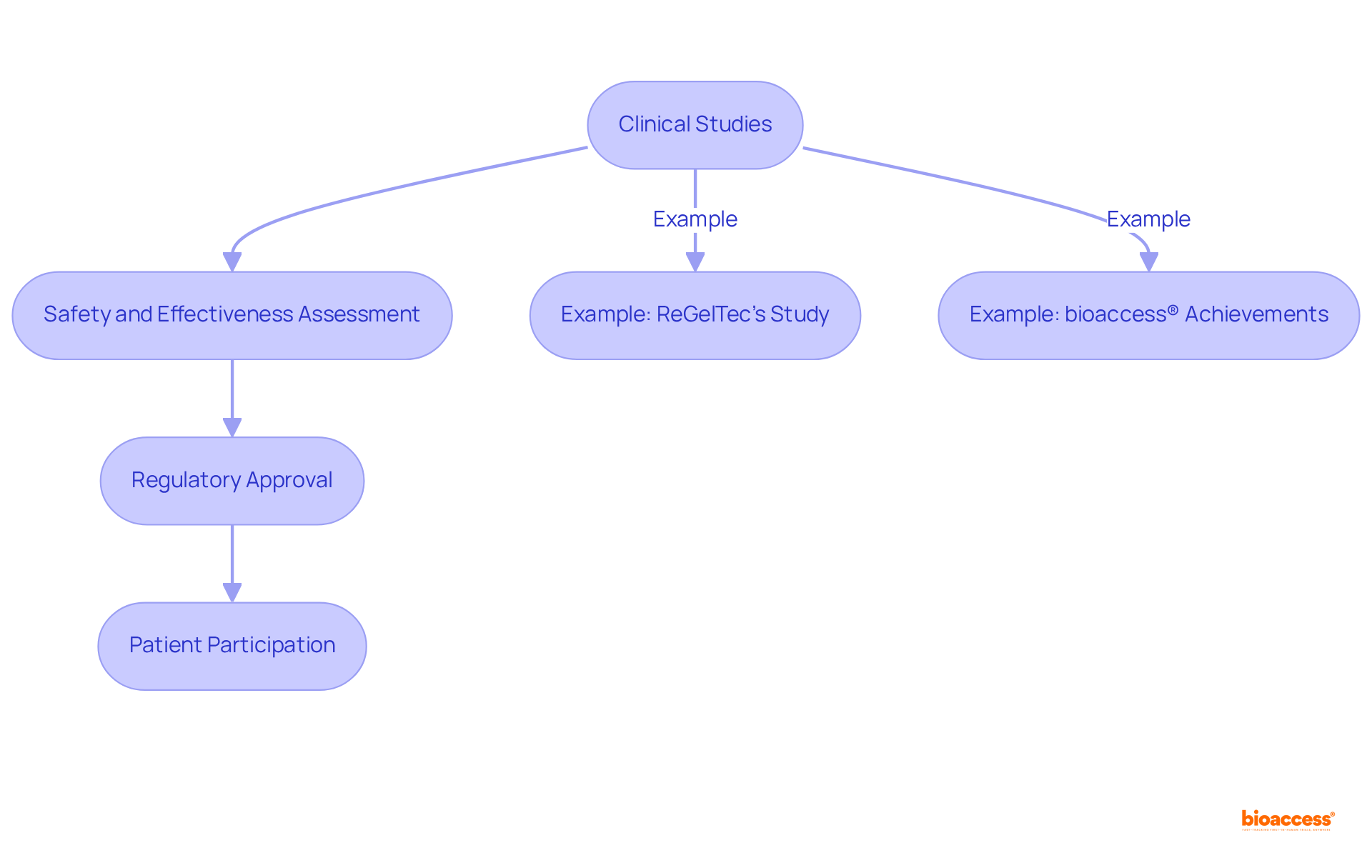
Clinical Trials: The Backbone of Drug Development and Innovation
are essential for the advancement of , serving as the primary method for assessing safety and effectiveness. They provide for approval, ensuring that new treatments meet safety standards before entering the market.
For instance, ReGelTec’s Early Feasibility Study on HYDRAFIL™ in Colombia effectively treated eleven patients suffering from chronic low back pain, underscoring the .
Additionally, bioaccess® collaborates with various organizations to enhance research services, achieving over a 50% reduction in and maintaining a 95% .
Participants in paid play a vital role in advancing and improving patient outcomes, making their involvement crucial to the success of new treatments.
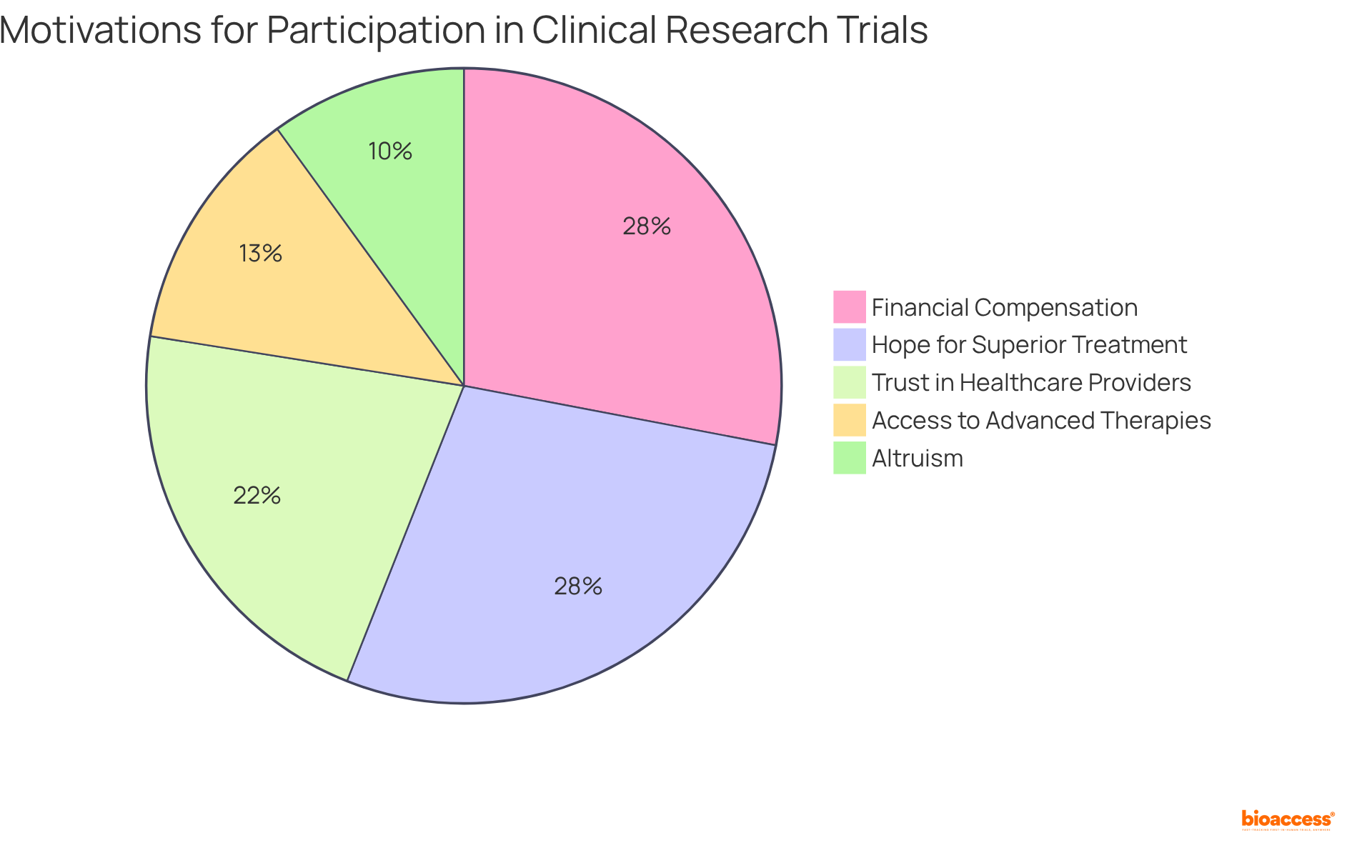
Diverse Motivations: Why People Choose to Participate in Paid Clinical Trials
Individuals are motivated to participate in paid for various reasons, with . Approximately 56% express hope that the test therapy may provide a superior treatment option, while 43% cite trust in their healthcare providers as a significant facilitator. Many individuals also seek , often through .
Altruism plays a vital role, as participants often hope their involvement will . Additionally, participation in paid can provide enhanced , which is particularly appealing to those with chronic conditions.
Notably, 31% of individuals believe that the of the study outweigh the risks, highlighting a complex interplay of motivations that drive people to engage in .
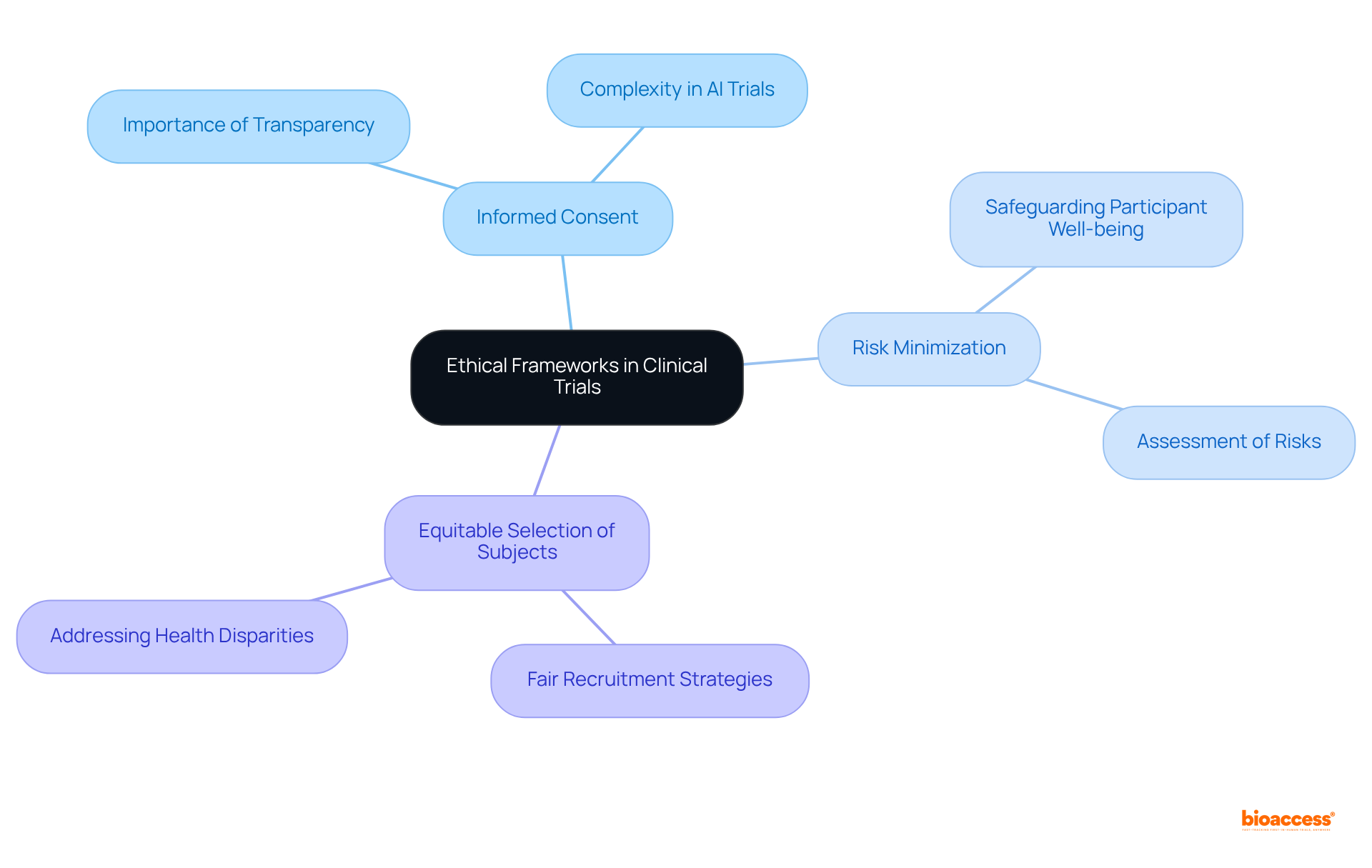
Ethical Frameworks: Safeguarding Participants in Clinical Trials
in , emphasizing the rights and welfare of individuals involved. assume a vital role in supervising study protocols, ensuring adherence to ethical standards such as:
In 2025, data indicates that compliance with remains high, with IRBs actively overseeing studies to safeguard the well-being of participants. As one IRB member articulated, ‘Our primary responsibility is to ensure that and that their involvement is both voluntary and informed.’ Such measures are essential for fostering , ultimately enhancing the .
Understanding Risks and Benefits: Key Considerations for Participants
Before participating in a research study, individuals must thoroughly assess the possible risks and advantages. Participating in paid can offer , including , enhanced medical supervision, and the opportunity to contribute to . However, participants should also be aware of the , which may encompass side effects from experimental therapies, the possibility of receiving a placebo, and the inherent uncertainty regarding treatment outcomes.
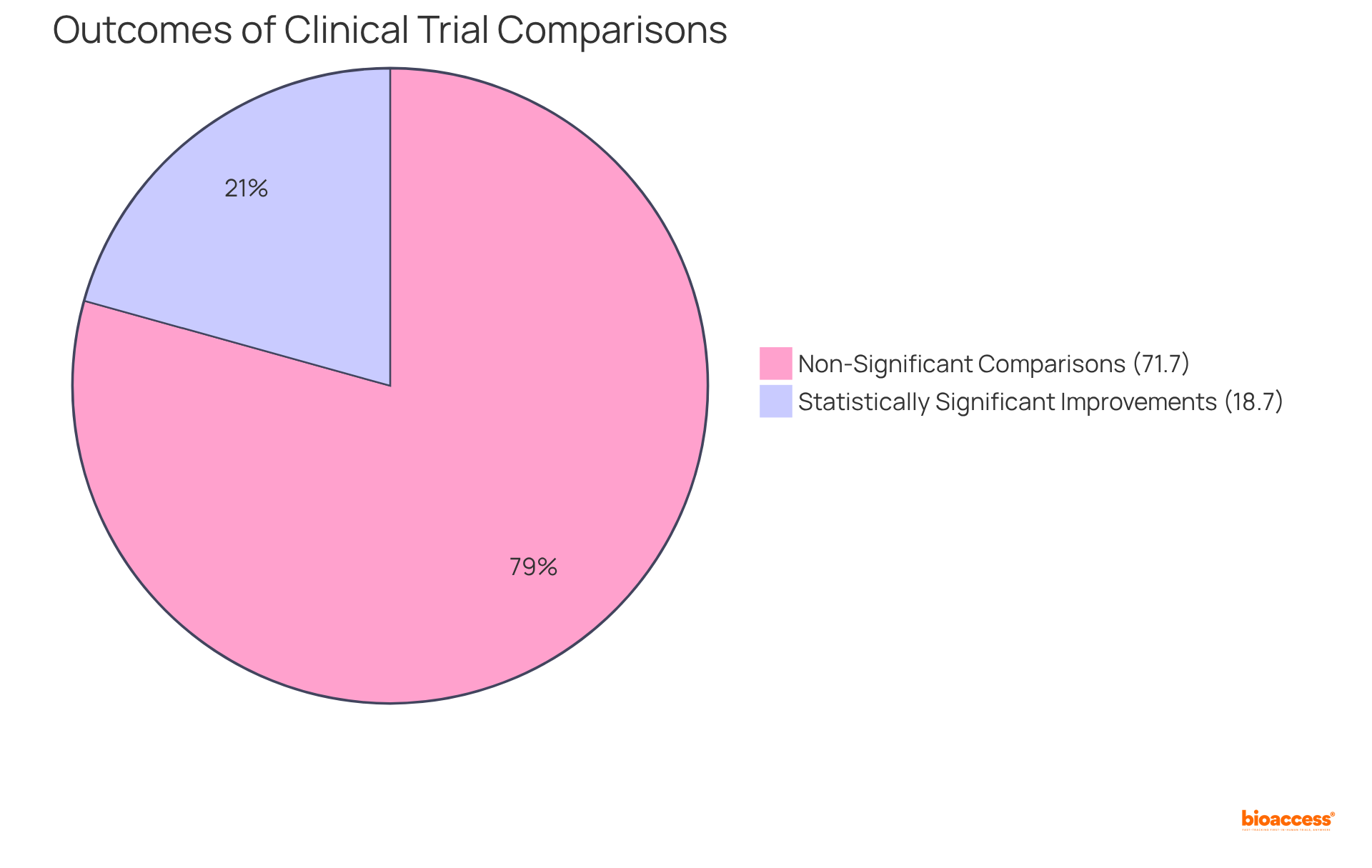
Data indicates that involvement in (RCTs) can lead to , with 18.7% of comparisons supporting participation and demonstrating statistically significant enhancements. Nonetheless, it is crucial to recognize that 71.7% of comparisons were not statistically significant, highlighting the variability in study results.
Healthcare professionals stress the importance of . For instance, Amira Bouzalmate-Hajjaj points out that provide essential information about potential benefits and risks, ensuring participants comprehend their commitments. Engaging in discussions with healthcare professionals is vital, as 92.7% of oncology patients indicate it is ‘important’ or ‘very important’ to communicate with the doctors involved in paid before joining the study. This dialogue can further elucidate these factors, enabling individuals to make well-informed choices about their involvement in clinical research.
Real-world experiences also shed light on the complexities of participation in studies. Many participants report that the close monitoring and access to new treatments can be transformative, while others voice concerns regarding the unknowns tied to experimental therapies. Ultimately, understanding these dynamics is essential for anyone in research studies.
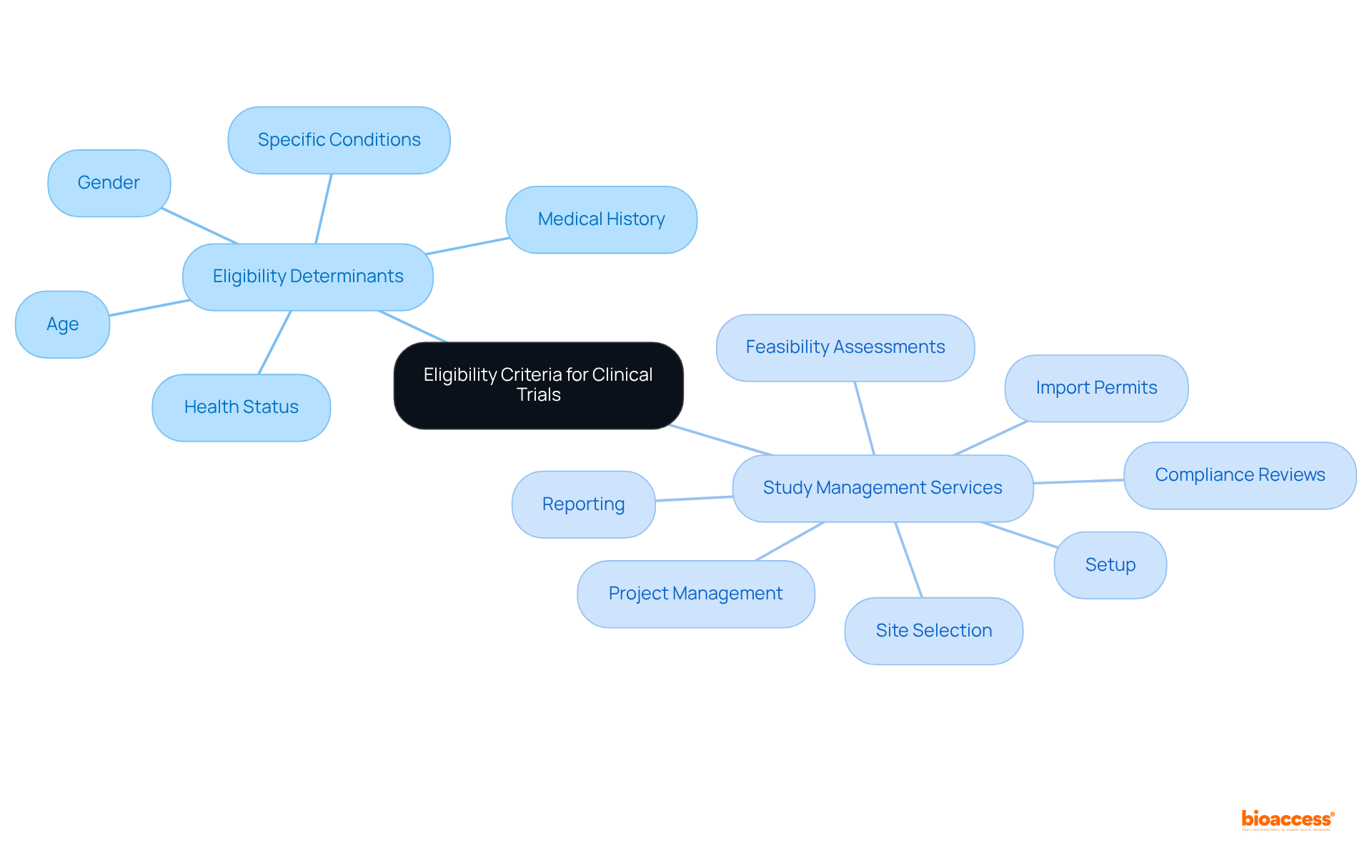
Eligibility Criteria: Who Can Participate in Paid Clinical Trials?
Eligibility criteria for are pivotal, varying significantly based on the study’s objectives and framework. Common determinants include age, gender, , and medical history. Some studies may require participants to possess , while others seek healthy volunteers. Understanding these criteria is essential for prospective candidates to and prepare for the .
At bioaccess®, we are at the forefront of supporting across Latin America. Our extensive encompass:
- Feasibility assessments
- Site selection
- Compliance reviews
- Setup
- Import permits
- Project management
- Reporting
This comprehensive approach ensures that every aspect of the study adheres to while prioritizing .
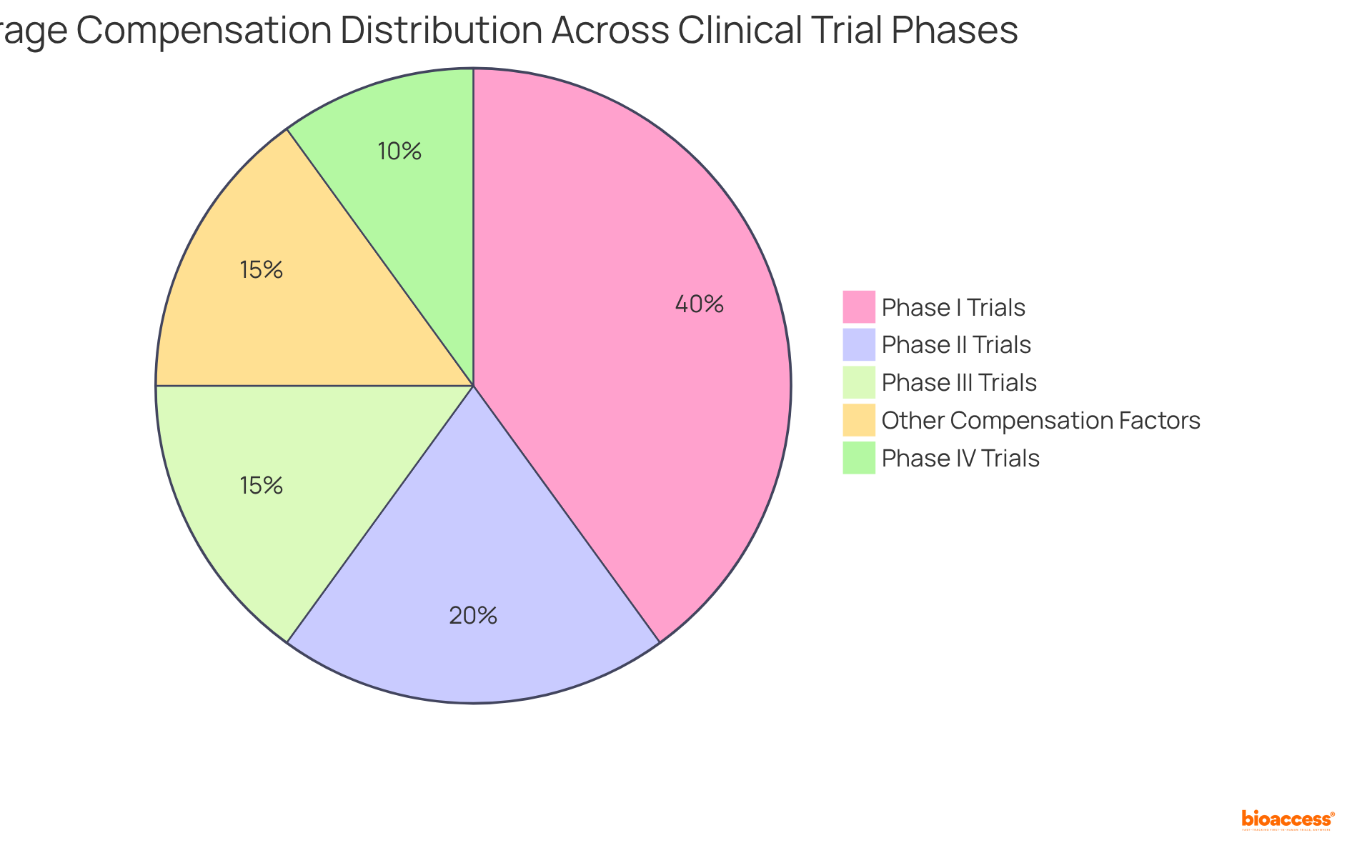
Compensation and Costs: Financial Aspects of Participating in Clinical Trials
Compensation for participation in varies significantly, influenced by factors such as the study’s complexity, duration, and phase. Participants may receive payments ranging from $75 to $4,500, with generally offering the highest compensation due to increased risks and time commitments. For instance, Phase I research averages around $2,000, while Phase IV examinations typically provide lower remuneration, averaging about $400.
In addition to direct payments, many studies cover , alleviating financial burdens on participants. Some studies may reimburse individuals for travel expenses or provide stipends for childcare, .
Understanding these is crucial for individuals considering enrollment in research studies, as they can significantly influence the decision to participate. Participants are encouraged to inquire about compensation details during the , ensuring clarity regarding expectations. This transparency not only helps manage expectations but also fosters trust between participants and study sponsors.
Moreover, the costs associated with . For example, conducting experiments in countries like Spain, Portugal, and Italy tends to incur lower operational expenses compared to more expensive nations such as Germany and the UK. This variability underscores the importance of thoroughly investigating potential studies to identify opportunities that align with both financial and personal circumstances.
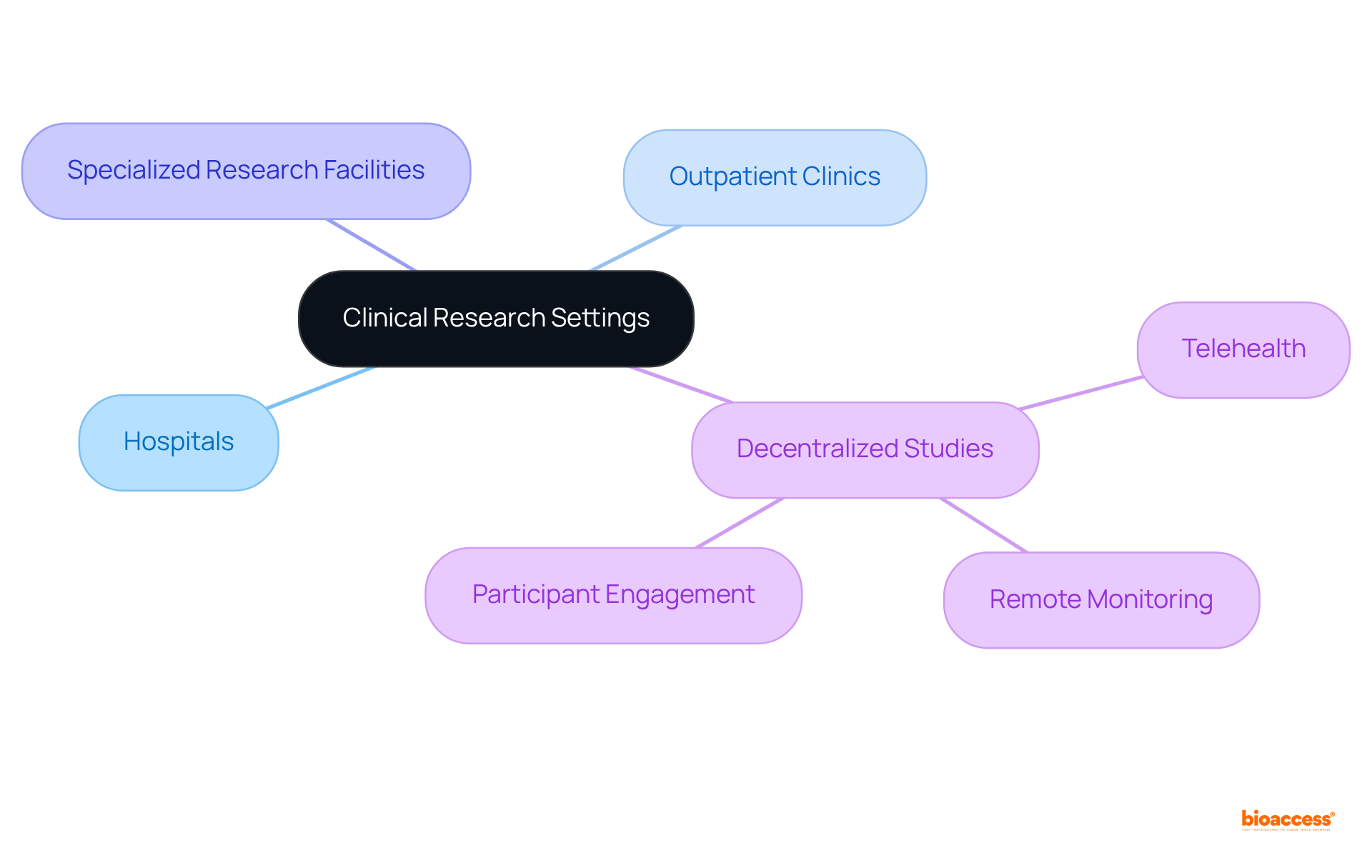
Trial Settings: Where Clinical Research Takes Place
are conducted across diverse settings, including hospitals, outpatient clinics, and specialized research facilities. Increasingly, these studies are adopting , which facilitate research in the comfort of individuals’ homes. This innovative approach leverages telehealth and remote monitoring technologies, significantly .
With nearly 70% of the population residing two hours or more from an academic medical facility, not only enable but also . Understanding the is crucial for participants, as it directly influences their experience and the level of support they receive throughout the research process.
Following the endorsing , the landscape of medical research is evolving to prioritize , ultimately improving recruitment and retention rates.
Participant Experience: What to Expect During a Clinical Trial
Participants in research with can anticipate a meticulously organized experience, commencing with a thorough screening process, informed consent, and continuous evaluations throughout the project. Our extensive encompass:
- Site selection
- Compliance reviews
- Study setup
- Import permits
- Project management
- Reporting
Individuals receive detailed information regarding the trial’s procedures, , and their rights as contributors. Effective communication with the is paramount, fostering a supportive environment that keeps individuals informed and engaged at every stage. Data indicates that , underscoring the importance of a positive experience. Feedback from previous participants reveals high satisfaction levels, with . As Viswanathan Mohan observes, “.” Moreover, 99.2% of individuals felt valued as research collaborators, highlighting the necessity of ongoing engagement with the . By understanding what to expect, participants can navigate their research journey with confidence, contributing to the advancement of and treatments while benefiting from the economic development and healthcare improvements that medtech studies bring to local communities.
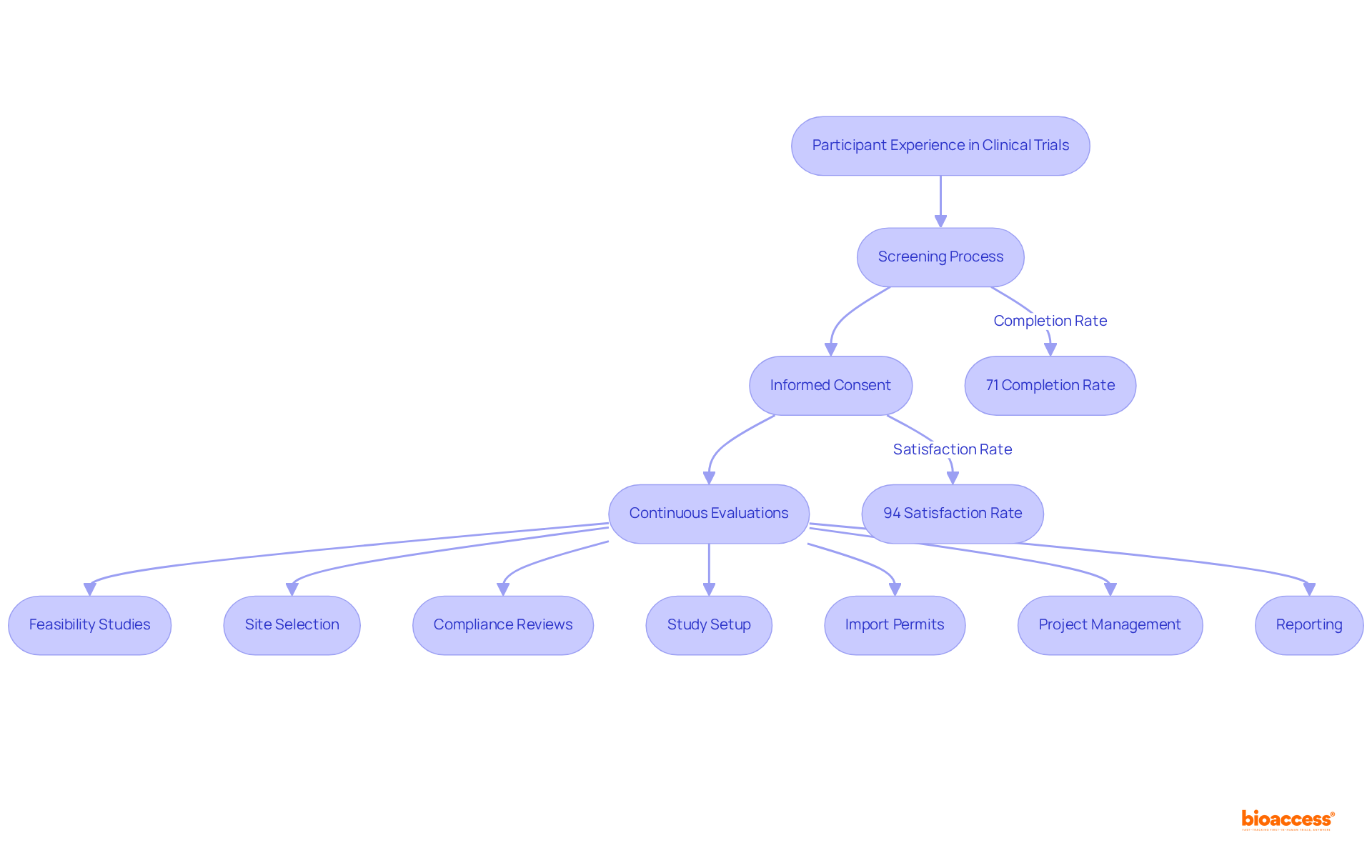
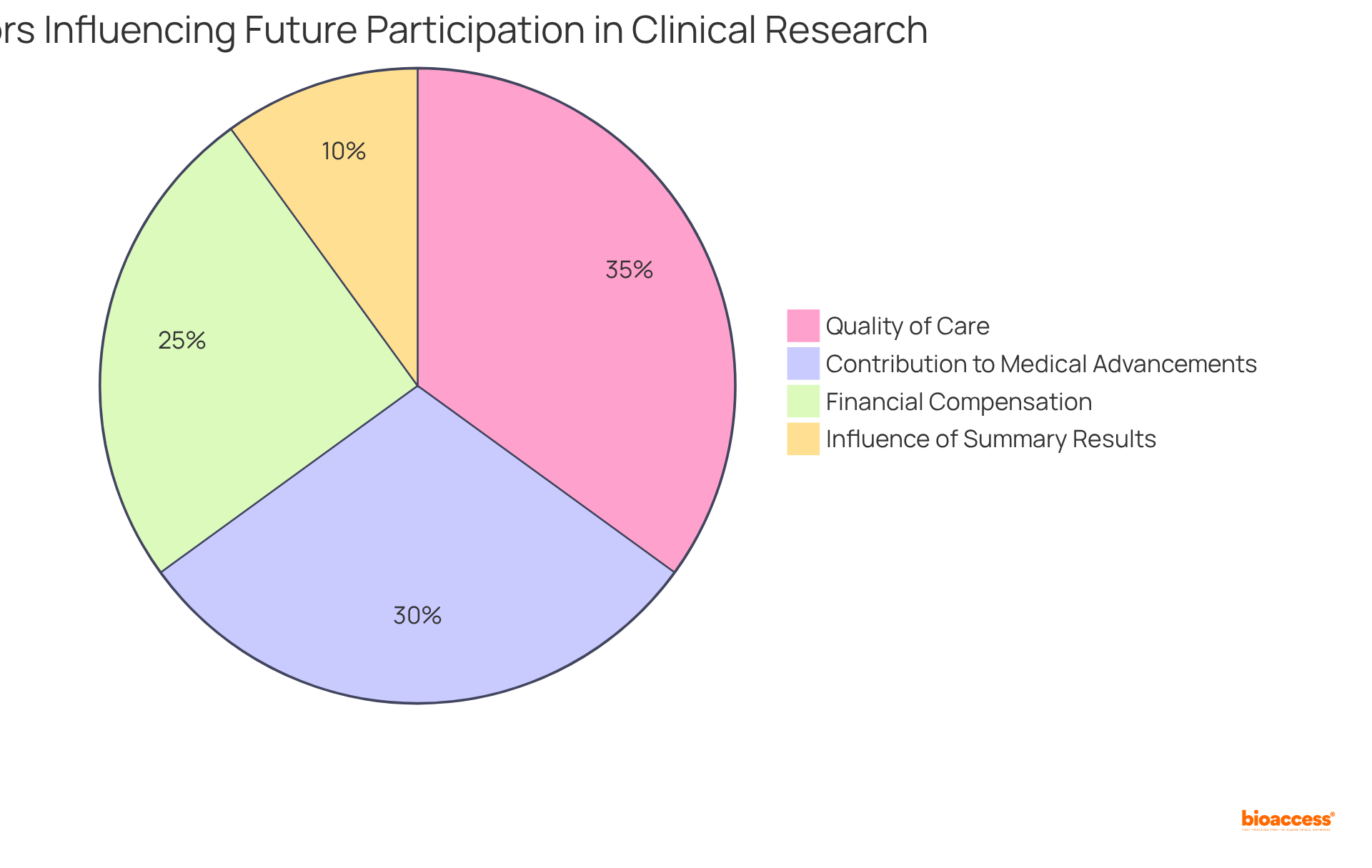
Participant Sentiment: Would They Do It Again?
, with an impressive 94% indicating a likelihood of future engagement, as evidenced by a recent survey. Key factors driving this willingness encompass:
- The high quality of care received
- The
The collaboration between bioaccess™ and the Caribbean Health Group to establish in Latin America, supported by Colombia’s Minister of Health, enhances the overall quality and accessibility of these studies. ; notably, 72% of respondents stated that receiving summary results would influence their decision to participate in future research. Furthermore, participants highlighted that the , underscoring the importance of logistical considerations.
The , which include:
- Feasibility analyses
- Site selection
- Compliance assessments
- Study setup
- Import permits
- Project management
- Reporting
contribute to a more streamlined experience for participants in paid clinical research trials. Overall, insights gathered from previous participants are essential for refining trial designs and improving the overall experience for future enrollees. As Rhonda G Kost aptly notes, “Research participants’ feedback about their participation experiences offers critical insights for improving programs.
Conclusion
Participation in paid clinical research trials represents a vital opportunity for individuals to contribute to medical advancements while potentially reaping significant benefits. This article has explored various aspects of clinical trials, emphasizing their crucial role in drug development and innovation. The insights provided illustrate not only the financial incentives and motivations for participants but also the ethical frameworks that safeguard their rights and well-being throughout the research process.
Key points discussed include:
- The accelerated timelines and cost efficiencies offered by organizations like bioaccess®, which enhance patient recruitment and access to cutting-edge treatments.
- The diverse motivations behind participation, ranging from financial compensation to altruistic desires to aid others, highlight the complex landscape of clinical trial involvement.
- Understanding the risks and benefits, eligibility criteria, and participant experiences is essential for individuals considering enrollment in these studies.
Ultimately, the significance of clinical trials extends beyond individual participation; they are foundational to healthcare advancements and the development of new treatments. As the landscape of clinical research continues to evolve, fostering awareness and understanding among potential participants is crucial. Engaging with healthcare professionals and staying informed about the latest trends can empower individuals to make informed decisions about their involvement in clinical trials, thereby contributing to the ongoing journey of medical innovation and improvement in patient care.
Frequently Asked Questions
What is bioaccess® and what role does it play in clinical research trials?
bioaccess® is an organization that leverages its extensive knowledge and strategically located sites across Latin America, particularly in Colombia, to expedite ethical approvals and patient recruitment in clinical research studies.
Why is Colombia considered a prime location for first-in-human studies?
Colombia is seen as a premier destination for first-in-human studies due to its over 30% cost savings compared to North America and Western Europe, along with a swift regulatory process that completes the total IRB/EC and MoH (INVIMA) review in just 90-120 days.
How quickly can bioaccess® conclude research studies?
bioaccess® can conclude research studies in as little as 4-6 weeks, significantly quicker than traditional markets.
What percentage of patients are willing to participate in research studies if informed?
Recent findings indicate that 75% of patients who were previously unaware of research studies expressed a willingness to participate if informed.
What is the significance of clinical trials in drug development?
Clinical trials are essential for the advancement of new medications and medical equipment, serving as the primary method for assessing safety and effectiveness, and providing critical data for regulatory approval.
Can you provide an example of a successful clinical study conducted by bioaccess®?
An example is ReGelTec’s Early Feasibility Study on HYDRAFIL™ in Colombia, which effectively treated eleven patients suffering from chronic low back pain.
How does bioaccess® enhance research services?
bioaccess® collaborates with various organizations to enhance research services, achieving over a 50% reduction in recruitment time and maintaining a 95% retention rate.
What motivates individuals to participate in paid clinical research trials?
Individuals are motivated by various factors, including financial compensation, hope for superior treatment options, trust in healthcare providers, access to advanced therapies, altruism, and the belief that the benefits of the study outweigh the risks.
What percentage of individuals believe the benefits of participating in a study outweigh the risks?
Approximately 31% of individuals believe that the perceived overall benefits of the study outweigh the risks.
List of Sources
- bioaccess®: Accelerating Paid Clinical Research Trials with Global Agility
- languageconnections.com (https://languageconnections.com/clinical-trials-in-latin-america)
- Latin American patients must wait an average of 4.7 years to access innovative or cutting-edge treatments (https://fifarma.org/en/waitindicator2024)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- statista.com (https://statista.com/statistics/1013599/latin-america-clinical-trials)
- Regulatory approval of clinical trials: is it time to reinvent the wheel? (https://gh.bmj.com/content/9/1/e013727)
- Clinical Trials: The Backbone of Drug Development and Innovation
- Clinical Trials Day 2025: Top 10 Statistics for Clinical Trials in 2025 (https://xtalks.com/clinical-trials-day-2025-top-10-statistics-for-clinical-trials-in-2025-4250)
- Clinical Trials Statistics and Facts (2026) (https://media.market.us/clinical-trials-statistics)
- 2025 Clinical Data Trend Report | Veeva (https://veeva.com/2025-clinical-data-trend-report)
- antidote.me (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- Diverse Motivations: Why People Choose to Participate in Paid Clinical Trials
- Clinical Trials Day 2025: Top 10 Statistics for Clinical Trials in 2025 (https://xtalks.com/clinical-trials-day-2025-top-10-statistics-for-clinical-trials-in-2025-4250)
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC4957899)
- Clinical Trials Statistics and Facts (2026) (https://media.market.us/clinical-trials-statistics)
- tandfonline.com (https://tandfonline.com/doi/full/10.1080/15265161.2019.1630498)
- Ethical Frameworks: Safeguarding Participants in Clinical Trials
- Ethical Considerations in the Design and Conduct of Clinical Trials of Artificial Intelligence – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11380101)
- Number of clinical trials by year, country, region and income group (https://who.int/observatories/global-observatory-on-health-research-and-development/monitoring/number-of-clinical-trials-by-year-country-who-region-and-income-group)
- Clinical Trials Statistics and Facts (2026) (https://media.market.us/clinical-trials-statistics)
- bmcmedethics.biomedcentral.com (https://bmcmedethics.biomedcentral.com/articles/10.1186/s12910-015-0022-z)
- Statistics and ethics: Models for strengthening protection of human subjects in clinical research | PNAS (https://pnas.org/doi/10.1073/pnas.0912882107)
- Understanding Risks and Benefits: Key Considerations for Participants
- abbvieclinicaltrials.com (https://abbvieclinicaltrials.com/resources/risks-and-benefits-of-participating-in-a-research-study)
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- Benefits of Participation in Clinical Trials: An Umbrella Review | MDPI (https://mdpi.com/1660-4601/19/22/15368)
- webmd.com (https://webmd.com/women/features/participating-in-clinical-trial-risks-benefits)
- statista.com (https://statista.com/statistics/621713/participation-in-clinical-trials-in-us-adults)
- Eligibility Criteria: Who Can Participate in Paid Clinical Trials?
- Clinical Trials Statistics and Facts (2026) (https://media.market.us/clinical-trials-statistics)
- 2025 Clinical Data Trend Report | Veeva (https://veeva.com/2025-clinical-data-trend-report)
- Clinical Trials Day 2025: Top 10 Statistics for Clinical Trials in 2025 (https://xtalks.com/clinical-trials-day-2025-top-10-statistics-for-clinical-trials-in-2025-4250)
- Trends and Charts on Registered Studies | ClinicalTrials.gov (https://clinicaltrials.gov/about-site/trends-charts)
- Rebooting the Statistic That 5% of Eligible Patients Participate in Clinical Trials | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/rebooting-the-statistic-that-5-of-eligible-patients-participate-in-clinical-trials)
- Compensation and Costs: Financial Aspects of Participating in Clinical Trials
- The Ultimate Guide to Clinical Trial Costs in 2025 (https://sofpromed.com/ultimate-guide-clinical-trial-costs)
- velocityclinicaltrials.com (https://velocityclinicaltrials.com/get-paid-participating-clinical-trial)
- statnews.com (https://statnews.com/2025/07/17/medical-research-trial-participants-payment-ethics-irbs)
- antidote.me (https://antidote.me/blog/how-to-find-and-take-part-in-paid-clinical-trials)
- payquicker.com (https://payquicker.com/how-much-do-clinical-trials-pay)
- Trial Settings: Where Clinical Research Takes Place
- qualtrics.com (https://qualtrics.com/blog/research-quotes)
- antidote.me (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- Clinical Trials Statistics and Facts (2026) (https://media.market.us/clinical-trials-statistics)
- Clinical Trials Day 2025: Top 10 Statistics for Clinical Trials in 2025 (https://xtalks.com/clinical-trials-day-2025-top-10-statistics-for-clinical-trials-in-2025-4250)
- Decentralized Clinical Trials – Key Trends and Statistics | Medidata Solutions (https://medidata.com/en/decentralized-clinical-trials-key-trends-and-statistics)
- Participant Experience: What to Expect During a Clinical Trial
- Strategies for participant retention in long term clinical trials: A participant –centric approaches – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10003583)
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S2590059523001097)
- antidote.me (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- Clinical Trials Statistics and Facts (2026) (https://media.market.us/clinical-trials-statistics)
- antidote.me (https://antidote.me/blog/7-clinical-trial-recruitment-and-retention-tips)
- Participant Sentiment: Would They Do It Again?
- mdpi.com (https://mdpi.com/2077-0383/14/10/3279)
- researchgate.net (https://researchgate.net/publication/387931077_What_Research_Participants_Say_About_Their_Research_Experiences_in_Empowering_the_Participant_Voice_Outcomes_and_Actionable_Data)
- What research participants say about their research experiences in Empowering the Participant Voice: Outcomes and actionable data – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11930633)
- wcgclinical.com (https://wcgclinical.com/insights/motivators-and-deterrents-to-clinical-trial-participation)
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S2590059523001097)

Leave a Reply