Introduction
Navigating the MedTech landscape in Brazil poses a distinctive array of challenges and opportunities for innovators. With a rapidly evolving regulatory framework and a burgeoning market, companies must implement strategic market access strategies to thrive in this competitive environment. This article delves into nine essential strategies that can empower MedTech firms to effectively penetrate the Brazilian market, enhance patient access to cutting-edge technologies, and ultimately drive growth.
How can companies align their offerings with local needs while surmounting regulatory hurdles? The following insights will illuminate this critical journey.
bioaccess®: Accelerate Clinical Research for MedTech in Brazil
bioaccess® excels in for MedTech companies in Brazil, employing over 15 years of industry expertise and a profound understanding of local regulations to develop effective . The organization secures , . This rapid approval process, combined with , establishes a to develop for MedTech in Brazil and swiftly bring their products to market.
In a competitive environment where time-to-market is critical, the not only enhance the likelihood of success but also position bioaccess® as an essential partner for companies eager to capitalize on the expanding medical technology sector. The strategic emphasis on highlights the significance of ethical approvals in fostering innovation and .
Understand Brazil’s Regulatory Framework for Medical Devices
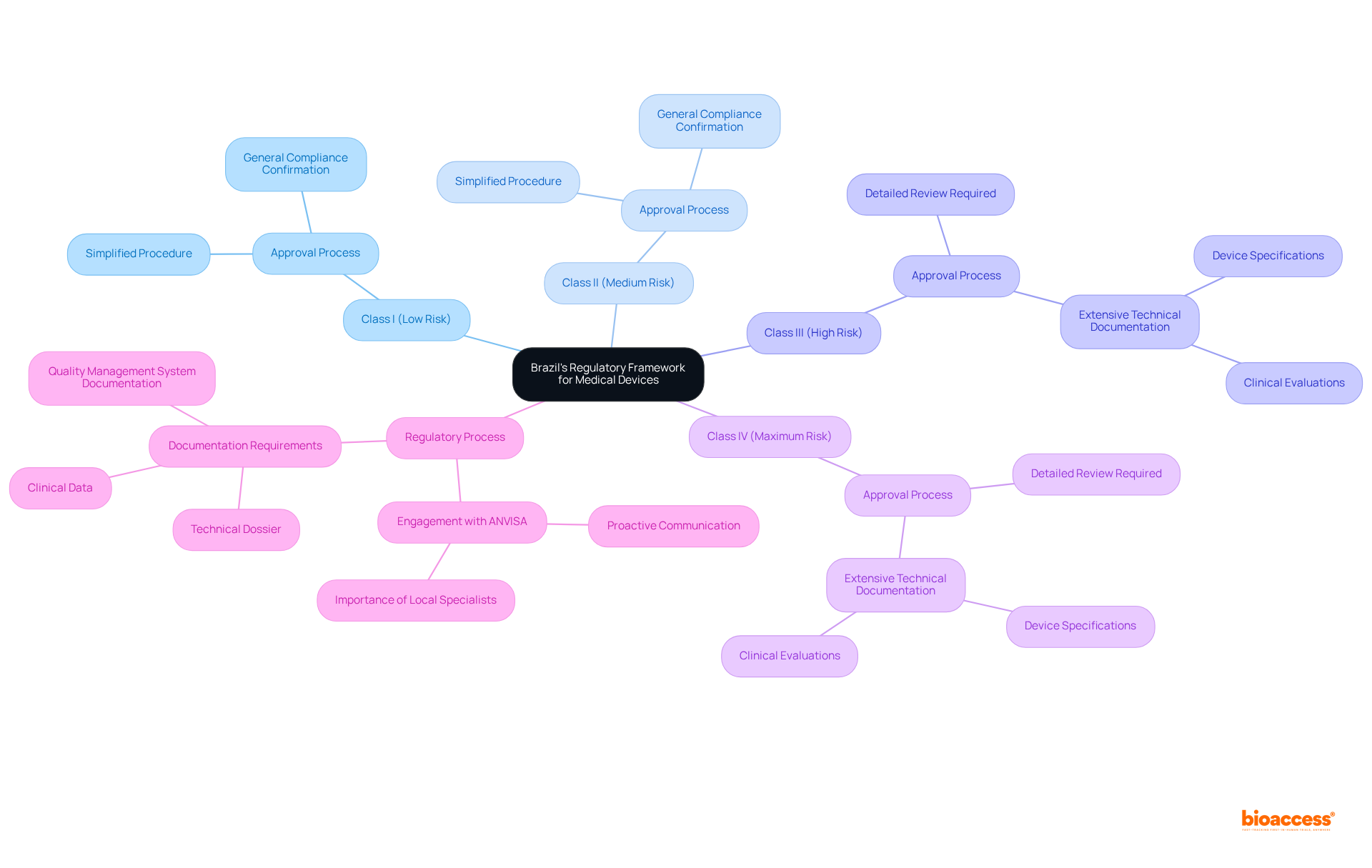
The in the country is primarily overseen by ANVISA (Agência Nacional de Vigilância Sanitária), which ensures that these devices meet stringent safety, efficacy, and quality standards. The classification of medical devices into four categories—Class I (low risk), Class II (medium risk), Class III (high risk), and Class IV (maximum risk)—is critical, as it dictates the regulatory pathway and documentation requirements for approval. For example, Class III and IV devices require a thorough review of extensive technical documentation, including device specifications and clinical evaluations, whereas Class I and II devices benefit from a simplified approval process that mandates general compliance confirmation.
In 2025, ANVISA updated its oversight agenda to adapt to emerging technologies, underscoring the necessity for manufacturers to remain informed about . Engaging with local regulatory specialists is essential for , which includes challenges such as and the demand for comprehensive documentation. Their expertise can significantly enhance and expedite the approval schedule, which typically sees ethical approvals in the country taking only 4-6 weeks. Furthermore, proactive communication with ANVISA can yield better outcomes, as nearly 67% of FDA 510(k) submissions resulted in requests for additional information during the review process, highlighting the importance of meticulous documentation and preparation.
By understanding the intricacies of pre-market approval, , and post-market monitoring, companies can implement for medtech in Brazil to position themselves for successful entry into the expanding . bioaccess® provides a that encompasses feasibility studies, research site selection, principal investigator (PI) selection, and comprehensive project management. This strategic partnership not only facilitates compliance but also fosters innovation, ultimately enhancing patient safety and access to advanced medical technologies. To effectively navigate the regulatory landscape of the country, consider engaging with bioaccess® for .
Build Strategic Partnerships for Effective Market Entry
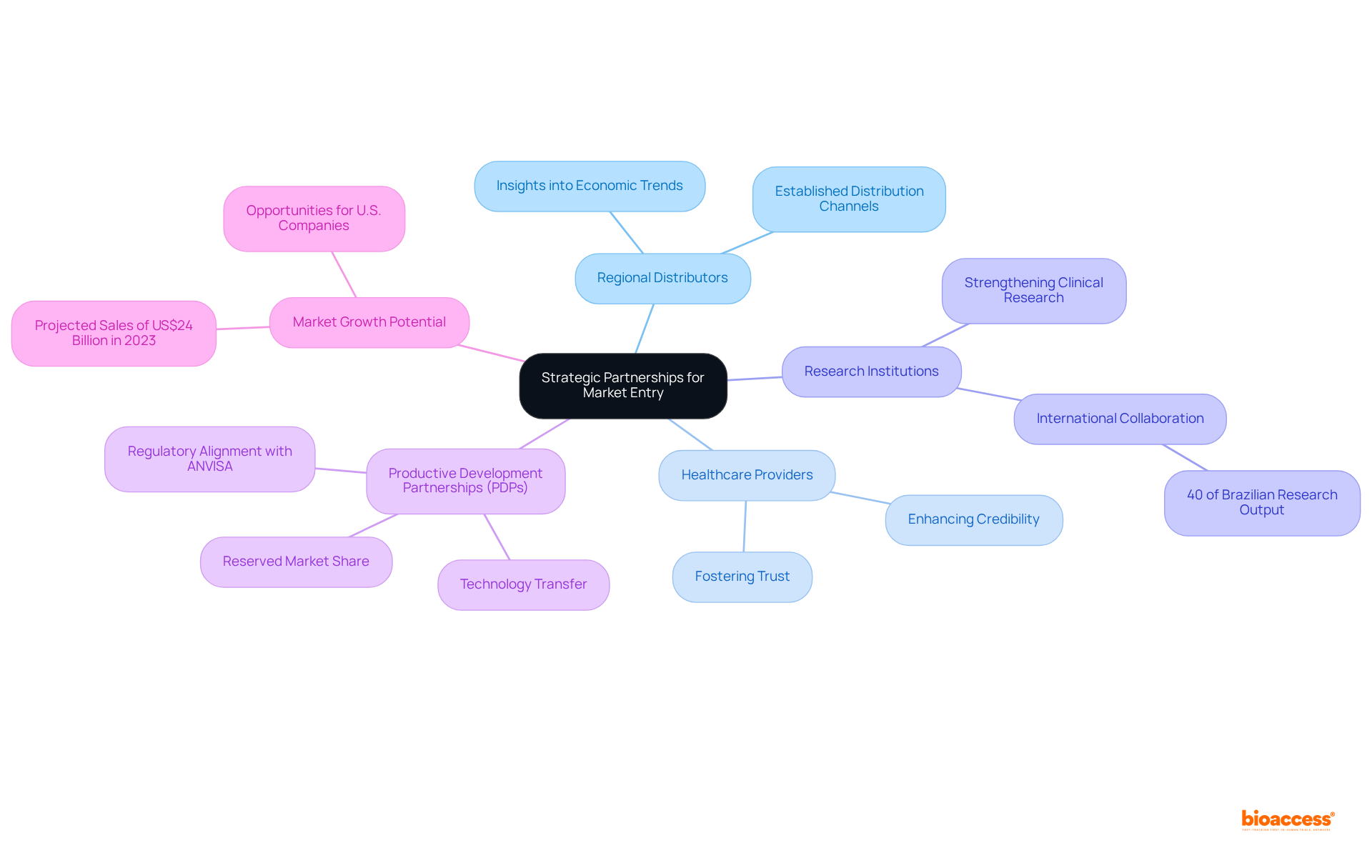
To develop effective for medtech in Brazil, forming , healthcare providers, and research institutions is essential. These partnerships offer crucial insights into , enabling businesses to tailor their strategies effectively. For example, the collaboration between bioaccess™ and Caribbean Health Group, announced on March 29, 2019, with the support of Colombia’s Minister of Health, illustrates how such partnerships can strengthen . By leveraging established distribution channels, MedTech firms can optimize their operations and expand their market reach. Furthermore, enhance credibility and foster trust among potential clients and stakeholders, which is vital in a competitive landscape.
(PDPs) allow international firms to collaborate with regional laboratories, facilitating and ensuring a reserved portion of the market share. Companies must thoroughly investigate all terms before entering into a PDP to maximize their advantages. This strategy not only boosts local production capabilities but also aligns with the country’s regulatory framework, overseen by ANVISA, akin to the FDA. As is projected to reach US$24 billion in 2023, the potential for growth through is substantial, especially given that approximately 40% of Brazilian research output is internationally collaborative. This underscores the critical role of partnerships in .
Leverage Local Market Insights to Tailor Products
Conducting in-depth research to understand the specific needs and preferences of Brazilian medical providers and patients is essential for devising . This involves evaluating , healthcare practices, and current market solutions. Notably, , underscoring the necessity of aligning products with regional expectations.
Additionally, MedTech companies must implement to navigate and sales tax regulations, which can present significant challenges in commercialization. By leveraging these insights, including the wisdom of Thomas A. Edison—”—MedTech companies can tailor their products to meet regional demands more effectively, thereby increasing their likelihood of successful adoption.
Firms such as bioaccess® exemplify this approach by offering comprehensive , including:
- Early-Feasibility Studies
- Compliance reviews
These services are crucial for . Companies that have modified their devices to align with local medical practices demonstrate a commitment to addressing the unique challenges faced by Brazilian providers. By implementing , MedTech innovators can position themselves for success in a competitive landscape.
Develop a Comprehensive Reimbursement Strategy
Developing necessitates a comprehensive understanding of the , particularly the interplay between . Engaging these payers early in the product development process is essential. This proactive engagement empowers companies to discern the , enabling them to craft compelling that underscore the . Such an approach not only facilitates but also aligns product offerings with the evolving dynamics of the country’s medical landscape, where approximately 60% of . By leveraging insights from payers, companies can refine their , which is essential for effective , ensuring alignment with the financial realities and priorities.
Engage Healthcare Stakeholders for Better Acceptance
Involving —physicians, hospital administrators, and patient advocacy groups—is essential for implementing . By hosting , relationships are built, and credibility within the healthcare community is established. further strengthens these connections, ensuring that products are developed with in mind.
As Majid U. emphasizes, are more readily adopted, highlighting the necessity of involving stakeholders throughout the development process. This strategy not only addresses concerns but also aligns innovations with the evolving trends in , thus paving the way for .
Generate Strong Clinical Evidence to Support Product Claims
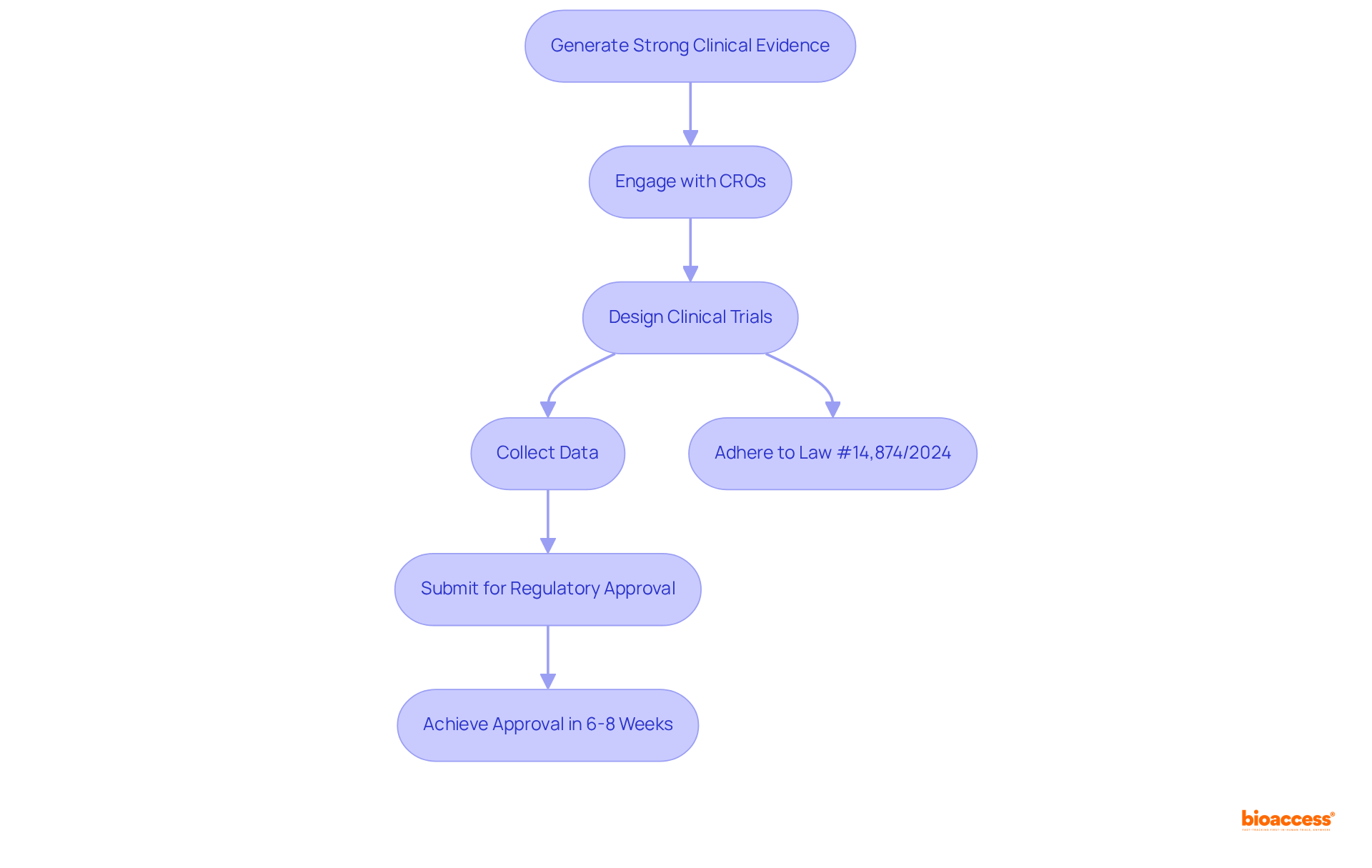
Producing robust is essential for MedTech firms seeking to demonstrate the safety and effectiveness of their products in the country. This endeavor necessitates the execution of meticulously designed clinical trials that adhere to the latest regulatory standards, such as those outlined in . Companies must prioritize the collection of data that not only fulfills regulatory requirements but also resonates with stakeholders involved in marketing and reimbursement discussions.
Engaging with like bioaccess® can significantly enhance this process. With over 15 years of expertise, bioaccess® specializes in in the region, ensuring that studies are conducted effectively and yield high-quality outcomes. Notably, bioaccess® employs a sprint approach that achieves regulatory approval in just 6-8 weeks, compared to the typical 6-12 months in the US and EU. This capability enables the enrollment of treatment-naive cardiology or neurology cohorts 50% faster than Western sites, leading to that demonstrate product safety and efficacy. Furthermore, bioaccess® facilitates $25K in and accelerates PMA data submission by 11 months, ultimately allowing for easier access within Brazil’s multi-billion dollar healthcare environment.
As the Brazilian industry evolves, particularly in light of recent legislative changes regarding clinical trial regulations, it is crucial for MedTech innovators to remain informed about and . This includes:
- Comprehending the current standards for
- Recognizing the necessity for
- Leveraging insights from CROs to optimize study protocols
By fostering cooperation among various stakeholders and implementing practical strategies, companies can produce compelling that supports their product claims and enhances their competitive edge.
Implement Effective Pricing Strategies for Market Success
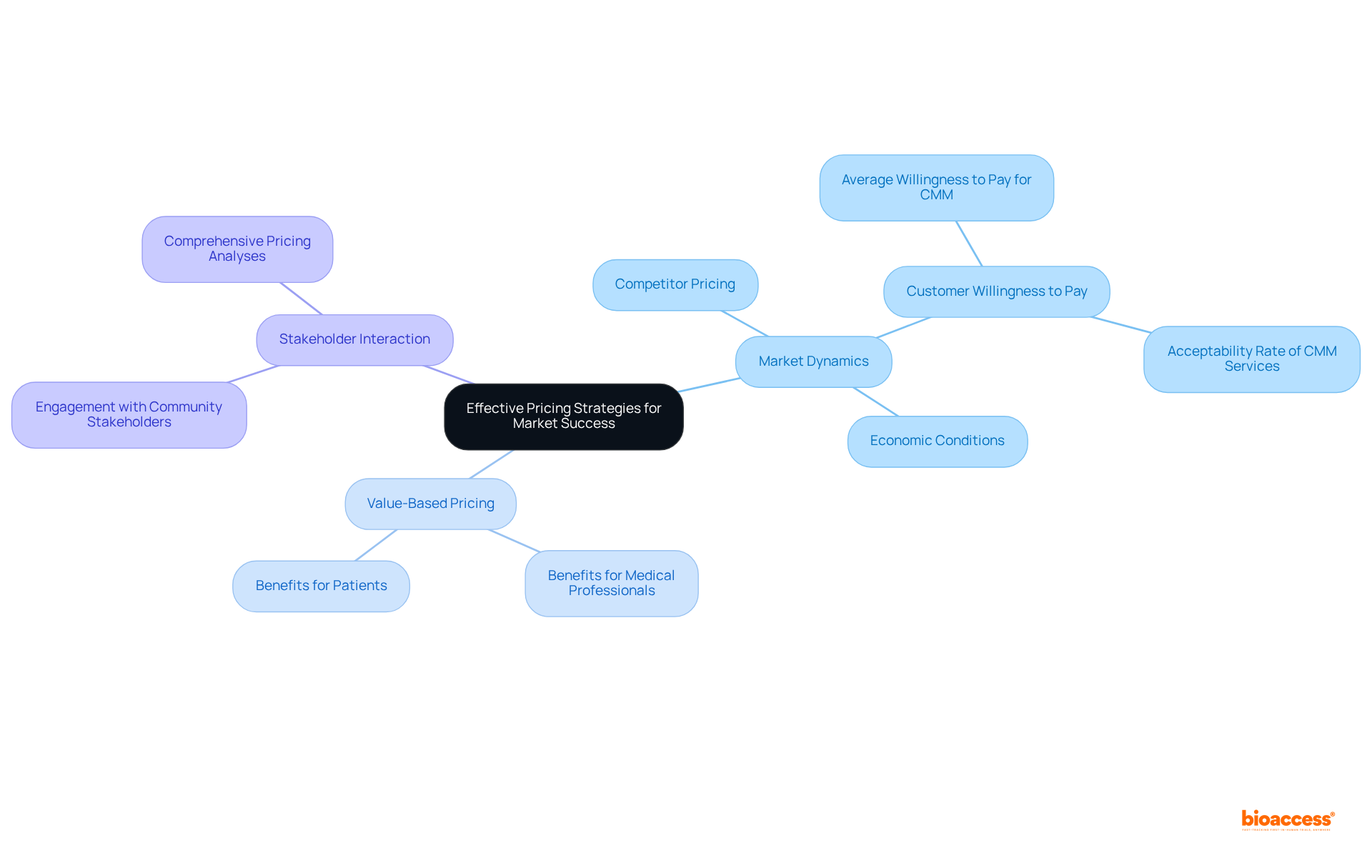
To attain success in Brazil, implementing effective is crucial. Companies must gain a comprehensive understanding of local market dynamics, including:
- Competitor pricing
- Customer willingness to pay
- Prevailing economic conditions
Notably, are particularly effective, as they align the pricing of with the tangible benefits they provide to medical professionals and patients. Recent studies indicate that the average (CMM) services is approximately $17.75 (40.00 BRL), reflecting a significant openness among consumers to invest in quality healthcare solutions.
Furthermore, the increasing demand for innovative in the country underscores the necessity for . Interacting with and performing can offer significant insights, allowing companies to establish optimal that achieve a balance between affordability and profitability.
Importantly, the , which allows for greater flexibility in . This approach not only improves access to the industry but also nurtures long-lasting connections with providers and individuals, ultimately propelling the in Brazil.
Adapt Marketing Strategies to Local Preferences
To effectively , it is imperative to implement that align with regional preferences. This requires a deep understanding of the prevalent among Brazilian medical professionals.
Businesses must create , integrating local languages and to cultivate connection and trust.
Collaborating with into , ultimately enhancing within this .
not only fortifies relationships but also guarantees that marketing efforts are culturally sensitive and impactful.
Monitor Market Trends and Regulatory Changes Continuously
Ongoing observation of industry trends and regulatory changes is crucial for developing effective . Staying informed about shifts in medical regulations, , and competitor activities is essential for organizations to develop effective . Establishing mechanisms for regular market analysis and engaging with local experts can significantly enhance and ensure compliance with evolving regulations. This proactive approach not only facilitates for medtech in Brazil but also helps maintain a competitive advantage in the country’s dynamic medical landscape.
With approximately 70% of , companies must prioritize and risk assessments to meet . Furthermore, organizations that have implemented comprehensive training initiatives report , compared to around 60% for those lacking such programs. This statistic underscores the importance of a knowledgeable workforce in adeptly navigating these regulatory changes. Additionally, starting in 2025, ANVISA will mandate a statement from plant suppliers confirming compliance with Good Agricultural and Collection Practices, marking a significant regulatory shift that MedTech companies must prepare for.
To support these efforts, bioaccess offers comprehensive , including:
- Feasibility studies
- Site selection
- Compliance reviews
- Trial setup
- Import permits
- Project management
- Reporting
These services not only streamline the clinical trial process but also contribute to and healthcare improvements, fostering international collaboration in the MedTech sector.
Conclusion
The landscape of MedTech in Brazil presents a multitude of opportunities for companies willing to navigate its complexities. By implementing tailored market access strategies, businesses can effectively position themselves within this growing sector. Understanding regulatory frameworks, forming strategic partnerships, and leveraging local market insights are pivotal components that enhance the likelihood of successful market entry.
Key strategies have been highlighted, including the importance of:
- Rapid ethical approvals
- Comprehensive reimbursement models
- The need for robust clinical evidence
Engaging with healthcare stakeholders and adapting marketing strategies to local preferences further solidify a company’s presence in this competitive environment. As Brazil’s MedTech market continues to evolve, proactive monitoring of trends and regulations will be essential for maintaining a competitive edge.
In conclusion, the MedTech sector in Brazil is ripe for innovation and growth. Companies must embrace a multifaceted approach that combines regulatory compliance, stakeholder engagement, and strategic market positioning. By doing so, they not only enhance their chances of success but also contribute to improving healthcare access and outcomes for Brazilian patients. The time to act is now—seize the opportunities that lie ahead in this dynamic market.
Frequently Asked Questions
What is bioaccess® and what services does it provide for MedTech companies in Brazil?
bioaccess® specializes in accelerating clinical research for MedTech companies in Brazil, leveraging over 15 years of industry experience and a deep understanding of local regulations to develop effective market access strategies.
How quickly can bioaccess® secure ethical approvals for clinical research?
bioaccess® can secure ethical approvals in an impressive 4-6 weeks, which is significantly faster than conventional sectors.
What advantages do MedTech companies gain by using bioaccess® for market access strategies?
By using bioaccess®, MedTech companies benefit from faster enrollment rates (50% quicker), streamlined pathways to market, and enhanced chances of success in the competitive medical technology sector.
Who oversees the regulatory framework for medical devices in Brazil?
The regulatory framework for medical devices in Brazil is primarily overseen by ANVISA (Agência Nacional de Vigilância Sanitária).
How are medical devices classified in Brazil and why is this important?
Medical devices in Brazil are classified into four categories: Class I (low risk), Class II (medium risk), Class III (high risk), and Class IV (maximum risk). This classification is important as it dictates the regulatory pathway and documentation requirements for approval.
What is the importance of engaging local regulatory specialists in Brazil?
Engaging local regulatory specialists is essential for navigating the complexities of the approval process, including prolonged timelines and comprehensive documentation demands, which can enhance compliance initiatives and expedite approvals.
What role do strategic partnerships play in market access strategies for MedTech in Brazil?
Strategic partnerships with regional distributors, healthcare providers, and research institutions are crucial for gaining insights into economic trends, optimizing operations, and expanding market reach, ultimately enhancing credibility and trust.
Can you provide an example of a successful partnership in the MedTech sector in Brazil?
An example is the collaboration between bioaccess™ and Caribbean Health Group, which strengthened clinical research capabilities and was supported by Colombia’s Minister of Health.
What are Productive Development Partnerships (PDPs) and how do they benefit international firms?
PDPs allow international firms to collaborate with regional laboratories, facilitating technology transfer and ensuring a reserved market share, thus boosting local production capabilities and aligning with Brazil’s regulatory framework.
What is the projected growth of Brazil’s pharmaceutical sector and how does it relate to market access strategies?
Brazil’s pharmaceutical sector is projected to reach US$24 billion in 2023, highlighting substantial growth potential through strategic partnerships, especially given that around 40% of Brazilian research output is internationally collaborative.
List of Sources
- bioaccess®: Accelerate Clinical Research for MedTech in Brazil
- bioaccessla.com (https://bioaccessla.com/br/blog/7-ways-croe-accelerates-clinical-research-for-medtech-startups)
- 10 Key Regulatory Pathways for Devices in Braz… | bioaccess® (https://bioaccessla.com/blog/10-key-regulatory-pathways-for-devices-in-brazil)
- bioaccessla.com (https://bioaccessla.com/br/blog/why-latin-america-is-a-medtech-hub-uncovering-the-key-drivers-of-growth)
- meddeviceonline.com (https://meddeviceonline.com/doc/succeeding-in-the-latin-american-medtech-market-three-tips-five-trends-0001)
- Latin America’s Landscape For Medtech Clinical Trials (https://clinicalleader.com/doc/latin-america-s-landscape-for-medtech-clinical-trials-0001)
- Understand Brazil’s Regulatory Framework for Medical Devices
- Master ANVISA Guidelines for Medtech Complianc… | bioaccess® (https://bioaccessla.com/blog/master-anvisa-guidelines-for-medtech-compliance-a-step-by-step-approach)
- regdesk.co (https://regdesk.co/resource-library/brazil)
- Approval of medical devices in Brazil (https://blog.johner-institute.com/regulatory-affairs/approval-from-medical-devices-in-brazil)
- 5 Medical Device Regulatory Approval Statistics You Need to Know – Arrotek | Medical Device Innovation (https://arrotek.com/5-medical-device-regulatory-approval-statistics-you-need-to-know)
- Build Strategic Partnerships for Effective Market Entry
- ir.clarivate.com (https://ir.clarivate.com/news-events/press-releases/news-details/2025/Clarivate-and-CAPES-Expand-Landmark-Partnership-to-Power-Research-and-Innovation-Across-Brazil/default.aspx)
- biospectrumasia.com (https://biospectrumasia.com/news/30/26097/brazil-and-gavi-reinforce-strategic-health-partnership-with-new-agreement.html)
- trade.gov (https://trade.gov/country-commercial-guides/brazil-healthcare)
- trial.medpath.com (https://trial.medpath.com/news/be9136bcd61b36b0/zydus-medtech-partners-with-braile-biomedica-to-expand-tavi-technology-across-europe-and-india)
- expresshealthcare.in (https://expresshealthcare.in/news/zydus-medtech-braile-biomedica-brazil-partner-to-commercialise-tavi-technology/448823)
- Leverage Local Market Insights to Tailor Products
- qualtrics.com (https://qualtrics.com/blog/research-quotes)
- Global Medical Device Podcast powered by Greenlight Guru (https://podcasts.apple.com/us/podcast/global-medical-device-podcast-powered-by-greenlight-guru/id1036394532)
- Develop a Comprehensive Reimbursement Strategy
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S2666560323000658)
- trade.gov (https://trade.gov/country-commercial-guides/brazil-healthcare)
- statista.com (https://statista.com/topics/5030/health-in-brazil)
- oecd.org (https://oecd.org/en/publications/2021/12/oecd-reviews-of-health-systems-brazil-2021_6797af6a.html)
- bmchealthservres.biomedcentral.com (https://bmchealthservres.biomedcentral.com/articles/10.1186/s12913-021-07376-2)
- Engage Healthcare Stakeholders for Better Acceptance
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC10012644)
- dokumen.pub (https://dokumen.pub/patent-games-in-the-global-south-pharmaceutical-patent-law-making-in-brazil-india-and-nigeria-9781509927395-9781509927425-9781509927401.html)
- tandfonline.com (https://tandfonline.com/doi/full/10.1080/24735132.2025.2506311)
- journals.plos.org (https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0209826)
- Generate Strong Clinical Evidence to Support Product Claims
- The Role of Clinical Research Organizations (CROs) in Nutraceutical Trials (https://sofpromed.com/the-role-of-clinical-research-organizations-cros-in-nutraceutical-trials)
- lickslegal.com (https://lickslegal.com/news/presidential-veto-on-the-law-of-clinical-trials-is-rejected-by-brazilian-congress)
- New Medical Device Regulations in 2025: Opportunities and Challenges | News | Pure Global (https://pureglobal.com/news/new-medical-device-regulations-in-2025-opportunities-and-challenges)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC7390662)
- Implement Effective Pricing Strategies for Market Success
- mystrategist.com (https://mystrategist.com/blog/article/brazil_benchmarks_device_prices.html)
- researchgate.net (https://researchgate.net/publication/337770303_Price-setting_strategies_and_practice_for_medical_devices_used_by_consumers)
- statista.com (https://statista.com/topics/13147/medical-technology-in-brazil)
- statista.com (https://statista.com/outlook/hmo/medical-technology/medical-devices/brazil)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC11044949)
- Adapt Marketing Strategies to Local Preferences
- mmm-online.com (https://mmm-online.com/home/channel/features/100-quotes-what-they-reveal-about-the-state-of-healthcare-advertising)
- cacm.acm.org (https://cacm.acm.org/research/digital-healthcare-in-latin-america)
- ajpe.org (https://ajpe.org/article/S0002-9459(23)01430-4/fulltext)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC11891519)
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S0969593114001607)
- Monitor Market Trends and Regulatory Changes Continuously
- mddionline.com (https://mddionline.com/business/brazil-s-medtech-market-to-hit-13-1-billion-in-7-years-report)
- 7 Key Insights on Post-Market Surveillance under ANVISA | bioaccess® (https://bioaccessla.com/blog/7-key-insights-on-post-market-surveillance-under-anvisa)
- bioaccessla.com (https://bioaccessla.com/blog/10-key-insights-on-class-ii-medical-devices-for-clinical-success)

Leave a Reply