Introduction
The landscape of clinical research is rapidly evolving, with electronic trial master file (eTMF) solutions at the forefront of this transformation. These advanced digital platforms serve as more than mere repositories for documents; they have emerged as essential tools that streamline operations, enhance compliance, and facilitate real-time collaboration among research teams.
As organizations navigate the complexities of regulatory requirements and strive for efficiency, a critical question arises: what are the key features that render eTMF solutions indispensable in today’s clinical trials? This article delves into ten essential functionalities that can significantly impact the success of clinical research, offering insights into how these tools can optimize processes and drive better outcomes.
bioaccess®: Accelerating Clinical Research with Integrated eTMF Solutions
bioaccess® harnesses to accelerate research processes in . By synergizing with diverse patient populations in the Balkans and , bioaccess® ensures that trials are not only compliant but also remarkably efficient. This strategic integration of the electronic trial master file etmf facilitates and fosters , both of which are crucial for adhering to stringent timelines in clinical research.
The Evolution of Electronic Trial Master File (eTMF) Solutions
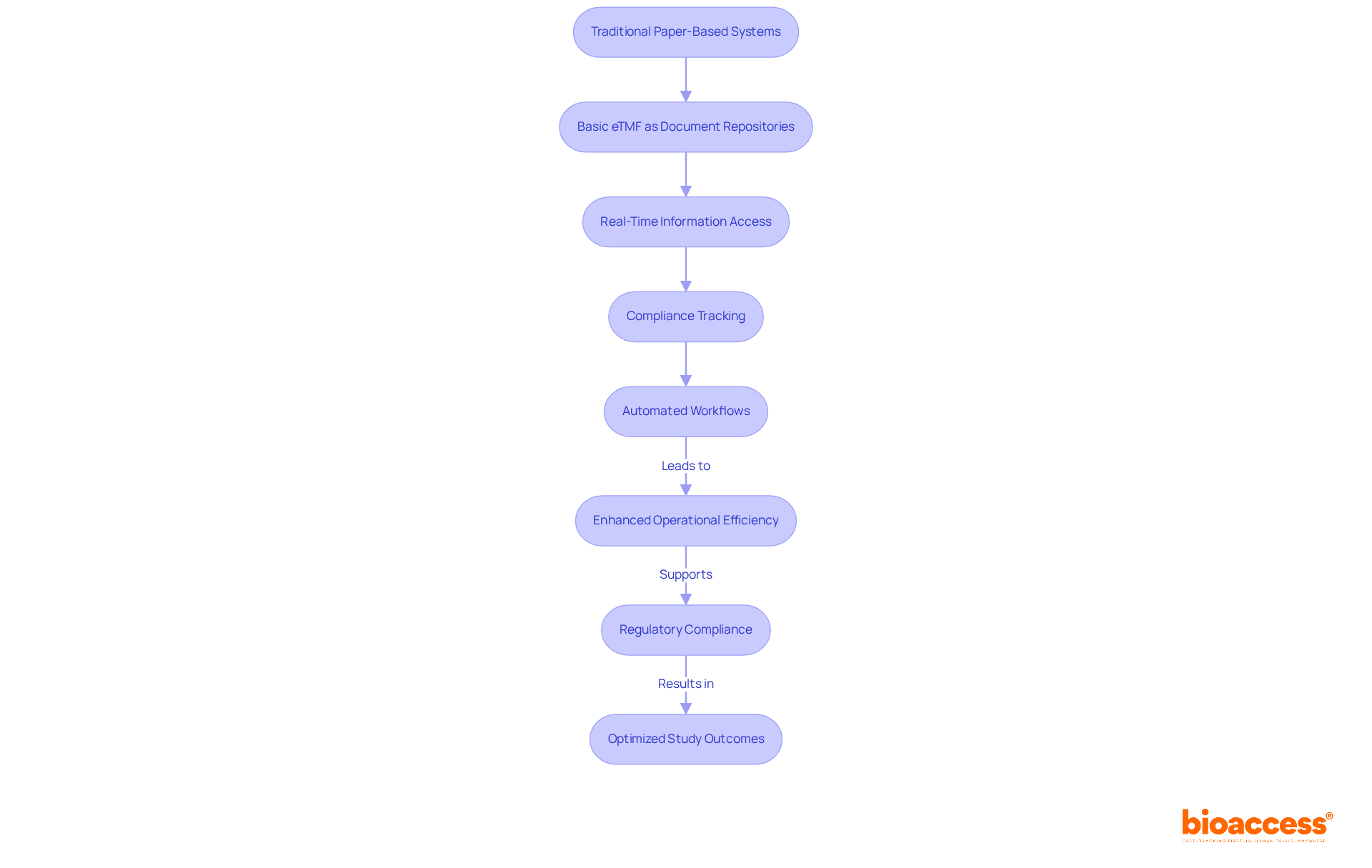
The has evolved significantly from traditional paper-based systems to sophisticated digital platforms, capturing the attention of . Initially, eTMFs served merely as document repositories. However, advancements in technology have transformed the into a dynamic system that facilitates real-time information access, compliance tracking, and automated workflows. This evolution is essential for addressing the growing demands of and ensuring the integrity of the study.
With , studies can enroll participants 50% quicker due to its advanced electronic document management features. This results in significant savings of $25K per patient through that eliminates rework and delays. These capabilities streamline testing procedures and assist startups in overcoming regulatory challenges, thereby accelerating approval processes and enhancing overall testing efficiency. The integration of such technology not only enhances operational efficiency but also positions organizations to meet the rigorous standards set by regulatory authorities.
In conclusion, the shift towards the represents a critical advancement in . Collaboration with innovative platforms like bioaccess® is vital for navigating the complexities of regulatory compliance and optimizing study outcomes. The next steps involve embracing these technologies to drive efficiency and effectiveness in .
Standalone vs. Integrated eTMF Systems: What You Need to Consider
When organizations are faced with the decision between standalone and integrated systems, it is crucial to consider several key factors:
- Scalability
- with other
Standalone systems may provide specialized features that cater to specific needs; however, they often result in that can hinder overall efficiency. In contrast, integrated systems offer a unified approach, significantly enhancing collaboration and data sharing across various platforms. The ultimate choice will depend on the unique requirements and resources of the organization, underscoring the importance of a tailored solution within the .
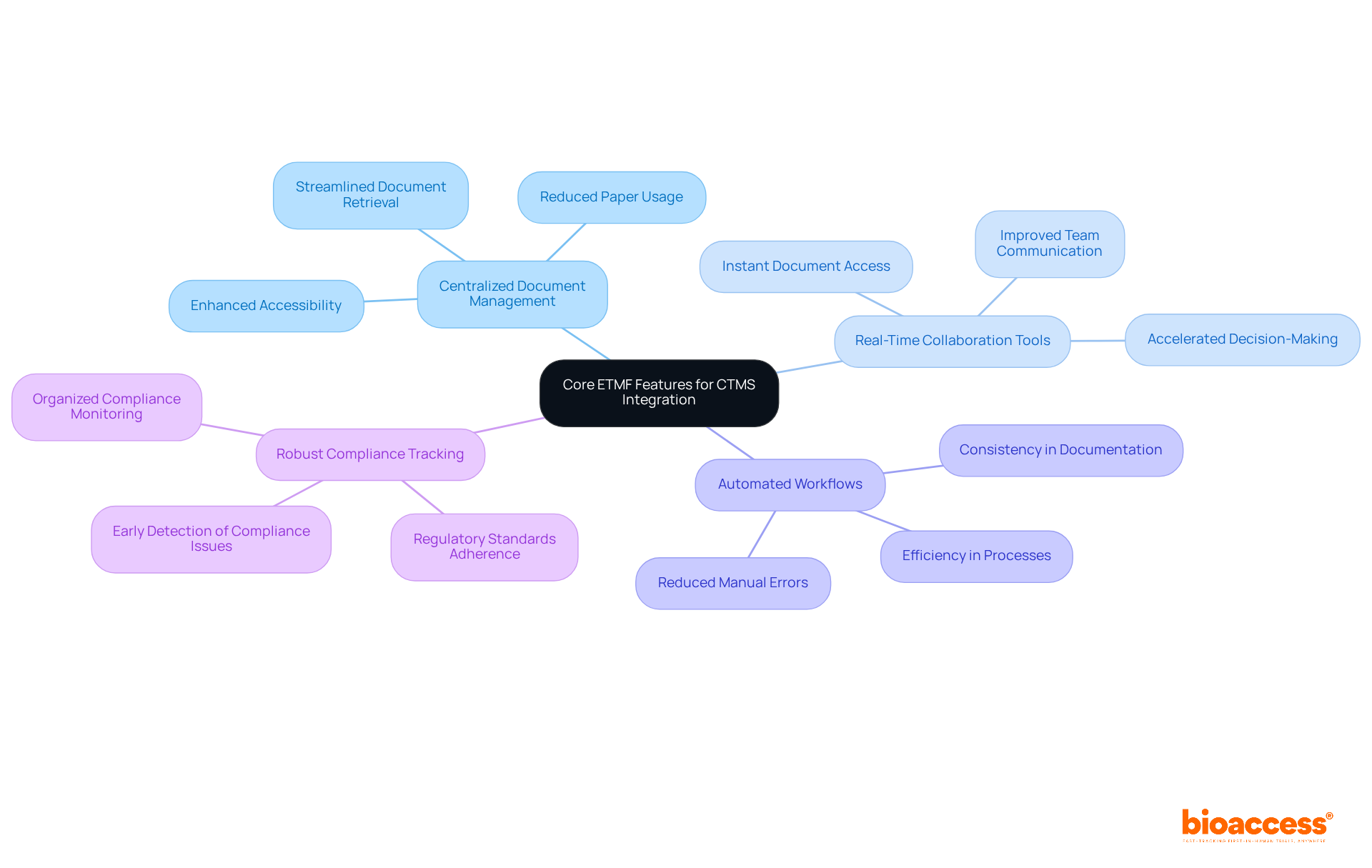
Core eTMF Features Essential for Effective CTMS Integration
, , , and robust are essential components of that significantly enhance . These functionalities ensure that all related documents are readily accessible and consistently updated, streamlining communication among teams and boosting overall efficiency.
Modern electronic management solutions facilitate instant access to documents, allowing teams to collaborate effectively regardless of location, thereby accelerating decision-making and minimizing delays in research processes. within the is crucial for meeting , providing an organized approach to monitoring and documenting compliance activities.
By leveraging these features, organizations can optimize their , ensuring they meet both operational and regulatory requirements efficiently. With bioaccess®’s solutions, organizations can enroll treatment-naive cardiology or neurology cohorts 50% faster than Western sites, achieving with —no rework, no delays.
Additional Features to Enhance Your eTMF and CTMS Solutions
Beyond core functionalities, additional features such as , customizable dashboards, and mobile access significantly enhance the eTMF and CTMS solutions. These characteristics not only enable improved information visualization but also facilitate and greater flexibility for users who require access to examination details while on the go. Implementing these enhancements can result in more efficient management of experiments and , underscoring their critical role in advancing .
Creating a Unified Clinical Trial Data Ecosystem with eTMF
(Electronic Capture of Information) and is vital for establishing a cohesive . This integration facilitates seamless information sharing and , providing all stakeholders with instant access to the latest details. By creating an integrated environment, organizations can significantly enhance operational effectiveness, ensuring compliance with regulatory standards and improving information integrity throughout the process.
For instance, increase by minimizing transcription errors, while streamlined workflows eliminate duplicate data entry and manual transfers. Successful implementations of the etmf with EDC and CTMS have showcased improved collaboration among study teams, leading to timely task completion and better overall management.
However, it is from teams adapting to new workflows, which can present challenges during the integration process. As Medha Datar observes, “The research landscape is constantly changing, with , handling information, and enhancing efficiency.
Evolving EDC Systems to Support Decentralized and Hybrid Clinical Trials
As clinical studies increasingly embrace decentralized and hybrid models, must adapt to meet these evolving requirements. Key characteristics essential for facilitating hybrid studies in 2025 include:
- Robust capabilities
- Electronic consent functionalities
These advancements enhance and streamline information collection processes, significantly improving overall study efficiency. For instance, bioaccess enables to be enrolled 50% faster than Western sites, resulting in $25K savings per patient with —no rework, no delays.
The integration of wearable devices allows for continuous monitoring of vital signs, enabling researchers to gather real-time information directly from participants. This shift towards is crucial, as research indicates that 80% of medical studies fail to meet their recruitment schedules, underscoring the necessity for .
can foster participation from typically underrepresented groups, enriching the diversity of research data. As Yvonne Chan, MD, states, ‘ and connected devices could facilitate valuable multi-dimensional, detailed, real-world medical data capture.’
By leveraging these features, EDC systems are poised to transform the landscape of , making studies more efficient and patient-centered.
Key Takeaways on Essential eTMF Features and Solutions
(ETMF) encompass:
- Centralized
These features are crucial for effective management of experiments and adherence to regulatory standards. Organizations must also contemplate additional functionalities, such as:
- Analytics
to enhance their electronic trial master file (ETMF) and (CTMS) solutions. A is vital for improving research procedures and achieving superior results.
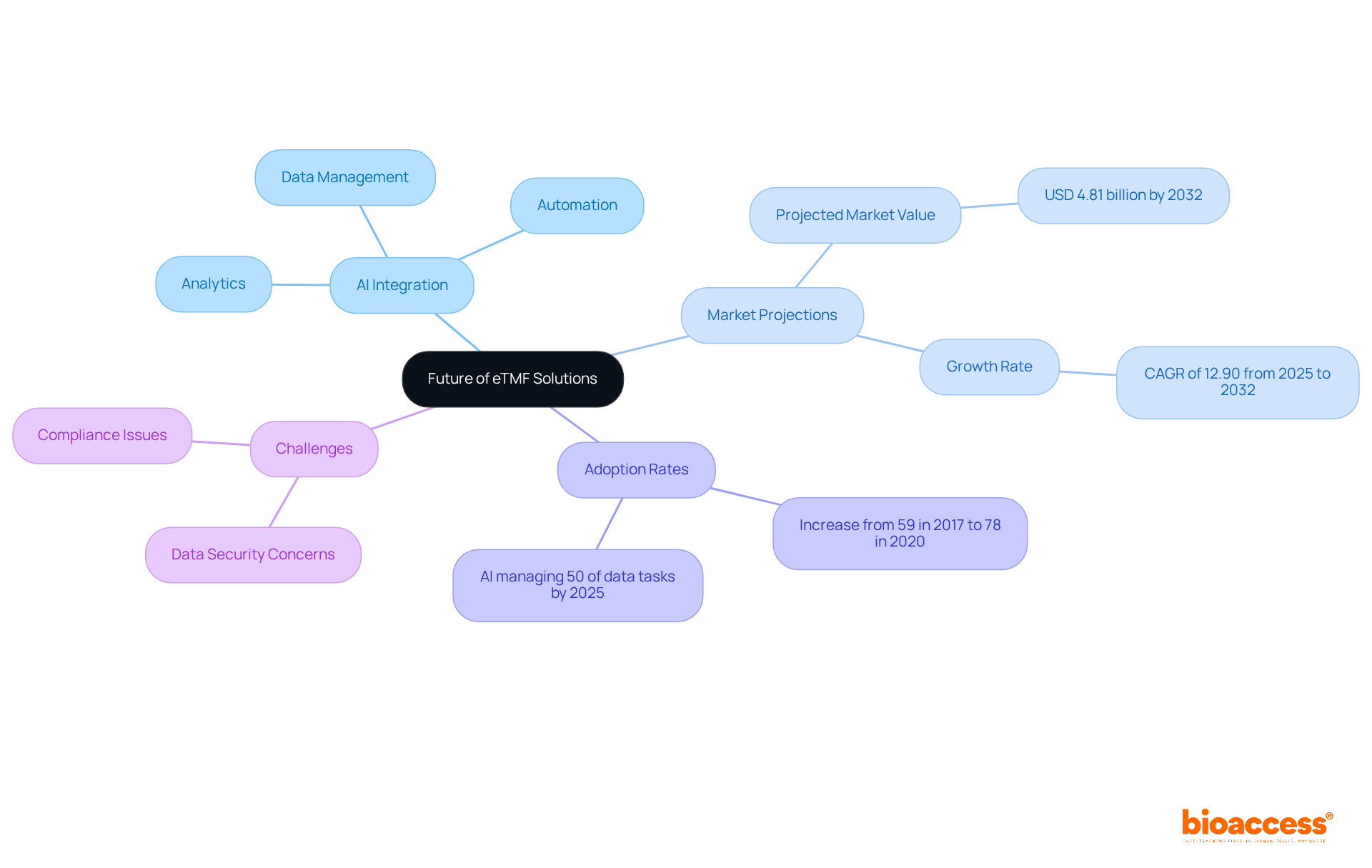
Looking Ahead: The Future of eTMF Solutions in Clinical Research
The future of the solutions in clinical research is poised for remarkable advancements, particularly through the integration of . These innovations are expected to greatly simplify testing procedures, enhance information quality, and . Notably, AI technologies are projected to manage up to 50% of research data tasks by 2025, by 20%.
Furthermore, the adoption of has surged, with the utilization rates of the increasing from 59% in 2017 to 78% in 2020. As Debashish Niyogi, Ph.D., noted, “the industry is requesting more efficient to create and manage .”
Organizations such as bioaccess, which offer —including feasibility studies, site selection, compliance reviews, setup, import permits, project management, and reporting—are strategically positioned to leverage these advancements. By proactively embracing these trends, they will be better equipped to navigate the complexities of modern , establishing themselves at the forefront of the industry.
Additionally, the electronic trial master file market is projected to reach USD 4.81 billion by 2032, underscoring its growing significance. However, challenges such as remain critical factors influencing the adoption of the systems.
Engage with bioaccess® for Expert eTMF Solutions and Support
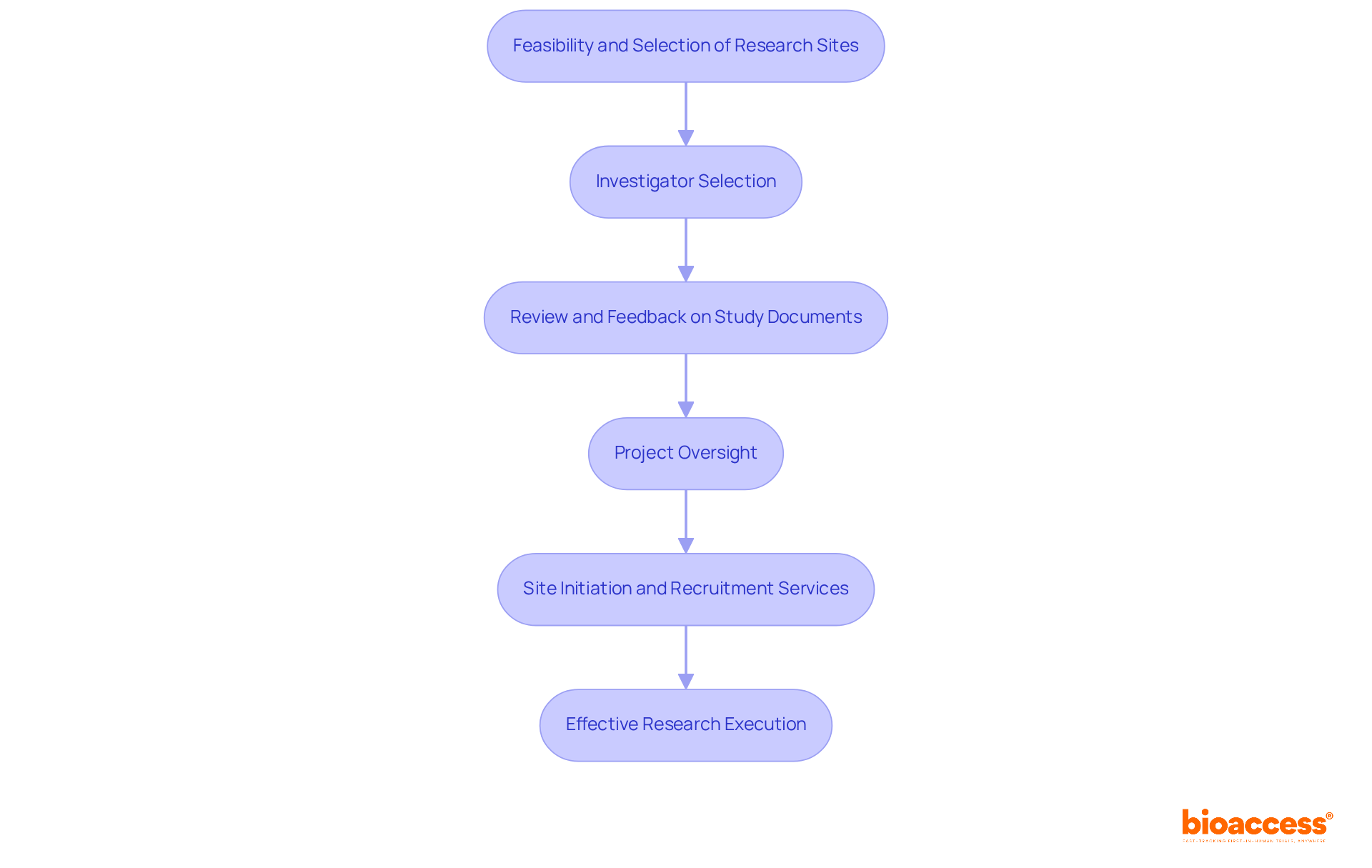
For organizations aiming to enhance their , collaborating with bioaccess® presents invaluable assistance. Our comprehensive approach encompasses:
- Feasibility and selection of
- Investigator selection
- Thorough review and feedback on to ensure compliance with country requirements and regulatory standards
Bioaccess® is adept at helping innovators streamline their research efforts.
Our expertise in project oversight and documentation guarantees effective study execution, complemented by expedited site initiation and tailored to the unique challenges of medical research in LATAM, Eastern Europe, and Australia. The importance of collaboration in cannot be overstated.
Book a meeting with bioaccess® to explore how we can support your effectively.
Conclusion
The transformation of electronic trial master file (eTMF) solutions signifies a pivotal shift in the clinical research landscape, enhancing both efficiency and compliance. Transitioning from traditional paper-based systems to advanced digital platforms enables organizations to leverage real-time collaboration, automated workflows, and centralized document management. This evolution allows stakeholders to navigate the complexities of regulatory requirements while streamlining research processes effectively.
Key insights reveal essential features of eTMF solutions, such as:
- Compliance tracking
- User-friendly interfaces
- Integration capabilities with other clinical trial management systems
The evolution of eTMF technology, particularly through platforms like bioaccess®, not only accelerates participant enrollment but also significantly reduces costs associated with clinical trials. Moreover, the integration of advanced analytics and mobile access is poised to enhance decision-making and optimize research outcomes.
Looking ahead, the proactive adoption of innovative eTMF solutions is crucial for organizations striving to remain competitive in the dynamic clinical research environment. As industry demands for efficiency and regulatory compliance continue to rise, embracing integrated eTMF systems will empower stakeholders to overcome challenges and achieve superior trial management. Engaging with expert solutions, such as those provided by bioaccess®, equips organizations with the necessary support to navigate this evolving landscape, ensuring that clinical trials are executed effectively and efficiently.
Frequently Asked Questions
What is bioaccess® and how does it enhance clinical research?
bioaccess® utilizes electronic trial master file (eTMF) solutions to accelerate research processes in clinical trials by integrating regulatory agility from Latin America, diverse patient populations in the Balkans, and optimized pathways in Australia. This ensures trials are compliant and efficient, facilitating expedited document management and real-time collaboration.
How has the electronic trial master file (eTMF) evolved over time?
The eTMF has evolved from traditional paper-based systems to advanced digital platforms that allow for real-time information access, compliance tracking, and automated workflows. This evolution is essential for meeting regulatory demands and ensuring study integrity.
What are the benefits of using bioaccess® for clinical trials?
bioaccess® enables studies to enroll participants 50% faster and provides FDA-ready data that eliminates rework and delays, resulting in significant cost savings of $25K per patient. These features streamline testing procedures and help startups overcome regulatory challenges, enhancing overall testing efficiency.
What factors should organizations consider when choosing between standalone and integrated eTMF systems?
Organizations should consider scalability, user experience, and integration capabilities with other clinical trial management systems (CTMS). Standalone systems may offer specialized features but can create data silos, while integrated systems enhance collaboration and data sharing.
Why is the shift towards electronic trial master file (eTMF) systems important in clinical research?
The shift to eTMF systems represents a critical advancement in clinical research, enabling organizations to navigate regulatory compliance complexities and optimize study outcomes. Embracing these technologies is essential for driving efficiency and effectiveness in clinical trials.
List of Sources
- Core eTMF Features Essential for Effective CTMS Integration
- cluepoints.com (https://cluepoints.com/centralized-monitoring-in-clinical-trials-everything-you-should-know)
- Top eTMF Features – Flex Databases (https://flexdatabases.com/blog/top-etmf-features)
- Premier Research | Data at Your Fingertips: The Case for Centralized Monitoring (https://premier-research.com/perspectives/data-at-your-fingertips-the-case-for-centralized-monitoring)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC10328794)
- quanticate.com (https://quanticate.com/blog/the-benefits-of-centralized-clinical-data)
- Creating a Unified Clinical Trial Data Ecosystem with eTMF
- evidentiq.com (https://evidentiq.com/library/clinical-trial-software)
- Modernizing Clinical Trials (https://credevo.com/articles/2025/08/25/modernizing-clinical-trials-the-role-of-etmf-ctms-and-edc-integration)
- cloudbyz.com (https://cloudbyz.com/resources/etmf/the-importance-of-integration-between-etmf-edc-and-ctms-systems)
- Evolving EDC Systems to Support Decentralized and Hybrid Clinical Trials
- informaconnect.com (https://informaconnect.com/clinical-trial-data-collection-technologies-industry-voices)
- milo-healthcare.com (https://milo-healthcare.com/en/significance-services-of-edc-systems-in-clinical-trials)
- mahalo.health (https://mahalo.health/insights/how-to-use-edc-for-clinical-trials)
- medidata.com (https://medidata.com/en/life-science-resources/medidata-blog/edc-system-innovation-and-future)
- Key Takeaways on Essential eTMF Features and Solutions
- Top eTMF Features – Flex Databases (https://flexdatabases.com/blog/top-etmf-features)
- milo-healthcare.com (https://milo-healthcare.com/en/understanding-etmf-systems-for-clinical-trials)
- blog.montrium.com (https://blog.montrium.com/blog/what-is-etmf-software-and-does-my-organization-need-to-implement-one)
- blog.cloudbyz.com (https://blog.cloudbyz.com/resources/the-essential-features-of-an-electronic-trial-master-file-etmf-for-clinical-trials)
- trialinteractive.com (https://trialinteractive.com/thought-leadership/etmf-features/527)
- Looking Ahead: The Future of eTMF Solutions in Clinical Research
- databridgemarketresearch.com (https://databridgemarketresearch.com/reports/global-electronic-trial-master-file-etmf-systems-market?srsltid=AfmBOopMQES2U2jzENsp_PYqTKVhO-yQs1O7uMM77zn9JjD0L9Q5Y9Cy)
- marketreportanalytics.com (https://marketreportanalytics.com/reports/electronic-trial-master-file-etmf-software-52319)
- dataintelo.com (https://dataintelo.com/report/etmf-software-market)
- Electronic Trial Master File Systems Market Size Report 2030 (https://grandviewresearch.com/industry-analysis/electronic-trial-master-file-etmf-systems-market-report)
- Electronic Trial Master File (eTMF) Market Size & Share,2032 (https://coherentmarketinsights.com/market-insight/electronic-trial-master-file-etmf-market-2092)

Leave a Reply