Introduction
In the rapidly evolving landscape of clinical research, the urgency for swift ethical approvals and innovative study designs has reached unprecedented levels. As organizations endeavor to expedite the introduction of groundbreaking therapies to the market, bioaccess® emerges as a transformative force. By leveraging its extensive regulatory knowledge across diverse regions, it streamlines the approval process effectively. With the capability to facilitate clinical trials in as little as four to six weeks, bioaccess® not only enhances research efficiency but also broadens access for diverse patient populations, ensuring that clinical studies yield relevant and impactful data.
This article explores the multifaceted advantages of investigator-initiated studies, highlighting how they:
- Reduce costs
- Foster collaboration
- Advance medical technologies
- Improve patient outcomes
All while navigating the complexities of regulatory frameworks. As the demand for innovative research solutions escalates, comprehending the pivotal role of organizations like bioaccess® is essential for propelling the future of medical science.
bioaccess®: Accelerate Clinical Research with Fast Ethical Approvals
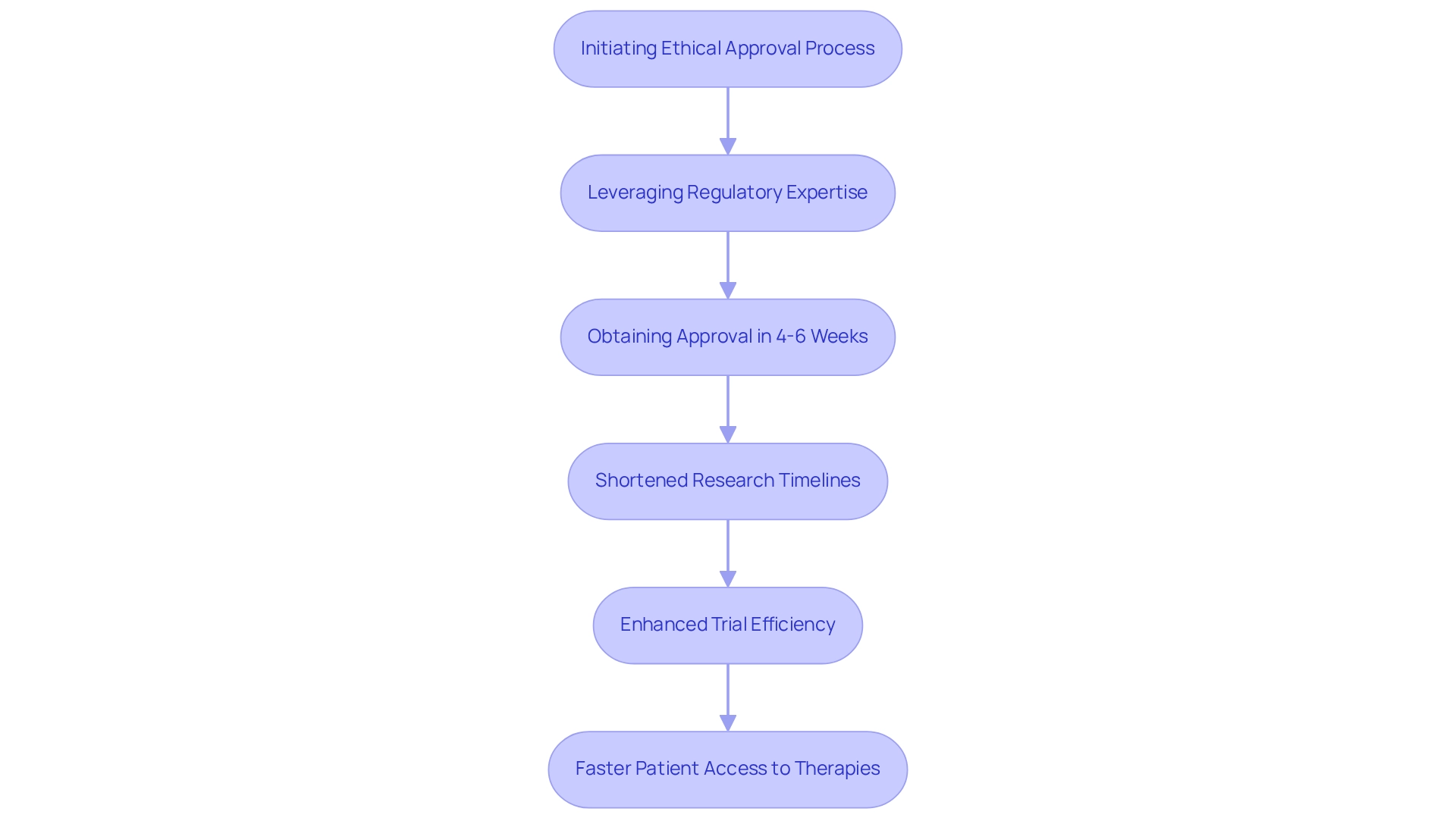
bioaccess® leverages its extensive expertise in across Latin America, the Balkans, and Australia to secure in an impressive 4-6 weeks. This expedited process is revolutionary for MedTech and Biopharma innovators, allowing them to engage in without the prolonged delays that are characteristic of conventional markets.
By streamlining the approval process, bioaccess® not only shortens but also significantly enhances , facilitating to groundbreaking therapies. In 2025, the remains a pivotal factor, as many organizations endeavor to shorten these timelines to boost competitiveness.
Recent statistics reveal that organizations that achieve , highlighting the critical nature of swift . Furthermore, the impact of on trial timelines is paramount; and, ultimately, quicker market entry for innovative solutions.
Recent case analyses, such as the scrutiny faced by a Chinese laboratory regarding data integrity, underscore the importance of a reliable approval process in ensuring patient safety and regulatory compliance. In this context, the ability to obtain rapid is not just advantageous but essential for the success of .
Access Diverse Patient Populations for Comprehensive Data Collection
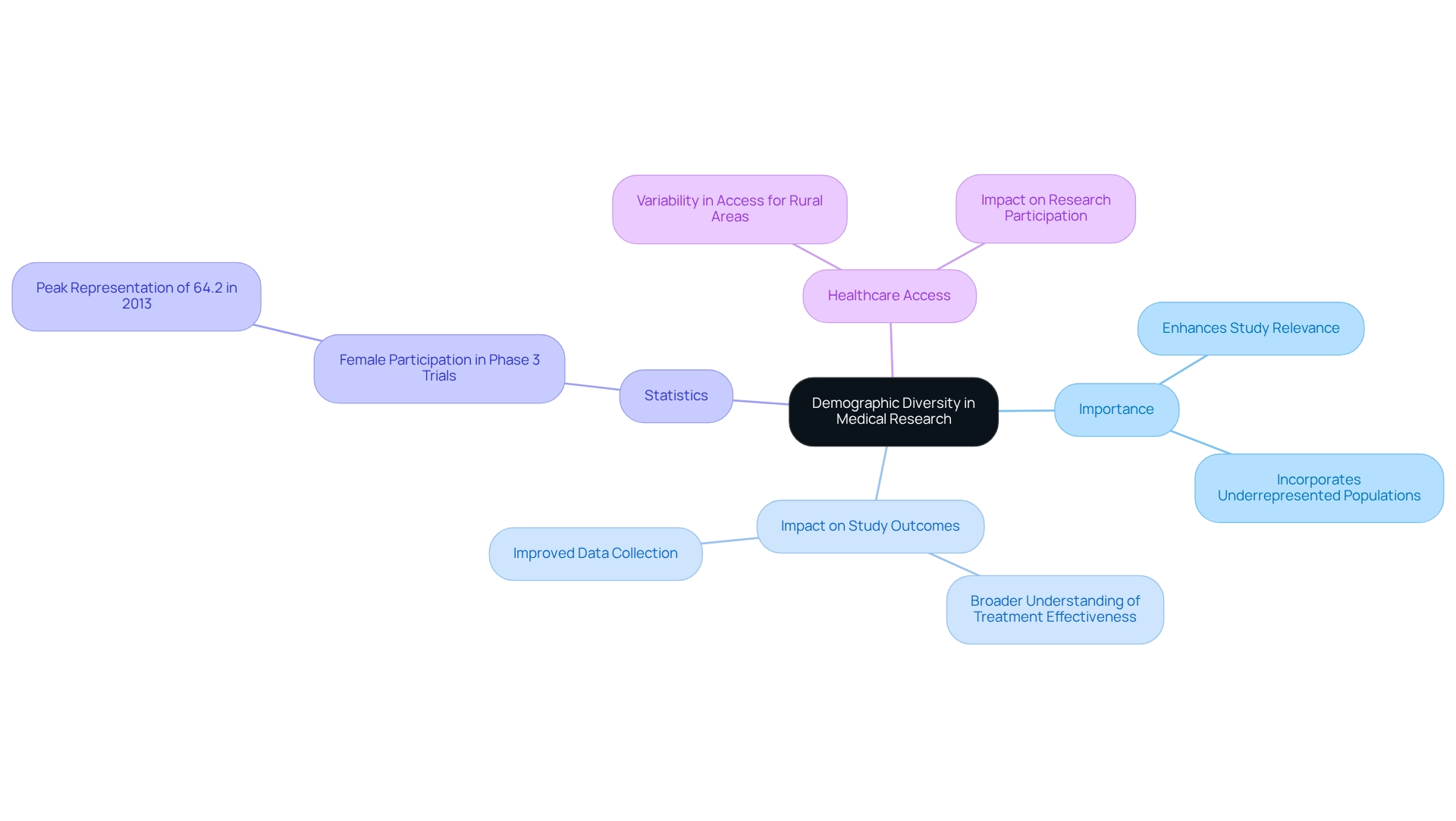
(IIS) conducted through bioaccess® leverage access to diverse patient groups across Latin America and the Balkans, significantly enhancing the quality of medical research. This facilitates that accurately reflects various populations, leading to results that are both relevant and applicable. By incorporating underrepresented populations, researchers gain crucial insights into how different demographics respond to treatments, which is essential for developing .
The importance of in research studies cannot be overstated. Research indicates that , as they provide a broader understanding of treatment effectiveness among diverse demographics. For instance, an analysis of female participation in Phase 3 studies revealed a peak representation of 64.2% in 2013, underscoring the necessity for .
Experts highlight that , especially for individuals in rural or remote areas, emphasizing the need to include diverse demographics in . By prioritizing , bioaccess®, in collaboration with Caribbean Health Group, not only but also contributes to the advancement of medical devices and biopharmaceuticals through that can better serve all segments of the population.
Reduce Research Costs Through Investigator-Initiated Studies
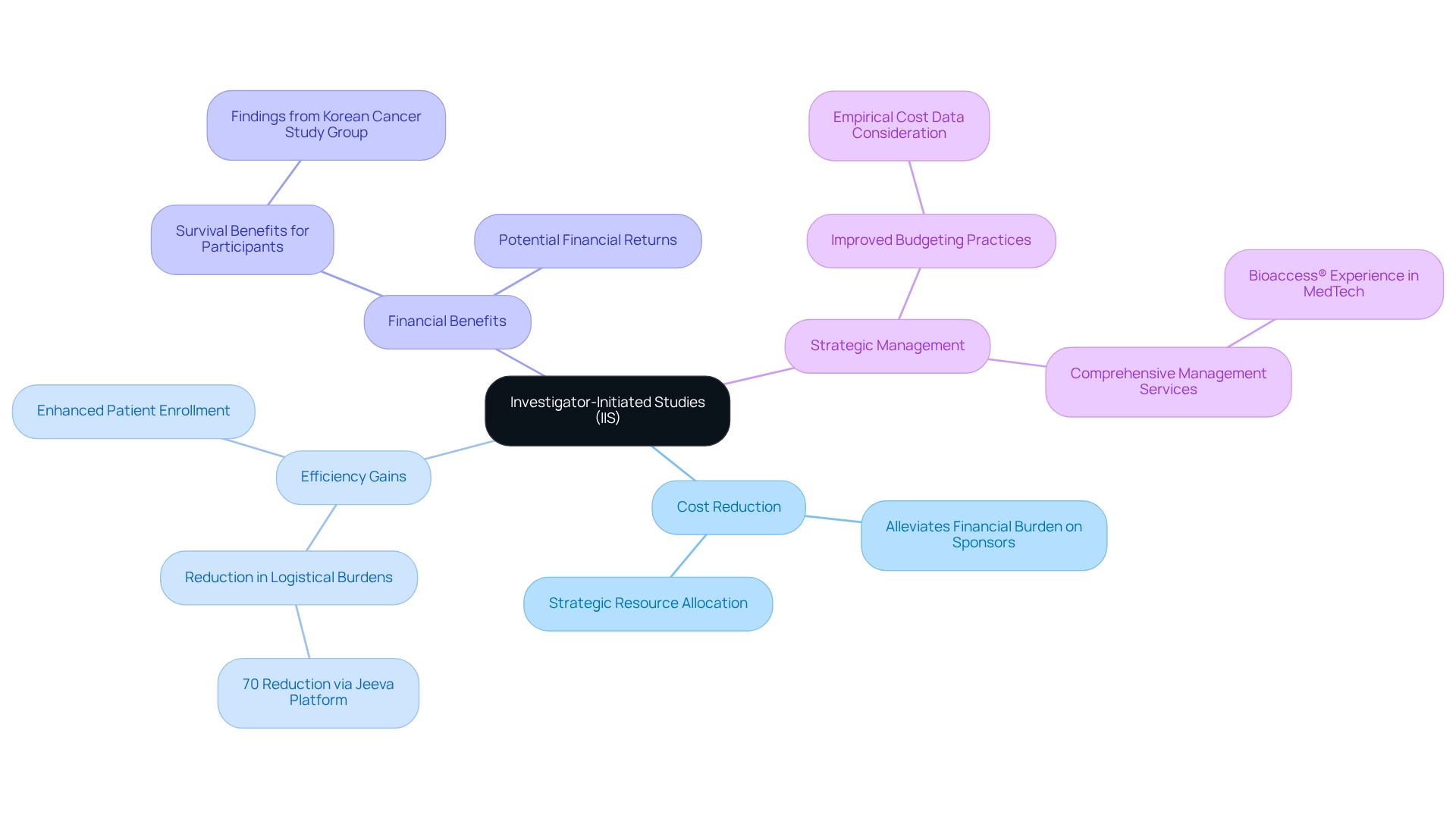
(IIS) offer a compelling alternative to conventional industry-sponsored trials, often providing a more . By empowering researchers to design and manage their own projects, on sponsors. This innovative model not only reduces initial costs but also allows researchers to allocate resources more strategically, ensuring that funding is directed toward the project’s most critical components. As a result, organizations can conduct without the excessive expenses typically associated with clinical trials.
The are underscored by the Jeeva eClinical Trial platform, which has demonstrated a by over 70%. This remarkable efficiency translates into substantial cost savings, enabling more research to be conducted within existing budget constraints. Moreover, a recent analysis has illuminated the shortcomings of traditional funding models, highlighting the urgent need for enhanced budgeting practices grounded in empirical cost data.
Recent studies have unequivocally shown that the financial benefits of are significant. Results indicate that these trials not only yield survival benefits for participants but also present potential . For instance, the Korean Cancer Study Group noted, “Our findings, derived from the analysis of published literature, indicate that IITs conducted by KCSG resulted in and, in some studies, might have offered financial gains by supplying investigational drugs.” This finding further emphasizes the critical role of IIS in advancing medical research through .
By leveraging the unique capabilities of bioaccess®, which boasts over 20 years of experience in Medtech and offers , organizations can execute cost-effective assessments in the MedTech and Biopharma sectors. This strategic approach not only fosters innovation but also effectively manages research expenditures, positioning Barranquilla as a .
Enhance Flexibility in Study Design and Protocol Adaptation
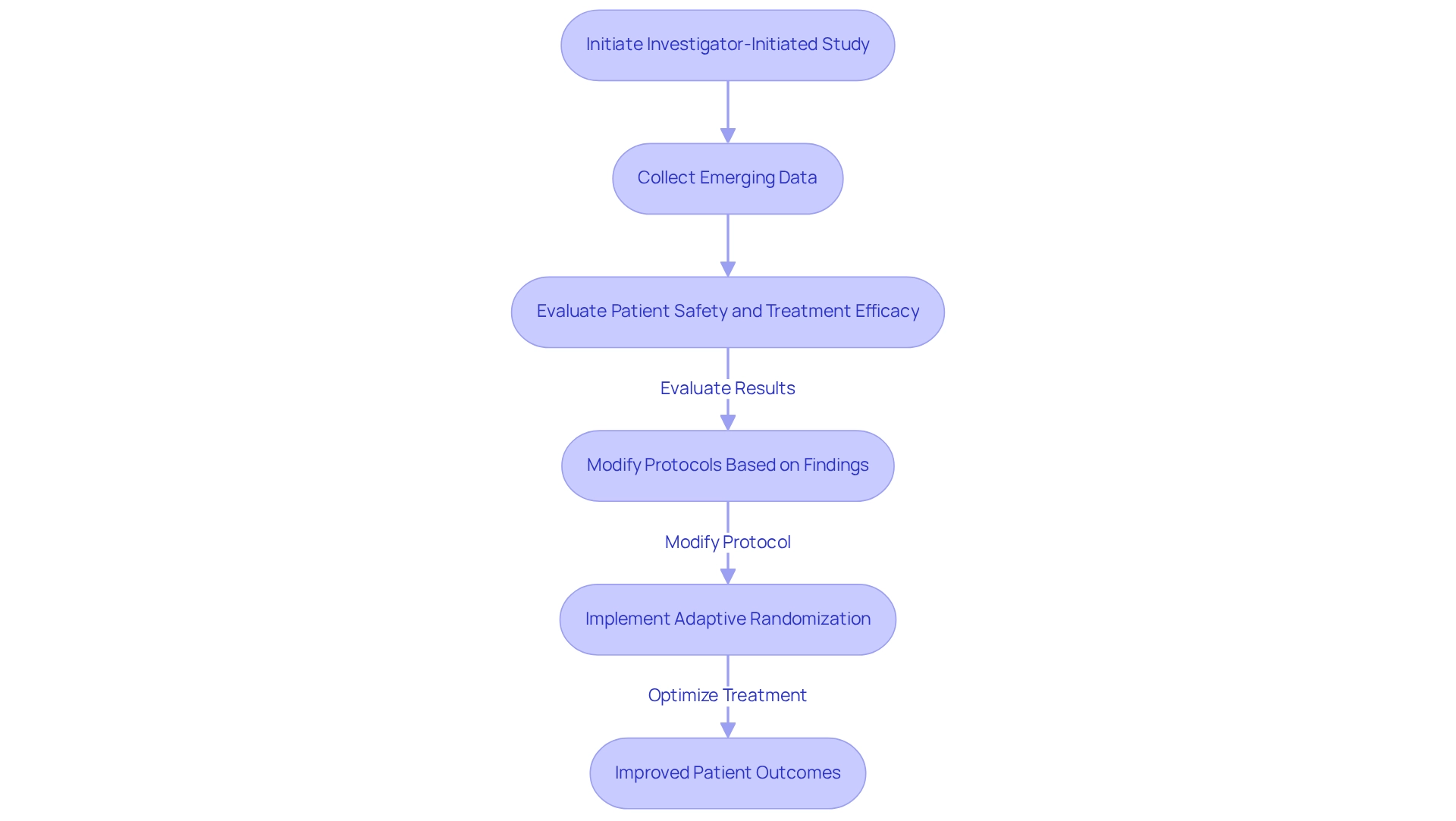
empower researchers to modify designs and protocols in response to emerging data, providing a crucial advantage in . For instance, the EWOC study, which enrolled 19 patients, illustrated the necessity of meticulous design to accurately determine the maximum tolerated dose (MTD) of 750 mg. This adaptability not only enhances patient safety but also optimizes by permitting .
A compelling illustration of this flexibility is found in a prospective, randomized study that assessed three troxacitabine-based regimens for patients aged 50 and older with untreated, adverse karyotype acute myeloid leukaemia. By employing a (RAR) design, the study updated randomization probabilities after each patient, resulting in the elimination of less effective treatment arms. This methodology led to varying success rates among the treatment arms, underscoring RAR’s potential to maximize patient benefit, which is further supported by that emphasize the , particularly in their capacity to respond to real-time data. As medical investigation advances in 2025, the emphasis on will be paramount, with data indicating that adaptable designs can yield improved health outcomes. By fostering an environment conducive to change, bioaccess® ensures that research remains relevant and aligned with the latest scientific advancements.
Explore Innovative Research Questions with Investigator-Initiated Studies
are essential for empowering researchers to delve into innovative inquiries that often diverge from the focus of traditional industry-sponsored projects. This autonomy not only nurtures creativity but also enables the exploration of new hypotheses, which can lead to . Recent trends indicate a growing interest in addressing through these investigations, as evidenced by a notable increase in the percentage of studies examining unique therapeutic approaches.
As we look towards 2025, the landscape of medical inquiry is witnessing a surge in innovative questions, signaling a shift towards more patient-centered and . Statistics reveal that a significant portion of investigator-initiated research is now focused on , underscoring the importance of creativity in . Industry experts highlight that the ability to conduct research internally enhances data management and quality, ultimately benefiting .
Case studies illustrate the challenges faced in estimating , emphasizing the necessity for comprehensive data collection and analytical methods. These insights reinforce the critical role of in generating reliable data that can inform healthcare practices and recommendations. By championing such initiatives, bioaccess® not only contributes to the advancement of medical science but also encourages the development of new therapeutic strategies that address pressing healthcare needs, paving the way for future innovations in the MedTech and Biopharma sectors.
Foster Collaboration Between Researchers and Sponsors
play a pivotal role in fostering cooperation between scientists and sponsors, cultivating a synergistic environment that significantly enhances the quality of . By facilitating open communication and aligning shared objectives, bioaccess™ ensures that all stakeholders remain synchronized throughout the study process. This not only streamlines the execution of but also fortifies the integrity of the research, leading to .
Statistics reveal that effective collaboration can result in a , highlighting the efficiency gained through these partnerships. A notable example is the , unveiled during a gathering on March 29, 2019, which aims to position Barranquilla as a leading hub for research studies in Latin America, supported by Colombia’s Minister of Health. This initiative exemplifies how strategic partnerships can enhance , achieving of 95%.
Furthermore, Dushyanth Surakanti, Founder & CEO of Sparta Biomedical, shared his positive experience with bioaccess® during its initial human study in Colombia, underscoring the practical benefits of such collaborations. The introduction of blockchain technology has also revolutionized among stakeholders, improving data integrity and study efficiency. Expert insights indicate that these synergistic environments not only benefit immediate stakeholders but also elevate the broader field of medical research by facilitating , ensuring that innovative therapies reach patients more swiftly and effectively. As industry leader Esserman aptly noted, “The whole point is that it does not hurt anyone, and it helps everyone,” underscoring the essential role of collaboration in advancing medical breakthroughs and enhancing patient outcomes.
Leverage Regulatory Advantages in Latin America for Faster Approvals
Latin America presents that can significantly accelerate . By effectively leveraging these local frameworks, bioaccess® streamlines approvals and simplifies the initiation of studies, which is especially advantageous for . Swift access to can profoundly impact project timelines, enabling innovations to reach the market more rapidly.
For instance, hospitals in Colombia must navigate a stringent , which guarantees that clinical studies adhere to . This certification not only bolsters the credibility of findings but also fosters a reliable environment for conducting investigations.
Moreover, the region’s rich linguistic diversity, encompassing approximately 65 indigenous languages and nearly 300 dialects, underscores the necessity of addressing . By effectively communicating and appreciating these differences, researchers can elevate study quality and enhance healthcare delivery.
Notably, bioaccess® has successfully partnered with companies like to facilitate the launch of , such as Celbrea®, in Colombia, demonstrating its proficiency in navigating the complexities of local regulations. By empowering researchers to realize their innovations more efficiently, bioaccess® capitalizes on the regulatory benefits that Latin America offers in 2025.
Improve Patient Outcomes Through Targeted Research Initiatives
are pivotal in implementing that address specific health challenges faced by diverse patient populations. By focusing on relevant issues, bioaccess® actively contributes to the development of , which can lead to significant improvements in . This patient-centered approach not only enhances the effectiveness of treatments but also fosters greater trust and engagement among participants. As a result, often yield , ultimately enhancing the overall landscape of clinical investigation.
For instance, the National Cancer Institute’s exemplifies how . However, the scope of such programs highlights an urgent need for in cancer prevention. With approximately 18 million cancer survivors in the United States, the importance of focused in improving cannot be overstated.
Advance Medical Technologies with Investigator-Initiated Research
Studies that are investigator initiated are pivotal in propelling forward by enabling researchers to test and validate innovative concepts in real-world environments. Through bioaccess®, investigators can conduct studies that rigorously assess the efficacy and safety of and therapies. This approach not only enriches the scientific community but also significantly improves , ultimately leading to enhanced health outcomes.
As we approach 2025, the landscape of medical studies is increasingly influenced by , offering essential insights into how new technologies operate outside regulated settings. This method is crucial for understanding the practical applications of medical innovations, as evidenced by the growing reliance on . Moreover, expert opinions underscore the necessity of . Innovators frequently highlight the importance of testing new devices in , allowing for a more comprehensive evaluation of their impact on patient outcomes.
A recent case analysis revealed that despite budget concerns, 48% of clinical sites showed a readiness to adopt new technologies, provided that sponsors support the necessary infrastructure. This collaborative approach not only facilitates the integration of innovative solutions but also ensures that advancements in healthcare are both effective and accessible, highlighting the growing importance of as . These research efforts not only aid in the progress of but also act as a basis for future innovations that can revolutionize patient care.
Enhance Researcher Skills and Knowledge Through Active Participation
Active involvement in significantly enhances researchers’ abilities and understanding, providing in . By participating in the design, execution, and evaluation of , researchers develop vital skills necessary for professional advancement.
Statistics indicate that a tends to reduce the probability of Type II errors, thereby enhancing the overall power of the investigation. This underscores the importance of robust , which bioaccess® actively supports through and resources.
Furthermore, case analyses illustrate that employing strategies such as pilot projects and can effectively address challenges in , leading to more reliable outcomes. This relationship is critical, as it ensures that genuine effects are identified rather than dismissed as random noise.
By fostering an environment where researchers can actively engage in , bioaccess® not only enhances their skills but also contributes to the and improved patient care.
Conclusion
Investigator-initiated studies, facilitated by bioaccess®, signify a pivotal advancement in the clinical research landscape, effectively addressing essential needs for efficiency, inclusivity, and innovation. By streamlining ethical approvals and granting access to diverse patient populations, bioaccess® elevates the quality and relevance of clinical trials, ultimately culminating in enhanced patient outcomes. The financial advantages of these studies empower researchers to allocate resources strategically, thereby minimizing costs while upholding rigorous standards of research integrity.
The inherent flexibility of investigator-initiated studies allows for real-time adaptations in study designs, nurturing a responsive research environment capable of addressing emerging medical inquiries more effectively. This adaptability, coupled with the collaborative framework that bioaccess® fosters, fortifies partnerships between researchers and sponsors, ensuring synchronized efforts toward shared objectives. Furthermore, leveraging regulatory advantages in Latin America expedites the approval process, enabling innovative therapies to reach the market with greater speed.
As the demand for groundbreaking medical technologies escalates, investigator-initiated studies will assume an increasingly critical role in shaping the future of clinical research. By empowering researchers to investigate novel hypotheses and enhance healthcare delivery, bioaccess® not only propels the advancement of medical science but also champions the development of targeted interventions tailored to the unique needs of diverse populations. The ongoing commitment to fostering collaboration, enhancing skills, and driving innovation will be indispensable in navigating the complexities of modern clinical trials and ultimately improving patient care on a global scale.
Frequently Asked Questions
What is bioaccess® and what expertise does it leverage?
bioaccess® is an organization that utilizes its extensive expertise in regulatory frameworks across Latin America, the Balkans, and Australia to secure ethical approvals for medical research.
How quickly can bioaccess® secure ethical approvals?
bioaccess® can secure ethical approvals in an impressive 4-6 weeks, which is significantly faster than traditional markets.
Why is the expedited approval process important for MedTech and Biopharma innovators?
The expedited process allows innovators to engage in investigator initiated studies without the prolonged delays typical of conventional markets, shortening research timelines and enhancing trial efficiency.
What impact does rapid ethical approval have on patient access to therapies?
Rapid ethical approvals can lead to quicker patient recruitment and faster market entry for innovative solutions, ultimately facilitating quicker access to groundbreaking therapies.
How much can enrollment speeds increase with rapid ethical approvals?
Organizations that achieve rapid ethical approvals can experience enrollment speeds increase by as much as 50%.
Why is demographic diversity important in investigator initiated studies (IIS)?
Demographic diversity enhances the quality of medical research by allowing comprehensive data collection that reflects various populations, leading to relevant and applicable results.
How does bioaccess® contribute to demographic diversity in research?
bioaccess® leverages access to diverse patient groups across Latin America and the Balkans, ensuring that underrepresented populations are included in clinical research.
What financial advantages do investigator initiated studies (IIS) offer?
IIS provide a cost-effective solution by allowing researchers to design and manage their own projects, which reduces initial costs and enables better allocation of resources.
What has the Jeeva eClinical Trial platform demonstrated regarding logistical burdens?
The Jeeva eClinical Trial platform has shown a reduction in logistical burdens for both patients and research teams by over 70%, resulting in substantial cost savings.
How does bioaccess® support organizations in conducting research?
With over 20 years of experience in MedTech, bioaccess® offers comprehensive management services for studies, enabling organizations to execute cost-effective assessments in the MedTech and Biopharma sectors.
List of Sources
- bioaccess®: Accelerate Clinical Research with Fast Ethical Approvals
- Factors influencing the time to ethics and governance approvals for clinical trials: a retrospective cross-sectional survey – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10693024)
- 5 Medical Device Regulatory Approval Statistics You Need to Know – Arrotek | Medical Device Innovation (https://arrotek.com/5-medical-device-regulatory-approval-statistics-you-need-to-know)
- medtechdive.com (https://medtechdive.com/news/fda-turnaround-510k-record-approval-waits/687476)
- Access Diverse Patient Populations for Comprehensive Data Collection
- clinicaltrialsarena.com (https://clinicaltrialsarena.com/news/fda-report-highlights-need-to-improve-diversity-in-clinical-trials)
- A statistical overview of diversity in clinical trials (https://antidote.me/blog/statistical-overview-of-diversity-in-clinical-trials)
- Key Trends in Demographic Diversity in Clinical Trials – Improving Representation in Clinical Trials and Research – NCBI Bookshelf (https://ncbi.nlm.nih.gov/books/NBK584392)
- Reduce Research Costs Through Investigator-Initiated Studies
- jeevatrials.com (https://jeevatrials.com/news/from-protocol-to-publication-reducing-costs-and-maximizing-efficiency-for-investigator-initiated-trials)
- e-crt.org (https://e-crt.org/journal/view.php?number=3638)
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S0895435624002920)
- Enhance Flexibility in Study Design and Protocol Adaptation
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC7677786)
- bmj.com (https://bmj.com/content/360/bmj.k698)
- Explore Innovative Research Questions with Investigator-Initiated Studies
- 2025 Clinical Data Trend Report | Veeva (https://veeva.com/2025-clinical-data-trend-report)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC7532587)
- academic.oup.com (https://academic.oup.com/biostatistics/article/20/2/273/4817524)
- Foster Collaboration Between Researchers and Sponsors
- Clinical Trials Statistics and Facts (2026) (https://media.market.us/clinical-trials-statistics)
- BENEFITS OF COLLABORATING – Extending the Spectrum of Precompetitive Collaboration in Oncology Research – NCBI Bookshelf (https://ncbi.nlm.nih.gov/books/NBK210038)
- Leverage Regulatory Advantages in Latin America for Faster Approvals
- languageconnections.com (https://languageconnections.com/clinical-trials-in-latin-america)
- bioaccessla.com (https://bioaccessla.com/es/blog/mastering-clinical-trial-regulation-benefits-in-latin-america-a-comprehensive-guide)
- Current barriers for developing clinical research in Latin America: A cross-sectional survey of medical oncologists (https://oatext.com/Current-barriers-for-developing-clinical-research-in-Latin-America-A-cross-sectional-survey-of-medical-oncologists.php)
- Improve Patient Outcomes Through Targeted Research Initiatives
- cancerprogressreport.aacr.org (https://cancerprogressreport.aacr.org/progress/cpr24-contents/cpr24-advancing-cancer-research-and-patient-care-through-evidence-based-policies)
- Predictive analytics and tailored interventions improve clinical outcomes in older adults: a randomized controlled trial – npj Digital Medicine (https://nature.com/articles/s41746-021-00463-y)
- Advance Medical Technologies with Investigator-Initiated Research
- Medical Technology and Innovation Statistics and Facts (2026) (https://media.market.us/medical-technology-and-innovation-statistics)
- numberanalytics.com (https://numberanalytics.com/blog/healthcare-analytics-pharma-innovation-stats)
- 7 Key Findings from the 2022 Clinical Trial Operations Technology Survey (https://florencehc.com/blog-post/7-key-findings-from-the-2022-clinical-trial-operations-technology-survey)
- Enhance Researcher Skills and Knowledge Through Active Participation
- numberanalytics.com (https://numberanalytics.com/blog/5-surprising-stats-effect-size-clinical-trials)

Leave a Reply