Introduction
Engaging participants in clinical studies is a pivotal aspect of advancing medical research. However, many struggle to understand the intricacies of this process. With only a fraction of individuals fully grasping the phases and significance of their involvement, the need for effective communication and tailored strategies becomes evident.
This article delves into best practices for enhancing participant engagement, including:
- Prioritizing informed consent
- Incorporating feedback
Ultimately, it raises the question: how can researchers transform the clinical trial experience to empower and retain participants effectively?
Understand the Fundamentals of Clinical Trials
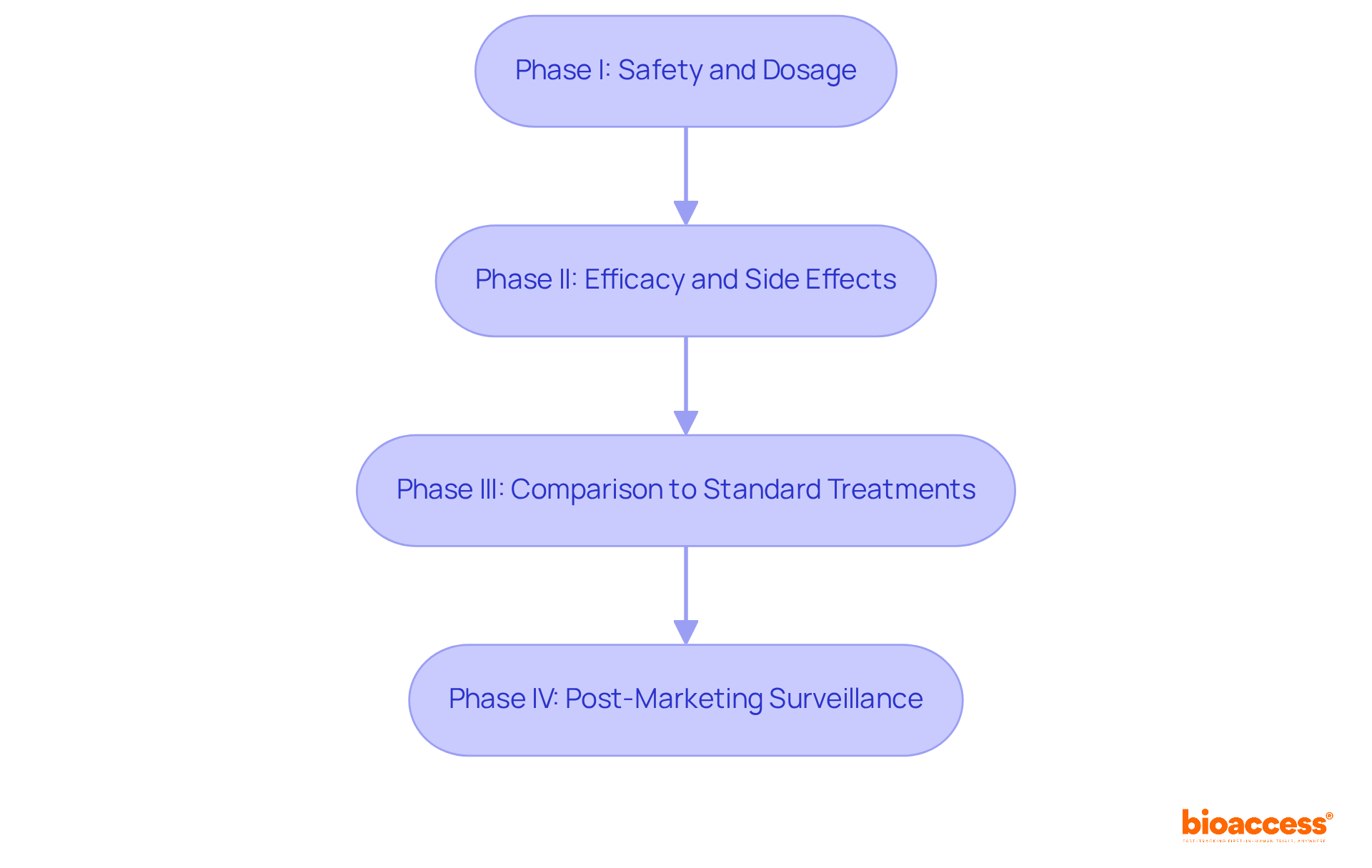
represent meticulously organized research studies designed to evaluate the . These studies progress through four distinct phases:
- .
- .
- Phase III compares the new intervention to standard treatments.
- Phase IV involves .
Understanding these phases is crucial for all participants, as it clarifies the purpose and , fostering a sense of contribution to medical advancements. For instance, individuals participating in Phase I studies are instrumental in determining safe dosage levels, which are vital for the success of subsequent phases.
Notably, studies indicate that only 47% of individuals fully comprehend the stages of , underscoring the need for . Implementing clear and accessible information regarding each stage and its significance can significantly enhance retention and satisfaction during the process. By equipping participants with essential information, can improve recruitment outcomes and ensure that studies are conducted effectively and ethically.
Prioritize Informed Consent and Transparency
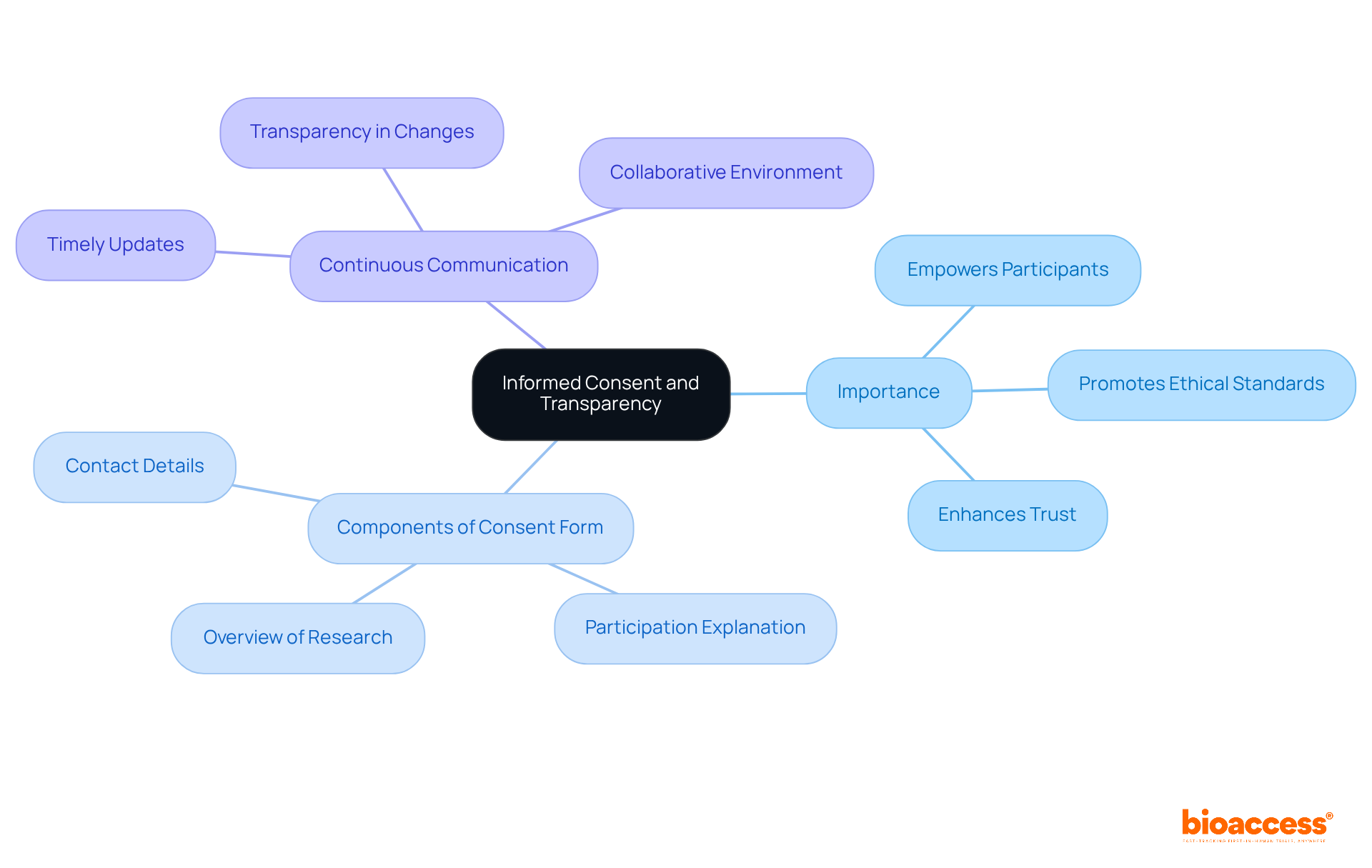
is paramount in ; it must be , thereby empowering participants to make informed decisions about their involvement. Researchers are tasked with providing , procedures, . A should include:
- A brief overview of the research
- A thorough explanation of what participation entails
- Contact details for any inquiries
Furthermore, maintaining is essential; individuals should receive on any changes to the research or new findings that may influence their decision to continue. This level of not only respects individuals’ autonomy but also cultivates a that significantly enhances participation and trust.
Implement Tailored Communication Strategies for Diverse Populations
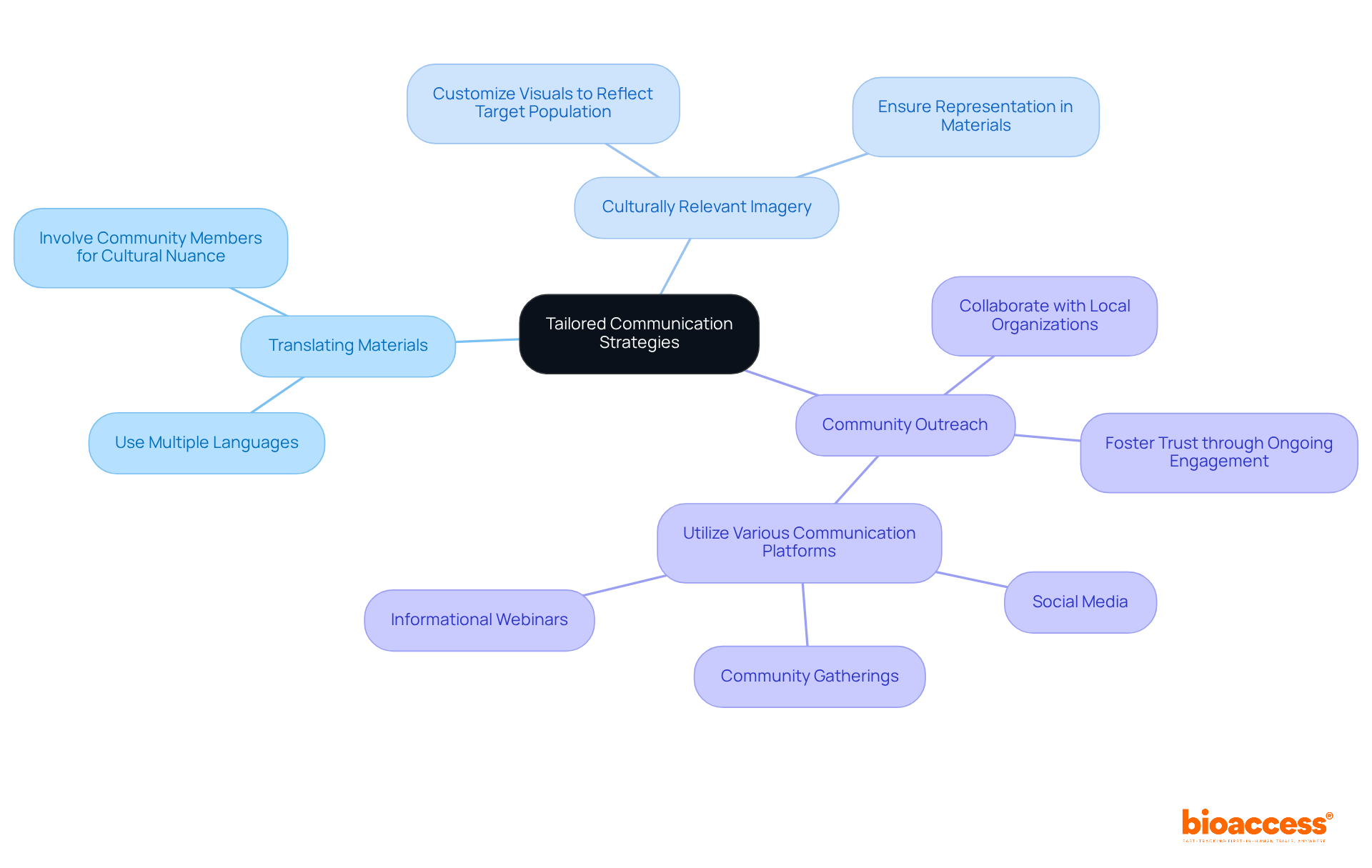
To effectively engage , researchers must develop that recognize . This involves:
- Translating materials into multiple languages
- Employing
- Fostering
For instance, collaborating with local organizations can enhance access to underrepresented groups, enabling researchers to deliver tailored information that addresses specific needs and concerns. Furthermore, utilizing various communication platforms—such as social media, community gatherings, and informational webinars—can significantly bolster outreach efforts, ensuring that potential attendees and are well-informed and empowered to make decisions regarding their participation.
Incorporate Participant Feedback to Enhance Engagement
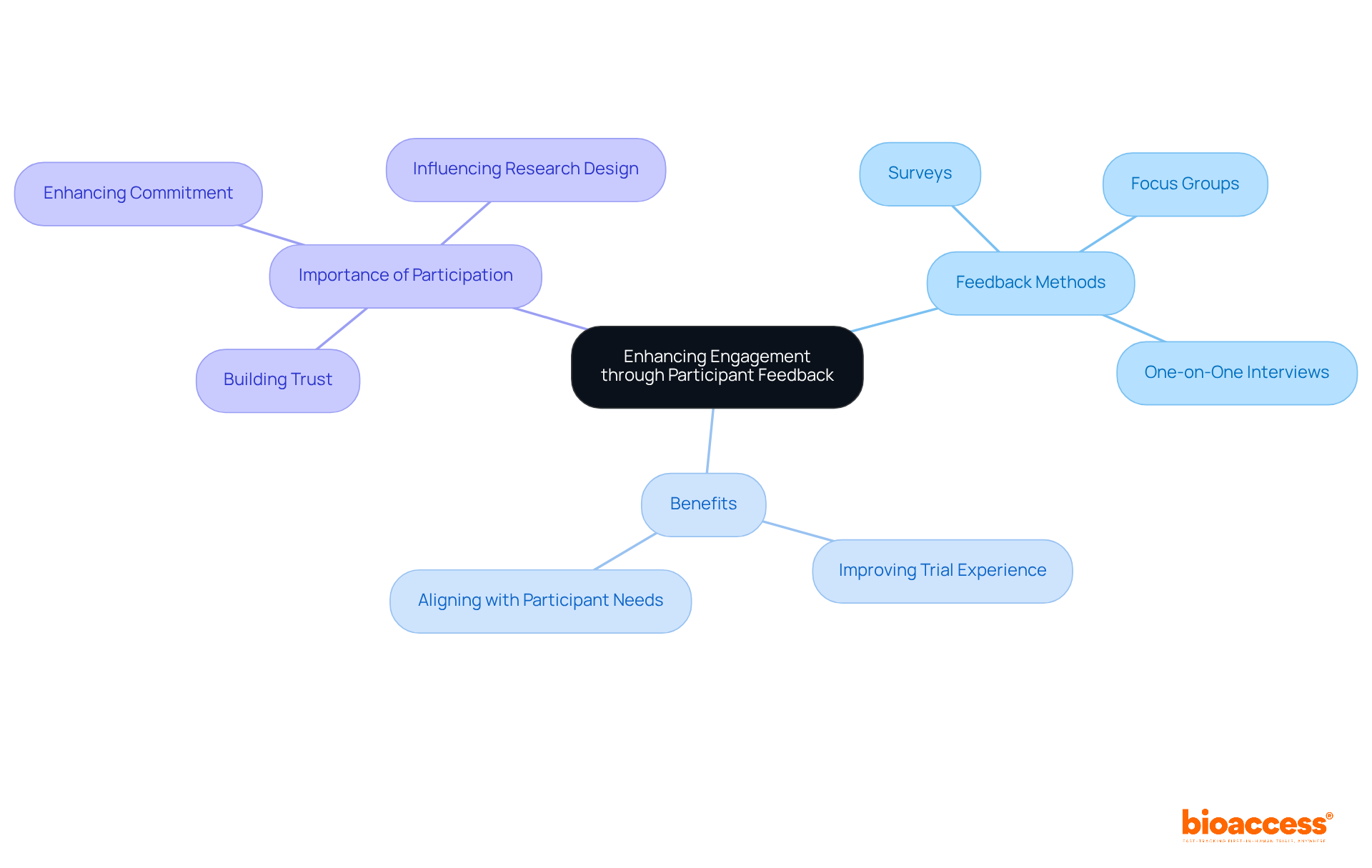
Actively seeking and addressing feedback from is essential for in . Researchers must implement various mechanisms, such as:
- Surveys
- Focus groups
- One-on-one interviews
to facilitate open communication regarding the experiences of . Collecting feedback on research procedures can unveil opportunities for improvement, such as or . The Transformation Initiative (CTTI) underscores that is crucial for the overall success of clinical studies. Furthermore, transparently communicating how has influenced research design strengthens their sense of value and commitment to the research process. Utilizing can further enhance the feedback process, making it more accessible for individuals involved. This iterative approach not only improves the trial experience but also significantly contributes to the overall success of the study by aligning it more closely with the needs and preferences of those involved. As Alicia Staley, senior director of patient engagement, asserts, “If our patient advocates haven’t looked at something rolling out the door, an explanation is expected as to why not.” This statement highlights the critical importance of incorporating the insights of into the research process.
Conclusion
Engaging clinical study participants is pivotal for the success of medical research, necessitating a comprehensive understanding of best practices that enhance their experience and involvement. By focusing on informed consent, transparent communication, tailored strategies for diverse populations, and actively incorporating participant feedback, researchers can create a more inclusive and effective research environment. This multifaceted approach not only respects the autonomy of participants but also fosters a collaborative atmosphere that can lead to more meaningful outcomes in clinical trials.
The article highlights the importance of clearly communicating the phases of clinical trials, ensuring participants are well-informed about what to expect. It emphasizes that informed consent should be thorough and accessible, empowering individuals to make educated decisions about their participation. Additionally, the necessity of culturally sensitive communication strategies is underscored, as these practices can significantly improve engagement with underrepresented populations. Finally, the integration of participant feedback is presented as a vital tool for enhancing the trial experience and aligning research with the needs of those involved.
As the landscape of clinical research continues to evolve, it is crucial for stakeholders to prioritize these best practices. By doing so, they can not only improve participant retention and satisfaction but also contribute to the advancement of medical knowledge and innovation. Embracing these strategies will ultimately lead to more successful clinical trials and better health outcomes for the communities they serve.
Frequently Asked Questions
What are clinical trials?
Clinical trials are meticulously organized research studies designed to evaluate the safety and effectiveness of new medical interventions.
What are the four phases of clinical trials?
The four phases of clinical trials are: – Phase I: Focuses on safety and dosage. – Phase II: Assesses efficacy and side effects. – Phase III: Compares the new intervention to standard treatments. – Phase IV: Involves post-marketing surveillance.
Why is it important for participants to understand the phases of clinical trials?
Understanding the phases is crucial for participants as it clarifies the purpose and potential impact of their involvement, fostering a sense of contribution to medical advancements.
What role do participants play in Phase I clinical trials?
Participants in Phase I studies are instrumental in determining safe dosage levels, which are vital for the success of subsequent phases.
What percentage of individuals comprehend the stages of clinical studies?
Studies indicate that only 47% of individuals fully comprehend the stages of clinical studies.
How can communication strategies improve participants’ experience in clinical trials?
Implementing clear and accessible information regarding each stage and its significance can enhance retention and satisfaction, improve recruitment outcomes, and ensure studies are conducted effectively and ethically.
List of Sources
- Understand the Fundamentals of Clinical Trials
- 10 Trends and Statistics for Clinical Trials in 2023 (https://xtalks.com/10-trends-and-statistics-for-clinical-trials-in-2023-3377)
- Clinical Trials Statistics and Facts (2026) (https://media.market.us/clinical-trials-statistics)
- Number of clinical trials by year, location, disease, phase, age and sex of trial participants (https://who.int/observatories/global-observatory-on-health-research-and-development/monitoring/number-of-trial-registrations-by-year-location-disease-and-phase-of-development)
- antidote.me (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- Prioritize Informed Consent and Transparency
- pharmafocusamerica.com (https://pharmafocusamerica.com/articles/enhancing-clinical-trial-participant-engagement)
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S1551714416300854)
- agingresearch.org (https://agingresearch.org/blog/new-york-times-article-on-clinical-trials-features-quote-from-the-alliance-for-aging-research)
- The Importance of Clinical Trial Transparency and FDA Oversight (https://fda.gov/news-events/fda-voices/importance-clinical-trial-transparency-and-fda-oversight)
- lindushealth.com (https://lindushealth.com/blog/best-practices-for-clinical-trial-execution)
- Implement Tailored Communication Strategies for Diverse Populations
- sciencedirect.com (https://sciencedirect.com/science/article/abs/pii/S1551714422000660)
- bioaccessla.com (https://bioaccessla.com/blog/8-strategies-to-enhance-diversity-in-clinical-trials-for-underrepresented-populations)
- Diversity in Clinical Trials: A Scientific Imperative for 2025 – Clinical Trial Listing Database & Insights | CenterWatch (https://wcgclinical.com/insights/diversity-in-clinical-trials-a-scientific-imperative-for-2025)
- Clinical trial participation statistics: 3 trends to keep an eye on (https://antidote.me/blog/clinical-trial-participation-statistics-3-trends-to-keep-an-eye-on)
- clinicaltrialsarena.com (https://clinicaltrialsarena.com/news/diversity-commitment-persists-in-clinical-trials-amidst-policy-shifts)
- Incorporate Participant Feedback to Enhance Engagement
- pharmafocusamerica.com (https://pharmafocusamerica.com/articles/enhancing-clinical-trial-participant-engagement)
- Patient Group Engagement – CTTI (https://ctti-clinicaltrials.org/about/ctti-projects/patients-groups-clinical-trials)
- totaldiversity.com (https://totaldiversity.com/clinical-trial-engagement-guide-biotech-pharma)
- Diversity in Clinical Trials – UW Research (https://washington.edu/research/hsd/guidance/dct)
- clinicalresearchnewsonline.com (https://clinicalresearchnewsonline.com/news/2021/03/16/award-winning-participant-engagement-strategies)

Leave a Reply