Introduction
The landscape of medical technology is rapidly evolving, with Class II medical devices playing a pivotal role in enhancing patient care and treatment outcomes. These devices, which encompass essential tools such as infusion pumps, surgical staplers, and defibrillators, are subject to rigorous regulatory scrutiny to ensure their safety and efficacy.
As innovators strive to bring these critical solutions to market, the challenge lies in navigating complex clinical research processes efficiently. Stakeholders must consider how to leverage advancements in clinical trial management to expedite the development and approval of these life-saving devices.
bioaccess®: Accelerating Clinical Research for Class II Medical Devices
bioaccess® excels in for by expertly navigating the , the Balkans, and Australia. This comprehensive approach includes:
- Feasibility and selection of research sites
- Trial set-up
With ethical approvals obtained in an impressive 4-6 weeks and than in conventional markets, bioaccess® provides a eager to introduce their products to market swiftly. This agility is crucial for companies striving to meet the rising demand for , ensuring .
Industry leaders recognize that is vital for fostering innovation, and bioaccess® stands at the forefront of this transformation, facilitating that ultimately improve outcomes for individuals. Furthermore, bioaccess®’s partnership with Caribbean Health Group aims to establish Barranquilla as a key location for , supported by Colombia’s Minister of Health.
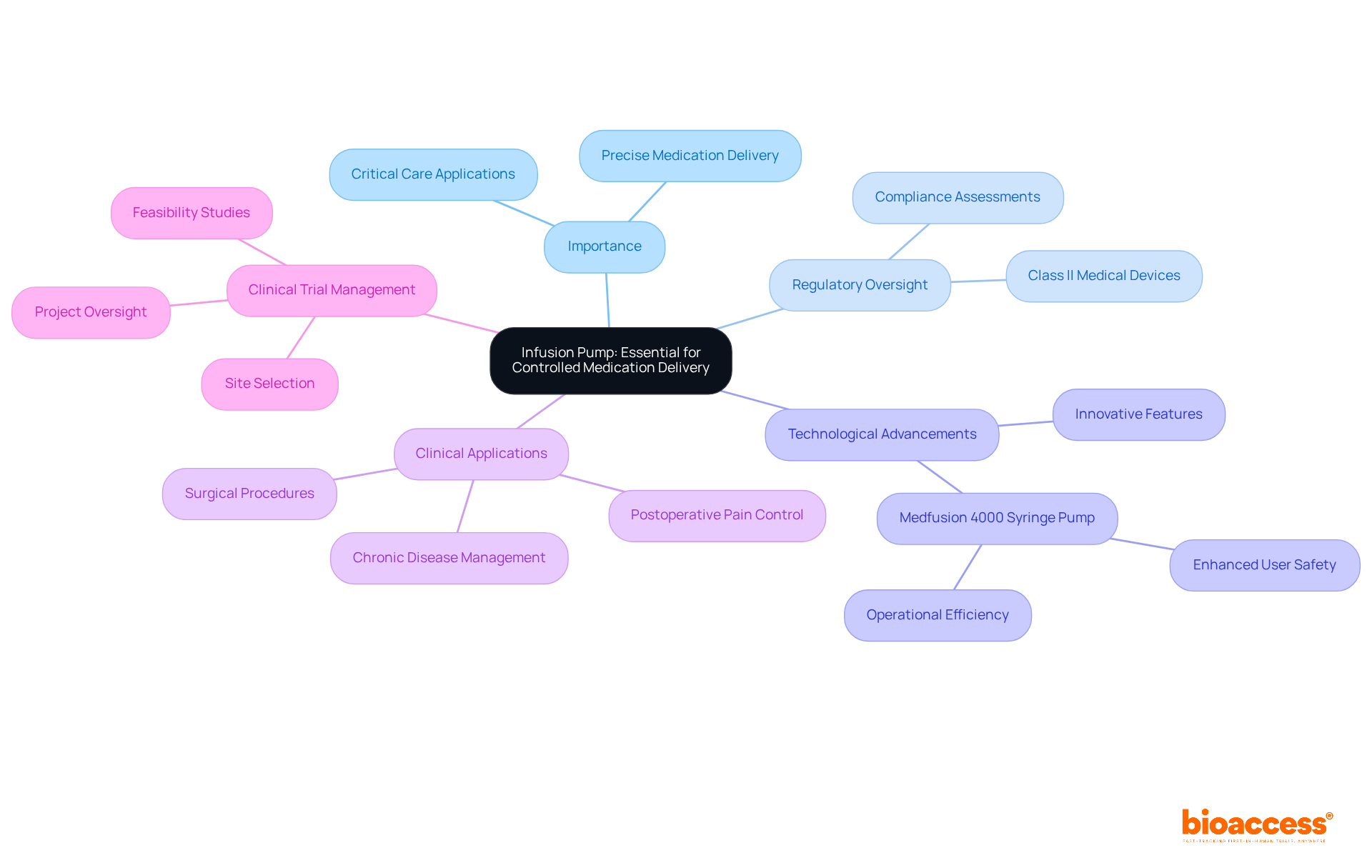
Infusion Pump: Essential for Controlled Medication Delivery
, meticulously designed to deliver medications, nutrients, and fluids to patients with precision. As examples of , they pose a moderate risk and necessitate extensive , including compliance assessments and setup processes. These pumps are pivotal in diverse medical scenarios, such as and chronic disease management, where is critical.
Recent studies highlight the , emphasizing usability to meet stringent regulatory standards. Notably, advancements in in 2025 have introduced . The Medfusion 4000 Syringe Pump exemplifies this progress, recognized for its precise medication delivery in critical care environments, including neonatal and pediatric intensive care units.
As the healthcare landscape evolves, the integration of innovative technologies continues to enhance the effectiveness of infusion pumps, ensuring they remain vital tools in patient care. Furthermore, research indicates a high success rate in postoperative pain control, underscoring the practical effectiveness of these devices.
Comprehensive , encompassing feasibility studies, site selection, and project oversight, are essential in navigating the regulatory landscape, ensuring that products like infusion pumps adhere to necessary safety and efficacy standards.
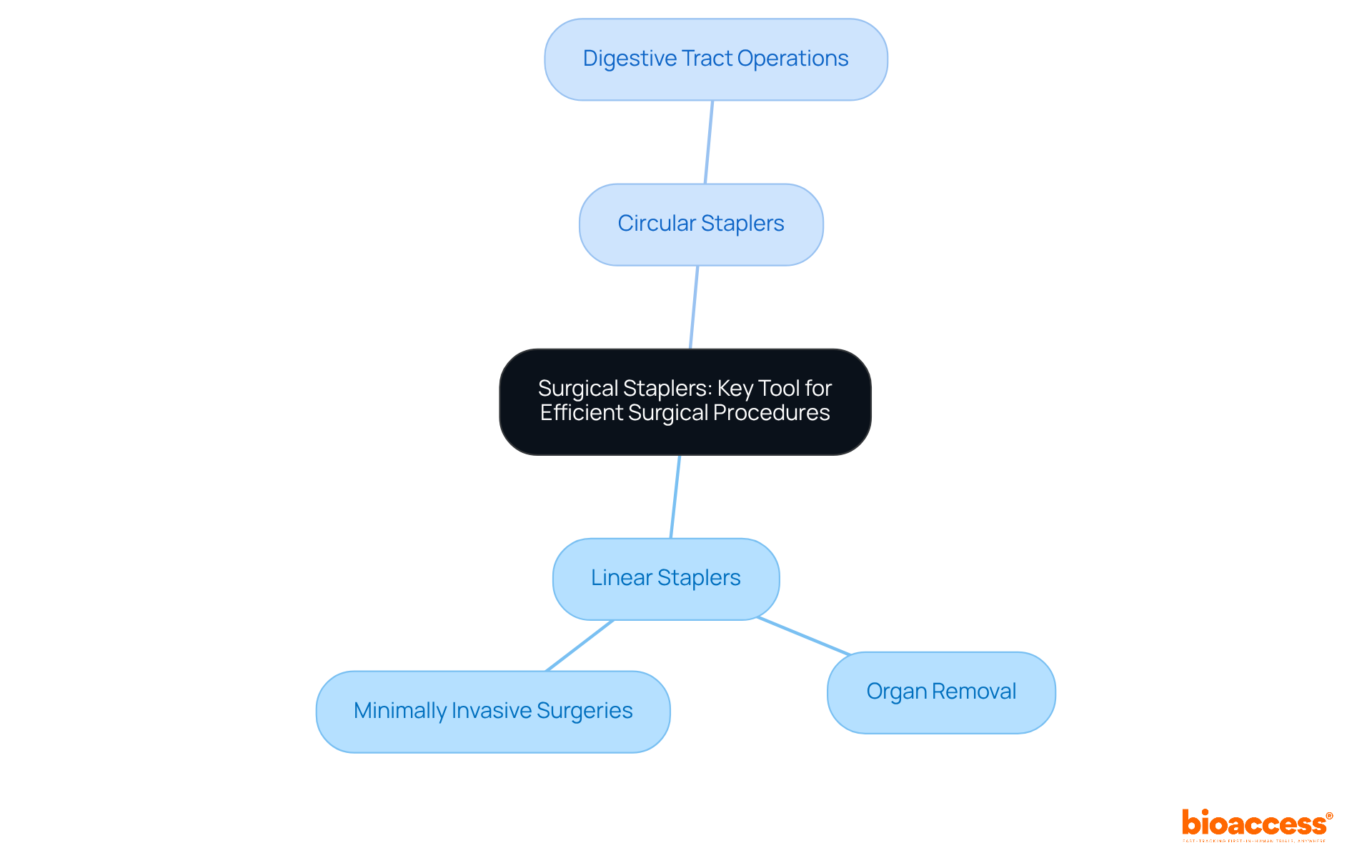
Surgical Stapler: Key Tool for Efficient Surgical Procedures
are indispensable tools in modern surgery, essential for efficiently closing wounds and connecting tissues. They are that can be either reusable or disposable, catering to diverse surgical applications. These instruments are primarily divided into two categories:
- Linear staplers, frequently employed for organ removal and minimally invasive surgeries.
- Circular staplers, typically utilized in operations involving the digestive tract.
Given their potential risks if not used correctly, are imperative.
At bioaccess®, we excel in accelerated medical device across Latin America, providing comprehensive that includes Early-Feasibility Studies, , and Post-Market Clinical Follow-Up Studies. This meticulous evaluation is vital for , a critical step for securing market approval. Recent innovations in in 2025 have significantly enhanced their functionality, resulting in improved precision and reduced surgical times. Surgeons have praised these advancements, noting that contemporary staplers not only streamline procedures but also contribute to enhanced outcomes for patients.
As the landscape of continues to evolve, the role of remains pivotal in augmenting and elevating patient care. Collaboration in clinical research is essential to navigate the challenges within the Medtech landscape, and bioaccess is committed to leading the way.
Catheter: Vital for Patient Monitoring and Treatment
Catheters, which are and fluids or to drain bodily fluids, are examples of due to their moderate risk profile. focus on their design, usability, and safety across various medical applications, including urinary drainage and intravenous therapy.
In this context, bioaccess® offers expert services that connect innovative with top-ranked research sites, facilitating accelerated trials for catheters. This approach not only ensures compliance with efficiently but also underscores the importance of .
With bioaccess®’s support, startups can confidently .
MRI Machine: Advanced Imaging for Accurate Diagnoses
MRI machines represent cutting-edge , delivering intricate visuals of the body’s internal structures. As , they require thorough to validate their imaging capabilities and ensure safety. These trials must evaluate the machine’s efficacy across diverse diagnostic scenarios, confirming compliance with the stringent standards established by . The continuous evolution of MRI technology demands persistent research and development, aimed at improving and addressing the challenges within the .
Blood Glucose Monitor: Critical for Diabetes Management
are indispensable tools for , allowing individuals to accurately track their blood sugar levels. Examples of are classified as medical instruments that are regulated due to their moderate risk. Recent have significantly enhanced the accuracy and usability of these devices, which are crucial for ensuring reliable readings. underscore the importance of these factors, as they directly influence individual adherence and health outcomes.
Effective blood glucose monitoring can lead to a and mitigate complications such as hypoglycemia and diabetic ketoacidosis. As diabetes management continues to evolve, the integration of is vital for enhancing care and achieving successful management outcomes.
Collaborating with bioaccess can accelerate the development and of these products, providing with expedited research services, including efficient patient recruitment and a faster route to market, ultimately improving .
Defibrillator: Life-Saving Device for Cardiac Emergencies
are life-saving instruments employed to restore normal heart rhythms during cardiac emergencies. As , they necessitate extensive to evaluate their efficacy and safety in real-world scenarios. These trials, which can be expertly managed by bioaccess® in Latin America, must evaluate the device’s performance across diverse settings, including hospitals and public spaces. This ensures that timely and effective treatment can be delivered during critical situations.
With bioaccess®’s specialized knowledge and experience in conducting , the pathway to and successful implementation of is expedited, ultimately enhancing health outcomes.
Ventilator: Essential for Respiratory Support in Critical Care
Ventilators serve a critical function in delivering to patients who cannot breathe adequately on their own. As examples of , they are subject to to ensure their safety and effectiveness. These trials assess ventilator performance in diverse clinical scenarios, including:
Recent advancements in have markedly enhanced outcomes for individuals in , improving both safety and efficacy. Adhering to is essential to ensure that these products deliver effective treatment while safeguarding patient health.
Endoscope: Key for Minimally Invasive Diagnostics and Treatments
Endoscopes serve as pivotal examples of that are essential for both diagnostic and therapeutic procedures within the body. Their classification necessitates to confirm , meticulously assessing the endoscope’s functionality across diverse healthcare applications. These evaluations guarantee that while minimizing discomfort for patients.
At bioaccess®, we excel in managing , including endoscopes, providing a suite of services such as:
- Site selection
- Compliance reviews
- Trial setup
- Import permits
- Project management
- Reporting
Recent , particularly the integration of suction tools with single-use scopes, have significantly enhanced procedural efficiency and . As the field progresses, are vital to further elevate the of these indispensable tools in .
Pacemaker: Essential for Heart Rhythm Management
Pacemakers serve as implantable instruments that play a crucial role in regulating heart rhythms for individuals experiencing arrhythmias. They are examples of , which require comprehensive studies to rigorously evaluate their safety and efficacy. Such assessments must scrutinize the pacemaker’s performance across diverse populations, ensuring compliance with .
Organizations like bioaccess® are at the forefront of this effort, providing extensive research management services that include:
These studies are vital for . Ongoing research and rigorous are indispensable for enhancing patient outcomes and propelling the evolution of pacemaker technology.
Conclusion
The exploration of Class II medical devices underscores their critical role in modern healthcare, highlighting the necessity for rigorous clinical studies to guarantee their safety and efficacy. Devices such as infusion pumps and pacemakers serve distinct purposes, significantly enhancing patient care and treatment outcomes. Technological advancements and streamlined processes provided by organizations like bioaccess® are pivotal in expediting the research and regulatory approval of these essential instruments.
Key insights from the article emphasize the significance of effective clinical trial management in navigating the complexities of regulatory landscapes. The examples presented, including surgical staplers and blood glucose monitors, illustrate how ongoing innovation and meticulous evaluation can lead to improved patient safety and healthcare delivery. Moreover, the collaboration between startups and research entities cultivates an environment where groundbreaking medical technologies can flourish.
In conclusion, as the demand for Class II medical devices continues to escalate, the commitment to enhancing research efficiency and regulatory compliance becomes paramount. Stakeholders in the medical technology field are urged to prioritize collaboration and innovation, ensuring that essential devices reach the market swiftly and safely. By doing so, the healthcare community can better meet the needs of patients and enhance health outcomes across diverse medical settings.
Frequently Asked Questions
What is bioaccess® and its role in clinical research for Class II medical devices?
bioaccess® specializes in accelerating clinical research for Class II medical devices by navigating regulatory landscapes in Latin America, the Balkans, and Australia. Their services include feasibility and selection of research sites, trial set-up, and meticulous study project management.
How quickly can bioaccess® obtain ethical approvals and enroll participants?
bioaccess® can obtain ethical approvals in approximately 4-6 weeks and achieve participant enrollment 50% faster than in conventional markets.
Why is the efficiency of regulatory processes important for Class II medical devices?
Enhancing regulatory efficiency is crucial for fostering innovation, allowing companies to meet the growing demand for Class II medical devices and ensuring timely access to essential healthcare solutions.
What partnership is bioaccess® involved in to enhance clinical studies in Latin America?
bioaccess® has partnered with Caribbean Health Group to establish Barranquilla as a key location for clinical studies in Latin America, with support from Colombia’s Minister of Health.
What are infusion pumps and their significance in healthcare?
Infusion pumps are essential devices designed to deliver medications, nutrients, and fluids to patients with precision. They are classified as Class II medical devices and are critical in various medical scenarios, including surgical procedures and chronic disease management.
What advancements have been made in infusion pump technology recently?
Recent advancements in infusion pump technology, particularly in 2025, have introduced enhanced features that improve user safety and operational efficiency, exemplified by the Medfusion 4000 Syringe Pump, known for its precise medication delivery in critical care settings.
What role do clinical trials play in the development of infusion pumps?
Comprehensive clinical trial management services, including feasibility studies and project oversight, are essential to ensure that infusion pumps meet necessary safety and efficacy standards.
What are surgical staplers and their applications in surgery?
Surgical staplers are key tools used in modern surgery to efficiently close wounds and connect tissues. They can be reusable or disposable and are categorized into linear staplers for organ removal and circular staplers for digestive tract operations.
Why is extensive study important for surgical staplers?
Given the potential risks associated with surgical staplers if used incorrectly, extensive studies are imperative to validate their safety and efficacy, which is essential for securing market approval.
How has surgical stapler technology evolved recently?
Innovations in surgical stapler technology in 2025 have significantly improved their functionality, resulting in enhanced precision and reduced surgical times, contributing to better patient outcomes.
What services does bioaccess® provide for surgical staplers?
bioaccess® offers accelerated clinical study services for surgical staplers, including Early-Feasibility Studies, First-In-Human Studies, and Post-Market Clinical Follow-Up Studies, to navigate the regulatory landscape effectively.
List of Sources
- bioaccess®: Accelerating Clinical Research for Class II Medical Devices
- Paul-Ehrlich-Institut – Information on procedures and timelines for submission of clinical trials (https://pei.de/EN/regulation/clinical-trials/procedures-timelines/procedures-timelines-node.html)
- Factors influencing the time to ethics and governance approvals for clinical trials: a retrospective cross-sectional survey – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10693024)
- Regulatory approval of clinical trials: is it time to reinvent the wheel? (https://gh.bmj.com/content/9/1/e013727)
- Infusion Pump: Essential for Controlled Medication Delivery
- (PDF) Artificial Intelligence, Machine Learning and Reasoning in Health Informatics—Case Studies (https://researchgate.net/publication/346113079_Artificial_Intelligence_Machine_Learning_and_Reasoning_in_Health_Informatics-Case_Studies)
- medonegroup.com (https://medonegroup.com/aboutus/blog/infusion-pumps-vital-tools-in-modern-healthcare)
- Surgical Stapler: Key Tool for Efficient Surgical Procedures
- drugwatch.com (https://drugwatch.com/surgical-staplers)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC4091615)
- Blood Glucose Monitor: Critical for Diabetes Management
- Demographic variation in continuous glucose monitoring utilisation among patients with type 1 diabetes from a US regional academic medical centre: a retrospective cohort study, 2018–2021 – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11907049)
- Ventilator: Essential for Respiratory Support in Critical Care
- jamanetwork.com (https://jamanetwork.com/journals/jama/fullarticle/2777716)
- Endoscope: Key for Minimally Invasive Diagnostics and Treatments
- ambuusa.com (https://ambuusa.com/blog?p=ambu-scores-high-for-safety-and-versatility-in-new-study)

Leave a Reply