Introduction
Navigating the complexities of telemedicine device approval presents a significant challenge, particularly within a regulatory landscape that is in a state of constant evolution. As innovative healthcare solutions continue to emerge, grasping the intricacies of INVIMA’s guidance is crucial for companies seeking to accelerate their market entry. This article explores nine essential strategies that not only increase the likelihood of successful approvals but also empower organizations to adapt to the ever-changing regulatory environment.
How can companies effectively leverage these strategies to not only achieve compliance but also excel in a competitive healthcare market?
bioaccess®: Accelerate Your Telemedicine Device Approval Process
bioaccess® is dedicated to accelerating the validation process for by leveraging extensive experience and a deep understanding of compliance frameworks, including , across Latin America, the Balkans, and Australia. By harnessing our pre-qualified networks and centralized monitoring systems, companies can anticipate ethical consent within a mere 4-6 weeks. This remarkable efficiency significantly shortens the time to market for innovative , ensuring adherence to .
Our comprehensive suite of services encompasses:
- Managing
- Pilot Studies
- Pivotal Studies
- Post-Market Clinical Follow-Up Studies
This expedited approval process is essential for maintaining a competitive edge in the swiftly evolving healthcare landscape, particularly as Directors of navigate the complexities of compliance.
Understand INVIMA’s Regulatory Requirements for Telemedicine Devices
Colombia’s National Food and Drug Surveillance Institute plays a crucial role in providing to regulate telemedicine devices and ensure compliance with established health standards. As an oversight body established in 1992 under Colombia’s Ministry of Health and Social Protection, this organization is tasked with inspecting and supervising the marketing and manufacturing of health products.
To meet the , companies must prepare and submit a comprehensive set of documentation, including:
- Thorough
Understanding the in Colombia is vital for navigating the regulatory framework and ensuring adherence to local standards. The Directorate for Medical Equipment and other Technologies within the agency oversees , proposing technical standards for production, promotion, and quality control.
Recent statistics indicate that adherence rates to regulatory standards have improved, with for medical equipment trials reaching approximately 75%. Staying informed about is essential, as these changes can significantly impact the submission process and overall compliance strategy for .
Build Local Partnerships to Enhance Approval Success
Establishing , research groups, and industry associations is paramount for enhancing the likelihood of . Such partnerships yield invaluable insights into , facilitating smoother communication throughout the approval journey by utilizing .
Involving local specialists not only aids in but also addresses . Successful case studies illustrate that following and approved through frequently achieve , underscoring the significance of tailored strategies that resonate with regional healthcare dynamics.
As highlighted by industry leaders, these collaborations are essential for fostering innovation and ensuring that new technologies align with the .
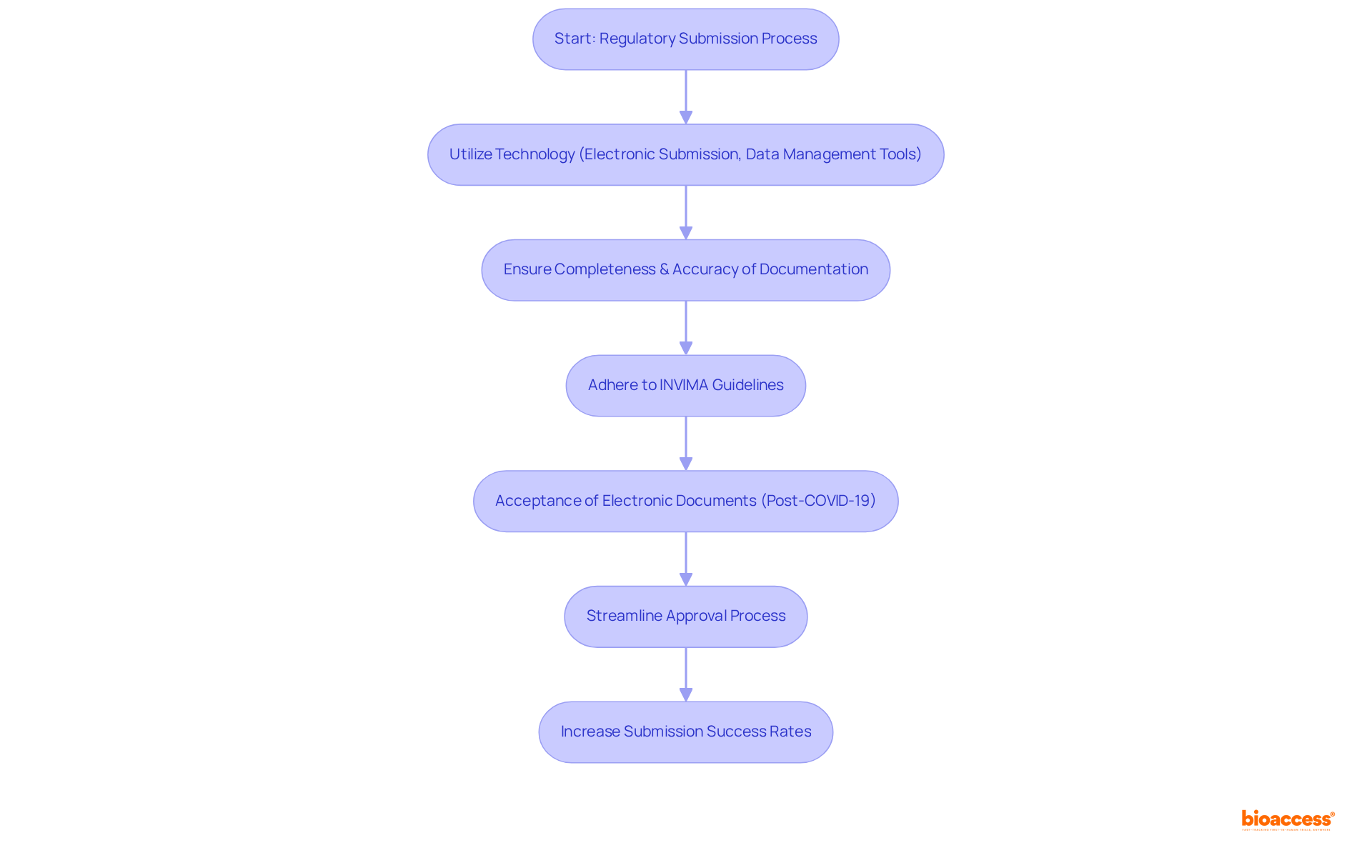
Leverage Technology for Efficient Regulatory Submissions
Utilizing technology, such as electronic submission systems and data management tools, is crucial for of to the , especially in relation to .
Established in 1992 under Colombia’s Ministry of Health and Social Protection, INVIMA plays a pivotal role in inspecting and supervising the marketing and manufacturing of health products, including the provision of .
Its is tasked with ensuring adherence to health standards while providing and overseeing the classification and authorization processes.
Notably, during the , policy flexibilities allowed for the acceptance of electronically generated documents, significantly .
These technological tools ensure that all required documentation is complete and accurately formatted, thereby reducing the likelihood of delays due to errors or missing information.
Industry specialists assert that can simplify the entire submission process, facilitating progress tracking and addressing inquiries from oversight bodies.
This proactive approach not only enhances submission success rates but also fosters a more adaptable oversight landscape, aligning with INVIMA’s commitment to ensuring the of through .
Conduct Market Research to Inform Your Strategy
Thorough is essential for navigating the competitive landscape and identifying unmet needs within the sector of . This research informs , , and . By analyzing current market trends, consumer preferences, and competitor offerings, companies can effectively tailor their submissions to align with market demands. Such strategic alignment not only enhances the likelihood of official approval but also supports the successful commercialization of in accordance with . Understanding these dynamics is crucial, particularly as the is projected to experience , propelled by the increasing demand for innovative and .
Train Your Team on INVIMA Guidelines and Best Practices
Educating your team on is and within the Latin American Medtech environment. Frequent workshops and training sessions not only keep team members informed about but also deepen their understanding of the submission process. This investment in training and within the organization, leading to .
For example, the partnership between bioaccess® and Welwaze Medical Inc. during the Celbrea® medical product launch illustrates how can accelerate compliance and market entry. Moreover, with participation in than in conventional markets, effective training is a crucial factor in navigating the complexities of the , particularly in securing vital approvals from health authorities and other oversight organizations.
Prepare Comprehensive Documentation for Regulatory Submissions
Creating thorough documentation is a crucial step in the regulatory submission process, particularly in accordance with . This documentation encompasses not only technical specifications and clinical data but also includes:
Successful submissions to the exemplify , demonstrating how well-prepared documentation can facilitate a smoother review process while reducing the likelihood of requests for additional information or clarifications.
INVIMA, classified as a Level 4 health authority by the Pan American Health Organization/World Health Organization, oversees the marketing and manufacturing of health products, including medical devices. Firms that adhere to best practices in organizing their report considerably quicker processing times.
As Dr. Tedros Adhanom Ghebreyesus has noted, is crucial for navigating the complexities of compliance procedures, ensuring that all necessary information is readily available for examination. By focusing on thorough documentation tailored for , companies can significantly enhance their chances of achieving prompt and effective regulatory outcomes.
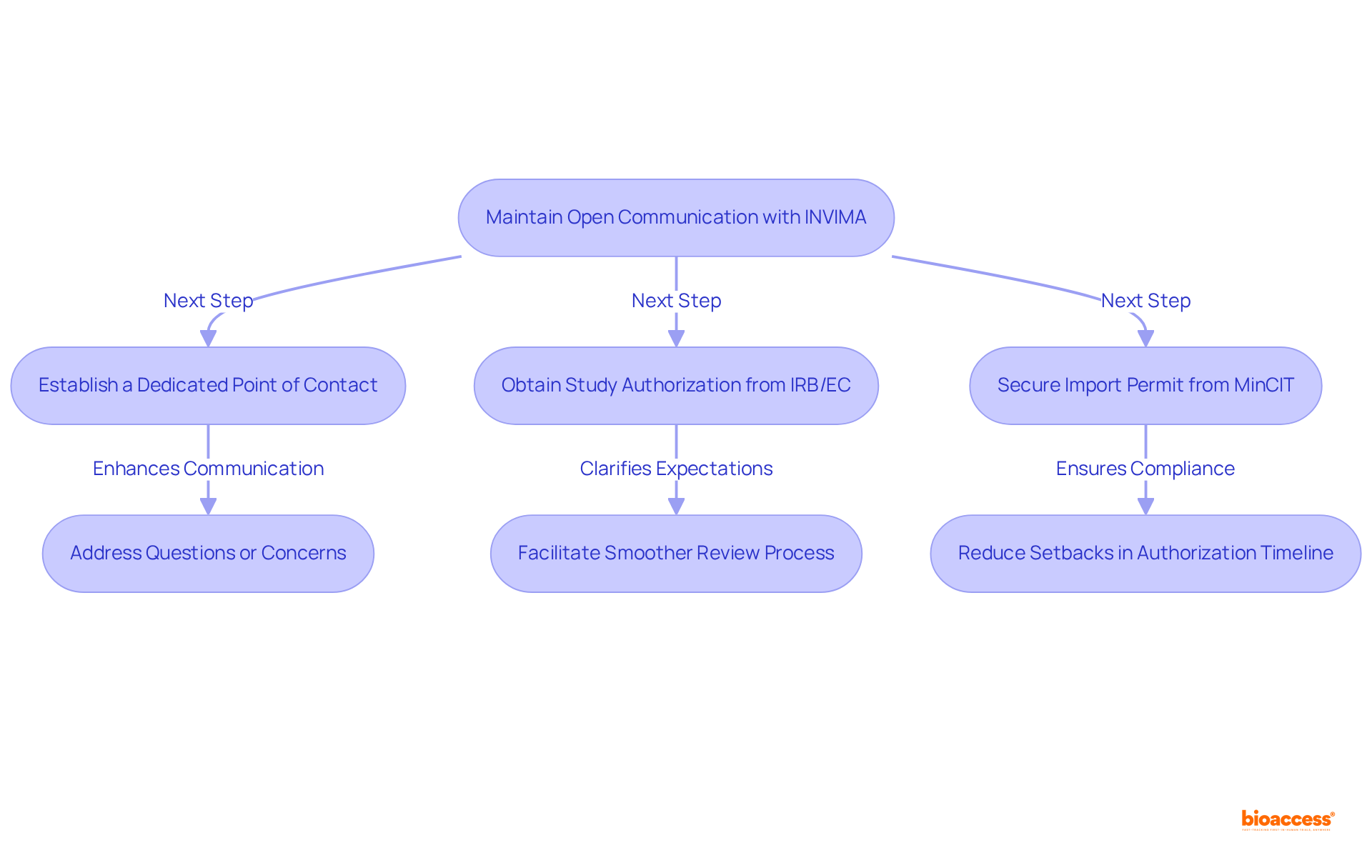
Maintain Open Communication with INVIMA During Approval
Maintaining clear channels of communication with the is crucial for effectively addressing any questions or concerns that may arise. Regular updates and proactive engagement not only clarify expectations but also facilitate a .
Establishing a significantly enhances communication, ensuring that any issues are resolved promptly. This proactive strategy reduces potential setbacks in the , particularly for , which have a timeline of approximately 300 days.
Furthermore, obtaining and securing an import permit from are essential steps in the process.
By , bioaccess® accelerates the path to market for , enabling startups to advance to the next phase of their clinical trials and ultimately achieve their objectives more swiftly.
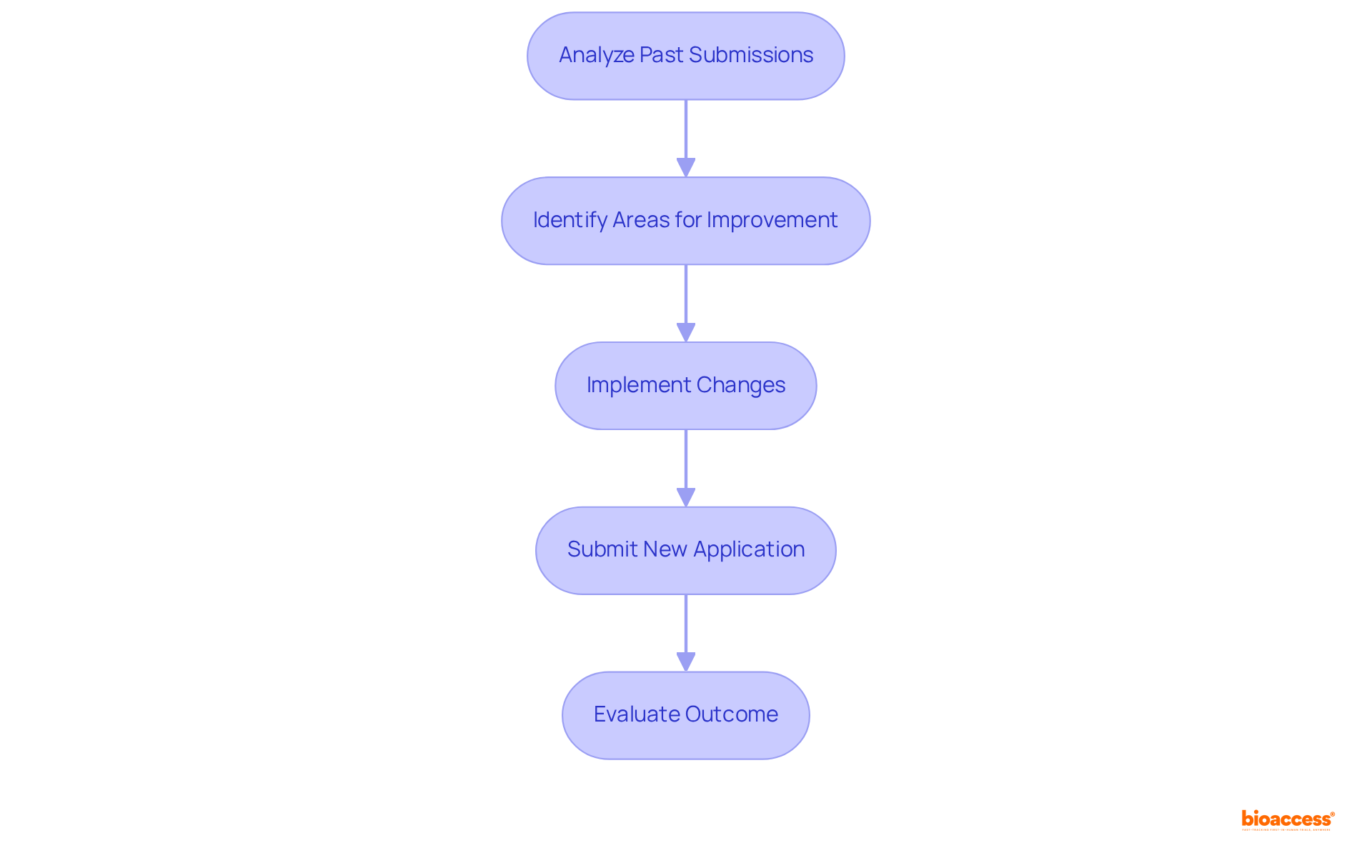
Utilize Feedback from Previous Submissions to Improve Future Applications
Utilizing is essential for enhancing . A thorough analysis of past submissions—especially those that faced —reveals . By proactively addressing these issues, companies can significantly elevate the quality of their submissions, thereby increasing their chances of approval in line with . This iterative process not only improves but also fosters a deeper understanding of , ultimately leading to more in . Adopting a culture of is crucial in forming an .
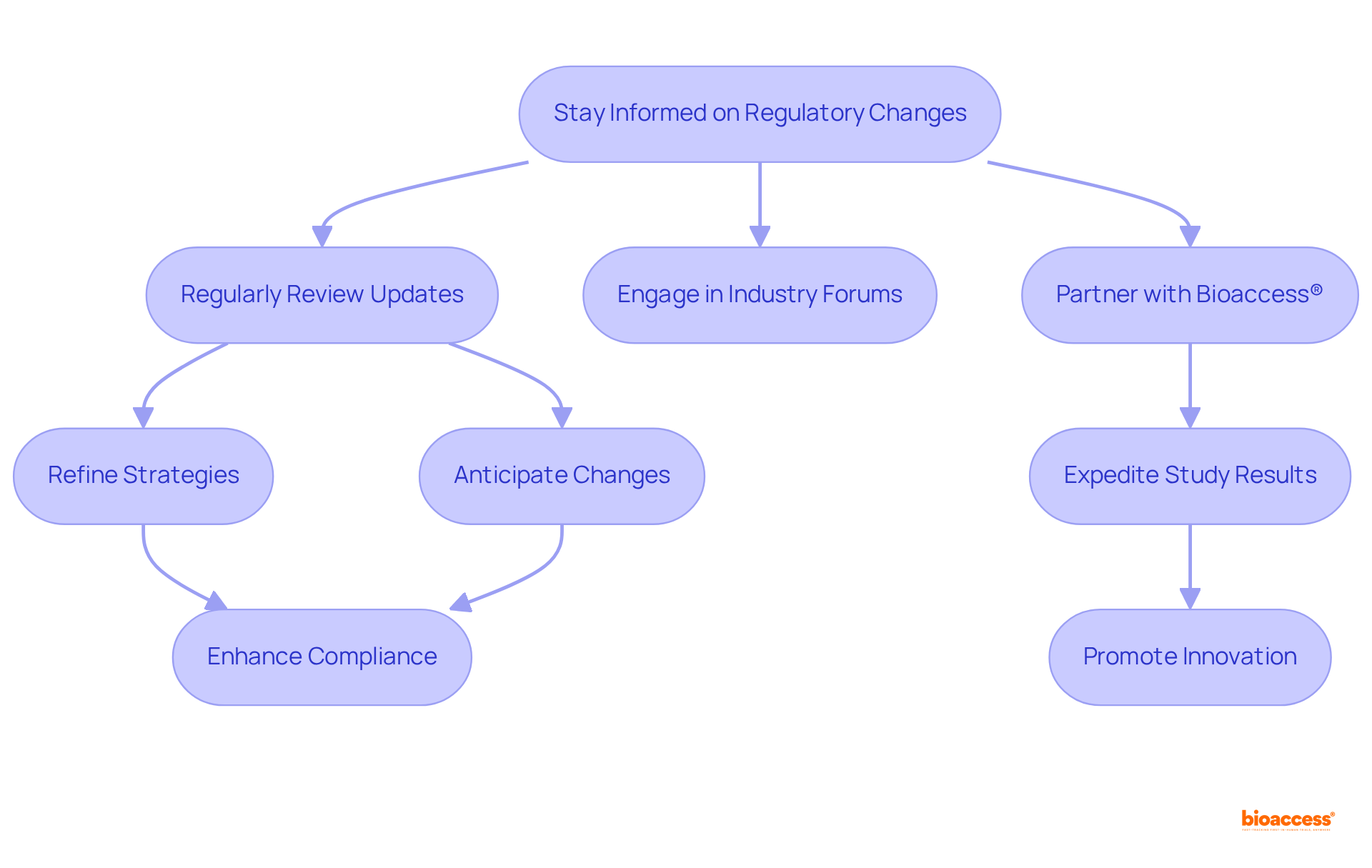
Stay Informed on Regulatory Changes Affecting Telemedicine Devices
Staying informed about legal changes affecting telemedicine devices is essential for compliance with and . With current turnaround times for applications often exceeding 400 days, companies must and engage in industry forums to anticipate changes and refine their strategies. This proactive approach not only ensures compliance with regulations but also allows organizations to capitalize on .
Firms that consistently can more effectively navigate , especially with the introduction of INVIMA’s new Contingency Plan aimed at enhancing oversight processes and reducing backlogs. Furthermore, can significantly expedite by offering comprehensive support in , clinical research site activation, patient recruitment, and trial data management.
By leveraging these initiatives, organizations can strengthen their market strategies and promote innovation in telemedicine. To maximize these advantages, consider scheduling a consultation with bioaccess® to explore tailored solutions for your .
Conclusion
Navigating the complexities of telemedicine device approval under INVIMA guidance is essential for companies aiming to thrive in the healthcare landscape. The strategies outlined in this article emphasize the importance of understanding regulatory requirements, leveraging technology, and fostering local partnerships to enhance the approval process. By adopting a comprehensive approach that includes thorough documentation, effective team training, and proactive communication with INVIMA, organizations can significantly improve their chances of successful submissions.
Key insights include:
- The necessity of conducting market research to align product offerings with consumer needs and regulatory expectations.
- Maintaining open lines of communication with regulatory bodies.
- Utilizing feedback from previous submissions to refine future applications.
These strategies not only streamline the approval process but also position companies to respond effectively to the evolving regulatory environment.
Ultimately, embracing these best practices for telemedicine device regulation accelerates the path to market and contributes to the overall advancement of healthcare solutions. Companies are encouraged to remain vigilant about regulatory changes and consider partnerships with experienced organizations like bioaccess® to navigate the complexities of compliance. By doing so, they can foster innovation and ensure that their telemedicine devices meet the needs of patients and healthcare providers alike.
Frequently Asked Questions
What is bioaccess® and how does it assist with telemedicine device approval?
bioaccess® accelerates the validation process for telemedicine devices by utilizing extensive experience and knowledge of compliance frameworks. They help companies anticipate ethical consent within 4-6 weeks, significantly reducing the time to market while ensuring adherence to FDA, EMA, and MDR standards.
What services does bioaccess® offer for telemedicine device approval?
bioaccess® offers a comprehensive suite of services that includes managing Early-Feasibility Studies, First-In-Human Studies, Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies.
Why is understanding INVIMA’s regulatory requirements important for telemedicine devices?
Understanding INVIMA’s regulatory requirements is crucial for ensuring compliance with established health standards in Colombia. It helps companies navigate the regulatory framework and meet the stringent requirements set by Colombia’s National Food and Drug Surveillance Institute.
What documentation is required to meet INVIMA’s regulatory standards for telemedicine devices?
Companies must prepare and submit a comprehensive set of documentation, including technical specifications, clinical evaluation reports, and thorough risk assessments.
How can companies enhance their chances of successful telemedicine device approval?
Companies can enhance their chances of approval by establishing collaborations with local healthcare entities, research groups, and industry associations. These partnerships provide insights into INVIMA’s expectations and facilitate smoother communication throughout the approval process.
What role do local specialists play in the approval process for telemedicine devices?
Local specialists help manage compliance challenges and address cultural subtleties that may affect product acceptance. Their involvement can lead to higher success rates for telemedicine tools that follow INVIMA’s guidance.
What recent trends have been observed in adherence to regulatory standards for telemedicine devices?
Recent statistics indicate improved adherence rates to regulatory standards, with ethical consent rates for medical equipment trials reaching approximately 75%. Staying informed about updates from the regulatory authority is essential for compliance strategy.
List of Sources
- bioaccess®: Accelerate Your Telemedicine Device Approval Process
- bioaccessla.com (https://bioaccessla.com/blog/designing-trials-for-approval-in-brazil-an-in-depth-tutorial-for-clinical-researchers)
- Current Trends in Argentina Medical Device Tri… | bioaccess® (https://bioaccessla.com/blog/current-trends-in-argentina-medical-device-trials-and-regulations)
- tandfonline.com (https://tandfonline.com/doi/full/10.1080/26410397.2023.2236780)
- bioaccessla.com (https://bioaccessla.com/blog/successfully-expanding-medtech-to-latin-america-a-step-by-step-guide)
- trade.gov (https://trade.gov/country-commercial-guides/brazil-healthcare)
- Understand INVIMA’s Regulatory Requirements for Telemedicine Devices
- bioaccessla.com (https://bioaccessla.com/es/blog/10-key-tests-for-ensuring-biocompatibility-in-medical-devices)
- INVIMA Colombia Medical Device Registration | Pure Global (https://pureglobal.com/markets/colombia)
- Understanding Anvisa Guidelines For Medical Device Trials An In Depth Tutorial | bioaccess® (https://bioaccessla.com/blog/understanding-anvisa-guidelines-for-medical-device-trials-an-in-depth-tutorial)
- magazine.medicaltourism.com (https://magazine.medicaltourism.com/article/stem-cell-therapy-for-cancer-an-overview-of-treatment-in-colombia)
- placidway.com (https://placidway.com/search-medical-centers/Stem-Cell-Therapy/Colombia/1)
- Build Local Partnerships to Enhance Approval Success
- trade.gov (https://trade.gov/country-commercial-guides/greece-healthcare)
- allergyasthmanetwork.org (https://allergyasthmanetwork.org/news/aadch-asthma-allergies-advocacy-day-2021)
- state.gov (https://state.gov/reports/2024-investment-climate-statements/indonesia)
- Fact Sheet: Telehealth | AHA (https://aha.org/fact-sheets/2025-02-07-fact-sheet-telehealth)
- mn.gov (https://mn.gov/dhs/partners-and-providers/news-initiatives-reports-workgroups/minnesota-health-care-programs/integrated-health-partnerships/index.jsp)
- Leverage Technology for Efficient Regulatory Submissions
-
Best Practices
Case Studies (https://ors.od.nih.gov/OD/OQM/benchmarking/bestpractice/Pages/case_studies.aspx) -
The Top 5 Challenges in Medical Device Regulatory Compliance and How to Overcome Them – Nerac (https://nerac.com/the-top-5-challenges-in-medical-device-regulatory-compliance-and-how-to-overcome-them)
-
CMC Requirements for New Drug Registration in Latin America (https://ispe.org/pharmaceutical-engineering/may-june-2023/cmc-requirements-new-drug-registration-latin-america)
-
apecusa.com (https://apecusa.com/case_studies/pharmaceutical)
-
- Conduct Market Research to Inform Your Strategy
- driveresearch.com (https://driveresearch.com/market-research-company-blog/how-to-conduct-a-physicians-survey-telemedicine-market-research-company)
- fortunebusinessinsights.com (https://fortunebusinessinsights.com/industry-reports/telemedicine-market-101067)
- health.economictimes.indiatimes.com (https://health.economictimes.indiatimes.com/news/health-it/strengthening-indias-healthcare-system-with-telemedicine/89111712)
- clearpointstrategy.com (https://clearpointstrategy.com/blog/pest-analysis-healthcare)
- Telehealth Systems – StatPearls – NCBI Bookshelf (https://ncbi.nlm.nih.gov/books/NBK459384)
- Train Your Team on INVIMA Guidelines and Best Practices
- freyrsolutions.com (https://freyrsolutions.com/blog/colombias-new-pharmacovigilance-regulations-what-mahs-need-to-know-are-you-up-to-date-with-colombias-latest-pharmacovigilance-regulations)
- bioaccessla.com (https://bioaccessla.com/blog/navigating-the-medtech-regulatory-landscape-in-latin-america-an-essential-guide)
- myshyft.com (https://myshyft.com/blog/cybersecurity-insurance-quotes-bridgeport-connecticut)
- onlinesafetytrainer.com (https://onlinesafetytrainer.com/190-quotes-to-motivate-your-co-workers-about-safety-training)
- Prepare Comprehensive Documentation for Regulatory Submissions
- accruent.com (https://accruent.com/resources/blog-posts/document-control)
- who.int (https://who.int/news/item/19-10-2023-who-outlines-considerations-for-regulation-of-artificial-intelligence-for-health)
- fda.gov (https://fda.gov/drugs/news-events-human-drugs/new-drugs-regulatory-program-modernization-integrated-assessment-marketing-applications-and)
- mfds.go.kr (https://mfds.go.kr/eng/wpge/m_39/denofile.do)
- Maintain Open Communication with INVIMA During Approval
- 130+ Compliance Statistics & Trends to Know for 2026 (https://secureframe.com/blog/compliance-statistics)
- freyrsolutions.com (https://freyrsolutions.com/blog/colombias-new-pharmacovigilance-regulations-what-mahs-need-to-know-are-you-up-to-date-with-colombias-latest-pharmacovigilance-regulations)
- INVIMA Medical Device Regulations & Registration Process in Colombia – Artixio (https://artixio.com/post/medical-device-regulations-in-colombia-a-comprehensive-guide)
- Understanding Statistical Analysis in Clinical Trials (https://lindushealth.com/blog/understanding-clinical-trial-statistics-a-comprehensive-guide)
- ir.biocryst.com (https://ir.biocryst.com/news-releases/news-release-details/orladeyor-berotralstat-approved-colombia)
- Utilize Feedback from Previous Submissions to Improve Future Applications
- FDA Proposes Framework to Advance Credibility of AI Models Used for Drug and Biological Product Submissions (https://fda.gov/news-events/press-announcements/fda-proposes-framework-advance-credibility-ai-models-used-drug-and-biological-product-submissions)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC6046667)
- fileandservexpress.com (https://fileandservexpress.com/why-was-my-filing-rejected)
- Understanding peer review – Author Services (https://authorservices.taylorandfrancis.com/publishing-your-research/peer-review)
- grants.nih.gov (https://grants.nih.gov/grants-process/submit/submission-policies/resubmission-applications)
- Stay Informed on Regulatory Changes Affecting Telemedicine Devices
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S1386505624001369)
- nsf.org (https://nsf.org/news/jan-2023-medical-device-ivd-update)
- freyrsolutions.com (https://freyrsolutions.com/blog/new-invima-contingency-plan-freyr-helps-you-navigate-this-regulatory-opportunity)
- bioaccessla.com (https://bioaccessla.com/blog/how-to-navigate-anvisa-regulations-for-medtech-companies-a-step-by-step-guide)

Leave a Reply