Introduction
The rise of digital technology has fundamentally transformed business operations, especially within the healthcare sector, where secure transactions are crucial. Argentina’s Administración Nacional de Medicamentos, Alimentos y Tecnología Médica (ANMAT) plays a vital role in ensuring that digital signatures adhere to stringent regulations, thus safeguarding public health and enhancing operational efficiency.
However, many stakeholders find navigating the complexities of ANMAT’s digital signature application process to be a daunting task. How can organizations effectively leverage this system to ensure compliance while steering clear of common pitfalls?
Understand ANMAT’s Role in Digital Signatures
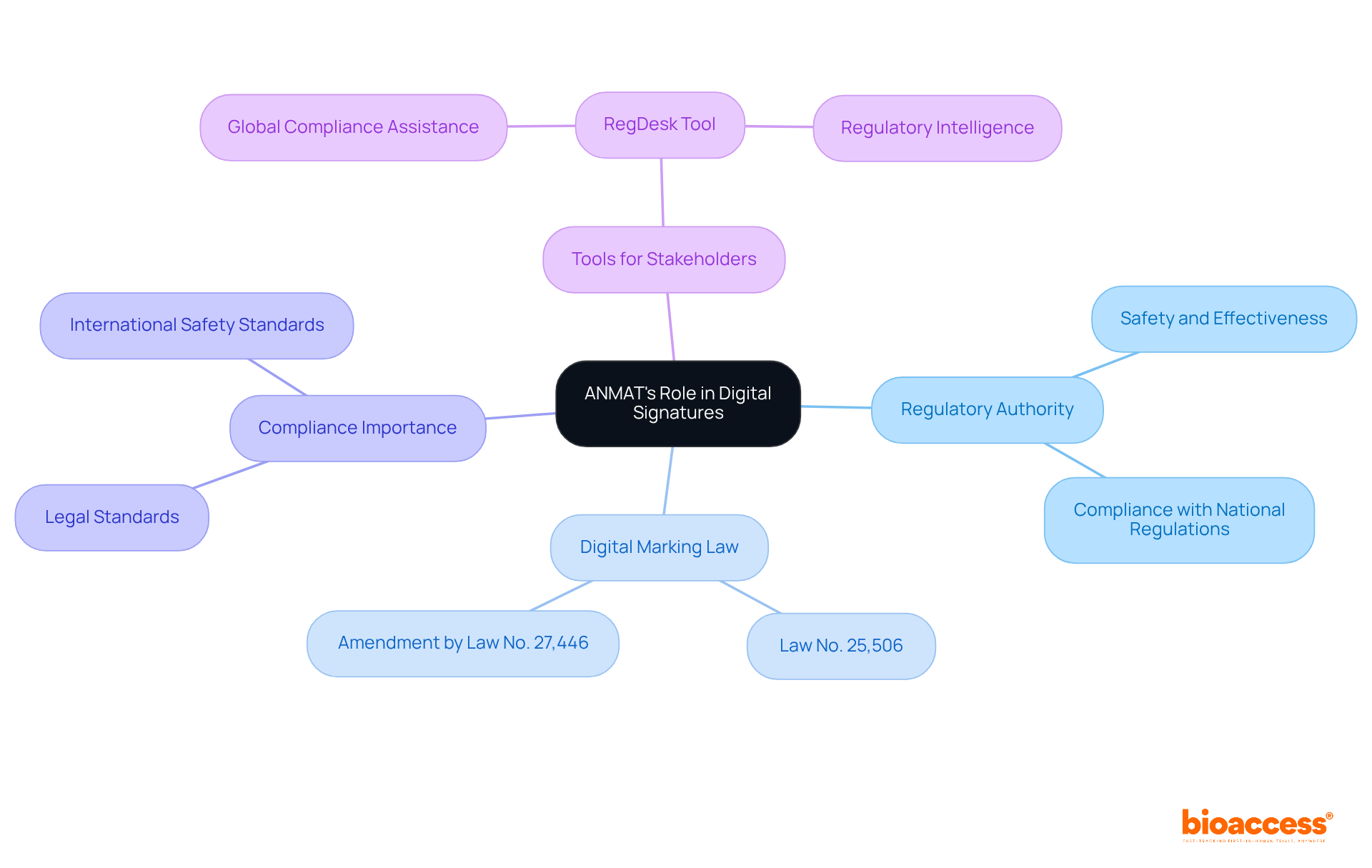
The serves as Argentina’s regulatory authority, ensuring the safety and effectiveness of , including the use of . This decentralized entity, operating under the Ministry of Health, plays a crucial role in enforcing , which is essential for secure and legally binding online transactions.
In Argentina, digital markings are governed by the Digital Marking Law No. 25,506, later amended by Law No. 27,446. Understanding the agency’s is vital for stakeholders aiming to navigate this landscape effectively, especially regarding to ensure their meet all necessary legal standards. Furthermore, adherence to international safety and quality standards is a cornerstone of the agency’s regulatory approach.
Recent updates to Argentina’s regulations underscore the necessity of compliance, enhancing operational efficiency and . Tools like RegDesk can facilitate smoother expansion into new markets, equipping stakeholders with practical resources to navigate effectively.
Gather Required Documentation for Digital Signature Application
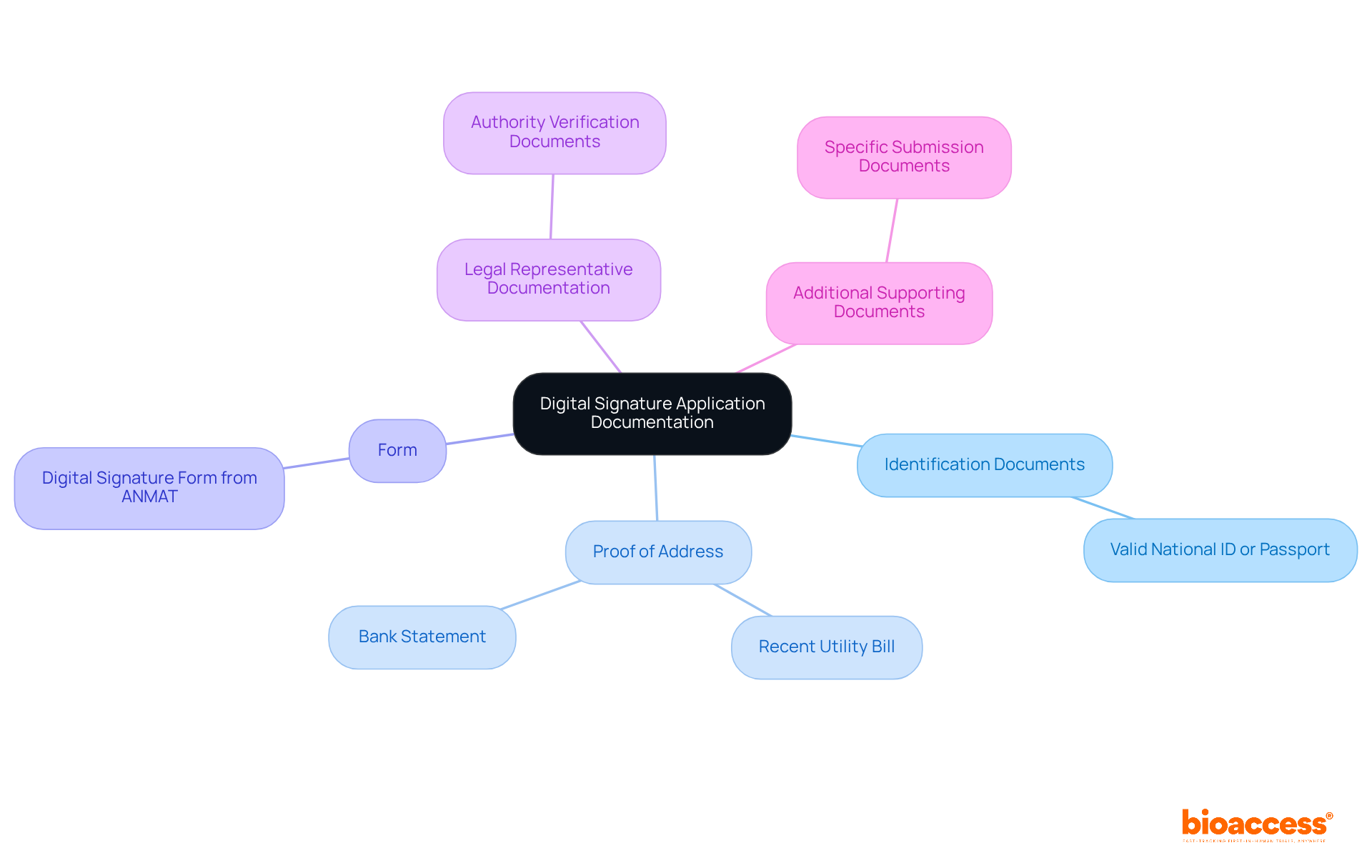
To successfully apply for a through ANMAT, you must compile the following documentation:
- Identification Documents: A valid national ID or passport is necessary to verify your identity.
- Proof of Address: A recent utility bill or bank statement that clearly displays your current address.
- Form: Complete the form, available on .
- Legal Representative Documentation: If applying on behalf of a company, include documentation that verifies your authority to act on its behalf.
- Additional Supporting Documents: Depending on your submission specifics, other documents may be required.
It’s crucial to ensure that all documents are up-to-date and formatted correctly to prevent any delays in processing your application. The at a , highlighting the growing significance of secure electronic validations across various sectors, including . As companies implement to combat identity theft online, ensuring the validity of your electronic mark becomes essential. Inadequate security measures can expose organizations to significant risks, making it vital to adhere to during the application process.
Apply for Your Digital Signature via ANMAT’s Online Portal
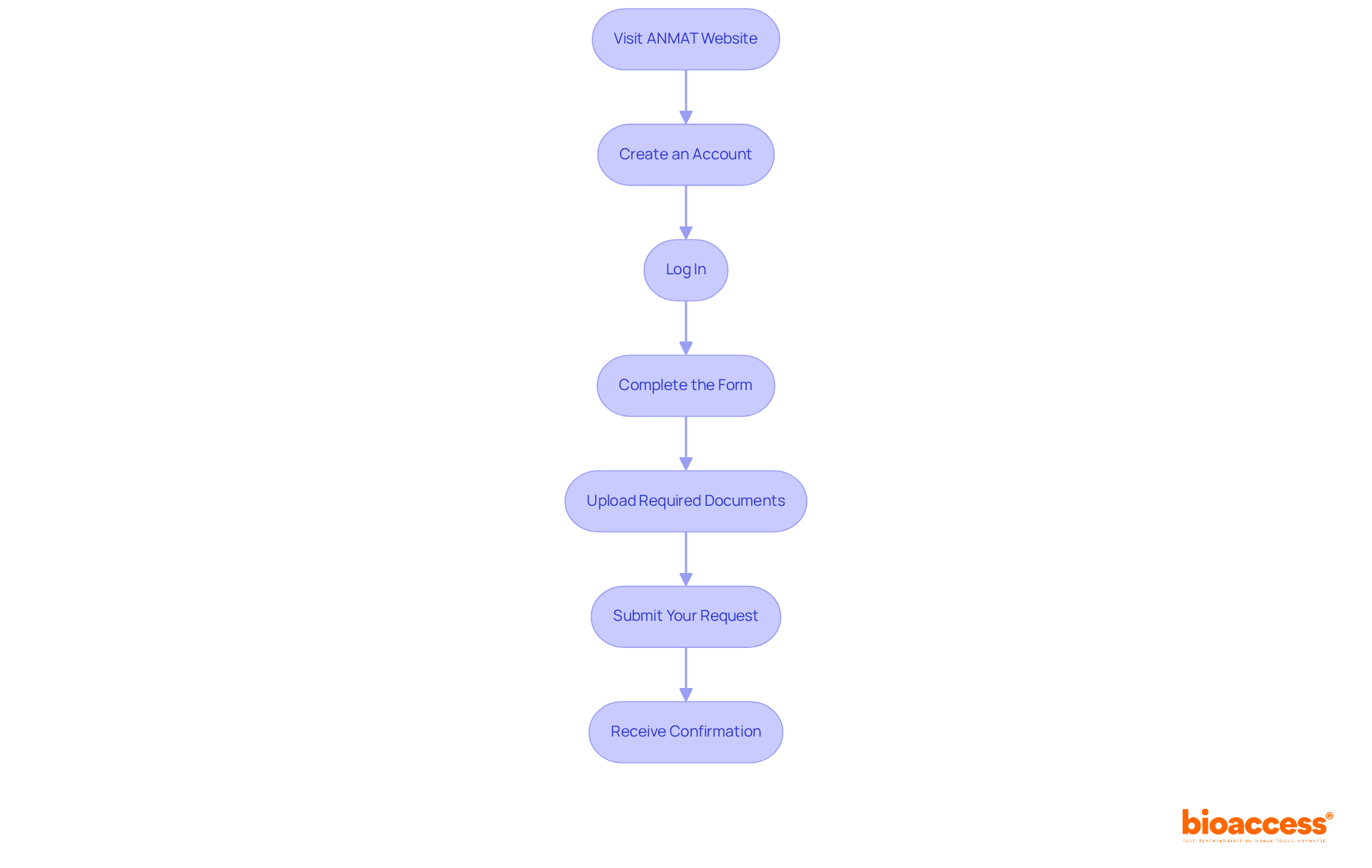
To apply for your digital signature through ANMAT’s online portal, follow these steps:
- Visit the ANMAT Website: Go to the official ANMAT site and find the .
- Create an Account: If you do not have an account, register by providing your email address and creating a password.
- Log In: Use your credentials to log into your account.
- Complete the Form: Fill out the with accurate information.
- Upload Required Documents: Attach the gathered documentation as specified in the previous section.
- Submit Your Request: Review all information for accuracy and submit your request.
- Receive Confirmation: After submission, you will receive a confirmation email. Keep this for your records.
Following these steps ensures that your application is submitted correctly and efficiently with Argentina ANMAT . As South America emerges as the , with a , adopting electronic signings is becoming increasingly essential for compliance and efficiency in . The growing need for underscores the significance of secure online processes, making it crucial for healthcare organizations to integrate these technologies into their operations.
Troubleshoot Common Issues in the Digital Signature Process
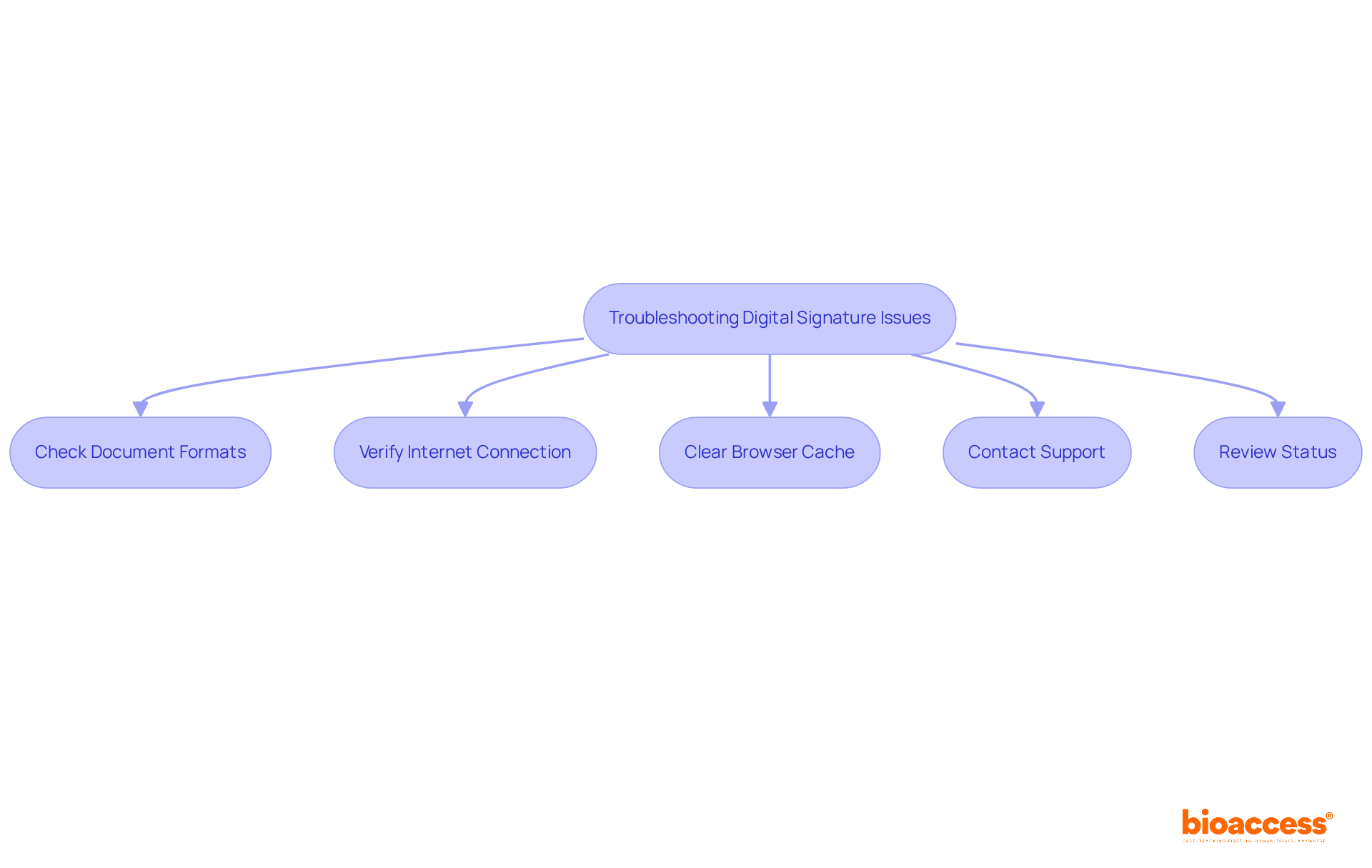
When facing challenges during the , consider these effective :
- Check Document Formats: Confirm that all uploaded documents adhere to the required format, typically PDF, and ensure they are not corrupted.
- Verify Internet Connection: A is essential for submitting your application. If you encounter connectivity issues, attempt the submission again later.
- Clear Browser Cache: Clearing your browser’s cache can often resolve loading issues on the portal, facilitating smoother navigation.
- Contact Support: Should problems persist, reach out to ANMAT’s . Providing detailed information about the issue can expedite the resolution process.
- Review Status: Log into your account to of your submission. If it shows as pending, double-check that all required documents were submitted correctly.
In the context of the expanding , where the need for is increasing due to security issues, these troubleshooting steps are essential. The absence of for electronic endorsements can impede market expansion, making it crucial to guarantee that your processing method is secure and effective. Additionally, insights from key players like i4pro and the partnership between SocialPath and Viafirma highlight the importance of robust in navigating these challenges. By implementing these troubleshooting steps, you can effectively navigate common issues and ensure your application remains on track.
Conclusion
Mastering the application process for Argentina’s ANMAT digital signatures is crucial for anyone navigating the complexities of electronic authentication in the healthcare sector. Understanding ANMAT’s regulatory role and the necessary documentation for application is vital. This guide outlines the step-by-step procedure to successfully obtain a digital signature, ensuring compliance with national regulations while enhancing operational efficiency.
Key insights highlight the importance of maintaining up-to-date documentation and meticulously following the application process. Additionally, troubleshooting strategies are essential for overcoming common challenges. As the electronic authentication market in South America expands, the significance of secure digital signatures becomes increasingly clear. Organizations must prioritize these practices to protect their operations and comply with evolving regulations.
Embracing the digital signature process streamlines transactions, safeguards public health, and enhances trust in online interactions. As the landscape of digital identity verification evolves, staying informed and proactive is vital. Engaging with ANMAT’s resources and leveraging available tools empowers stakeholders to navigate this essential aspect of the healthcare industry effectively.
Frequently Asked Questions
What is ANMAT and what role does it play in digital signatures in Argentina?
ANMAT, or the Administración Nacional de Medicamentos, Alimentos y Tecnología Médica, is Argentina’s regulatory authority responsible for ensuring the safety and effectiveness of health products, including the use of digital signatures.
What laws govern digital signatures in Argentina?
Digital signatures in Argentina are governed by the Digital Marking Law No. 25,506, which was later amended by Law No. 27,446.
Why is understanding ANMAT’s regulatory framework important for stakeholders?
Understanding ANMAT’s regulatory framework is essential for stakeholders to navigate the landscape effectively and ensure that their electronic authentication applications meet necessary legal standards.
What is the significance of compliance with international safety and quality standards for ANMAT?
Adherence to international safety and quality standards is a cornerstone of ANMAT’s regulatory approach, ensuring that health products and digital signatures are reliable and secure.
What recent updates have been made to Argentina’s electronic authentication regulations?
Recent updates to Argentina’s electronic authentication regulations emphasize the necessity of compliance, which enhances operational efficiency and safeguards public health.
How can tools like RegDesk assist stakeholders in navigating regulatory compliance?
Tools like RegDesk can facilitate smoother expansion into new markets by equipping stakeholders with practical resources to navigate regulatory compliance effectively.
List of Sources
- Understand ANMAT’s Role in Digital Signatures
- What is ANMAT? (https://argentina.gob.ar/anmat/anmat-en/what-anmat)
- Cosmetic Regulatory Requirements in Argentina: ANMAT Guide (https://freyrsolutions.com/blog/understanding-the-cosmetic-regulatory-landscape-in-argentina)
- regdesk.co (https://regdesk.co/blog/anmat-regulation-on-technovigil-overviewance)
- Gather Required Documentation for Digital Signature Application
- South America Digital Signature Market Size & Growth [2030] (https://fortunebusinessinsights.com/south-america-digital-signature-market-107412)
- Apply for Your Digital Signature via ANMAT’s Online Portal
- South America Digital Signature Market Size & Growth [2030] (https://fortunebusinessinsights.com/south-america-digital-signature-market-107412)
- Troubleshoot Common Issues in the Digital Signature Process
- South America Digital Signature Market Size & Growth [2030] (https://fortunebusinessinsights.com/south-america-digital-signature-market-107412)

Leave a Reply