Introduction
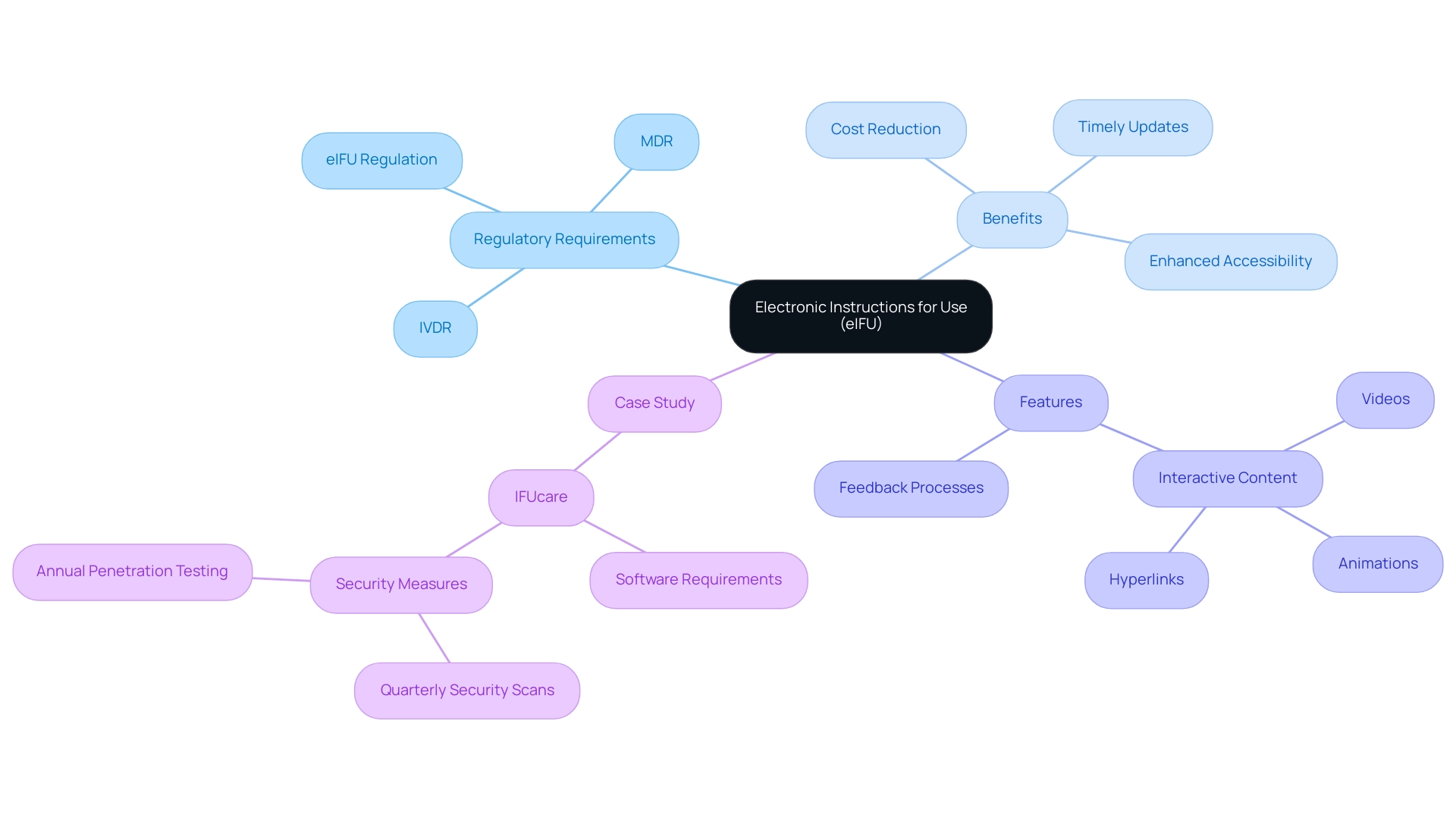
The landscape of medical device regulations is undergoing a significant transformation with the advent of Electronic Instructions for Use (eIFUs). Governed by stringent frameworks such as the Medical Device Regulation (MDR) and In Vitro Diagnostic Regulation (IVDR), these digital documents are not merely a compliance requirement; they represent a pivotal shift towards enhancing user experience and patient safety.
By mandating accessible, clear, and comprehensive eIFUs, regulators aim to empower healthcare professionals and patients alike, ensuring that critical information regarding medical devices is readily available. As manufacturers navigate the complexities of these regulations, understanding the implications of recent updates and the diverse applications of eIFUs across various medical device categories becomes essential.
This article delves into the key requirements, benefits, and future trends surrounding eIFUs, providing a roadmap for manufacturers to enhance compliance and ultimately improve patient care.
Overview of eIFU Regulations in Medical Devices
are governed by as outlined in ) 2017/745 and the . These regulations require that producers offer clear, accessible, and compliant to users, thereby enhancing user understanding and improving . The regulations specify particular requirements for , which must be easily accessible for download and must contain essential information regarding the equipment’s functionality, installation, and maintenance.
Compliance with the eifu is essential, as it reflects a manufacturer’s commitment to both legal obligations and in medical device usage. Particularly since the evaluation will pay special attention to the , especially for small and medium-sized enterprises, understanding these dynamics is crucial. Furthermore, recent statistics show that 53% of businesses leave 1,000 or more files containing sensitive information accessible to all employees, emphasizing the necessity of .
Furthermore, insights reveal that 60% of risk and compliance professionals intend to incorporate over the next two to three years, underscoring the increasing focus on . As the aimed at healthcare professionals, the feedback gathered will further inform the effectiveness of the , which is crucial for maintaining high standards in medical safety.
Key Requirements and Benefits of Electronic Instructions for Use
The application of electronic Instructions for Use in medical equipment necessitates adherence to the eIFU as well as the (MDR) and In Vitro Diagnostic Regulation (IVDR). It is imperative that the information provided within electronic instructions for use is not only accurate and comprehensive but also user-friendly to meet the needs of healthcare professionals and patients alike. As per the regulations, to ensure unconditional access to the instructions for use in electronic form, these instructions should be available on the manufacturer’s website in an in which the device is made available.
The advantages of are substantial, including:
- Significant reductions in printing and distribution costs
- The capacity for timely updates
Notably, electronic Instructions for Use support , which greatly enhance the user experience. Additionally, manufacturers gain from , fostering continuous improvement in product usage and safety outcomes.
A case study involving IFUcare, Qarad’s eIFU software service, highlights the necessary requirements and to assist operators in successfully implementing electronic instructions for use while addressing associated challenges. Furthermore, it’s essential to recognize that Regulation 207/2012 remains applicable to legacy equipment until the end of validity of certificates issued under Directives 90/385/EEC and 93/42/EEC. By , the is set to improve both adherence and participant engagement, ultimately resulting in .
Recent Updates in eIFU Regulations: What Manufacturers Need to Know
Recent updates to , particularly through the eifu 2021/2226, have introduced significant new requirements that producers must prioritize. These regulations require that be available in various languages, addressing a diverse audience. Additionally, producers must guarantee compatibility among different electronic devices, such as smartphones and tablets, to improve accessibility for individuals.
A critical aspect of the eifu is the necessity for , which can be effectively implemented by incorporating QR codes on product packaging. According to the cumulative success rates of , which are up to 98%, adherence to these updates is imperative, as failure to comply could result in non-conformance penalties and pose risks to . Therefore, it is crucial that manufacturers create that include these new requirements to uphold eifu and ensure the well-being of individuals.
As noted by Ana Criado, our Director of Regulatory Affairs and an expert in health economics and cannabis regulation, who has held various executive roles at Colombia’s INVIMA and has served as a professor at Universidad Javeriana and Universidad de los Andes:
- ‘The implications of extend beyond mere compliance; they are pivotal in safeguarding and enhancing user experience.’
Her extensive experience in Regulatory Affairs provides critical insights into the necessity of these updates. Additionally, the case study titled ‘Building a Partner Ecosystem to Beat Opioid Withdrawal’ illustrates how collaboration can lead to innovative compliance strategies, further emphasizing the importance of working together to meet regulatory demands.
eIFU Applications Across Different Medical Device Categories
have proven to be versatile tools applicable across a variety of , including software, diagnostic tools, and traditional . For in vitro diagnostics, are mandated to provide , handling procedures, and interpretations of test results. In software applications, can enhance the experience of individuals by incorporating interactive tutorials and troubleshooting guides, which facilitate easier navigation and understanding.
When discussing implantable devices, it is crucial for to include thorough to ensure patient safety. Grasping the specific requirements for each category is crucial for manufacturers, as it allows them to not only meet compliance standards but also greatly enhance engagement and satisfaction. Recent data indicates , with an average rating of 4.5 out of 5, reflecting their effectiveness in enhancing usability and compliance.
As Christopher Seib noted, “Not sure if a comma would really change the meaning – if it was there after ‘addition’ and before ‘to’, then I would completely agree with you,” highlighting the nuances in regulatory language that can impact user interpretation. With the recent implementation of the , which emphasizes the need for , the proper integration of the eifu medical device regulation guidelines will be key to maintaining safety and efficacy across . Ana Criado, Director of Regulatory Affairs and CEO of Mahu Pharma, has been instrumental in shaping regulatory practices in Colombia.
Her extensive experience in biomedical engineering and health economics has influenced her contributions to the creation of that adhere to new regulations, ensuring that producers can navigate the evolving landscape effectively. As manufacturers adapt to these changes, insights from experts like Ana will be crucial for advancing the standards of in Colombia.
The Future of eIFU Regulations: Trends and Predictions
The evolution of eifu medical device regulation is set to closely align with advancements in , particularly in telehealth and remote patient monitoring. As emphasized by Dr. Tess Skyrme, Senior Technology Analyst at IDTechEx, the incorporation of digital health can considerably improve the efficiency and accessibility of electronic instructions for use. Notably, , underscoring the anticipated impact of these technologies.
Manufacturers must prepare to adapt their electronic instructions for use to include tailored to individuals’ needs. An example of this integration can be seen in the case study of Sensor-Based COAst, where digital measures are used for remote patient assessments, validated to monitor meaningful aspects of patient health. Furthermore, to encompass advanced features, such as AI-driven support tools, which can offer users immediate assistance and information.
TATEEDA Global’s observation of healthcare IT market trends emphasizes the need for producers to align their electronic instructions for use with industry demands. To stay ahead of these impending changes, manufacturers should invest in adaptable digital solutions and engage in proactive communication with regulatory authorities to ensure compliance with the eifu medical device regulation and emerging guidelines. As we look to the future, strategies to integrate eifs with will be crucial in streamlining and enhancing patient outcomes.
Conclusion
The transition to Electronic Instructions for Use (eIFUs) represents a critical advancement in the medical device sector, driven by the rigorous frameworks of the Medical Device Regulation (MDR) and In Vitro Diagnostic Regulation (IVDR). By mandating clear, accessible, and user-friendly eIFUs, regulators aim to enhance both user experience and patient safety. The article has outlined the essential requirements for compliance, emphasizing the importance of accurate and comprehensive information that can be easily accessed by healthcare professionals and patients alike.
The benefits of eIFUs extend beyond mere compliance; they significantly reduce printing costs, allow for timely updates, and foster interactive user experiences, ultimately leading to improved patient care. As manufacturers adapt to recent regulatory updates, including those introduced by Regulation 2021/2226, they must ensure compatibility across various devices and consider the diverse needs of their user base. This adaptability is not only necessary for compliance but also vital for safeguarding patient safety and enhancing the overall efficacy of medical devices.
Looking ahead, the integration of eIFUs with digital health technologies presents exciting opportunities for further improving healthcare delivery. As the landscape continues to evolve, manufacturers are encouraged to invest in innovative digital solutions that align with emerging trends and regulatory expectations. By embracing these changes, the medical device industry can enhance compliance, user engagement, and ultimately, patient outcomes in a rapidly advancing healthcare environment.
Frequently Asked Questions
What regulations govern electronic Instructions for Use (eIFU) for medical devices?
Electronic Instructions for Use are governed by the eIFU medical device regulation as outlined in the Medical Device Regulation (MDR) 2017/745 and the In Vitro Diagnostic Regulation (IVDR) 2017/746.
What are the requirements for electronic Instructions for Use according to the regulations?
The regulations require that producers offer clear, accessible, and compliant electronic instructions for use, which must be easily downloadable and contain essential information regarding the equipment’s functionality, installation, and maintenance.
Why is compliance with the eIFU medical device regulation important?
Compliance reflects a manufacturer’s commitment to legal obligations and patient safety in medical device usage, and it is particularly important for managing costs and administrative burdens, especially for small and medium-sized enterprises.
What statistics highlight the need for secure management of electronic instructions for use?
Recent statistics indicate that 53% of businesses leave 1,000 or more files containing sensitive information accessible to all employees, underscoring the necessity of secure management.
What trend is emerging among risk and compliance professionals regarding cybersecurity?
Insights reveal that 60% of risk and compliance professionals intend to incorporate cybersecurity into their training agendas over the next two to three years, emphasizing the growing focus on secure management of electronic instructions for use.
How should electronic Instructions for Use be made accessible to users?
To ensure unconditional access, electronic instructions for use should be available on the manufacturer’s website in an official language determined by the Member State in which the device is made available.
What are the advantages of adopting electronic Instructions for Use?
Advantages include significant reductions in printing and distribution costs, the capacity for timely updates, and enhanced accessibility across healthcare environments.
What interactive features do electronic Instructions for Use support?
Electronic Instructions for Use can support interactive features such as videos, animations, and hyperlinks, which enhance the user experience.
What case study is mentioned regarding the implementation of electronic Instructions for Use?
A case study involving IFUcare, Qarad’s eIFU software service, highlights necessary requirements and solutions to assist operators in successfully implementing electronic instructions for use.
What regulation remains applicable to legacy equipment?
Regulation 207/2012 remains applicable to legacy equipment until the end of validity of certificates issued under Directives 90/385/EEC and 93/42/EEC.
List of Sources
- Overview of eIFU Regulations in Medical Devices
- casusconsulting.com (https://casusconsulting.com/eu-commission-survey-on-electronic-ifus-for-all-professional-use-medical-devices-mdr)
- 100+ Compliance Statistics You Should Know in 2025 (https://sprinto.com/blog/compliance-statistics)
- 24 Stats Every Chief Compliance Officer Should Know in 2024 (https://complianceandrisks.com/blog/24-stats-every-chief-compliance-officer-should-know-in-2024)
- 130+ Compliance Statistics & Trends to Know for 2026 (https://secureframe.com/blog/compliance-statistics)
- Key Requirements and Benefits of Electronic Instructions for Use
- makrocare.com (https://makrocare.com/blog/eifu-for-medical-devices-country-specific-regulations-and-benefits)
- orielstat.com (https://orielstat.com/blog/eu-mdr-eifu)
- ifucare.io (https://ifucare.io/blog/ifucare-integrates-eus-implementing-regulation-2021-2226-on-eifu-for-medical-devices)
- ifucare.io (https://ifucare.io/blog/eifu-versus-paper-based-ifu-what-are-the-benefits-of-going-digital-for-manufacturers)
- Recent Updates in eIFU Regulations: What Manufacturers Need to Know
- mdpi.com (https://mdpi.com/2079-6412/15/1/109)
- researchgate.net (https://researchgate.net/publication/250380548_The_scientific_assessment_of_the_impact_of_housing_on_animal_welfare_A_critical_review)
- greenlight.guru (https://greenlight.guru/site-map)
- eIFU Applications Across Different Medical Device Categories
- blog.johner-institute.com (https://blog.johner-institute.com/regulatory-affairs/electronic-instructions-for-use-medical-devices-207-2012)
- blog.johner-institute.com (https://blog.johner-institute.com/regulatory-affairs/instructions-for-use)
- casusconsulting.com (https://casusconsulting.com/eu-commission-survey-on-electronic-ifus-for-all-professional-use-medical-devices-mdr)
- FAQs about Investigational Device Exemption (https://fda.gov/medical-devices/investigational-device-exemption-ide/faqs-about-investigational-device-exemption)
- orielstat.com (https://orielstat.com/blog/eu-mdr-eifu)
- The Future of eIFU Regulations: Trends and Predictions
- statista.com (https://statista.com/topics/2409/digital-health)
- Digital Health Trends 2024 (https://iqvia.com/insights/the-iqvia-institute/reports-and-publications/reports/digital-health-trends-2024)
- edge-ai-vision.com (https://edge-ai-vision.com/2024/01/digital-health-and-artificial-intelligence-2024-2034-trends-opportunities-and-outlook)
- tateeda.com (https://tateeda.com/blog/healthcare-technology-statistics-and-market-share)

Leave a Reply