Introduction
Understanding the clinical trial process is crucial for anyone considering volunteering in medical research. With an increasing number of studies seeking participants, the opportunity to contribute to groundbreaking advancements in healthcare has never been more significant. However, navigating the complexities of clinical trials can be daunting, leaving potential volunteers with numerous questions and concerns. What essential insights can empower individuals to make informed decisions about their participation and enhance their overall experience?
bioaccess®: Accelerate Your Clinical Trial Experience with Expert Guidance
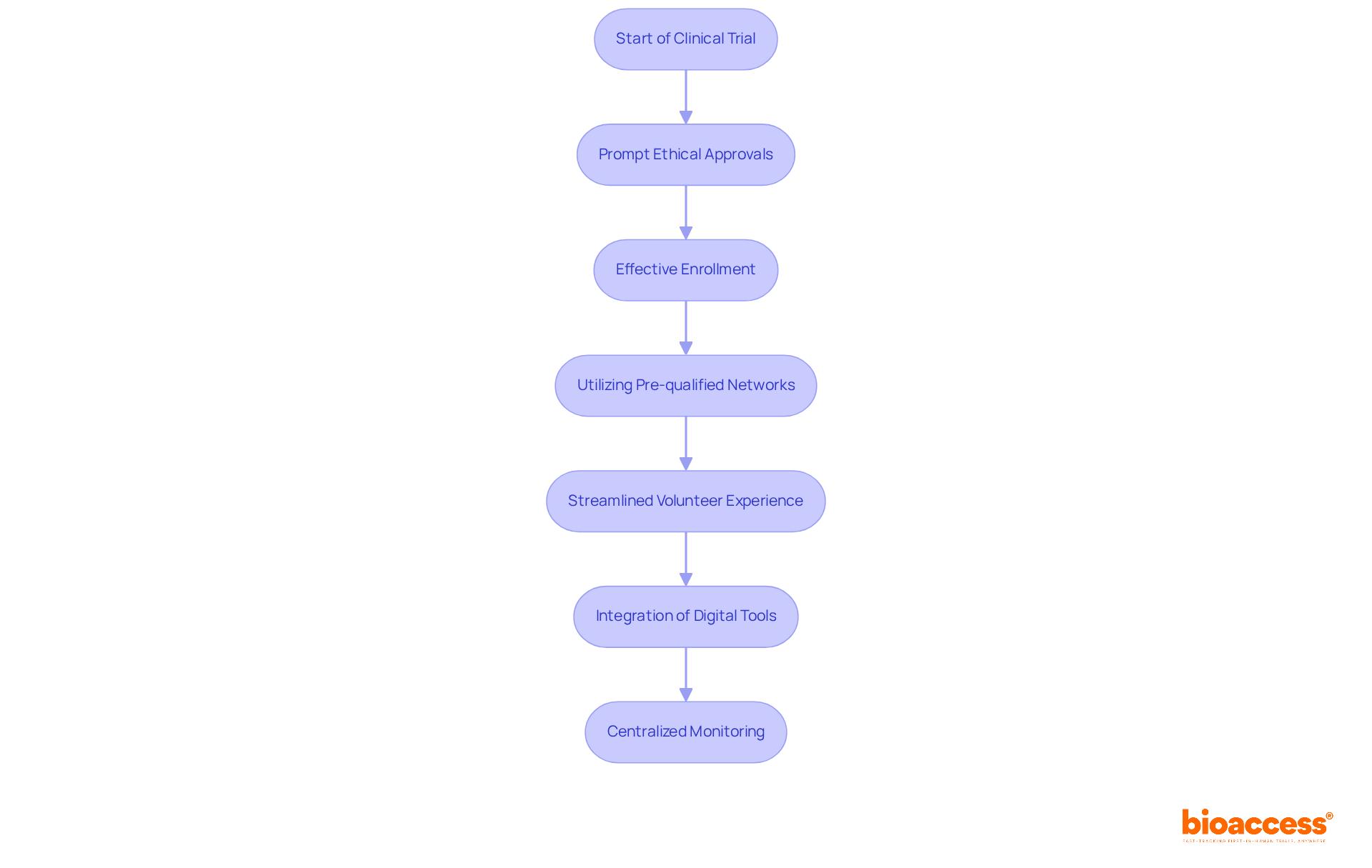
At bioaccess®, we excel in enhancing the , ensuring that volunteers receive exceptional guidance throughout their journey. With over 15 years of experience in , our team is adept at facilitating , leveraging our pre-qualified networks of more than 50 activated sites in under eight weeks.
Volunteers who serve as in studies managed by bioaccess® can expect a streamlined and organized experience, supported by a dedicated team committed to advancing responsibly and ethically. Recent advancements in study management, including the integration of digital tools and patient-focused designs, further enhance the recruitment process, making participation more accessible and engaging.
Notably, around , underscoring the significance of our role in improving this process. Our and centralized monitoring capabilities ensure that our expert guidance not only enhances the overall study experience but also significantly impacts outcomes, fostering a collaborative environment where volunteers feel valued and informed.
As Samruddhi Yardi aptly states, ‘research studies are the cornerstone of medical advancement,’ highlighting the essential nature of our efforts in this area.
Learn the Clinical Trial Process: Key Steps Every Volunteer Should Know
Learn the :
Understanding the is essential for volunteers. Key steps include:
- Screening: Assessing eligibility based on specific criteria, which is crucial as many patients lack knowledge about the recruitment process.
- : Reviewing and signing documents that describe the project’s purpose, procedures, risks, and benefits. This step is vital, as 93.6% of patients with chronic conditions want assurance they can complete the trial. Dr. Neal Thomas emphasizes, “The participant will have a , what the potential risks are, and what the potential benefits are.”
- Randomization: Assigning participants to various research groups, if relevant, which helps ensure impartial results.
- : Receiving the intervention or placebo as part of the research, allowing researchers to understand how therapies function in healthy individuals.
- Follow-Up: Attending scheduled visits for monitoring and data collection, which is and ensuring accurate data. Notably, 70% of the population lives two hours or more from an academic medical center, highlighting the many face in participating.
To enhance your experience as a clinical study volunteer, it is important to familiarize yourself with these steps and consider managing your consent preferences effectively. Understanding your can empower you throughout the trial process. Furthermore, if you have any worries regarding your involvement, feel free to contact the coordinators for clarification.
. Follow the arrows to understand how each step leads to the next, ensuring you know what to expect as a volunteer. Each box represents a step in the clinical trial process. Follow the arrows to understand how each step leads to the next, ensuring you know what to expect as a volunteer.](https://images.tely.ai/telyai/zcjqzwwr-each-box-represents-a-step-in-the-clinical-trial-process-follow-the-arrows-to-understand-how-each-step-leads-to-the-next-ensuring-you-know-what-to-expect-as-a-volunteer.webp)
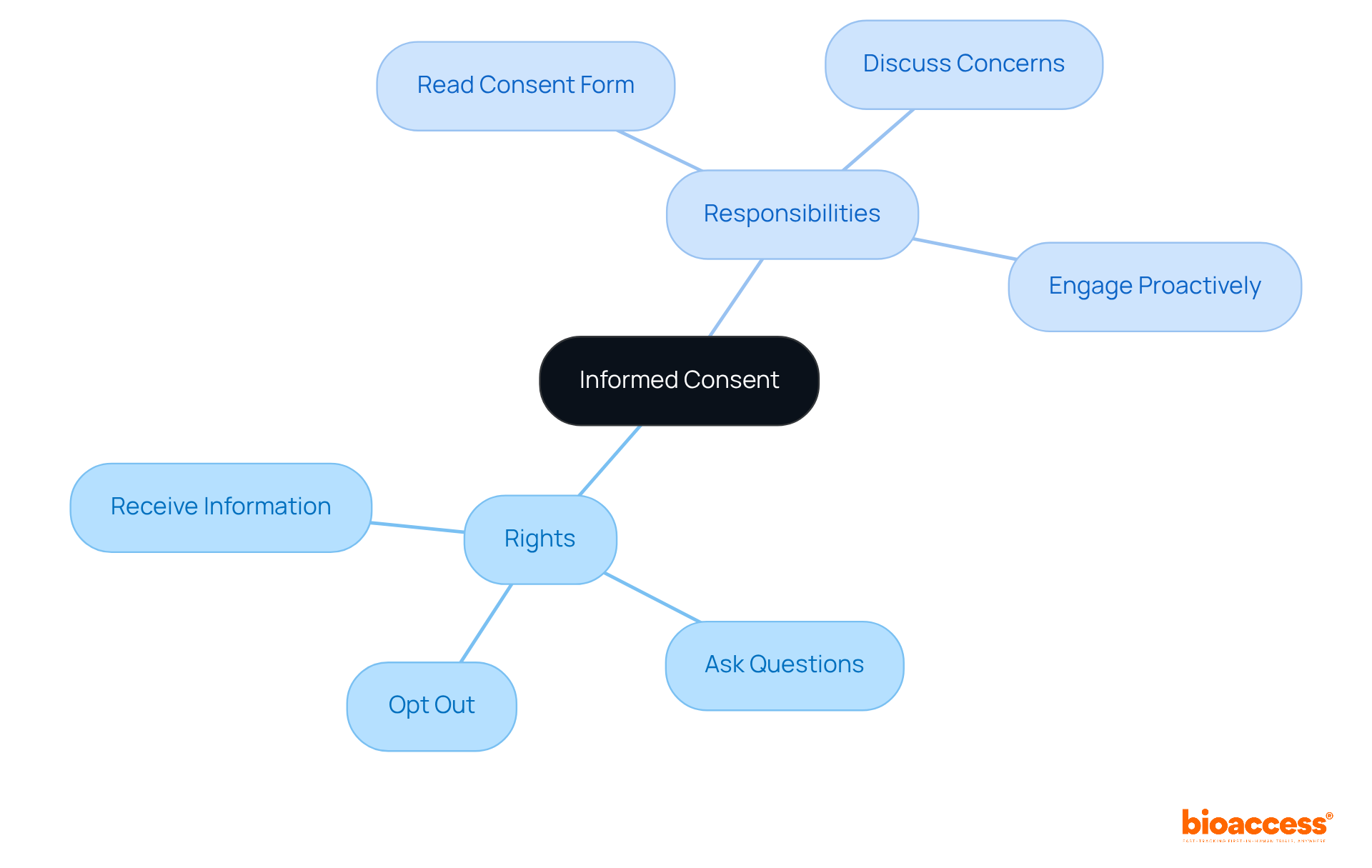
Understand Informed Consent: Your Rights and Responsibilities as a Volunteer
is an essential process that ensures volunteers are comprehensively in . As a volunteer, you possess the right to receive , procedures, risks, and benefits. You are encouraged to and seek clarification on any aspect of the research. Importantly, you can at any moment without facing any penalties.
It is your responsibility to thoroughly read the consent form and discuss any concerns with the research team. This but also fosters a . Effective practices, including the use of , can significantly enhance your understanding and comfort level with the research. By being well-informed, you contribute to the integrity of the research process and ensure that your involvement aligns with your values and preferences.
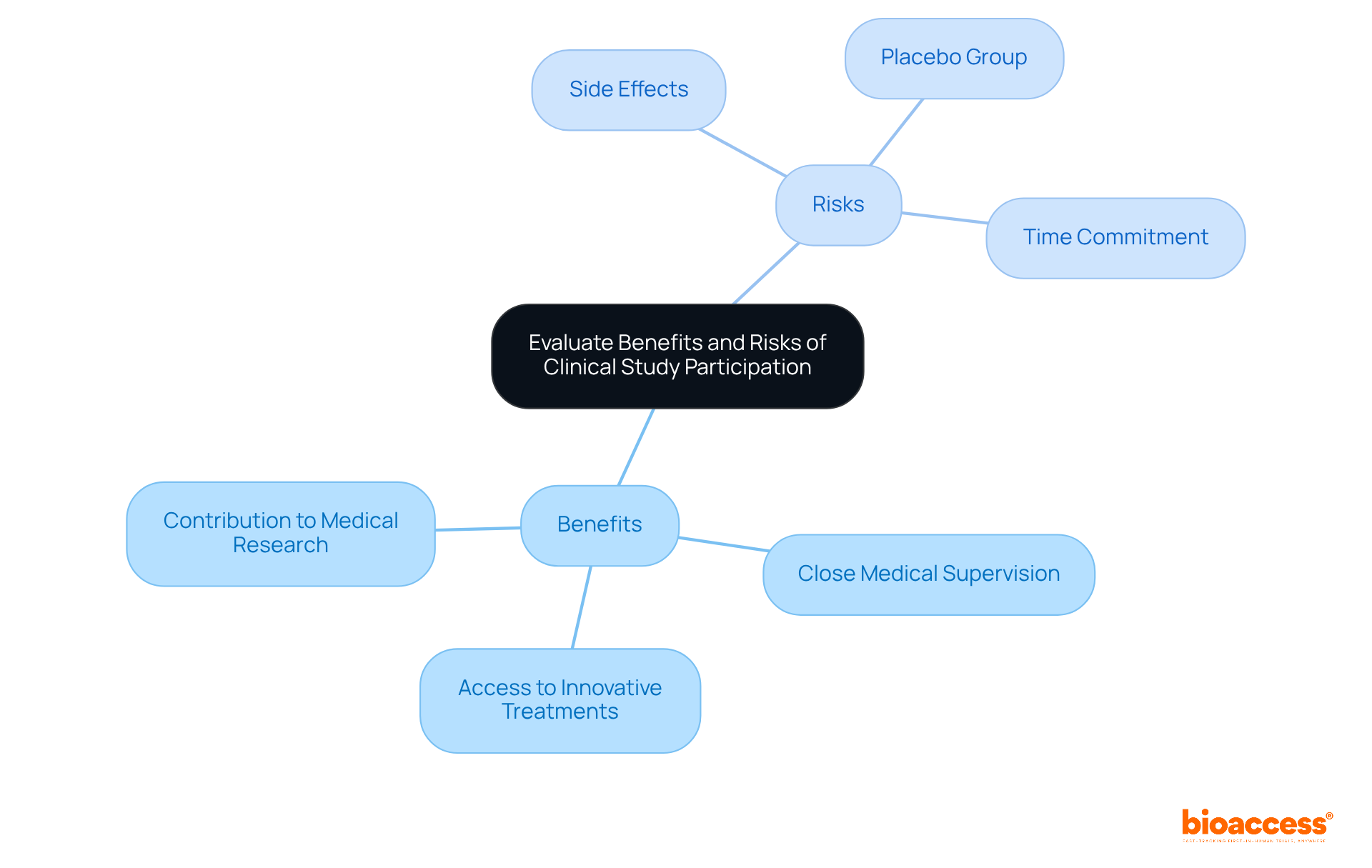
Evaluate Benefits and Risks: What You Need to Consider Before Joining a Study
Before a participates in a , it is essential to evaluate both the potential advantages and disadvantages involved. Benefits for a often include:
- Close medical supervision
- The opportunity to contribute to
For instance, involvement in (RCTs) has been linked to , as research indicates that RCT participants frequently experience higher survival rates than non-participants.
However, the must not be overlooked. Participants may encounter:
- Side effects from experimental therapies
- The possibility of being assigned to a
- The significant required for research protocols
Statistics reveal that logistical barriers considerably impact involvement rates, with many potential volunteers living more than two hours from study centers. Moreover, the necessitates that individuals fully understand what participation entails.
Carefully evaluating these factors will empower you to make an informed decision that aligns with your health goals and personal circumstances. As emphasized by medical researchers, a comprehensive understanding of the risks and benefits is crucial for potential s, ensuring they are well-prepared for the journey ahead.
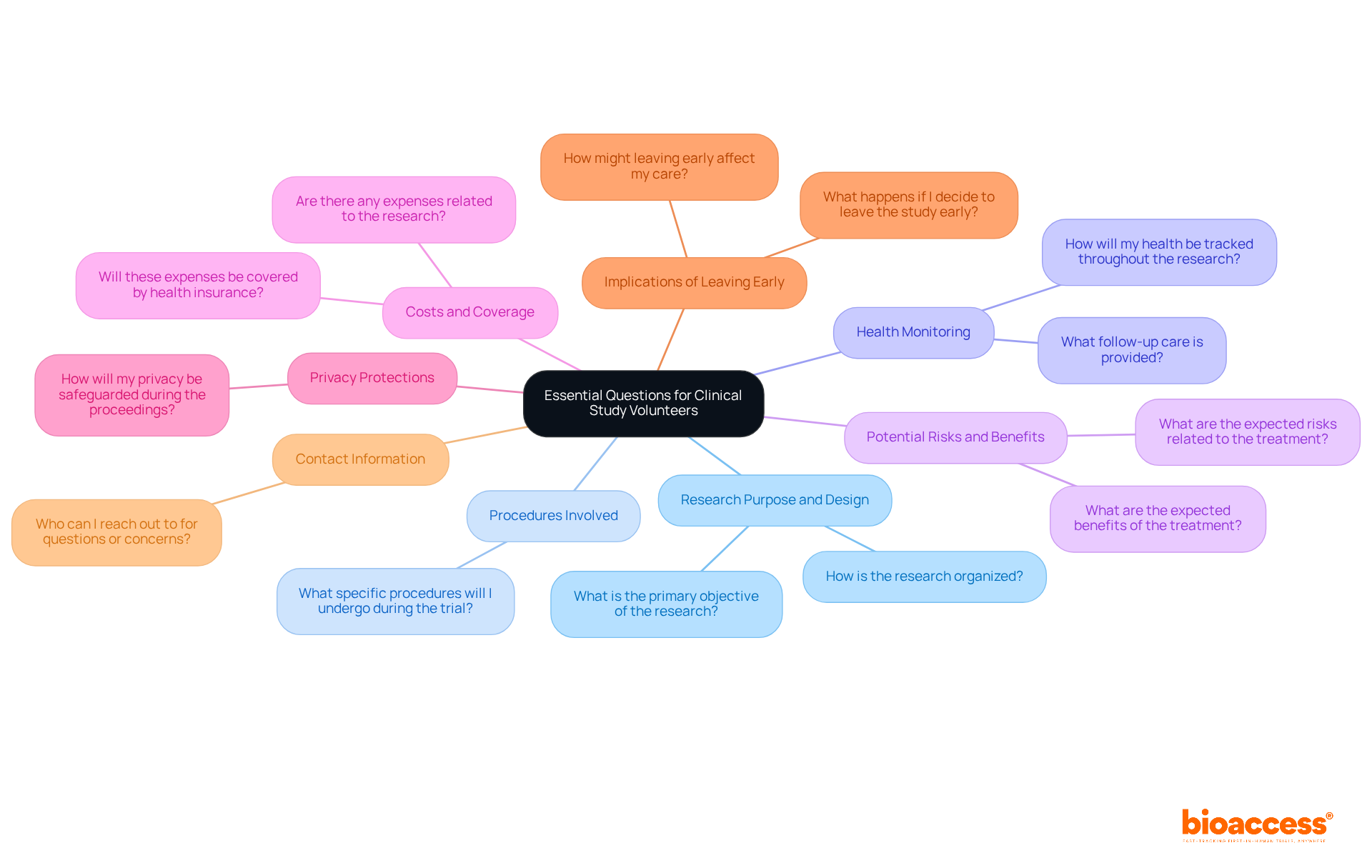
Ask the Right Questions: Essential Inquiries for Clinical Study Volunteers
As a , asking the right questions is crucial for understanding your role and the trial’s implications. Consider inquiring about the following key aspects:
- : What is the primary objective of the research, and how is it organized?
- : What specific procedures will I undergo during the trial?
- : How will my health be tracked throughout the research, and what follow-up care is provided?
- : What are the expected risks and advantages related to the treatment drug or therapy?
- : Are there any expenses related to the research, including tests, procedures, or research drugs, and will these be covered by health insurance?
- : How will my privacy and personal information be safeguarded during the proceedings?
- Implications of Leaving Early: What happens if I decide to leave the study early, and how might that affect my care?
- : Who can I reach out to for questions or concerns during the testing period?
These questions not only clarify your responsibilities as a but also help you make about your involvement. Participating in open discussion with the research team can greatly improve your comprehension and ease, ultimately aiding in the study’s success.
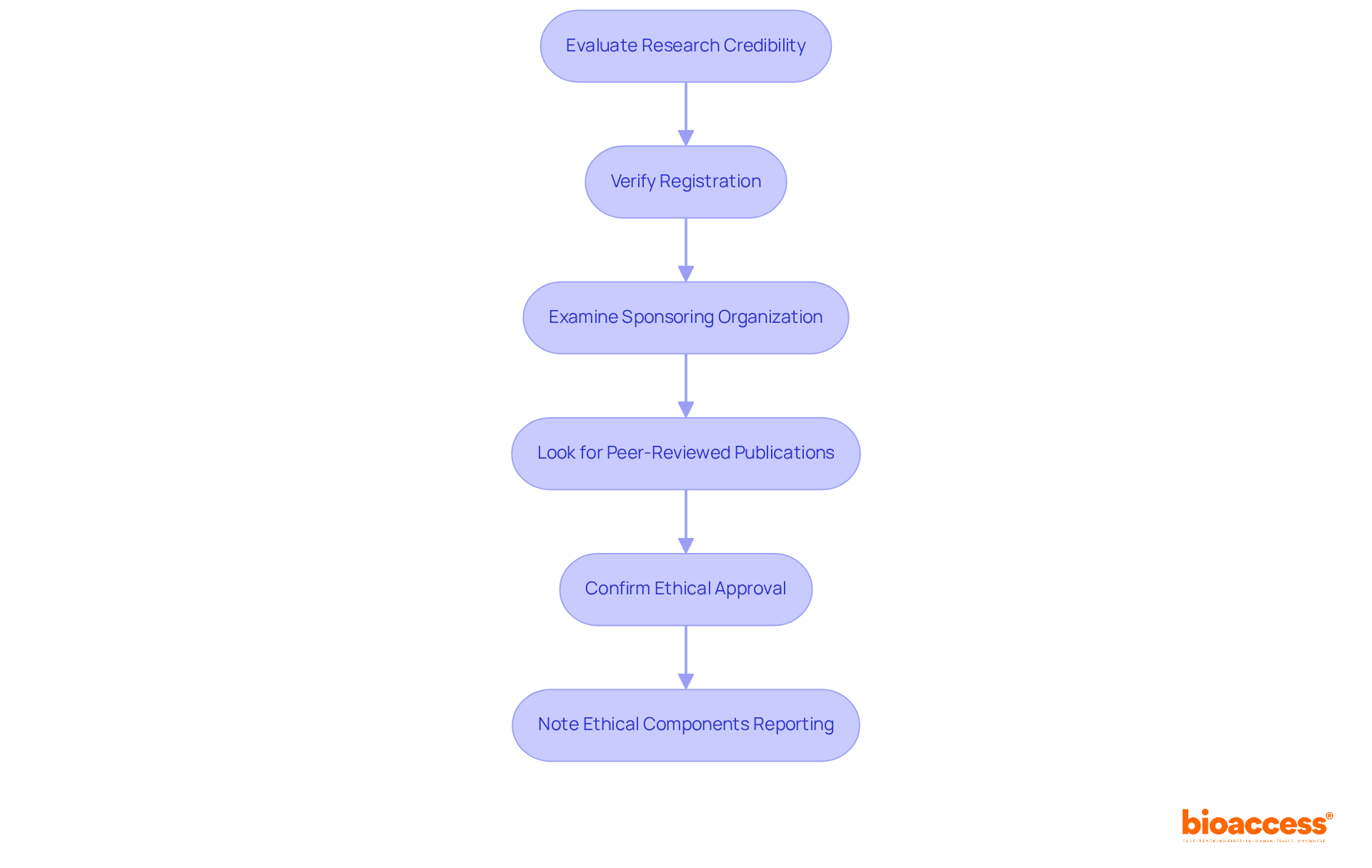
Research Trial Credibility: How to Identify Reputable Clinical Studies
To ensure involvement in a trustworthy research initiative, consider the following steps:
- Verify the registration of the research on , such as ClinicalTrials.gov, which currently lists over 551,947 projects worldwide.
- Examine the reputation and history of the sponsoring organization; established entities are more likely to adhere to ethical standards and provide comprehensive , including feasibility assessments, site selection, compliance reviews, and trial setup.
- Look for related to the research, as these indicate a level of scrutiny and validation by the scientific community.
- Confirm that the research has received from an , which protects participant rights and ensures research integrity.
- Note that only 5.4% of studies report all three (IRB approval, Declaration of Helsinki, and informed consent), highlighting .
By conducting this thorough research, including understanding the , you can confidently evaluate the legitimacy of the study and its potential contributions to medical knowledge.
Communicate Effectively: Building a Relationship with Your Clinical Research Team
Establishing a strong relationship with your is paramount for a fulfilling . regarding your , concerns, and feedback throughout the study cultivates a . Regular updates and check-ins from the research team not only keep you informed about the study’s progress but also reinforce your role as a valued participant.
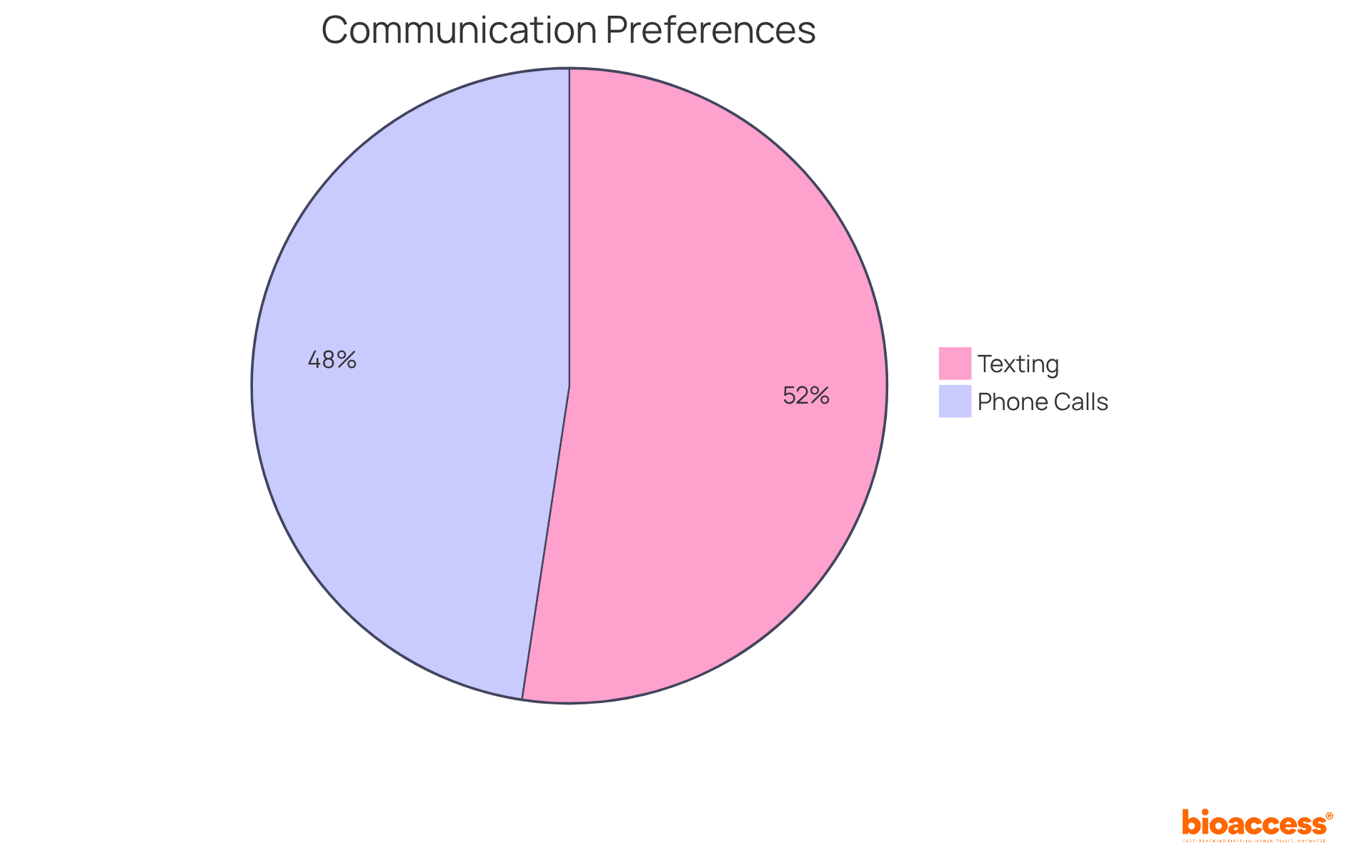
Research indicates that patients who feel connected to their are more likely to adhere to protocols and accurately report their symptoms. For example:
- 80% of participants prefer texting for communication, which can .
- 72.7% of participants favored phone calls when communicating with healthcare providers, underscoring the significance of .
Your perspectives are vital to the project’s success; thus, a can significantly enhance both your experience and the outcomes.
Consider Future Medical Care: How Participation May Affect Your Health Options
Becoming a clinical study volunteer can profoundly impact your future medical treatment. It is crucial to evaluate whether the research offers or .
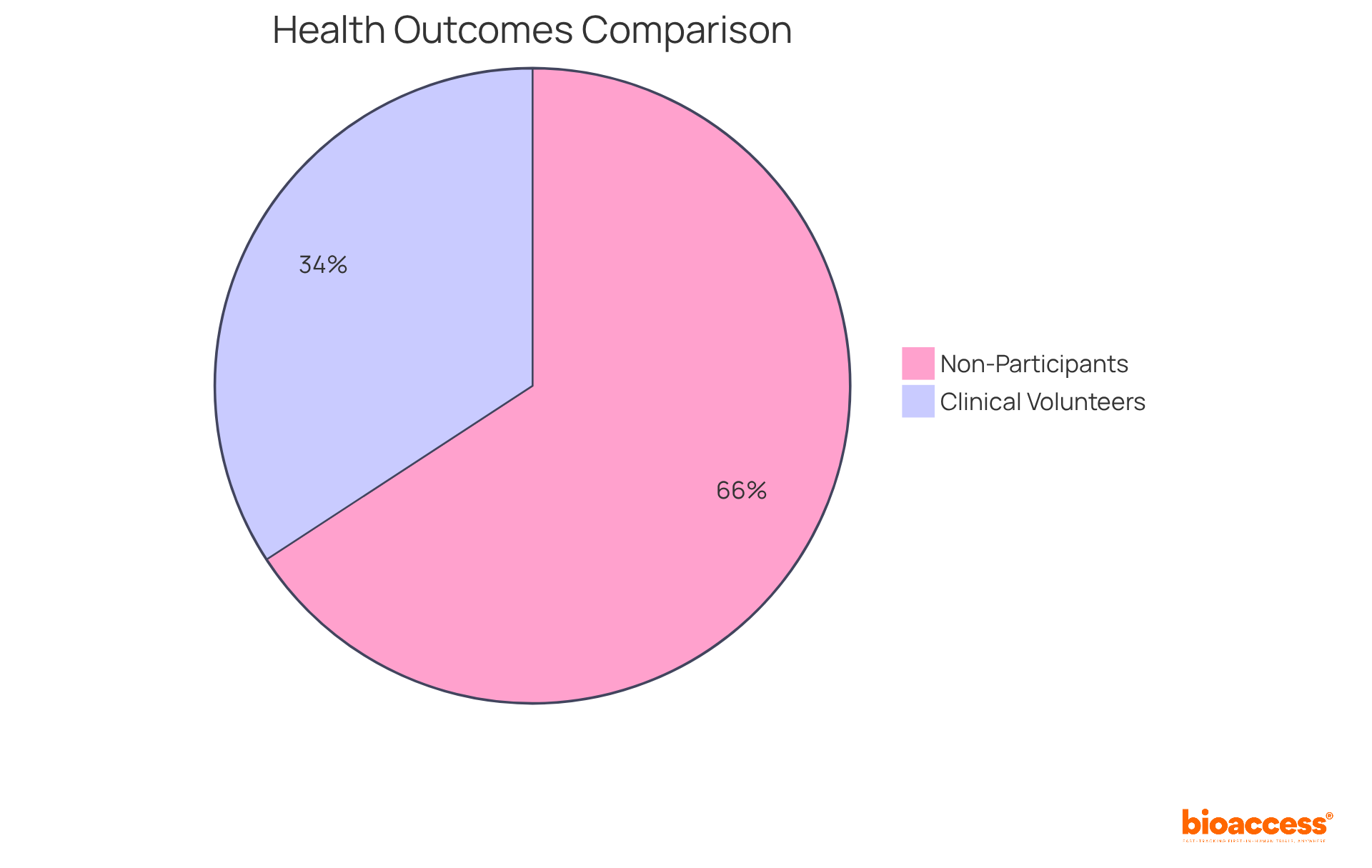
Studies indicate that involved in (RCTs) often experience compared to non-participants. Investigations reveal that:
- 50% of non-participants faced mortality or cancer recurrence
- Only 26% of involved in RCTs experienced similar outcomes
is vital, as it may influence your treatment options moving forward. By proactively addressing these considerations, you empower yourself to , ultimately enhancing your overall care experience.
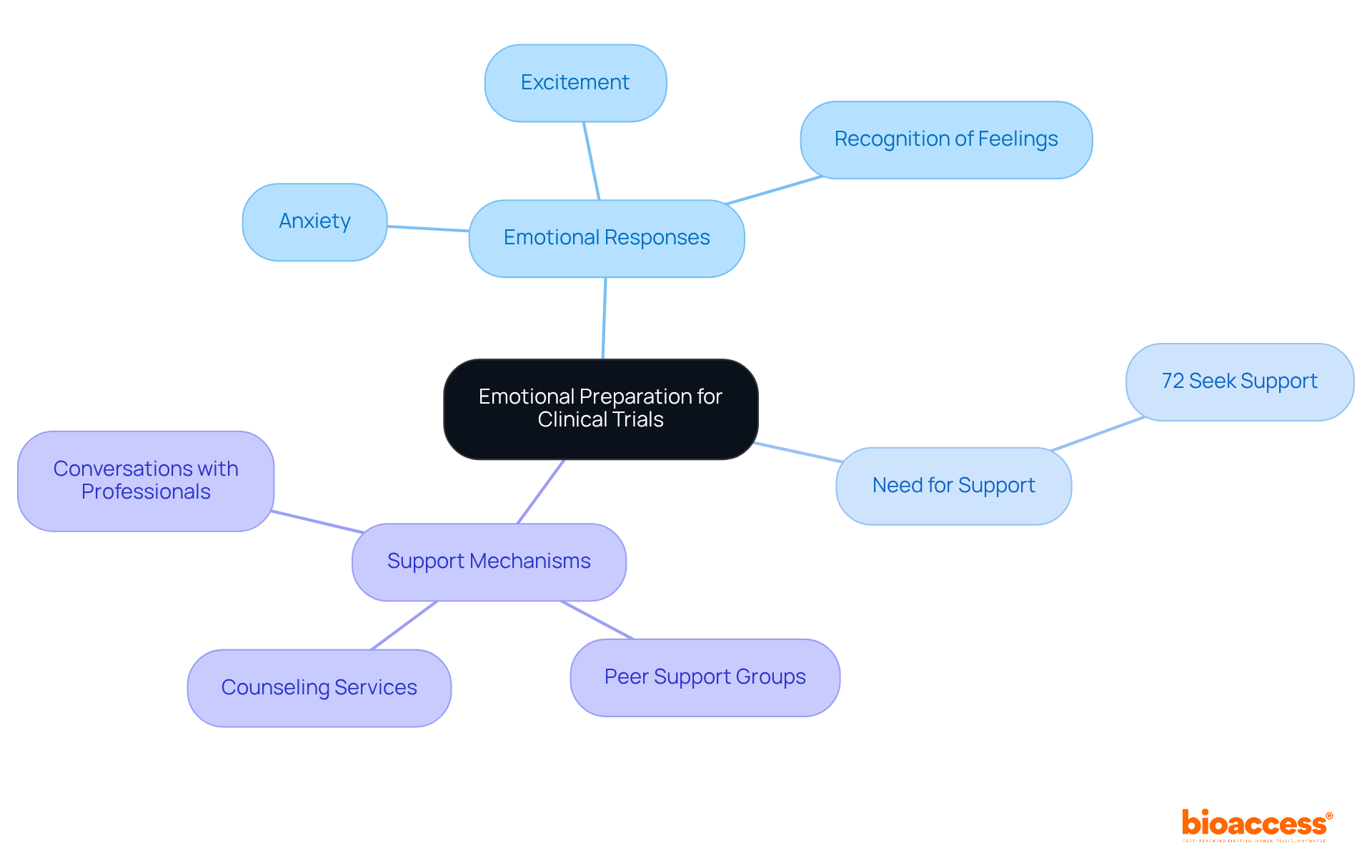
Prepare Emotionally: Understanding the Psychological Impact of Clinical Trials
Being a can evoke a spectrum of emotions, ranging from excitement to anxiety. Notably, research indicates that approximately 72% of seek during their involvement, underscoring the critical need to address these feelings.
Emotional preparation entails recognizing these responses and actively pursuing support. Engaging in conversations with friends, family, or proves beneficial. Initiatives such as and specifically designed for provide essential emotional assistance.
Mental health experts emphasize that understanding is a fundamental aspect of the experience for a , significantly enhancing their ability to navigate the challenges they may face. As we approach 2025, cultivating will be paramount for the , ensuring they remain supported and informed throughout their journey.
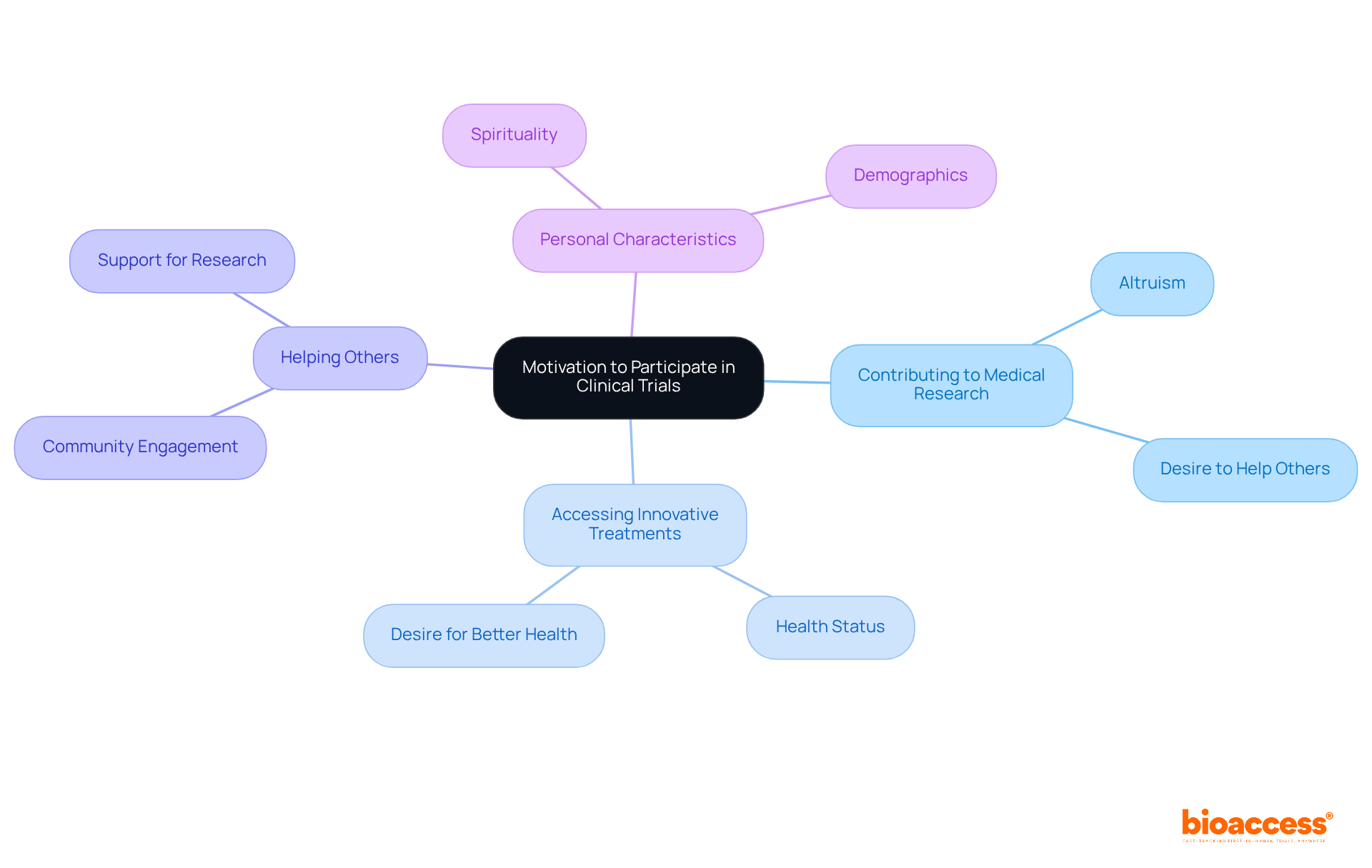
Reflect on Your Motivation: Why You Want to Participate in a Clinical Trial
Before becoming a , it is essential to consider your motivations. Are you driven by a desire to contribute to , gain access to , or assist others in need? Comprehending your motives for involvement not only offers clarity and purpose but also enhances your experience.
Research indicates that a significant percentage of s are motivated by altruism, with many of them expressing a and improve . However, motivations can vary significantly among participants, influenced by personal characteristics such as health status and spirituality.
As one clinical investigator observed, ‘Understanding may assist research teams in enlisting the widest and most representative cohort of patients.’ This self-reflection can guide your interactions with the research team, fostering a more engaged and in the study.
Conclusion
Engaging in clinical studies as a volunteer represents a vital contribution to medical research and innovation. The insights shared throughout this article underscore the importance of understanding the clinical trial process, ensuring informed consent, evaluating risks and benefits, and fostering effective communication with research teams. Each step in this journey is essential for enhancing the experience of volunteers and advancing healthcare outcomes.
Key points discussed emphasize:
- The significance of being well-informed about the clinical trial process.
- The necessity of understanding one’s rights and responsibilities.
- The importance of evaluating the credibility of studies before participation.
Volunteers are encouraged to ask pertinent questions and communicate openly with their research teams, as these actions foster trust and improve overall engagement. Furthermore, reflecting on personal motivations can lead to a more meaningful and fulfilling experience.
Ultimately, participation in clinical trials not only offers the potential for personal health benefits but also plays a crucial role in shaping the future of medical care. By understanding the intricacies of clinical studies and preparing emotionally for the journey, individuals can empower themselves to make informed decisions that align with their health goals. Taking these steps enhances the volunteer experience and contributes to the collective advancement of medical knowledge, underscoring the profound impact that each volunteer can have on the future of healthcare.
Frequently Asked Questions
What is bioaccess® and what services do they provide for clinical trials?
bioaccess® specializes in enhancing the clinical trial process by providing expert guidance to volunteers. With over 15 years of experience, they facilitate prompt ethical approvals and effective enrollment through a network of more than 50 activated sites, ensuring a streamlined experience for participants.
What can volunteers expect when participating in studies managed by bioaccess®?
Volunteers can expect a well-organized experience supported by a dedicated team focused on advancing medical innovation ethically. Recent advancements in study management, including digital tools and patient-focused designs, make participation more accessible and engaging.
What challenges do clinical trials face regarding recruitment?
Approximately 80% of research studies are postponed or terminated due to recruitment challenges, highlighting the importance of bioaccess®’s role in improving this process.
What are the key steps in the clinical trial process that volunteers should know?
Key steps include:
- Screening: Assessing eligibility based on specific criteria.
- Informed Consent: Reviewing and signing documents outlining the trial’s purpose, procedures, risks, and benefits.
- Randomization: Assigning participants to different research groups to ensure unbiased results.
- Treatment: Receiving the intervention or placebo.
- Follow-Up: Attending scheduled visits for monitoring and data collection.
Why is informed consent important for clinical trial volunteers?
Informed consent ensures that volunteers are fully informed about the project, including its purpose, procedures, risks, and benefits. It allows participants to ask questions, seek clarification, and opt out at any time without penalties.
What rights do volunteers have during the informed consent process?
Volunteers have the right to receive detailed information about the study, ask questions, and discuss any concerns with the research team. They can also opt out of the research at any time without facing penalties.
How can volunteers enhance their understanding and comfort level with the research?
Volunteers can enhance their understanding by thoroughly reading the consent form, asking questions, and engaging proactively with the research team. Effective informed consent practices, including clear language and multimedia tools, can also improve comprehension.
List of Sources
- bioaccess®: Accelerate Your Clinical Trial Experience with Expert Guidance
- Clinical Trials Statistics and Facts (2026) (https://media.market.us/clinical-trials-statistics)
- Rebooting the Statistic That 5% of Eligible Patients Participate in Clinical Trials | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/rebooting-the-statistic-that-5-of-eligible-patients-participate-in-clinical-trials)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC11265487)
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- antidote.me (https://antidote.me/blog/medical-research-volunteers-clinical-trial-day-statistics)
- Learn the Clinical Trial Process: Key Steps Every Volunteer Should Know
- How researchers are enlisting volunteers to help shape clinical trials (https://news.vumc.org/2025/02/13/how-researchers-are-enlisting-volunteers-to-help-shape-clinical-trials)
- antidote.me (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- Rebooting the Statistic That 5% of Eligible Patients Participate in Clinical Trials | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/rebooting-the-statistic-that-5-of-eligible-patients-participate-in-clinical-trials)
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- pennstatehealthnews.org (https://pennstatehealthnews.org/2018/09/medical-minute-healthy-volunteers-clinical-trials)
- Understand Informed Consent: Your Rights and Responsibilities as a Volunteer
- generalsurgerynews.com (https://generalsurgerynews.com/In-the-News/Article/04-21/Court-Ruling-Alters-Informed-Consent-Process/63144)
- Informed Consent – StatPearls – NCBI Bookshelf (https://ncbi.nlm.nih.gov/books/NBK430827)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- researchsupport.admin.ox.ac.uk (https://researchsupport.admin.ox.ac.uk/governance/ethics/resources/consent)
- Evaluate Benefits and Risks: What You Need to Consider Before Joining a Study
- Rebooting the Statistic That 5% of Eligible Patients Participate in Clinical Trials | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/rebooting-the-statistic-that-5-of-eligible-patients-participate-in-clinical-trials)
- nia.nih.gov (https://nia.nih.gov/health/clinical-trials-and-studies/clinical-research-benefits-risks-and-safety)
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- Benefits of Participation in Clinical Trials: An Umbrella Review | MDPI (https://mdpi.com/1660-4601/19/22/15368)
- dementiastatistics.org (https://dementiastatistics.org/statistics/clinical-trials)
- Ask the Right Questions: Essential Inquiries for Clinical Study Volunteers
- hhs.gov (https://hhs.gov/ohrp/education-and-outreach/about-research-participation/questions-to-ask)
- antidote.me (https://antidote.me/blog/research-studies-questions)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC3059317)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC2823735)
- Research Trial Credibility: How to Identify Reputable Clinical Studies
- alasdairmunro.substack.com (https://alasdairmunro.substack.com/p/why-bad-research-is-worse-than-no)
- Trends and Charts on Registered Studies | ClinicalTrials.gov (https://clinicaltrials.gov/about-site/trends-charts)
- onlinelibrary.wiley.com (https://onlinelibrary.wiley.com/doi/10.1002/hcs2.113)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC4073550)
- iavi.org (https://iavi.org/iavi-report/the-future-of-antibody-based-hiv-prevention)
- Communicate Effectively: Building a Relationship with Your Clinical Research Team
- Five Medical Communication Strategies Backed by Patient Insights | PPD (https://ppd.com/blog/medical-communication-strategies-patient-insights)
- systematicreviewsjournal.biomedcentral.com (https://systematicreviewsjournal.biomedcentral.com/articles/10.1186/s13643-019-1065-x)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC11789515)
- Improving Communication in Clinical Research – SOCRA Blog (https://socra.org/blog/improving-communication-in-clinical-research)
- Consider Future Medical Care: How Participation May Affect Your Health Options
- niddk.nih.gov (https://niddk.nih.gov/about-niddk/research-areas/obesity/longitudinal-assessment-bariatric-surgery)
- Benefits of Participation in Clinical Trials: An Umbrella Review | MDPI (https://mdpi.com/1660-4601/19/22/15368)
- nap.nationalacademies.org (https://nap.nationalacademies.org/catalog/11900/long-term-health-effects-of-participation-in-project-shad-shipboard-hazard-and-defense)
- ascopubs.org (https://ascopubs.org/doi/10.1200/EDBK_156686)
- link.springer.com (https://link.springer.com/article/10.1007/s00125-023-06048-6)
- Prepare Emotionally: Understanding the Psychological Impact of Clinical Trials
- nature.com (https://nature.com/articles/s41598-023-47840-z)
- journals.plos.org (https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0284268)
- clinicaltrialvanguard.com (https://clinicaltrialvanguard.com/clinicaltrials/reducing-patient-burden-in-clinical-trials-how-to-make-trials-more-feasible-for-patients)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC10124833)
- researchgate.net (https://researchgate.net/publication/5532475_Who_volunteers_for_phase_I_clinical_trials_Influences_of_anxiety_social_anxiety_and_depressive_symptoms_on_self-selection_and_the_reporting_of_adverse_events)
- Reflect on Your Motivation: Why You Want to Participate in a Clinical Trial
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC4870048)
- researchinvolvement.biomedcentral.com (https://researchinvolvement.biomedcentral.com/articles/10.1186/s40900-019-0145-3)
- trialsjournal.biomedcentral.com (https://trialsjournal.biomedcentral.com/articles/10.1186/s13063-021-05818-0)
- healthaffairs.org (https://healthaffairs.org/doi/10.1377/hlthaff.2022.00520)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC8825950)

Leave a Reply