Introduction
Understanding the methodologies of in vivo and in vitro research is crucial for unraveling the complexities of modern science. These two distinct approaches not only shape the landscape of clinical research but also provide unique insights into drug development and biological interactions.
How do researchers navigate the strengths and limitations of each method to optimize their findings and drive medical breakthroughs? This exploration delves into the nuances of in vivo and in vitro studies, illuminating their critical roles in the pursuit of effective therapies.
Define In Vivo and In Vitro
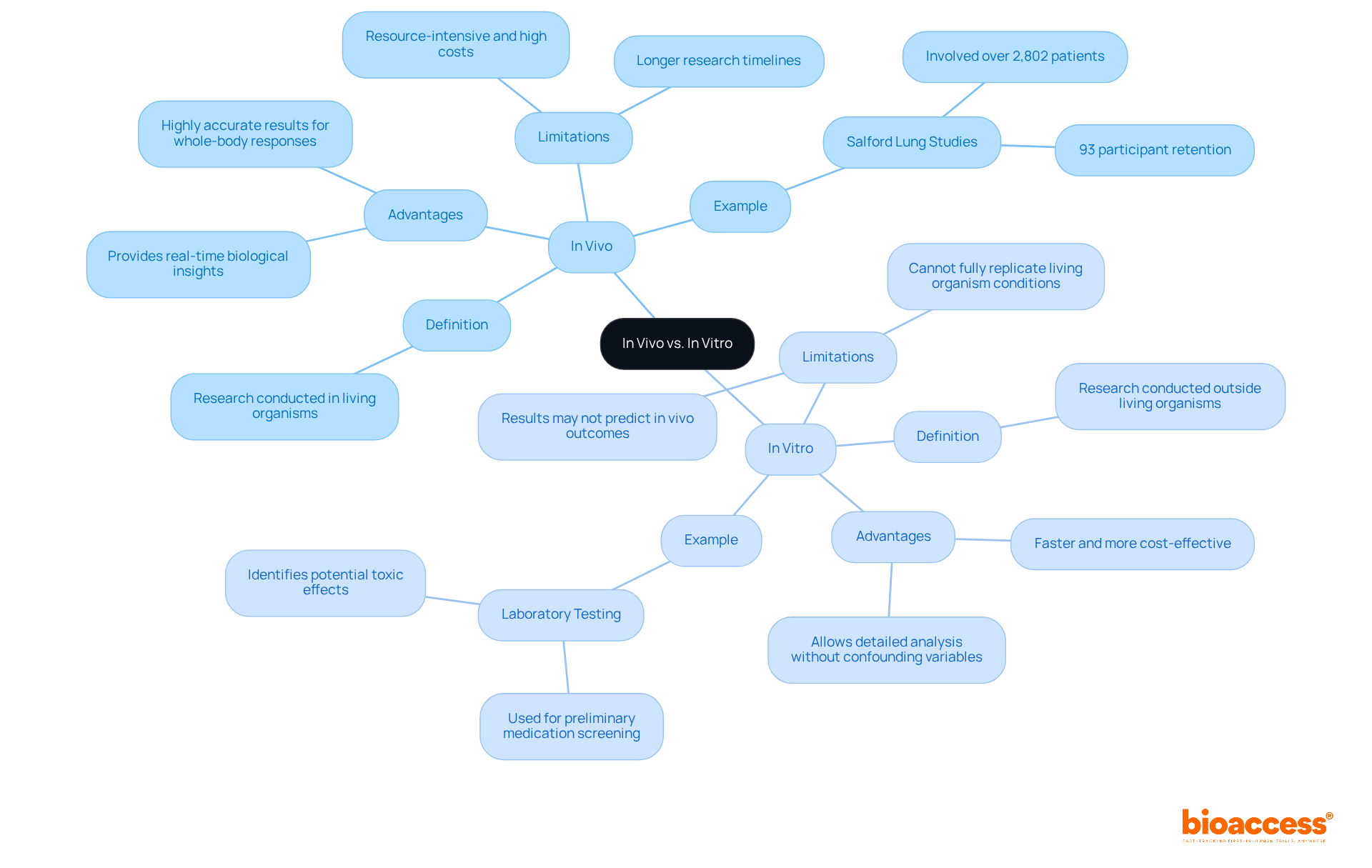
The difference between refers to two distinct methods of scientific investigation, with both playing a crucial role in .
- ‘,’ meaning ‘within the living,’ pertains to research conducted in living entities, such as humans or animals. This method allows researchers to observe biological processes in their natural context, providing invaluable insights into the physiological relevance of their findings. However, in living studies can be resource-intensive, often incurring high costs due to the use of live subjects and requiring longer research timelines. For instance, it is anticipated that around 70% of in 2026 will employ in-body methods, underscoring their essential role in understanding intricate biological interactions. A notable example is the Salford Lung Studies, which involved over 2,802 patients treated by their own GPs, showcasing the effectiveness of .
- Conversely, ” refers to experiments conducted outside of living organisms, typically in controlled laboratory environments using test tubes or petri dishes. Laboratory experiments often involve isolated cells or biological molecules, enabling researchers to manipulate variables in a controlled setting. These investigations are generally faster and more cost-effective, making them ideal for preliminary medication screening and toxicity evaluations. For example, laboratory testing has been pivotal in identifying potential harmful effects of new medications before they advance to .
Understanding the difference between is integral to , as each offers unique advantages and limitations. In living experiments provide comprehensive, real-time information essential for medication development and safety assessment, while controlled environments facilitate detailed cellular examination without the confounding factors present in living beings. Recent advancements, such as organ-on-chip technologies, are enhancing the capabilities of both approaches, leading to more ethical and accurate research outcomes. As Jean-Francois Denault, a professional consultant, notes, ‘Recognizing the strengths and limitations of both models can assist researchers in accelerating breakthroughs in medication development and personalized medicine.
Context and Importance in Clinical Research
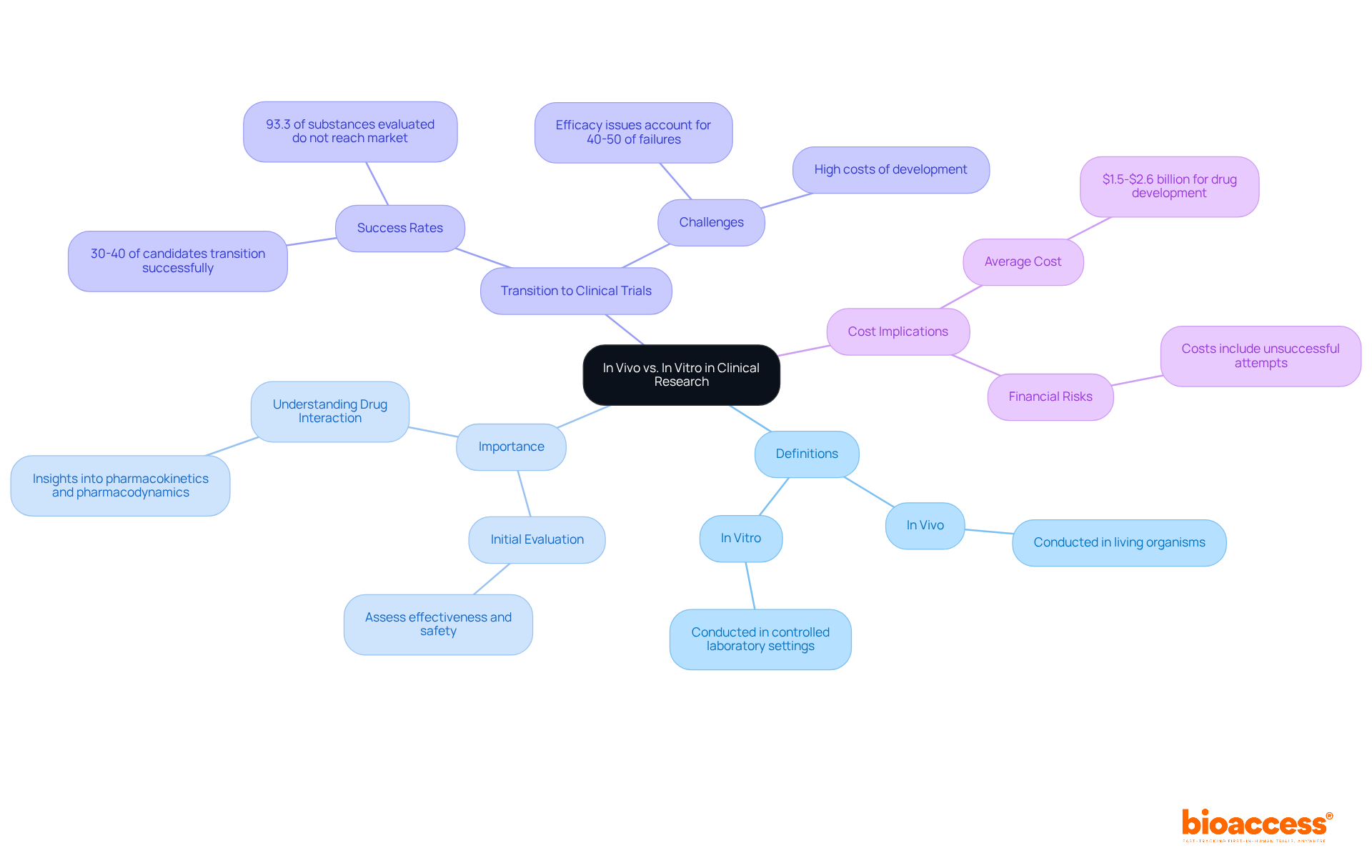
In clinical research, it is essential to recognize the difference between experiments as they are vital components of the . between , as they serve as the initial stage for evaluating new medications and enable researchers to assess the effectiveness and safety of substances in a controlled laboratory setting. This foundational information is crucial for determining which candidates progress to in-life testing, where the of the substance are analyzed within a living organism.
Living organism studies provide a deeper understanding of a medication’s interaction with biological systems, illustrating the difference between , as well as offering insights into . Promising therapeutic candidates, such as specific oncology treatments, underscore the importance of this transition, as they often show potential in laboratory studies before advancing to clinical trials. Notably, the emphasizes the difference between , with only about 30-40% of candidates making this leap. Furthermore, it’s essential to recognize that 93.3% of substances evaluated in humans do not reach the market, highlighting the challenges faced in medication development.
, is estimated at $1.5-$2.6 billion, illustrating the financial implications of rigorous testing protocols. Additionally, , emphasizing the significance of laboratory examinations in identifying promising candidates. At bioaccess, our comprehensive – including feasibility assessments, site selection, compliance reviews, trial setup, import permits, project management, and reporting – are designed to effectively address these challenges. By integrating both methodologies, we enhance the safety and efficacy of new treatments, ultimately leading to improved patient outcomes.
Historical Development of In Vivo and In Vitro
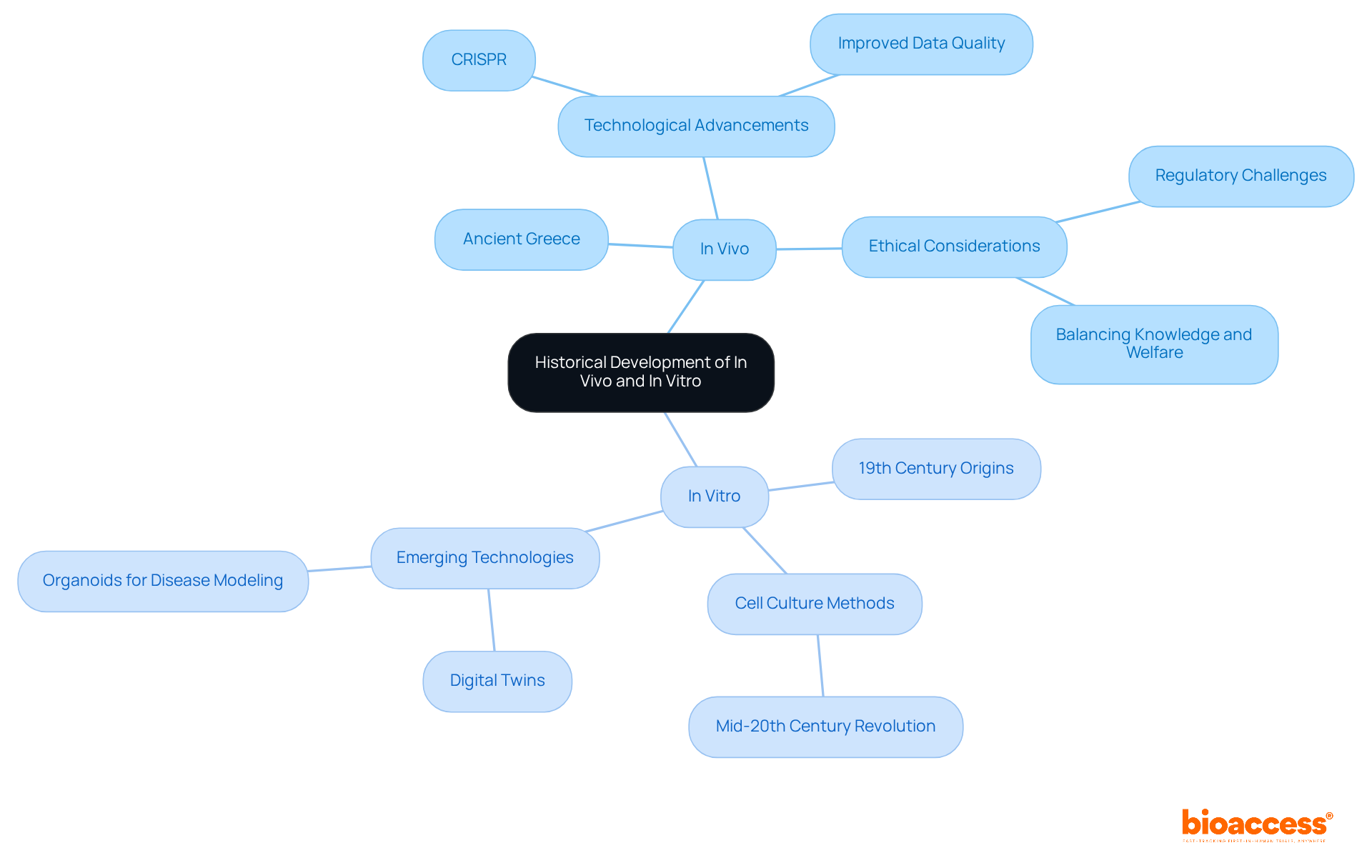
The historical evolution of experimental methods, both within and outside the organism, showcases a rich tapestry of scientific inquiry that spans centuries. trace their roots back to ancient Greece, where early philosophers and physicians meticulously documented the effects of various treatments on living animals. This foundational work laid the groundwork for future research, emphasizing the importance of understanding . However, as regulations governing tighten, scientists now face the challenge of demonstrating that no alternative methodologies can be employed. This balance between the knowledge gained and the surrounding animal welfare is crucial in today’s research landscape.
The phrase ‘in glass’ emerged in the 19th century, marking a pivotal shift as scientists began isolating cells and tissues for experimentation outside of living organisms. This transition was accelerated by the advent of cell culture methods in the mid-20th century, which revolutionized laboratory research. These methods allowed for more advanced studies into cellular behavior and drug interactions, significantly enhancing our understanding of . Emerging technologies, such as CRISPR, are further transforming pre by simplifying complex animal models, making them more affordable and quicker to implement.
Over the decades, the difference between in vivo and has become evident as both have undergone substantial evolution, driven by technological advancements and an increasingly nuanced understanding of . As Sarah Moore observes, experiments conducted in living organisms address the significant limitations of laboratory tests by showcasing the effects of medications on the entire body. This evolution not only enhances the applications of in situ and in vitro research in but also paves the way for more of scientific exploration. The importance of collaboration in this field cannot be overstated, as it fosters innovation and drives progress toward more humane research practices.
Key Characteristics and Differences
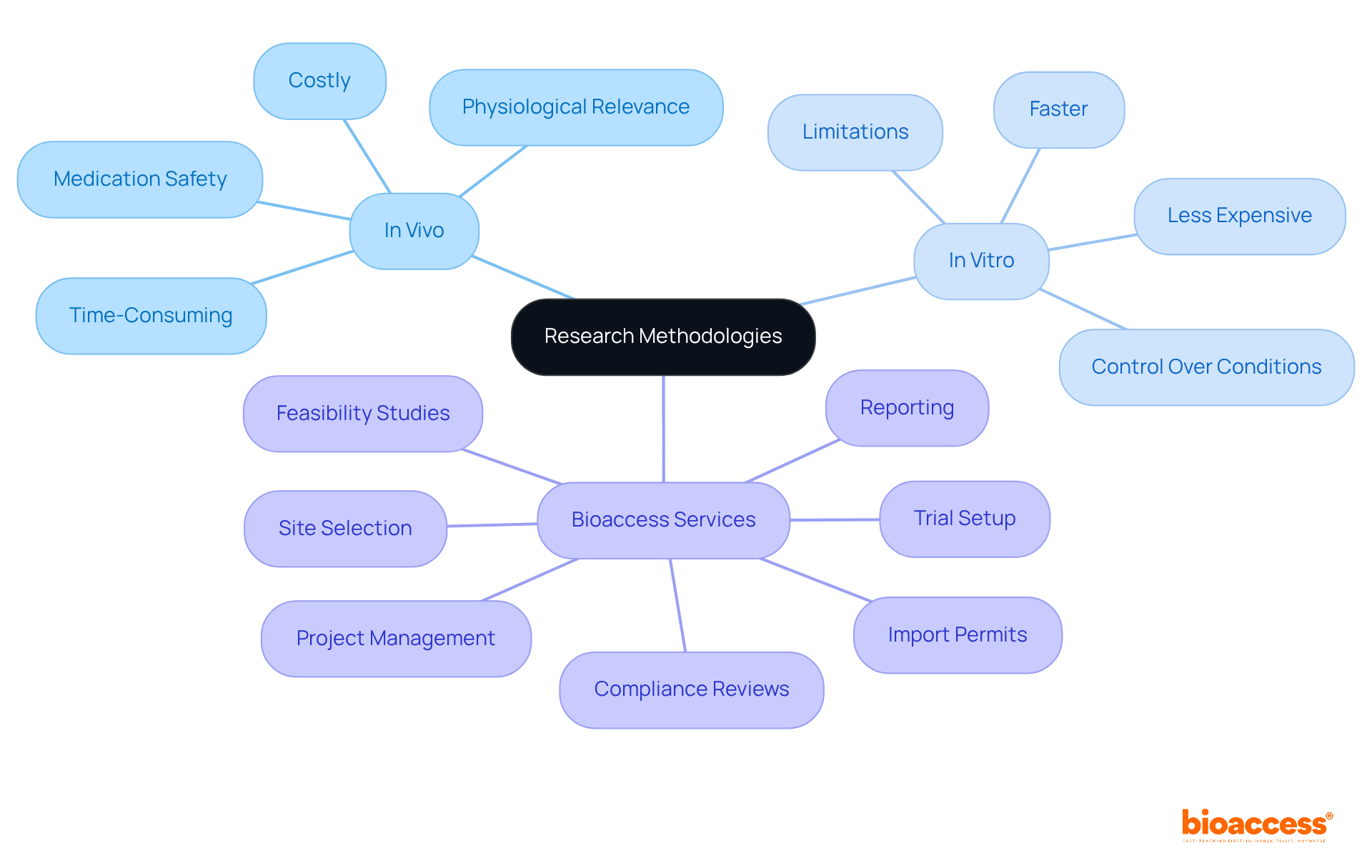
Experiments conducted within living organisms hold significant physiological relevance, offering insights into complex biological interactions that are crucial for human health. These investigations yield information that reflects the intricate interplay of various biological systems. For instance, , as they replicate real-life conditions that laboratory tests often fail to emulate. However, it’s important to note that can be more time-consuming and costly, requiring ethical considerations and regulatory approvals that may extend timelines considerably.
Conversely, , allowing for the isolation of specific variables and high-throughput screening of drug candidates. They are typically faster and less expensive, making them ideal for early-stage research. Yet, a notable limitation of is their inability to fully replicate the complexities of living organisms, which can lead to discrepancies in predicting human responses.
when designing studies and interpreting results. While laboratory experiments can deliver quick insights, are indispensable for validating the physiological significance of those findings. Experts in the field emphasize that both methodologies are necessary for a comprehensive evaluation of the safety and efficacy of experimental drugs, with each approach offering unique strengths to the research process.
At bioaccess, we provide comprehensive , including:
- site selection
- compliance reviews
- trial setup
- import permits
- project management
- reporting
Our services are tailored to tackle the challenges associated with both . By streamlining the research process, we enhance the efficiency and effectiveness of , ultimately contributing to local economies through job creation, economic growth, and healthcare improvement, while fostering international collaboration.
Conclusion
Understanding the distinction between in vivo and in vitro methodologies is crucial for advancing scientific research, especially in clinical applications. These approaches serve unique purposes, enhancing our understanding of biological processes and the development of effective medications. In vivo studies provide valuable insights into how treatments interact with living organisms, while in vitro experiments offer a controlled environment for preliminary testing. Together, they create a comprehensive framework that boosts the efficacy and safety of new therapies.
The article delved into the definitions, advantages, and limitations of both in vivo and in vitro research methods. It underscored the significance of in vivo studies in revealing the physiological relevance of findings, alongside the cost-effectiveness and speed of in vitro experiments for initial drug screening. Furthermore, the historical context and evolution of these methodologies highlight ongoing advancements in research techniques and ethical considerations surrounding animal testing. The integration of both approaches is essential for successfully transitioning drug candidates from laboratory settings to clinical trials.
Reflecting on the importance of these methodologies, it’s evident that a balanced approach leveraging the strengths of both in vivo and in vitro research is vital for the future of medication development. As scientific inquiry evolves, embracing innovations such as organ-on-chip technologies can lead to more ethical and effective research practices. Researchers and stakeholders must recognize the value of both methods in driving breakthroughs in personalized medicine and improving patient outcomes, ultimately fostering a more informed and innovative healthcare landscape.
Frequently Asked Questions
What do the terms in vivo and in vitro refer to in scientific research?
In vivo refers to research conducted in living organisms, such as humans or animals, while in vitro refers to experiments conducted outside of living organisms, typically in controlled laboratory environments using test tubes or petri dishes.
What are the advantages of in vivo research?
In vivo research allows researchers to observe biological processes in their natural context, providing insights into physiological relevance. It is essential for understanding intricate biological interactions and medication development, although it can be resource-intensive and costly.
Can you provide an example of in vivo research?
An example of in vivo research is the Salford Lung Studies, which involved over 2,802 patients treated by their own general practitioners, demonstrating the effectiveness of in vivo methodologies in real-world settings.
What are the advantages of in vitro research?
In vitro research is generally faster and more cost-effective, making it ideal for preliminary medication screening and toxicity evaluations. It allows researchers to manipulate variables in a controlled setting without the confounding factors present in living beings.
How does in vitro research contribute to medication development?
Laboratory testing in vitro has been pivotal in identifying potential harmful effects of new medications before they advance to clinical trials, ensuring safety and efficacy.
What is the significance of understanding the differences between in vivo and in vitro methodologies?
Understanding the differences is integral to advancing medical research, as each method offers unique advantages and limitations that can aid researchers in accelerating breakthroughs in medication development and personalized medicine.
What recent advancements are enhancing in vivo and in vitro research?
Recent advancements, such as organ-on-chip technologies, are enhancing the capabilities of both approaches, leading to more ethical and accurate research outcomes.
How can recognizing the strengths and limitations of in vivo and in vitro models benefit researchers?
Recognizing the strengths and limitations of both models can assist researchers in accelerating breakthroughs in medication development and personalized medicine.
List of Sources
- Define In Vivo and In Vitro
- Real World Evidence in clinical trials: 3 case studies of successful implementation (https://informaconnect.com/real-world-evidence-clinical-trials-case-studies)
- liveonbiolabs.com (https://liveonbiolabs.com/in-vivo-vs-in-vitro-models)
- In vivo vs. in vitro: What is the difference? (https://medicalnewstoday.com/articles/in-vivo-vs-in-vitro)
- Context and Importance in Clinical Research
- biospace.com (https://biospace.com/fda/opinion-addressing-the-regulatory-reality-of-replacing-in-vivo-models-in-drug-development)
- linkedin.com (https://linkedin.com/pulse/95-drug-failure-rate-whats-really-going-wrong-its-you-stefano-1ujzf)
- Clinical Trial Success Rates: How Many Drugs Make It to Market? (Latest Approval Stats) (https://patentpc.com/blog/clinical-trial-success-rates-how-many-drugs-make-it-to-market-latest-approval-stats)
- The Probability of Technical Success in Drug Development (https://blog.biobide.com/the-probability-of-technical-success-in-drug-development)
- zeclinics.com (https://zeclinics.com/blog/differences-between-in-vitro-in-vivo-and-in-silico-assays-in-preclinical-research)
- Historical Development of In Vivo and In Vitro
- news-medical.net (https://news-medical.net/life-sciences/In-Vitro-vs-In-Vivo-Preclinical-Studies.aspx)
- Key Characteristics and Differences
- In Vitro vs. In Vivo: What’s the Difference? (https://newlifefertility.com/blog/in-vitro-and-in-vivo)
- liveonbiolabs.com (https://liveonbiolabs.com/in-vivo-vs-in-vitro-models)
- uhnresearch.ca (https://uhnresearch.ca/news/vitro-vs-vivo-one-better)

Leave a Reply