Introduction
Early feasibility studies (EFS) in Chile offer a promising yet challenging landscape for MedTech and biopharma companies. With expedited regulatory approval timelines and a diverse patient demographic, Chile stands out as a strategic hub for conducting EFS. This environment enables startups to gather crucial safety and efficacy data swiftly. However, navigating these complexities can be daunting for many companies. The local regulatory framework and potential approval delays raise pressing questions:
- What steps can you take to master early feasibility studies in Chile?
- How can you unlock the region’s potential for clinical innovation?
Exploring these essential strategies reveals the path to success in this competitive arena.
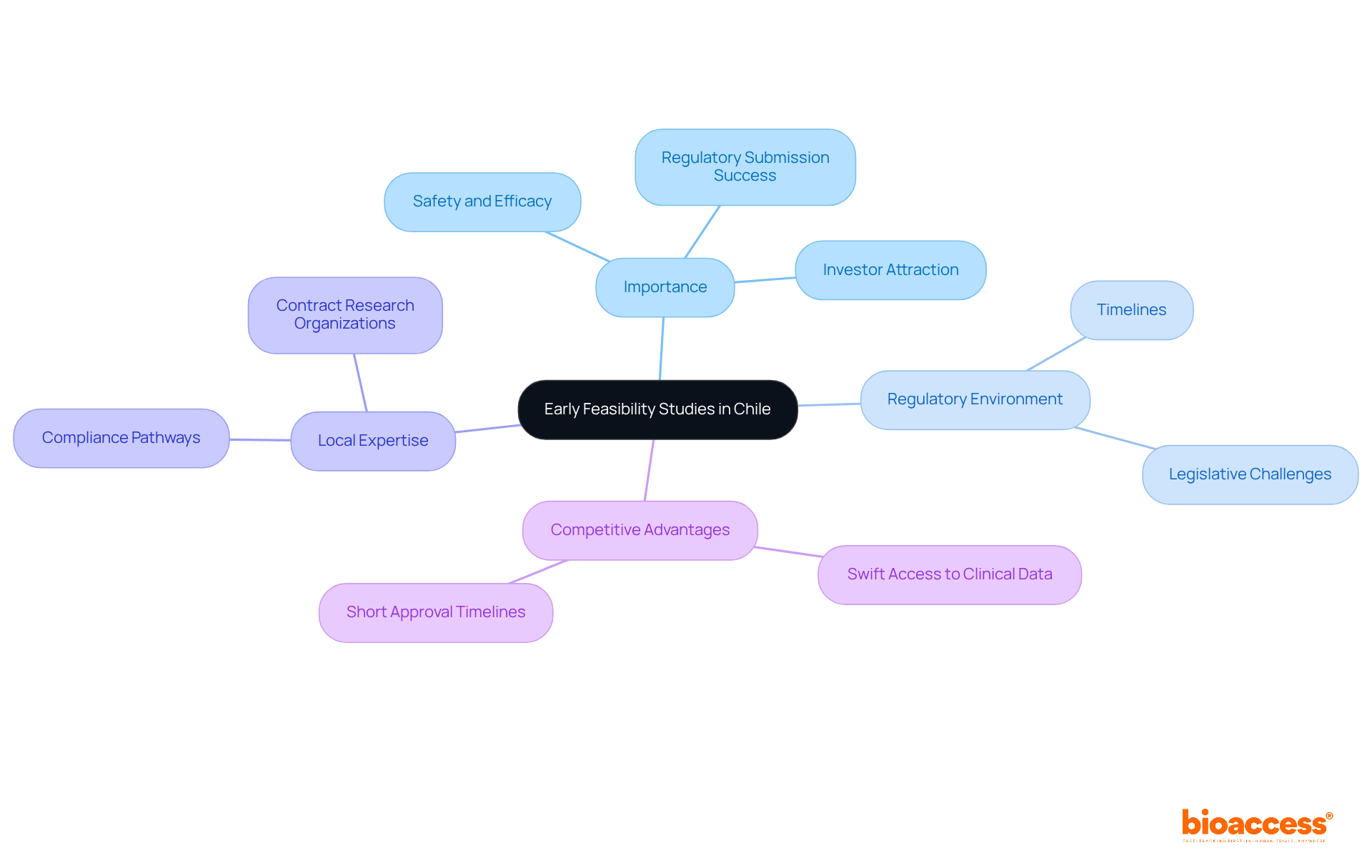
Understand Early Feasibility Studies and Their Importance in Chile
In the fast-evolving landscape of clinical research, an early feasibility study in Chile is essential for assessing the viability of new medical devices or treatments. How can the effective governance framework and diverse patient demographics in this South American nation enhance the benefits of EFS? These studies allow companies to gather vital information on safety and efficacy, greatly enhancing the likelihood of successful regulatory submissions.
The early feasibility study Chile holds strategic significance in the region, especially because of the swift access to clinical data. This is crucial for startups aiming to attract investors and secure funding. Regulatory approval timelines in this region can be as short as 30-90 days, enabling companies to achieve their first-in-human milestones more swiftly than in traditional markets. This competitive advantage positions Chile as an ideal site for an early feasibility study in MedTech, especially for startups navigating the complexities of compliance pathways effectively.
The Chilean legislative framework, unchanged since 2006, poses unique challenges. However, these can be mitigated through careful planning and collaboration with local experts. By utilizing the expertise of specialized contract research organizations like bioaccess®, startups can ensure adherence to ICH-GCP standards and facilitate smoother interactions with oversight authorities such as ANVISA. This method not only improves the reliability of the clinical data produced but also conforms to the FDA’s acceptance standards for international clinical trial data, further reinforcing the region’s status as a favorable destination for early feasibility study Chile. By leveraging local expertise, startups can not only navigate regulatory challenges but also enhance their chances of success in the competitive MedTech arena.
Navigate Regulatory Requirements for Early Feasibility Studies in Chile
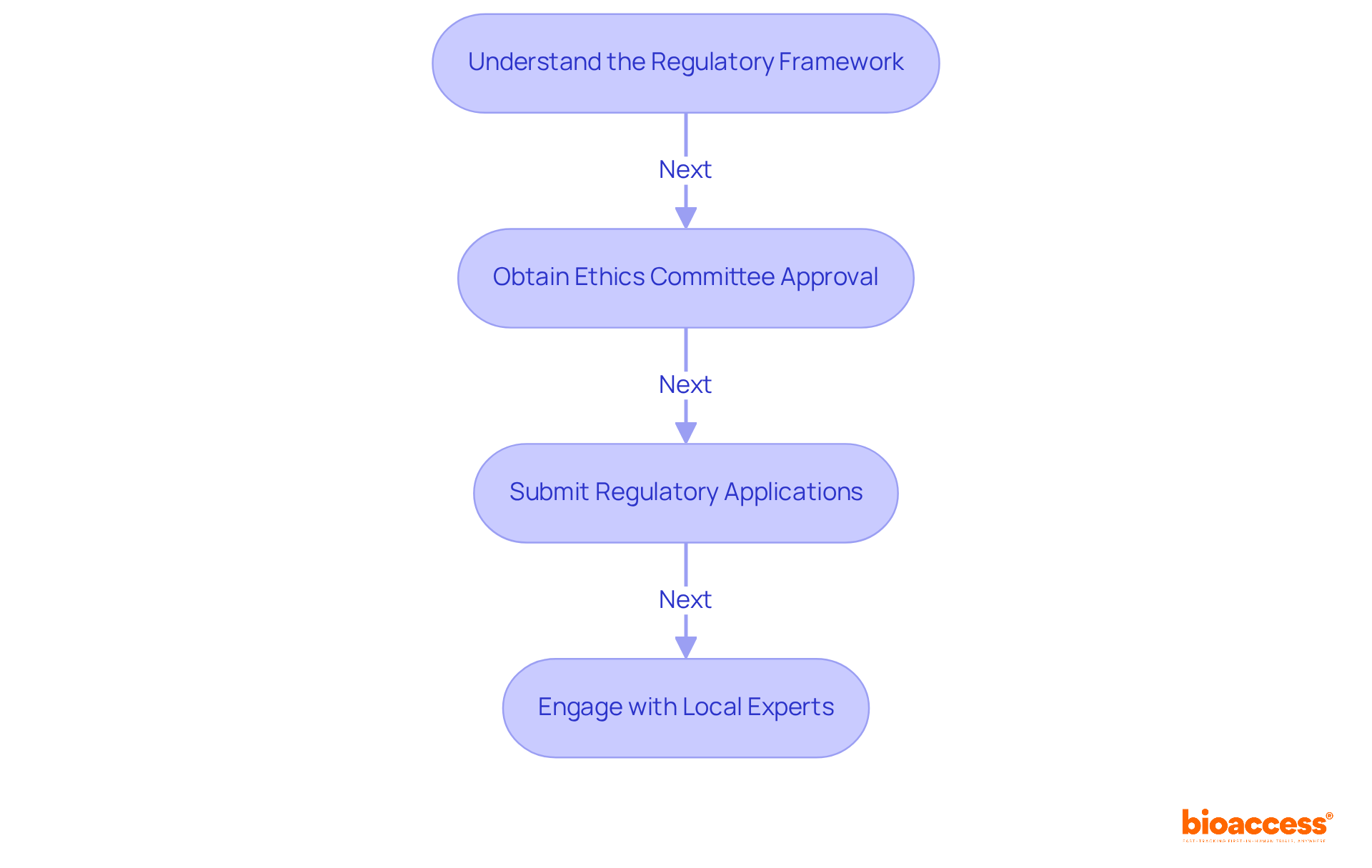
Navigating the compliance framework is crucial for conducting clinical research in Chile, where regulatory oversight is stringent and complex. To effectively carry out an initial feasibility assessment in the region, understanding the compliance framework is essential. The Instituto de Salud Pública (ISP) is the main oversight body supervising clinical trials in the country. Here are the essential steps to ensure compliance:
- Understand the Regulatory Framework: Familiarize yourself with Decree 3/2010, which details the requirements for medical device registration and clinical trials in Chile. Compliance with ICH-GCP standards is mandatory. Understanding the complexity of the certification and approval process is vital for success. Staying updated on regulatory changes from bioaccess® can provide essential insights into any modifications that may influence your research.
- Obtain Ethics Committee Approval: Before commencing your research, submit your research protocol to an Institutional Review Board (IRB) for ethical evaluation. This step is vital for ensuring participant safety and adherence to ethical standards, which is a key focus for bioaccess® in facilitating first-in-human trials. Notably, ethics approvals in Chile can be accelerated, allowing for quicker project initiation compared to other regions.
- Submit Regulatory Applications: Prepare and send your application to the ISP, including all necessary documentation such as study protocols, informed consent forms, and safety information. Align your submission with the ISP’s guidelines to prevent delays, as the average approval duration can exceed six months. Working with bioaccess® can simplify this process. Their expertise in compliance can help ensure your data meets FDA and EMA standards.
- Engage with Local Experts: Collaborating with local CROs or compliance consultants can provide valuable insights into the intricacies of the Chilean approval environment, facilitating a smoother approval process. Utilizing the strategic benefits of executing clinical trials in Latin America, such as quicker activation and cost-effectiveness, can greatly enhance your research. Bioaccess® offers tailored support to help MedTech startups navigate these complexities effectively.
By following these steps, sponsors can navigate the complexities of the Chilean regulatory framework. This ensures timely and compliant execution of early feasibility study Chile assessments. Embracing these steps not only ensures compliance but also positions your research for success in a competitive landscape.
Plan and Execute Your Early Feasibility Study: Key Steps and Strategies
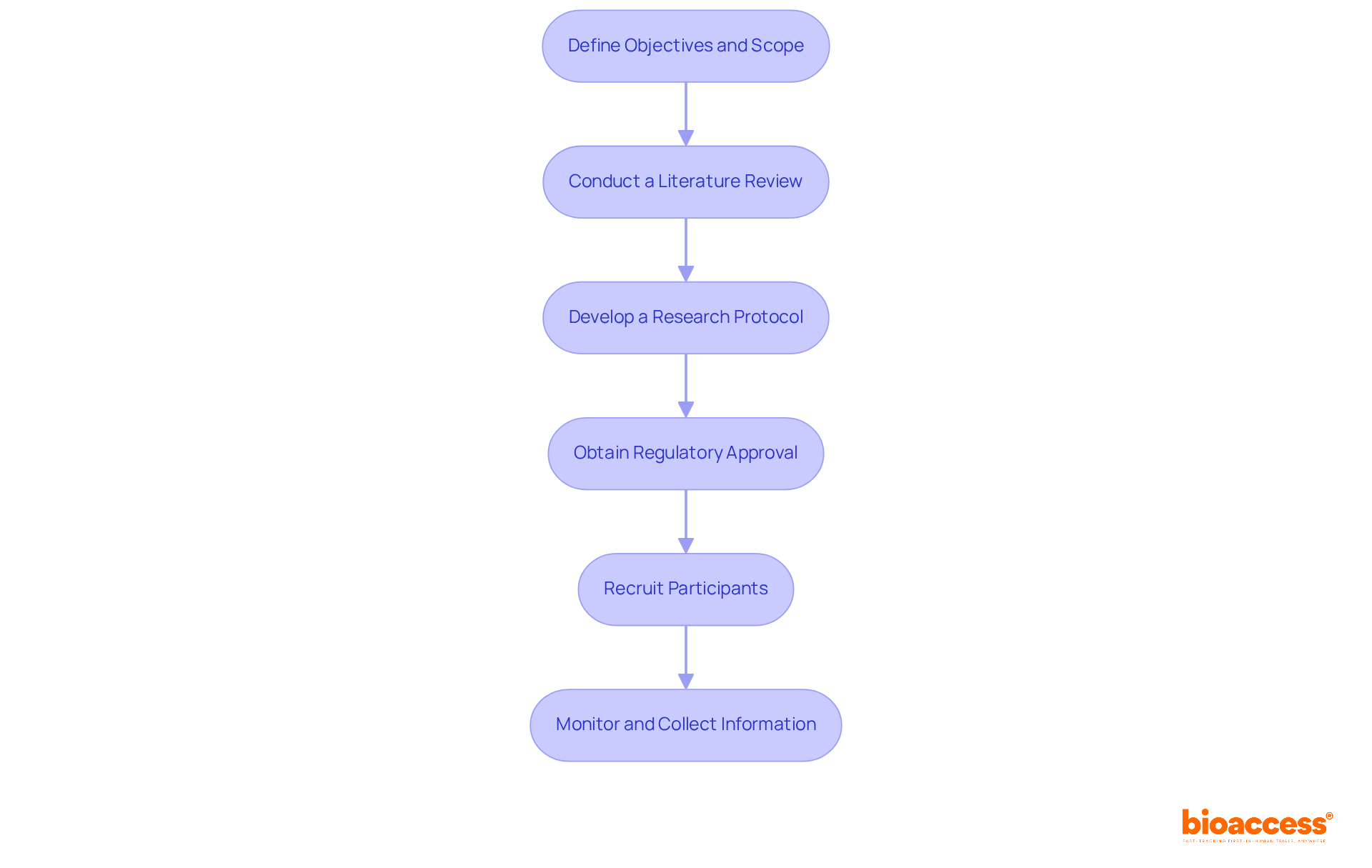
Successfully planning and executing an early feasibility study in Chile requires a strategic approach to overcome regulatory challenges and ensure participant safety. Here are several key steps to consider:
- Define Objectives and Scope: Clearly outline the objectives of your research, including specific questions regarding the device’s safety and efficacy. This clarity will guide how you design your research and develop your protocol.
- Conduct a Literature Review: Collect existing research and information related to your device and comparable examinations. Identifying gaps in knowledge will inform your research design and enhance its relevance.
- Develop a Research Protocol: Create a detailed research protocol that includes methodology, participant criteria, and data collection methods. Ensure that your protocol aligns with regulatory requirements, including compliance with ICH-GCP standards and FDA acceptance criteria.
- Obtain Regulatory Approval: Submit your research protocol to the Instituto de Salud Pública (ISP) for approval. This step is crucial for ensuring compliance and protecting participant safety. The CEC review and approval typically takes 4-8 weeks, depending on device complexity.
- Recruit Participants: Develop a robust recruitment strategy targeting the appropriate patient population. Engage with local healthcare providers and leverage community networks to enhance recruitment efforts. Notably, Chile’s patient referral networks are physician-driven, allowing for faster recruitment through established relationships.
- Monitor and Collect Information: Implement a rigorous monitoring plan to ensure information integrity and participant safety throughout the study. Consistently review information and make modifications as needed to address any emerging issues. Ensure that 100% of critical information points undergo source verification documentation to maintain compliance and quality.
Navigating the complex regulatory landscape can be daunting for sponsors. However, by following these steps, sponsors not only streamline their processes but also enhance their chances of successful market entry. This methodical approach not only mitigates risks but also positions sponsors for a competitive edge in the clinical research landscape.
Overcome Challenges in Conducting Early Feasibility Studies in Chile
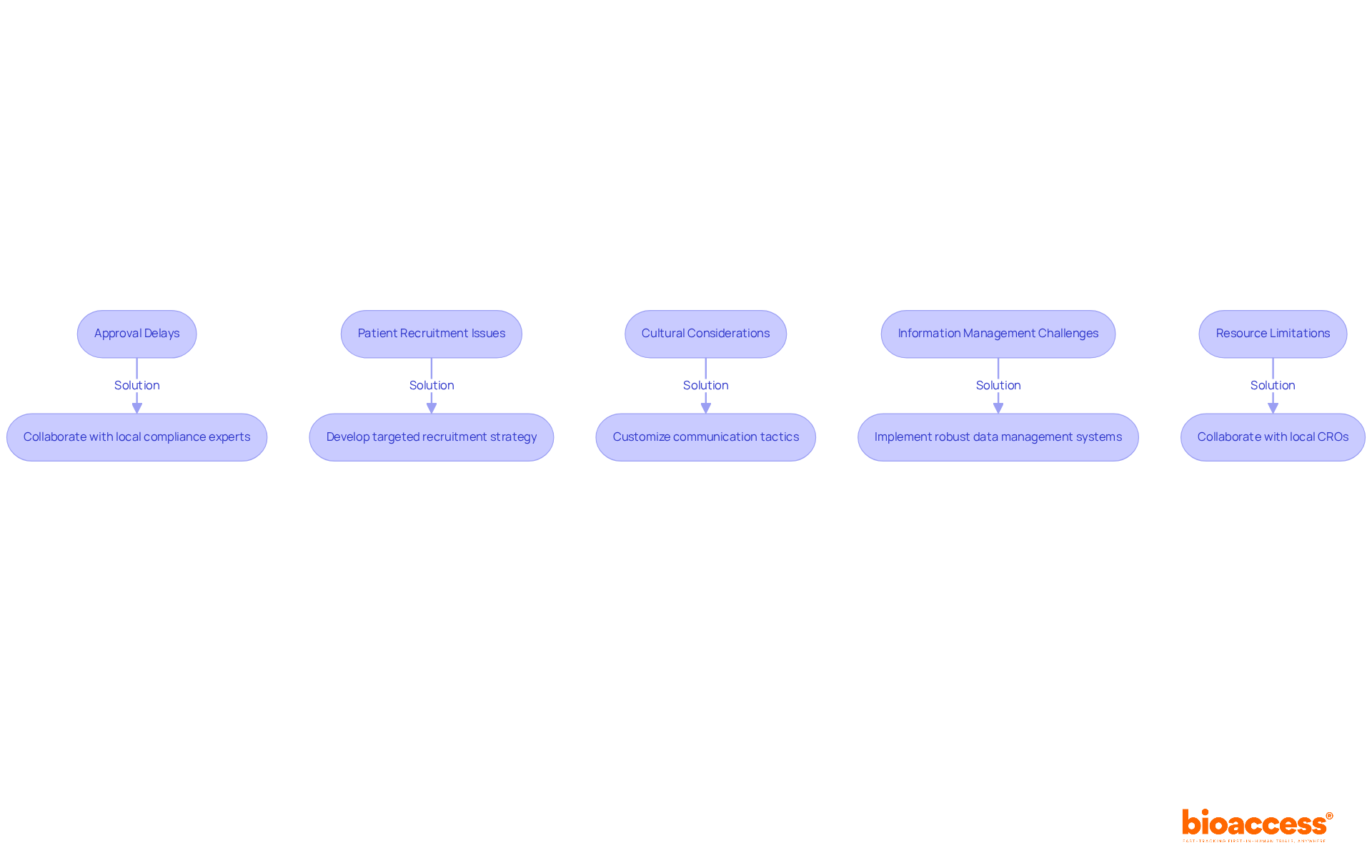
Conducting early feasibility study Chile can be fraught with challenges that require strategic navigation to ensure success. Here are some key hurdles and how to effectively manage them:
- Approval Delays: Navigating the approval landscape in Chile can be a daunting task, often leading to frustrating delays that hinder progress. Collaborating with local compliance experts is essential. They can provide guidance and improve communication with the Instituto de Salud Pública (ISP). Proactive engagement with the ISP can streamline the approval process, which typically lasts about 30 business days.
- Patient Recruitment Issues: Recruiting participants, particularly from niche populations, can be a significant hurdle. Without a targeted recruitment strategy, you risk falling short on participant numbers, which could jeopardize the success of your study. Developing a targeted recruitment strategy is crucial. This includes outreach to local healthcare providers and community organizations, enhancing visibility and fostering trust within the community. Utilizing online platforms for advertising can also improve recruitment outcomes.
- Cultural Considerations: Grasping the cultural nuances can make a real difference in how you engage and retain participants. Customizing communication and recruitment tactics to connect with the local community ensures that potential participants feel at ease and well-informed about the research. Engaging with local patient advocacy groups can further enhance recruitment efforts.
- Information Management Challenges: Preserving information integrity and adherence to regulatory standards can be overwhelming. Implementing robust data management systems and conducting regular monitoring are essential to ensure high-quality data throughout the research. Compliance with ICH-GCP standards is mandatory, and meticulous documentation is required for all submissions to the ISP.
- Resource Limitations: Startups often face constraints in funding and personnel. Collaborating with local CROs or research institutions can offer additional resources and expertise to aid your project. Partnering with established organizations like bioaccess® can facilitate access to a network of pre-qualified clinical trial sites and experienced personnel, enhancing the overall efficiency of the study.
By addressing these challenges head-on, you can position your study for success and contribute valuable insights to the clinical research landscape in Chile.
Conclusion
The potential for MedTech and biopharma companies in Chile is vast, yet navigating the complexities of clinical trials requires strategic foresight. By leveraging the unique regulatory environment, organizations can maximize their chances of success. Swift approval timelines and diverse patient demographics further enhance this opportunity, making Chile a strategic location for early feasibility studies.
Throughout this article, we outlined essential steps for conducting successful early feasibility studies. Understanding the regulatory framework, obtaining necessary approvals, and developing effective recruitment strategies are key. Collaborating with local experts and contract research organizations like bioaccess® is crucial for streamlining processes and mitigating potential challenges. By focusing on compliance with ICH-GCP standards and engaging with local healthcare providers, sponsors can enhance their study’s reliability and efficiency.
In conclusion, the potential for success in the Chilean clinical trial landscape is immense. Startups and established companies alike must embrace the outlined best practices. As the demand for innovative medical solutions continues to grow, the strategic advantages offered by early feasibility studies in Chile present an invaluable opportunity. It’s crucial for companies to take action, tap into local expertise, and prioritize compliance. This approach ensures their research meets regulatory requirements and adds valuable insights to the global MedTech ecosystem.
Frequently Asked Questions
What is the purpose of an early feasibility study (EFS) in Chile?
An early feasibility study in Chile assesses the viability of new medical devices or treatments, allowing companies to gather vital information on safety and efficacy, which enhances their chances of successful regulatory submissions.
Why is Chile considered a strategic location for early feasibility studies?
Chile is strategic for early feasibility studies due to its swift access to clinical data, with regulatory approval timelines as short as 30-90 days. This enables companies to achieve first-in-human milestones more quickly than in traditional markets.
How can startups benefit from the governance framework in Chile?
The effective governance framework and diverse patient demographics in Chile enhance the benefits of early feasibility studies by providing a competitive advantage, particularly for startups looking to attract investors and secure funding.
What challenges do startups face in Chile’s regulatory environment?
Startups may encounter challenges due to the Chilean legislative framework, which has remained unchanged since 2006. However, these challenges can be mitigated through careful planning and collaboration with local experts.
How can specialized contract research organizations assist startups in Chile?
Specialized contract research organizations, like bioaccess®, can help startups ensure adherence to ICH-GCP standards, facilitate smoother interactions with regulatory authorities such as ANVISA, and improve the reliability of clinical data produced.
What are the compliance requirements for conducting clinical trials in Chile?
Compliance with ICH-GCP standards and alignment with FDA acceptance criteria for international clinical trial data are essential for conducting clinical trials in Chile.
What role does local expertise play in the success of early feasibility studies?
Leveraging local expertise allows startups to navigate regulatory challenges effectively, enhancing their chances of success in the competitive MedTech arena in Chile.
List of Sources
- Understand Early Feasibility Studies and Their Importance in Chile
- Latin America’s Landscape For Medtech Clinical Trials (https://clinicalleader.com/doc/latin-america-s-landscape-for-medtech-clinical-trials-0001)
- Early Feasibility Studies in Latin America (https://greenlight.guru/blog/early-feasibility-studies-in-latin-america)
- Navigate Regulatory Requirements for Early Feasibility Studies in Chile
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Case Studies – QPS Custom-Built Research (https://qps.com/resources/case-studies)
- Plan and Execute Your Early Feasibility Study: Key Steps and Strategies
- First-in-Human Medical Device Trial in Chile | bioaccess® (https://bioaccessla.com/blog/first-in-human-medical-device-trial-chile)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Early Feasibility Studies in Latin America (https://greenlight.guru/blog/early-feasibility-studies-in-latin-america)
- Overcome Challenges in Conducting Early Feasibility Studies in Chile
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Early Feasibility Studies: Top 6 Considerations | MED Institute (https://medinstitute.com/blog/early-feasibility-studies-top-6-considerations)
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Keys to Maximize Patient Recruitment (https://linical.com/articles-research/keys-to-maximize-patient-recruitment)

Leave a Reply