Introduction
Navigating the complex landscape of clinical trials in Belize presents a unique set of challenges and opportunities that can significantly influence research outcomes. Understanding the local regulatory environment, healthcare infrastructure, and patient demographics is essential for selecting the most suitable trial sites. But how can sponsors ensure that their chosen locations not only meet compliance standards but also foster successful participant recruitment and engagement? This article explores effective strategies for navigating clinical trial site selection in Belize, equipping researchers with the insights needed to enhance their study’s success.
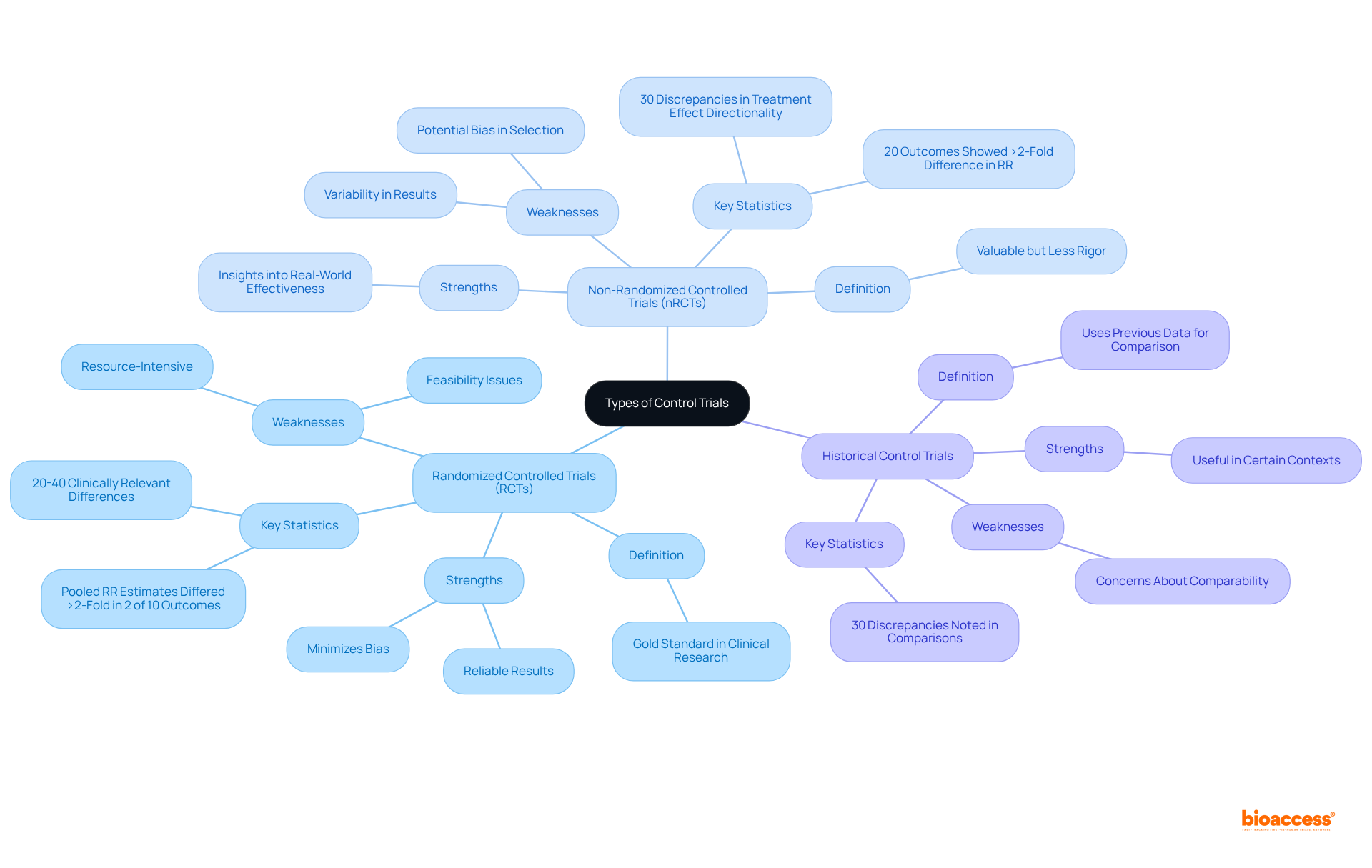
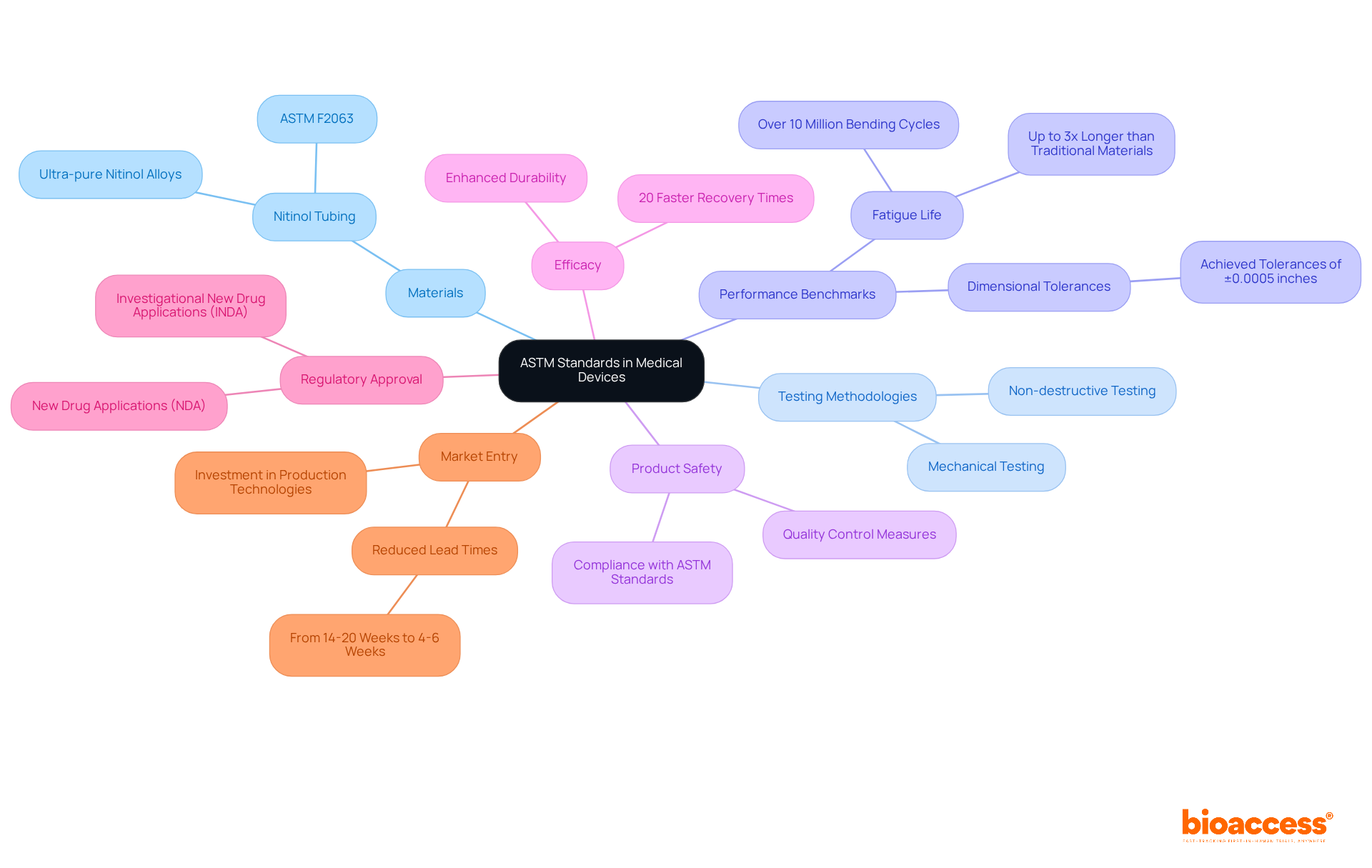
Understand the Clinical Trial Landscape in Belize
Navigating the selection of a clinical trial site in Belize requires a solid understanding of the local study landscape. This knowledge is crucial for ensuring successful outcomes at the clinical trial site Belize. Here are several key areas to consider:
- Regulatory Environment: The Ministry of Health and Wellness is the governing body for clinical trial site Belize. Familiarizing yourself with the guidelines and regulations of the clinical trial site Belize is vital for compliance and smooth operations. Insights from Global Trial Accelerators™ can provide timely updates on these regulations, ensuring your studies meet the latest compliance standards.
- Healthcare Infrastructure: Belize boasts a blend of public and private healthcare providers. Evaluating the capabilities of these facilities is essential to identify which locations are adequately equipped to conduct research.
- Patient Demographics: Understanding the demographics of the Belizean population is critical. By analyzing local health conditions and treatment needs, you can identify a clinical trial site Belize that is suitable for your research, ensuring that your participant group aligns with the study’s objectives.
- Current clinical studies in Belize: Investigating ongoing and past clinical trial site Belize can shed light on the competitive landscape. Resources like the WHO International Clinical Trials Registry Platform (ICTRP) offer valuable insights into active studies, aiding in the assessment of location performance and participant recruitment potential.
- Cultural considerations are significant factors in patient participation and operations at the clinical trial site Belize. Engaging with local communities and understanding their perspectives can enhance recruitment efforts and contribute to the overall success of research studies in Belize. Insights from Global Trial Accelerators™ emphasize the importance of aligning study goals with community interests.
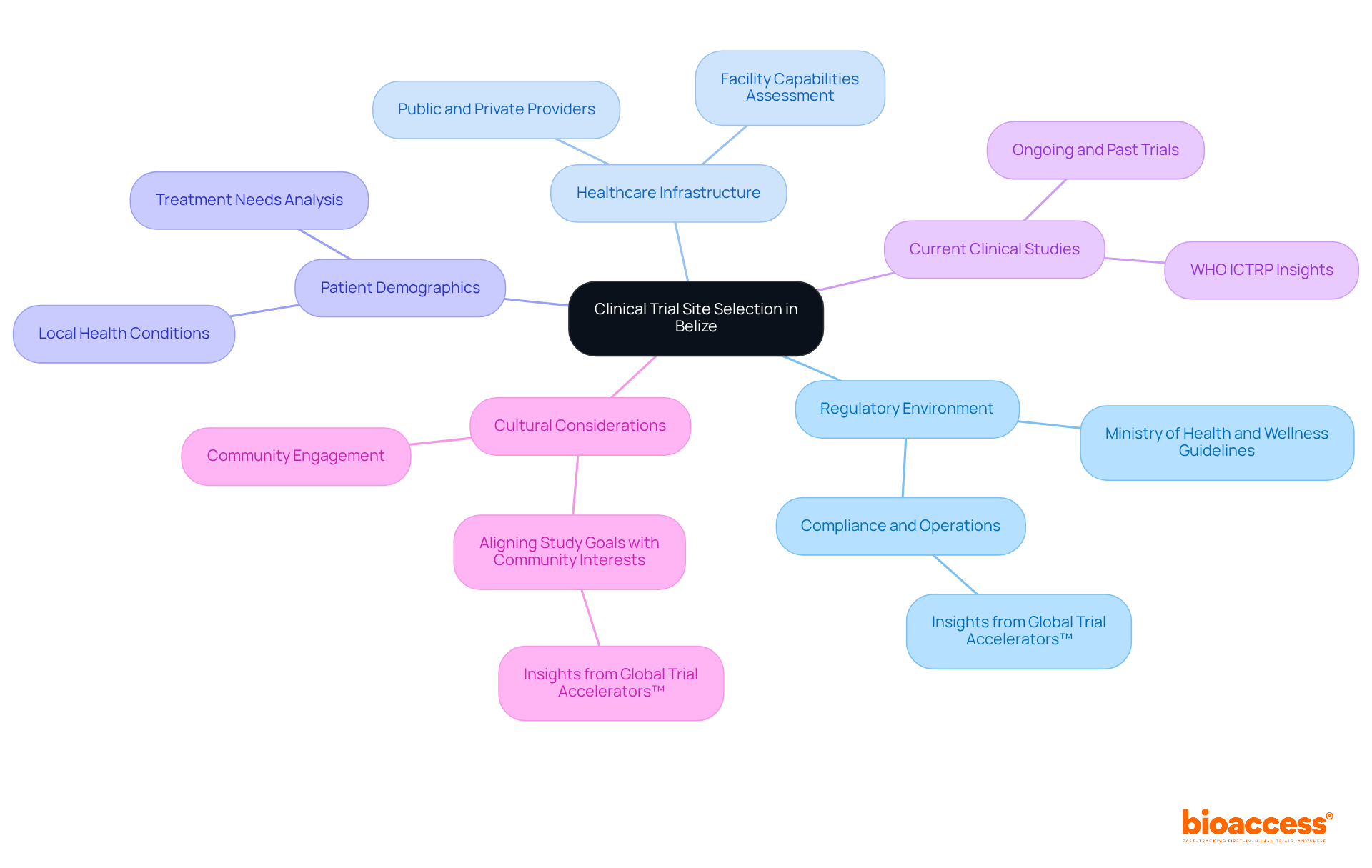
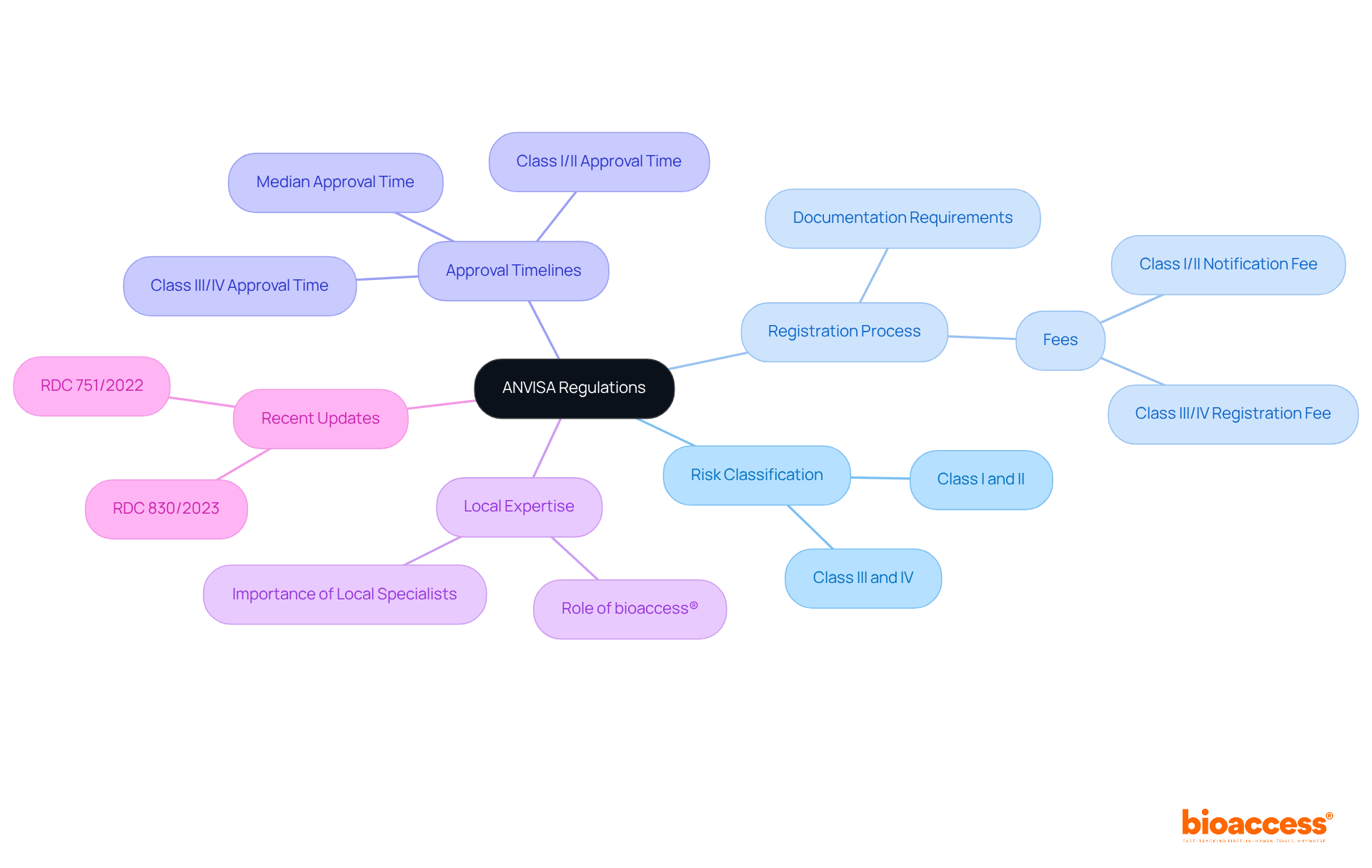
Evaluate Key Criteria for Site Selection
When selecting clinical trial sites in Belize, it’s crucial to consider several key criteria to ensure the success of your study:
- Principal Investigator (PI) Experience: The qualifications and experience of the PI and their team are paramount. A knowledgeable PI can significantly impact the case’s success, as their expertise often correlates with better management and adherence to protocols.
- Location Infrastructure: Evaluate the location’s facilities, including equipment, technology, and staff availability. A robust infrastructure is essential for accommodating the specific requirements of your research protocol, ensuring that all operational aspects are efficiently managed.
- Participant Recruitment Capabilities: Examine the location’s past performance in enrolling individuals. Research shows that more than 30% of clinical trial site Belize locations do not enroll a single participant, highlighting the necessity for clinical trial sites with a demonstrated history of successful participant enrollment in comparable studies.
- Regulatory Compliance: Confirm that the location adheres to local and international regulatory standards, including ICH-GCP compliance. This adherence is essential for maintaining data integrity and securing approval. Insights from Global Trial Accelerators™ can provide valuable updates on regulatory changes that directly impact compliance, helping to navigate potential hurdles that could delay timelines.
- Geographic Accessibility: Consider the location of the area in relation to the target clientele. Locations that are easily reachable can improve participant engagement and retention, which is vital for study success.
- Community Engagement: Assess the organization’s relationships with local communities. Strong community ties can facilitate patient recruitment and improve trial acceptance, fostering a supportive environment for the study.
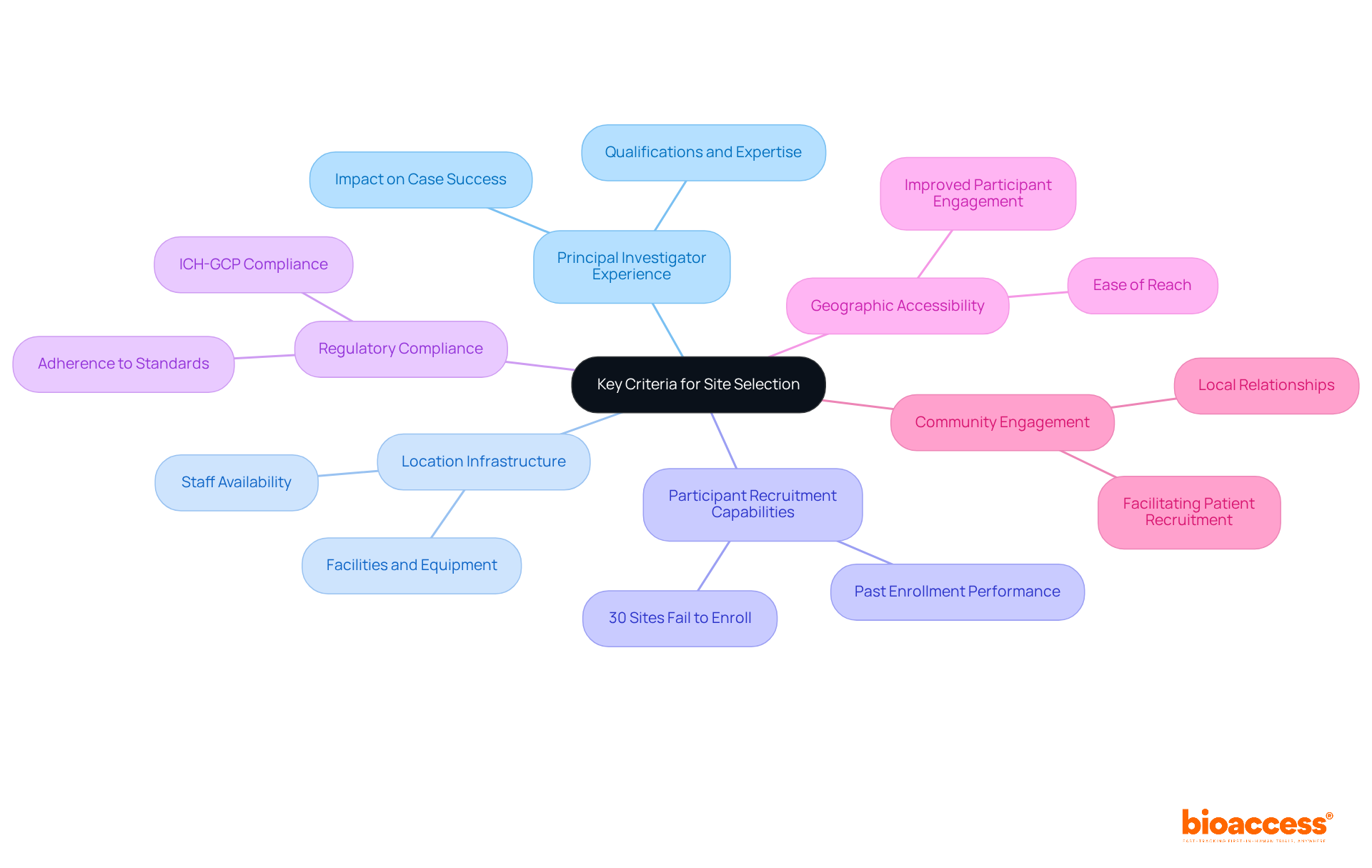
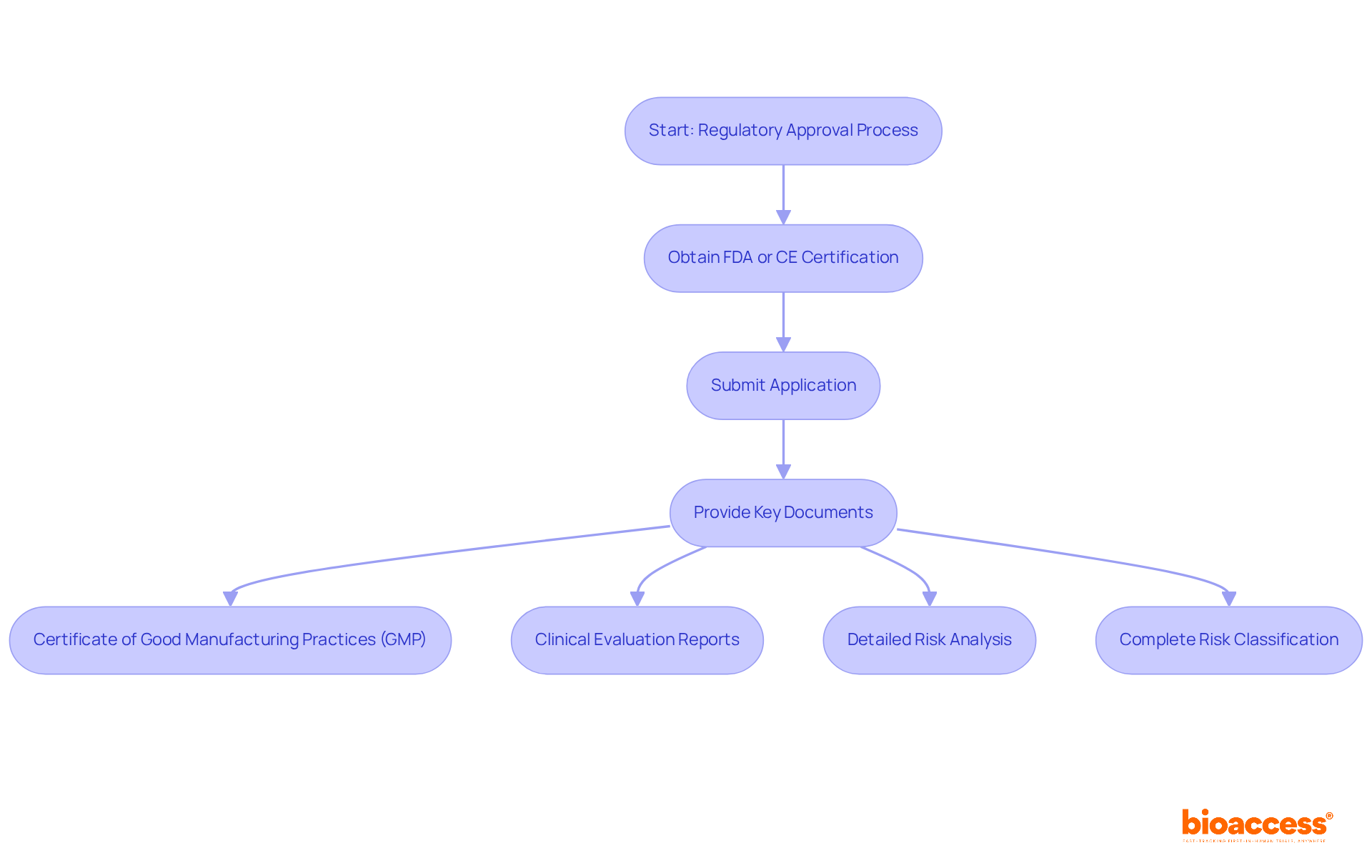
Engage and Negotiate with Selected Sites
Once potential sites have been identified, the next step is to engage and negotiate effectively:
- Initial Communication: Start by reaching out to selected locations to introduce your study and express interest in collaboration. Clear communication from the outset establishes a positive tone for the relationship and sets expectations.
- Visits: Conduct visits to assess facilities and meet the research team. This personal interaction fosters rapport and trust, which are essential for a successful partnership.
- Discuss Study Requirements: Clearly outline the study’s requirements, including timelines, patient enrollment goals, and specific protocol needs. Ensuring that the site comprehends these expectations is crucial for smooth execution.
- Negotiate Terms: Engage in discussions to negotiate the clinical research agreement conditions, including budget, timelines, and responsibilities. Transparency regarding funding and resource allocation promotes a collaborative environment. As Jeff Parke notes, “To consistently get what we need in a negotiation, we must marry the substance to the process.”
- Establish Communication Channels: Set up regular communication channels to keep all parties informed throughout the process. This can include scheduled meetings, progress reports, and updates on patient recruitment. Biweekly or monthly check-ins are advised to address ongoing needs and provide updates on progress, which are essential for maintaining alignment.
- Build a Partnership: Approach the relationship as a partnership rather than a transactional agreement. Encourage a cooperative atmosphere by including personnel in decision-making processes and appreciating their contributions, which boosts commitment and involvement. Active locations can decrease patient dropout rates and limit recruitment delays, ultimately enhancing the study’s success.
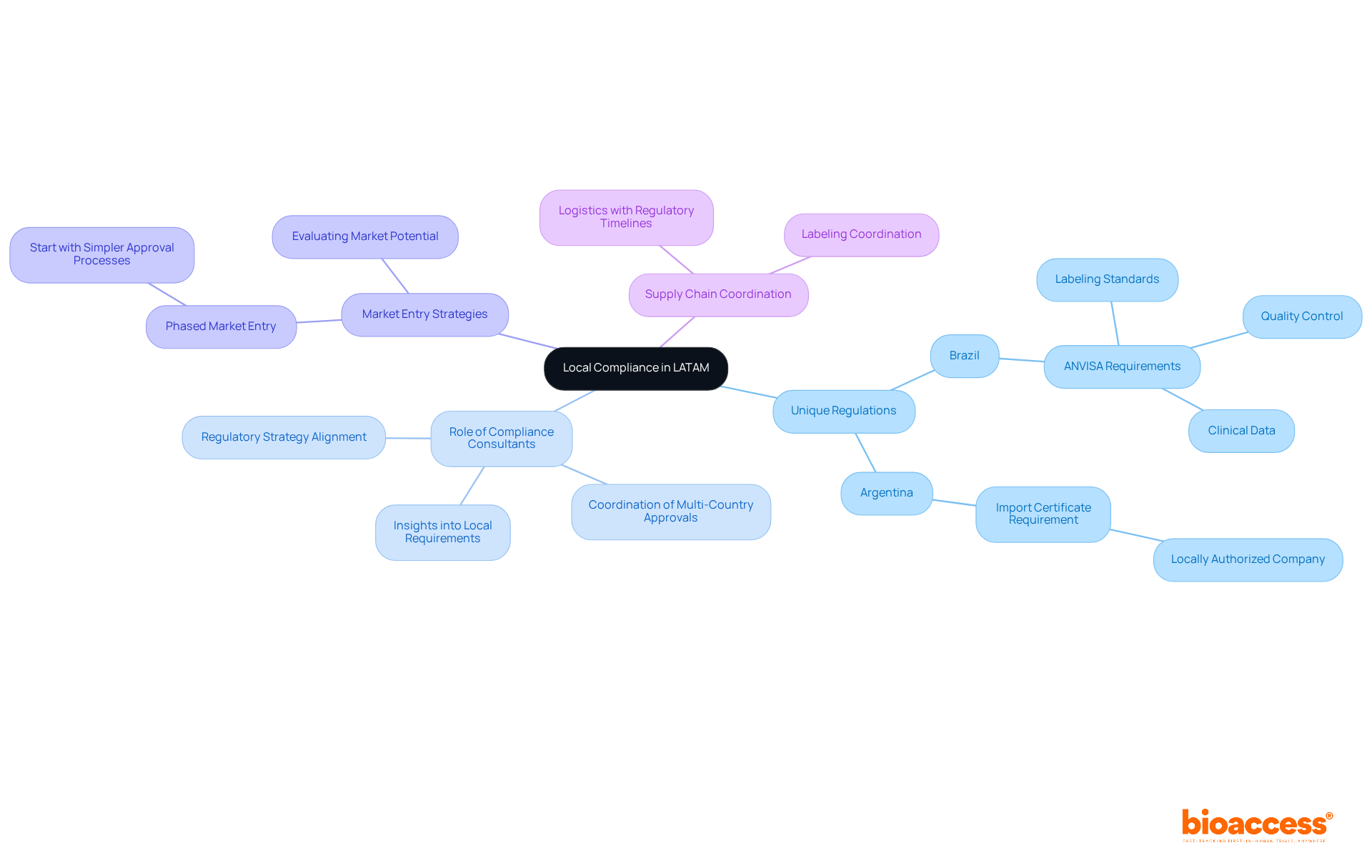
Establish a Partnership for Successful Trials
To ensure the success of your clinical trial site Belize, establishing a robust partnership with your selected sites is crucial. Leveraging insights from bioaccess’s Global Trial Accelerators™, you can enhance your trial’s effectiveness.
- Define Roles and Responsibilities: Clearly delineate the roles and responsibilities of both the sponsor and the site. This clarity reduces misunderstandings and promotes accountability, which is vital for seamless procedural operations.
- Foster Open Communication: Maintain transparent communication throughout the process. Regular updates and feedback sessions facilitate prompt issue resolution and keep the trial on schedule.
- Offer Training and Assistance: Conduct targeted training sessions for personnel on the study protocol and any new technologies. This support enhances platform performance and ensures adherence to study requirements, ultimately improving data quality.
- Monitor Progress Together: Collaborate with the site to track patient recruitment and data collection. By assessing progress together, you can recognize obstacles early and make prompt modifications, improving the efficiency of the process.
- Celebrate Milestones: Acknowledge and celebrate milestones accomplished during the experiment. Recognizing the efforts of site staff boosts morale and strengthens the partnership, fostering a collaborative environment.
- Plan for Future Collaborations: Initiate discussions about potential future collaborations early in the process. Establishing long-term relationships can lead to more effective experiments and enhanced results in subsequent studies. By utilizing the essential clinical trial insights and market access strategies provided by Bioaccess, you can significantly enhance your success at the clinical trial site Belize.
Conclusion
Navigating the complexities of clinical trial site selection in Belize is crucial for achieving successful research outcomes. A comprehensive understanding of the local landscape – including regulatory frameworks, healthcare infrastructure, and community dynamics – forms the bedrock for effective site selection. By acknowledging these factors, researchers can align their clinical trials with the unique characteristics of Belize, ensuring a more tailored and impactful approach.
Key insights from the article underscore the significance of evaluating:
- Principal investigator experience
- Location infrastructure
- Participant recruitment capabilities
- Community engagement
Each of these elements is vital not only for selecting the right site but also for fostering a collaborative environment that enhances trial success. Establishing clear communication and strong partnerships with selected sites further solidifies the foundation for effective clinical research.
Ultimately, the success of clinical trials in Belize relies on a strategic approach that marries thorough preparation with robust engagement practices. By prioritizing these best practices, researchers can navigate the site selection process with confidence, paving the way for meaningful contributions to the field of clinical research. Embracing this comprehensive methodology will not only improve trial outcomes but also strengthen the healthcare landscape in Belize, benefiting both researchers and the communities they serve.
Frequently Asked Questions
What is the significance of understanding the clinical trial landscape in Belize?
Understanding the clinical trial landscape in Belize is crucial for ensuring successful outcomes at clinical trial sites, as it helps in navigating the selection process and compliance with local regulations.
Who governs clinical trials in Belize?
The Ministry of Health and Wellness is the governing body for clinical trials in Belize, overseeing guidelines and regulations that must be followed for compliance and smooth operations.
How can I stay updated on regulations for clinical trials in Belize?
Insights from Global Trial Accelerators™ can provide timely updates on regulations, ensuring that your studies meet the latest compliance standards.
What type of healthcare infrastructure is available in Belize for clinical trials?
Belize has a mix of public and private healthcare providers. Evaluating the capabilities of these facilities is essential to determine which locations are equipped to conduct research.
Why is it important to understand patient demographics in Belize?
Understanding the demographics of the Belizean population helps in analyzing local health conditions and treatment needs, allowing for the identification of suitable clinical trial sites that align with study objectives.
How can I find information on current clinical studies in Belize?
Resources like the WHO International Clinical Trials Registry Platform (ICTRP) can provide valuable insights into ongoing and past clinical trials in Belize, aiding in the assessment of location performance and participant recruitment potential.
What cultural considerations should be taken into account for clinical trials in Belize?
Cultural considerations are significant for patient participation and operations at clinical trial sites. Engaging with local communities and understanding their perspectives can enhance recruitment efforts and contribute to the success of research studies.
List of Sources
- Understand the Clinical Trial Landscape in Belize
- who.int (https://who.int/observatories/global-observatory-on-health-research-and-development/monitoring/number-of-clinical-trials-by-year-country-who-region-and-income-group)
- Clinical Trial Country Coverage in Latin America | H Clinical (https://hclinical.com/clinical-trial-country-coverage)
- Belize Marks World Health Day with Call for Vaccination, Healthy Lifestyles, and Science-Based Health Action (https://paho.org/en/news/7-4-2026-belize-marks-world-health-day-call-vaccination-healthy-lifestyles-and-science-based)
- Frontiers | International Clinical Trials in Latin American and Caribbean Countries: Research and Development to Meet Local Health Needs (https://frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2017.00961/full)
- MOH Metadata (https://sib.org.bz/data-portals/documentation/moh-metadata)
- Evaluate Key Criteria for Site Selection
- Top 5 Alarming Statistics You Must Know About Clinical Trial Site Selection (https://linkedin.com/pulse/top-5-alarming-statistics-you-must-know-clinical-trial-kgfce)
- Optimizing Site Selection and Management for Clinical Trial Success (https://studypages.com/blog/optimizing-site-selection-and-management-for-clinical-trial-success)
- Role of Principal Investigator in Trials | CCRPS (https://ccrps.org/clinical-research-blog/role-of-a-principal-investigator-in-clinical-trials)
- Top 5 Alarming Statistics on Clinical Trial Site Selection (https://biobostonconsulting.com/top-5-alarming-statistics-you-must-know-about-clinical-trial-site-selection)
- What Inspires and Sustains Long-Term Principal Investigators? – ACRP (https://acrpnet.org/2024/08/29/what-inspires-and-sustains-long-term-principal-investigators)
- Engage and Negotiate with Selected Sites
- Enhancing Clinical Trials: Why Site Engagement Matters for Sponsors (https://studypages.com/blog/enhancing-clinical-trials-why-site-engagement-matters-for-sponsors)
- Enhancing Clinical Trials: Best Practices for Site Collaboration (https://iqvia.com/blogs/2025/06/enhancing-clinical-trials-best-practices-for-site-collaboration)
- Strategies to Elevate Your Study at Saturated Sites (https://precisionformedicine.com/blog/strategies-to-elevate-your-study-at-saturated-sites)
- Negotiating Effective Clinical Trial Agreements and Study Budgets with Research Sites | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/negotiating-effective-clinical-trial-agreements-and-study-budgets-research-sites)
- Streamlining Clinical Trial Agreement Negotiations at Your Site: Five Strategies for Success | WCG (https://wcgclinical.com/insights/streamlining-clinical-trial-agreement-negotiations-at-your-site-five-strategies-for-success)
- Establish a Partnership for Successful Trials
- Clinical Trial Success Rates: How Many Drugs Make It to Market? (Latest Approval Stats) (https://patentpc.com/blog/clinical-trial-success-rates-how-many-drugs-make-it-to-market-latest-approval-stats)
- Topic: Clinical trials (https://statista.com/topics/6756/clinical-trials?srsltid=AfmBOoqCcYpU6pJO2uvzZpZN5BZypKxIkbCy8t5az6eIokeVTyrunxm7)
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- Strengthening Clinical Trial Site Relationships (https://premier-research.com/perspectives/strengthening-clinical-trial-site-relationships-defining-sponsor-and-cro-roles)
- Site Training for Clinical Trial success | ICON plc (https://iconplc.com/insights/blog/2024/03/25/site-training-clinical-trial-success)