Introduction
The investigational brochure (IB) is a cornerstone in the realm of clinical trials, encapsulating vital information that guides researchers and regulatory bodies alike. A well-structured IB possesses the potential to make or break a study; it not only enhances participant safety but also fosters clarity in communication among all stakeholders involved. As the complexities of clinical research continue to evolve, researchers must ask: how can their investigational brochures remain relevant, compliant, and effective? This article delves into the seven key elements that every investigational brochure must include to navigate the challenges of clinical trials successfully.
Investigator’s Brochure: Core Document for Clinical Trials
The (IB) serves as a pivotal document summarizing , encompassing its pharmacological characteristics, preclinical data, and study protocols. This serves as a vital reference for both investigators and regulatory authorities, ensuring alignment on the study’s objectives and methodologies.
A meticulously crafted is essential for the effective initiation and execution of , as it lays the groundwork for informed decision-making throughout the research process. Notably, insufficient s contribute to approximately 80% of studies facing delays or shutdowns, highlighting the .
Successful instances of s in Medtech illustrate that a can , ultimately leading to improved outcomes. As highlighted in recent discussions, it is imperative that the be and maintain its relevance, thereby ensuring it continues to support effective risk assessments and safeguard participant welfare.
Regulatory Compliance: Key Requirements for Investigator’s Brochure
The must adhere to . Essential requirements include:
- A clear description of the investigational product
- A
Ensuring that the meets these standards is crucial for securing ethical approvals and maintaining the confidence of participants and stakeholders throughout the study. Moreover, a thorough procedure for advancing medical device evaluations encompasses:
- Site selection
These are vital for addressing and achieving successful evaluation outcomes. Bioaccess offers expertise in these areas, providing support in:
- Import permits
- Reporting
All of which are .
Essential Components: What to Include in an Investigator’s Brochure
A well-organized investigational brochure is crucial for the success of research trials, serving as a . are:
- Summary of the Investigational Product: This should detail the product’s name, identity, and a confidentiality statement, ensuring clarity on what is being studied.
- Preclinical and Clinical Data: Summarize findings from both non-clinical and clinical research, including pharmacological, toxicological, and pharmacokinetic information, to provide a thorough understanding of the product’s safety and efficacy.
- : Clearly outline the aims of the research and the methods to be employed, facilitating a shared understanding among the research team.
- : Include a section on adverse drug reactions and safety monitoring procedures, preparing investigators for possible challenges during the study.
- : Outline the methods for acquiring informed consent, highlighting the ethical factors involved in the research.
- Roles and Responsibilities: Clearly define the roles of the research team members, ensuring accountability and effective collaboration.
- Contact Information: Provide contact details for key personnel, enabling quick communication for any queries or issues that may arise.
In addition to these elements, it is vital to consider the that support the creation of a successful investigational brochure. Services such as feasibility studies, site selection, compliance reviews, testing setup, and project management play a significant role in ensuring that the IB is not only informative but also aligned with regulatory requirements. Typically, contain around 80 to 100 pages, balancing detail with conciseness, which is crucial for effective communication. Expert opinions indicate that a in medical research, enhancing the overall integrity of the study process. Furthermore, the investigational brochure must be reviewed and updated at least annually or whenever significant new data is received to ensure its relevance. It is also important to note that the should be submitted along with the investigational brochure to national competent authorities. Examples of comprehensive IBs can be found in various , showcasing best practices in documentation and stakeholder engagement. To maximize the effectiveness of the IB, consider leveraging the expertise and services offered by bioaccess, which can streamline the process and ensure compliance with all regulatory requirements.
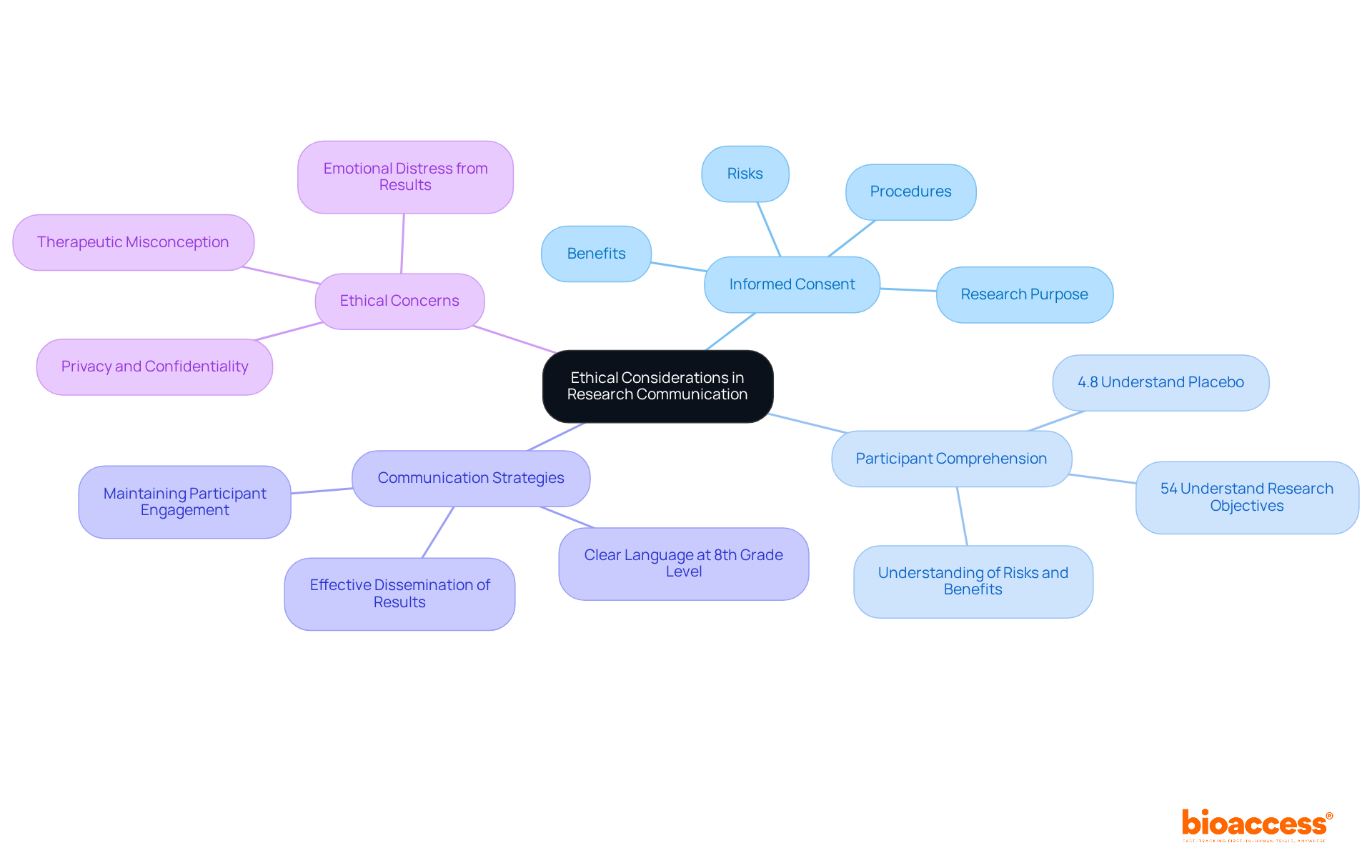
Ethical Considerations: Informing Participants Through the Investigator’s Brochure
The Investigator’s Brochure (IB) must prioritize . It is essential that participants are thoroughly informed about the research’s purpose, procedures, risks, and benefits, including their right to withdraw from . Effective communication through the is crucial for fostering trust and transparency, which are vital elements for successful and retention. To enhance comprehension, researchers should utilize in the , ideally at an 8th-grade reading level or lower. This approach not only supports but also positively influences rates, as individuals are more likely to remain engaged when they fully understand the implications of their involvement.
Statistics indicate that comprehension levels for elements can be low, with only 54% of participants grasping research objectives and a mere 4.8% understanding the concept of placebo. Furthermore, maintaining a data file that tracks all signed ancillary consent forms is essential for . As one researcher noted, ” and context can result in more questions…without a process to address these issues, participants may be left with serious concerns without any way to address them.
Updates and Revisions: Maintaining Accuracy in the Investigator’s Brochure
To uphold the integrity of the Investigator’s Brochure (IB), a is crucial. Frequent evaluations of the IB in response to new medical information, changing compliance demands, and alterations to the research protocol are essential. Statistics indicate that outdated IBs can significantly compromise , underscoring the necessity of .
For instance, maintaining an accurate IB can enhance the success rates of , with Phase III completion rates rising to 66% when local compliance knowledge is utilized. Moreover, professional perspectives emphasize that a well-maintained not only ensures adherence but also safeguards participant welfare by providing the and protocols.
in the revision process can streamline updates and mitigate potential risks, reinforcing the commitment to and research integrity.
Submission Process: Preparing the Investigator’s Brochure for Regulatory Review
Preparing the for regulatory review demands meticulous attention to detail and strict adherence to established guidelines. It is crucial to confirm that the is comprehensive, incorporating essential elements such as:
- Research objectives
Additionally, supporting documents, including the and , must be compiled and organized effectively. Engaging in and selecting qualified research sites and principal investigators (PIs) is vital at this stage to ensure compliance with country-specific requirements. Moreover, considering the trial arrangement, import authorizations, and project oversight is essential to facilitate a seamless compliance process.
Submitting a complete and well-organized package to the relevant oversight authority is imperative, as it can significantly reduce . Statistics reveal that applications receiving more substantial oversight had median of:
- 46 days for full reviews
- 84.5 days for those with less assistance
This underscores the importance of thorough preparation and adherence to legal standards to enable timely approval for , including acquiring necessary approvals from ethics committees and health ministries. Regular updates to the are equally essential to reflect any changes in the investigational product’s safety profile and recent research findings, ensuring that the document remains relevant and compliant.
Staff Training: Educating Researchers on the Investigator’s Brochure
Educating researchers about the is essential for ensuring that all team members grasp its contents and significance. Training sessions must comprehensively cover the , along with . Studies indicate that 60% of researchers express confidence in their understanding of the IB, underscoring the need for . Ongoing education is vital to keep staff updated on revisions and new information related to the , as it should be regularly updated to reflect changes in the investigational product’s safety profile and recent research findings.
By promoting a , research groups can significantly improve the quality and integrity of studies, ultimately resulting in enhanced patient safety and study effectiveness. As Sarah Lee observed, ‘The is an essential document that describes the details of the investigational product, including its composition, pharmacology, toxicology, and research data.’
Furthermore, the number of participants achieving increased from 17 to 33 following the , illustrating the effectiveness of training programs. The should also be shared with all relevant stakeholders, ensuring that everyone involved in the process has access to the most current information.
bioaccess: Streamlining Investigator’s Brochure Development for Clinical Trials
bioaccess® excels in enhancing the creation of for research studies, ensuring that all are meticulously included. Our group of experienced experts is committed to and tackling ethical issues, which are essential for the .
With more than 20 years of experience in the Medtech field, we utilize our extensive knowledge of the clinical research environment to create high-quality that not only assist in gaining approval but also enhance the overall success rates of . Research shows that effective IB development can greatly enhance testing results, with success rates for tests facilitated by bioaccess® exceeding industry averages.
By incorporating comprehensive details and adhering to the latest , including guidelines outlined in Resolution No. 466/2012, we enable researchers to of with confidence. Our commitment to excellence in the creation of the investigational brochure is further underscored by our proactive approach to integrating feedback and updates, ensuring that each brochure remains relevant and aligned with evolving research needs.
Additionally, we provide thorough review and feedback on study documents to comply with country requirements, enhancing the overall quality of the trial process.
Common Challenges: Navigating the Development of an Investigator’s Brochure
Creating an presents various challenges, including the necessity for thorough , , and the . A significant hurdle lies in effectively communicating complex information in a manner accessible to all stakeholders. Notably, research indicates that 63% of participants are familiar with the CTFG Q&A document; however, many still find the summary of data and guidance for the investigator (Section 7) in need of improvement.
To navigate these challenges, establishing among team members is crucial. Engaging can enhance the quality of the , as evidenced by 55% of participants . Continuous review and revision of the is essential for maintaining its relevance and accuracy as new information emerges.
Strategies such as utilizing can significantly improve documentation quality, ensuring that all required elements are included and enhancing compliance. By proactively addressing these typical obstacles, researchers can create a well-organized that facilitates the effective execution of research studies.
Collaboration: Engaging Sponsors and Investigators in Investigator’s Brochure Development
in the development of the (IB) is essential for capturing diverse perspectives and ensuring . This collaboration fosters a more robust IB that effectively addresses the needs and concerns of all stakeholders involved.
To facilitate this collaboration, regular meetings and are vital. These interactions not only allow for the but also enable , ultimately enhancing the quality and effectiveness of the IB.
By actively involving sponsors and investigators in the IB process, organizations can significantly improve the of the information presented, leading to better in .
Conclusion
The investigational brochure (IB) is an essential document that plays a pivotal role in the success of clinical trials. It serves as a comprehensive resource, outlining the investigational product and its associated risks while ensuring that all stakeholders are informed and aligned throughout the research process. The significance of a well-constructed IB cannot be overstated, as it directly impacts participant safety, regulatory compliance, and the overall integrity of the clinical study.
Key elements of an effective investigational brochure have been explored, including:
- The necessity for regulatory compliance
- Ethical considerations
- Essential components to include
- The importance of regular updates
By adhering to established guidelines and fostering collaboration among sponsors and investigators, researchers can create a robust IB that enhances communication and facilitates a smoother approval process. Evidence highlights that a meticulously crafted IB not only aids in meeting regulatory requirements but also contributes to higher success rates in clinical trials.
In light of these insights, it is imperative for researchers and organizations involved in clinical trials to prioritize the development and maintenance of the investigational brochure. By investing time and resources into creating a high-quality IB, stakeholders can ensure compliance with regulatory standards and demonstrate their commitment to safeguarding participant welfare. Embracing best practices in IB development will ultimately lead to more effective clinical trials, improved outcomes, and a stronger foundation for future medical advancements.
Frequently Asked Questions
What is an Investigator’s Brochure (IB)?
The Investigator’s Brochure is a pivotal document that summarizes essential information about an investigational product, including its pharmacological characteristics, preclinical data, and study protocols. It serves as a vital reference for investigators and regulatory authorities.
Why is a well-crafted Investigator’s Brochure important?
A meticulously crafted Investigator’s Brochure is essential for the effective initiation and execution of research studies. It lays the groundwork for informed decision-making and helps prevent delays or shutdowns in studies, as insufficient brochures contribute to approximately 80% of such issues.
What are the key regulatory compliance requirements for an Investigator’s Brochure?
The Investigator’s Brochure must adhere to regulations established by authorities like the FDA and EMA, which include a clear description of the investigational product, detailed information on the research design, and a comprehensive risk evaluation.
What essential components should be included in an Investigator’s Brochure?
Essential components include:
- Summary of the Investigational Product
- Preclinical and Clinical Data
- Research Objectives and Methodology
- Potential Risks
- Informed Consent Process
- Roles and Responsibilities
- Contact Information
How often should the Investigator’s Brochure be updated?
The Investigator’s Brochure should be reviewed and updated at least annually or whenever significant new data is received to ensure its relevance.
What role do comprehensive research management services play in creating an Investigator’s Brochure?
Comprehensive research management services, such as feasibility studies, site selection, compliance reviews, testing setup, and project management, support the creation of a successful Investigator’s Brochure and ensure alignment with regulatory requirements.
What is the typical length of a successful Investigator’s Brochure?
Successful Investigator’s Brochures typically contain around 80 to 100 pages, balancing detail with conciseness for effective communication.
What should be submitted alongside the Investigator’s Brochure to regulatory authorities?
The Clinical Trial Application should be submitted along with the Investigator’s Brochure to national competent authorities.
How can Bioaccess assist with the Investigator’s Brochure process?
Bioaccess offers expertise in study setup, import permits, project management, and reporting, which are essential for navigating the complexities of research studies and ensuring compliance with regulatory requirements.
List of Sources
- Investigator’s Brochure: Core Document for Clinical Trials
- antidote.me (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC7857342)
- The Investigator’s Brochure – A multidisciplinary document – Trilogy Writing & Consulting (https://trilogywriting.com/document/investigators-brochure-multidisciplinary-document)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC5886385)
- bioaccessla.com (https://bioaccessla.com/blog/9-essential-elements-for-your-investigators-brochure)
- Regulatory Compliance: Key Requirements for Investigator’s Brochure
- bioaccessla.com (https://bioaccessla.com/blog/9-essential-elements-for-your-investigators-brochure)
- numberanalytics.com (https://numberanalytics.com/blog/ultimate-guide-investigators-brochure-clinical-trials)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC6662388)
- editverse.com (https://editverse.com/?p=36121)
- Essential Components: What to Include in an Investigator’s Brochure
- vaia.com (https://vaia.com/en-us/explanations/medicine/pharmacy/investigators-brochure)
- toolbox.eupati.eu (https://toolbox.eupati.eu/resources/investigators-brochure)
- What is an Investigator’s Brochure? | Novotech CRO (https://novotech-cro.com/faq/what-investigators-brochure)
- celero.global (https://celero.global/blog/investigator-brochure)
- editverse.com (https://editverse.com/?p=36121)
- Ethical Considerations: Informing Participants Through the Investigator’s Brochure
- abbvieclinicaltrials.com (https://abbvieclinicaltrials.com/resources/risks-and-benefits-of-participating-in-a-research-study)
- spirit-statement.org (https://spirit-statement.org/ethics-and-dissemination-24-31)
- Mastering Patient Recruitment in Clinical Trials (https://clinicalleader.com/topic/patient-recruitment-and-enrollment)
- Participants’ understanding of informed consent in clinical trials: A systematic review and updated meta-analysis (https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0295784)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC7263438)
- Updates and Revisions: Maintaining Accuracy in the Investigator’s Brochure
- bioaccessla.com (https://bioaccessla.com/blog/9-essential-elements-for-your-investigators-brochure)
- Investigator’s Brochure Writing: 5 Tips to Streamline Your SOC (https://certara.com/blog/5-tips-for-authoring-an-investigators-brochure-summary-of-changes)
- linkedin.com (https://linkedin.com/pulse/update-investigators-brochure-tim-hardman)
- research.colostate.edu (https://research.colostate.edu/safety-and-compliance/qa/investigators-brochure)
- Submission Process: Preparing the Investigator’s Brochure for Regulatory Review
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S2452302X1600036X)
- Effects of Regulatory Support Services on Institutional Review Board Turnaround Times – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC5546085)
- bioaccessla.com (https://bioaccessla.com/blog/9-essential-elements-for-your-investigators-brochure)
- Staff Training: Educating Researchers on the Investigator’s Brochure
- bioaccessla.com (https://bioaccessla.com/blog/9-essential-elements-for-your-investigators-brochure)
- Clinical Investigator Training Program (CITP) – A practical and pragmatic approach to conveying clinical investigator competencies and training to busy clinicians (https://sciencedirect.com/science/article/pii/S2451865420300739)
- qualtrics.com (https://qualtrics.com/blog/research-quotes)
- numberanalytics.com (https://numberanalytics.com/blog/ultimate-guide-investigator-brochures-ophthalmic-trials)
- bioaccess: Streamlining Investigator’s Brochure Development for Clinical Trials
- bioaccessla.com (https://bioaccessla.com/blog/designing-trials-for-approval-in-brazil-an-in-depth-tutorial-for-clinical-researchers)
- bioaccessla.com (https://bioaccessla.com/blog/9-essential-elements-for-your-investigators-brochure)
- Common Challenges: Navigating the Development of an Investigator’s Brochure
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC7857342)
- qualtrics.com (https://qualtrics.com/blog/research-quotes)
- bioaccessla.com (https://bioaccessla.com/blog/9-essential-elements-for-your-investigators-brochure)
- freyrsolutions.com (https://freyrsolutions.com/blog/investigators-brochure-ib-and-challenges-for-medical-writers)
- Collaboration: Engaging Sponsors and Investigators in Investigator’s Brochure Development
- bioaccessla.com (https://bioaccessla.com/blog/9-essential-elements-for-your-investigators-brochure)
- BENEFITS OF COLLABORATING – Extending the Spectrum of Precompetitive Collaboration in Oncology Research – NCBI Bookshelf (https://ncbi.nlm.nih.gov/books/NBK210038)
- 60 Collaboration Quotes: Inspiring Teamwork & Business Success | ITD World (https://itdworld.com/blog/human-resources/collaboration-quotes)
- inc.com (https://inc.com/gordon-tredgold/50-quotes-on-the-importance-and-benefits-of-teamwork.html)
- teamwork.com (https://teamwork.com/blog/25-quotes-inspire-collaboration)

Leave a Reply