Introduction
Ensuring the safety and efficacy of medical devices is paramount in the healthcare industry, where even the slightest oversight can lead to dire consequences. Cleaning validation stands at the forefront of this crucial process, serving as a systematic approach to verify that sanitation procedures effectively eliminate contaminants that could jeopardize patient health. As regulatory scrutiny intensifies, manufacturers face the pressing challenge of navigating complex compliance landscapes while implementing best practices for cleaning validation.
How can organizations balance the need for rigorous sanitation with the demands of efficiency and regulatory adherence, all while safeguarding their reputation and ensuring patient safety? This question underscores the critical intersection of compliance and operational efficiency in the Medtech landscape. By addressing these challenges head-on, organizations can not only meet regulatory requirements but also enhance their commitment to patient safety.
Define Cleaning Validation and Its Importance in Medical Devices
is a systematic and documented process that verifies the effectiveness of through for medical devices. This procedure is crucial for ensuring that all residues, contaminants, and microorganisms are reduced to levels that do not compromise or interfere with subsequent sterilization methods. The significance of cannot be overstated; it is a fundamental requirement for and plays a vital role in preventing cross-contamination, ensuring quality, and maintaining the integrity of .
For example, the FDA requires manufacturers to demonstrate that their sanitation methods, including , consistently produce devices that meet established cleanliness standards. This is not just a regulatory formality; it is essential for protecting patient health and enhancing the credibility of the medical device industry. Recent updates from the FDA underscore the necessity of , with statistics indicating that inadequate hygiene can lead to serious health risks and product recalls. In fact, in 2022, the FDA issued 42 warning letters regarding deficiencies in sanitation verification, highlighting the .
Industry leaders stress that involves balancing thoroughness with efficiency. As one expert noted, ” is far more than a technical formality; it is essential to ensuring quality and .” This perspective underscores the need for meticulous documentation and tailored sanitation methods that address specific product risks, ensuring compliance and safeguarding patient health through .
Understand Regulatory Standards for Cleaning Validation
Regulatory standards for are established by various organizations, including the , ISO, and EMA. A cornerstone of these regulations is the ‘s , which specifies the in manufacturing processes. This regulation mandates that equipment and utensils must be regularly cleaned, maintained, and sanitized to that could compromise drug safety, quality, or purity. Furthermore, offers extensive guidelines for the processing of medical devices, highlighting the importance of and validated cleaning methods designed for specific items.
Adherence to these standards is not merely a regulatory formality; it is essential for protecting integrity and ensuring patient safety. Non-compliance can lead to serious consequences, including recalls, legal repercussions, and substantial harm to a company’s reputation. For instance, in fiscal year 2022, the issued 42 Warning Letters to pharmaceutical manufacturers citing associated with sanitation checks, underscoring the increased scrutiny during inspections. The presence of noticeable residues or contamination can trigger inquiries, as seen in cases like Chem-Tech, where a disastrous failure in sanitation procedures resulted in a warning letter due to visible dirt and remaining pharmaceutical substance.
Moreover, the ‘s emphasis on has intensified, enforcing a between industrial and pharmaceutical items. This regulatory environment necessitates that manufacturers implement strict and maintain thorough documentation of sanitation activities, sampling, and testing outcomes. By adhering to these guidelines, companies can mitigate risks and ensure compliance, ultimately safeguarding their products and reputation in the marketplace.
Implement Effective Cleaning Validation Methodologies
To implement effective methodologies for , manufacturers must adopt a structured approach that encompasses several critical steps.
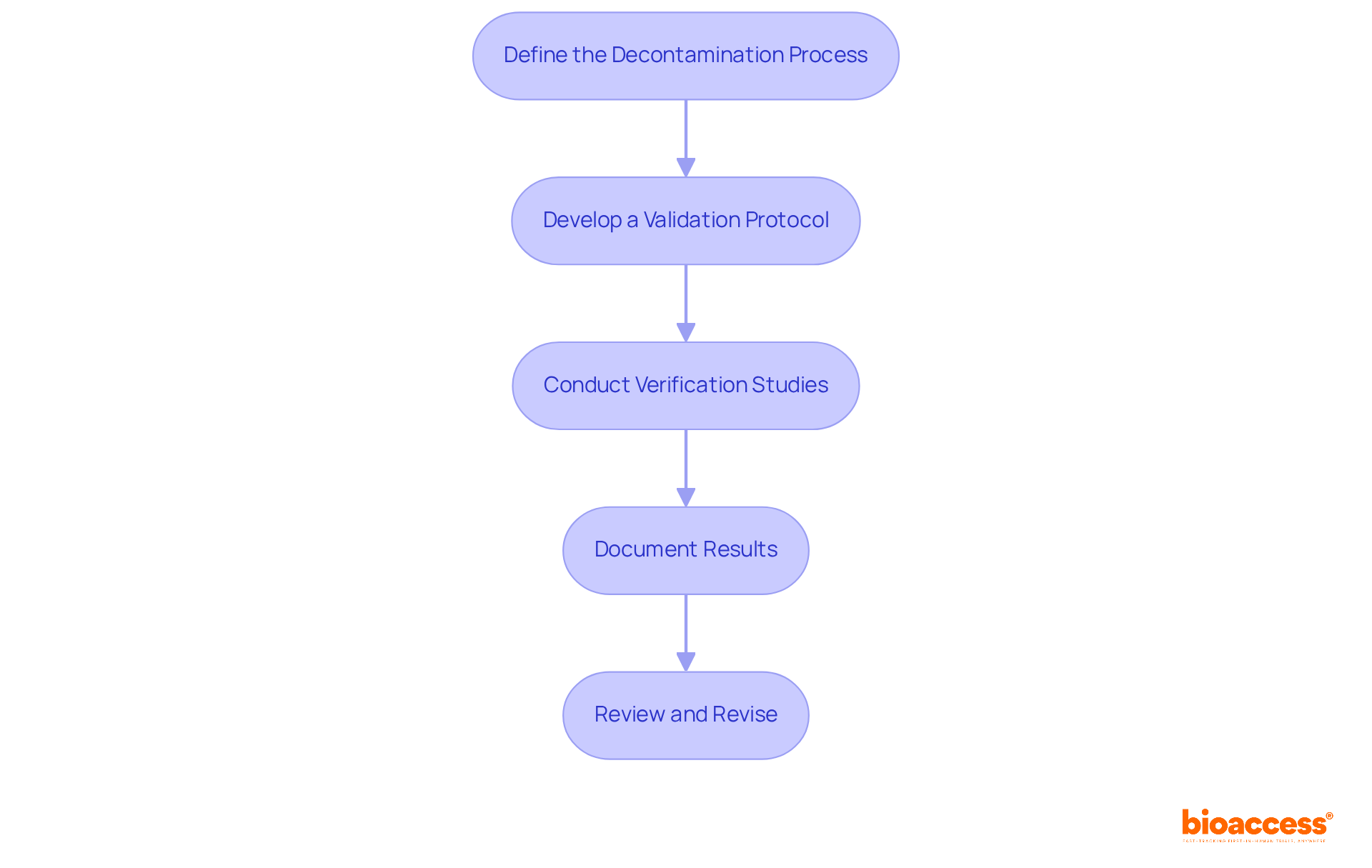
- Define the : Clearly outline the agents, equipment, and procedures utilized. This essential step in ensures that all elements of the sanitation procedure are comprehended and documented, thereby laying the groundwork for effective verification.
- Develop a : Create a comprehensive protocol detailing acceptance criteria, sampling methods, and . This protocol serves as a guide for the process, ensuring adherence to and fostering confidence in the results.
- Conduct : Execute cleaning using representative soil loads and conditions to confirm the effectiveness of the cleaning process. Advanced , such as High-Performance Liquid Chromatography (HPLC), Gas Chromatography (GC), and Atomic Absorption Spectroscopy (AAS), are essential for as they significantly enhance the detection of residues, ensuring compliance with stringent cleanliness standards.
- Document Results: Maintain meticulous records of all verification activities, including test outcomes and any deviations encountered. This documentation is vital for , especially in light of the FDA sending 42 warning letters to manufacturers in 2022 regarding deficiencies. It also provides a clear audit trail for future reference.
- Review and Revise: Regularly evaluate and modify hygiene verification protocols in response to new regulations or technological advancements. Continuous improvement in sanitation verification methods, particularly in , is crucial for preserving product integrity and ensuring patient safety. Organizations that adopt these best practices in often report higher success rates in their cleaning assessments, which leads to enhanced compliance and reduced risk profiles. Investing in cleaning and process confirmation has been shown to result in fewer deviations and significantly lower risk profiles.
Address Challenges in Cleaning Validation and Solutions
Challenges in pose significant hurdles for manufacturers, which are crucial to address in the context of .
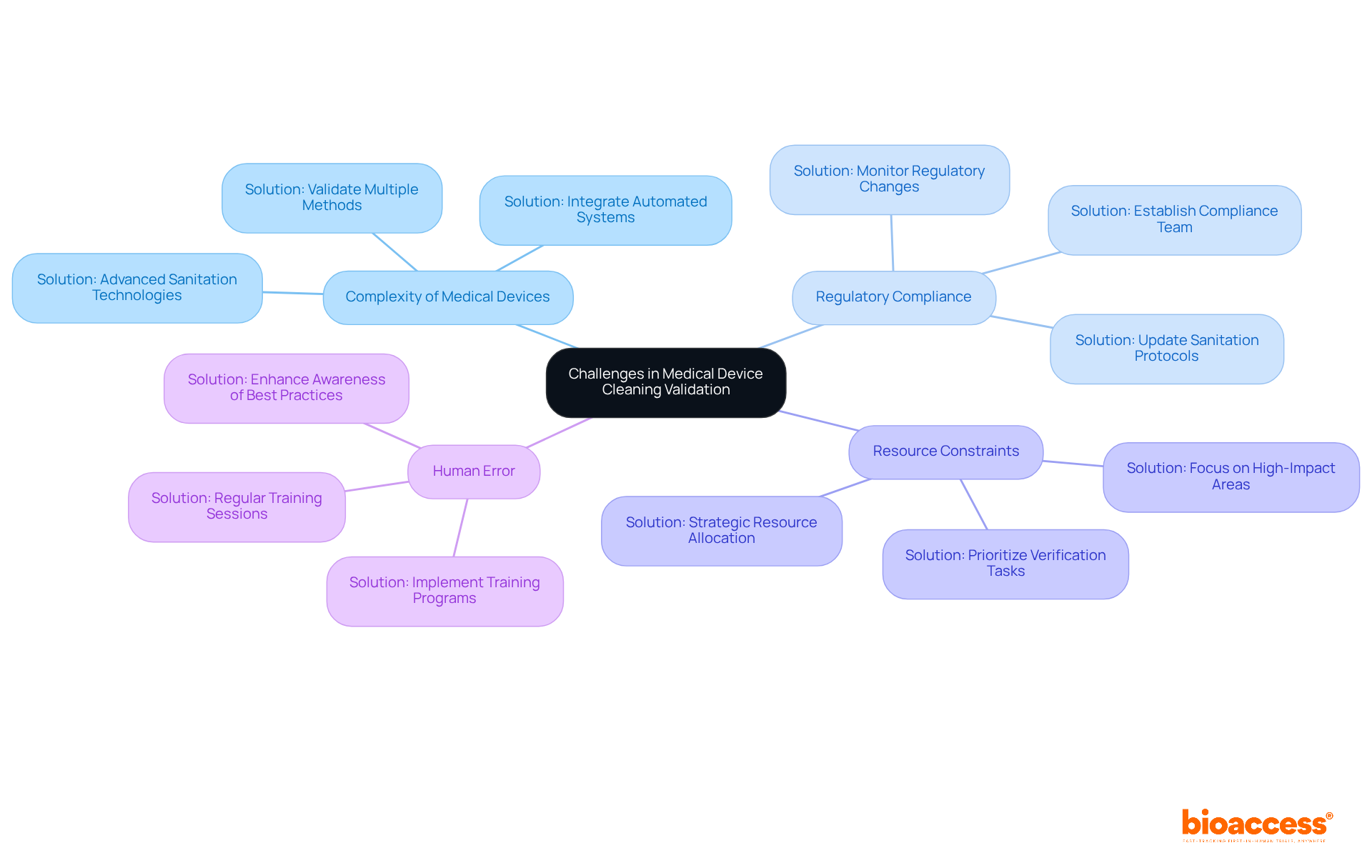
- Complexity of Medical Devices: Many devices feature intricate geometries that complicate the sanitization process. Solution: Employ and validate multiple methods to ensure effectiveness. For instance, integrating automated systems can significantly enhance sanitation efficiency and consistency. Industry reports indicate that the is projected to grow from USD 1.2 billion in 2024 to USD 2.5 billion by 2033, highlighting the increasing importance of effective sanitization verification practices.
- : Navigating evolving regulations can be daunting for manufacturers. Solution: Establish a dedicated compliance team to monitor regulatory changes and update accordingly. This proactive approach mitigates risks associated with , which can lead to costly delays and penalties. According to the European Centre for Disease Prevention and Control, around 4.3 million patients are affected by , underscoring the critical need for stringent compliance.
- Resource Constraints: Limited time and budget can hinder comprehensive sanitation verification efforts. Solution: Prioritize sanitation verification tasks based on risk evaluations, focusing on high-impact areas that pose the greatest threat to patient safety and product integrity. This strategic allocation of resources enhances verification processes without compromising quality. The rising incidence of HAIs adds an estimated USD 25 billion to U.S. healthcare costs annually, emphasizing the necessity for .
- Human Error: Inconsistent maintenance practices can lead to . Solution: Implement robust training programs for staff to ensure adherence to and enhance awareness of best practices. Regular training sessions can significantly reduce variability in sanitation practices, thereby improving overall compliance. A case study on North America’s market illustrates how effective training and compliance measures have resulted in improved outcomes in infection control.
By effectively addressing these challenges, manufacturers can streamline their processes, ensuring compliance with regulatory standards while enhancing product safety and efficacy.
Conclusion
Ensuring the effectiveness of cleaning validation in medical devices is not just a regulatory obligation; it’s a cornerstone of patient safety and product integrity. A comprehensive understanding of cleaning validation, along with its pivotal role in compliance with standards set by organizations like the FDA and ISO, underscores its significance in the medical device industry. Manufacturers must adopt robust cleaning validation practices to safeguard against contamination, uphold quality, and maintain the trust of healthcare providers and patients alike.
Key insights from the article highlight the necessity of:
- Defining cleaning processes
- Developing thorough validation protocols
- Conducting rigorous verification studies
By addressing challenges such as the complexity of medical devices, evolving regulatory landscapes, and resource constraints, manufacturers can implement effective methodologies that enhance compliance and minimize risks. Continuous training and adaptation to new technologies further ensure that sanitation practices are not only compliant but also efficient and effective.
In light of these considerations, a commitment to stringent cleaning validation practices will yield significant benefits, including:
- Reduced risk of product recalls
- Improved patient outcomes
The medical device industry must prioritize these best practices to navigate the complexities of compliance and enhance overall safety in healthcare. Engaging in proactive cleaning validation strategies is not merely a regulatory necessity; it is a vital step towards fostering a safer medical environment for all.
Frequently Asked Questions
What is cleaning validation in the context of medical devices?
Cleaning validation is a systematic and documented process that verifies the effectiveness of sanitation procedures for medical devices, ensuring that residues, contaminants, and microorganisms are reduced to safe levels.
Why is cleaning validation important for medical devices?
Cleaning validation is crucial for regulatory compliance, preventing cross-contamination, ensuring quality, and maintaining the integrity of clinical trials, all of which are essential for patient safety.
What regulatory body requires manufacturers to demonstrate cleaning validation?
The FDA (Food and Drug Administration) requires manufacturers to demonstrate that their sanitation methods, including cleaning validation, consistently produce devices that meet established cleanliness standards.
What are the consequences of inadequate cleaning validation?
Inadequate cleaning validation can lead to serious health risks, product recalls, and regulatory actions, as evidenced by the FDA issuing 42 warning letters in 2022 regarding deficiencies in sanitation verification.
How do industry leaders view the role of cleaning validation?
Industry leaders emphasize that cleaning validation is essential for ensuring quality and patient safety, requiring meticulous documentation and tailored sanitation methods to address specific product risks.
List of Sources
- Define Cleaning Validation and Its Importance in Medical Devices
- Statistics in Cleaning Validation? (https://cleaningvalidation.com/memos/statistics-for-cleaning-validation-january-2021)
- What is “cleaning validation” for medical devices? (https://cleancontrolling.com/en/news/newsdetails/what-is-cleaning-validation-for-medical-devices)
- The Importance of Cleaning Validation | QMS Templates (https://qmsdoc.com/2026/01/13/the-importance-of-cleaning-validation)
- Cleaning Validation: Increasingly Under the Regulatory Spotlight (https://bioprocessintl.com/validation/cleaning-validation-increasingly-under-the-regulatory-spotlight)
- The Importance of Cleaning Validation | Epista (https://epista.com/knowledge/the-importance-of-cleaning-validation)
- Understand Regulatory Standards for Cleaning Validation
- Cleaning in 6th Place in the FDA Warning Letter Statistics – ECA Academy (https://gmp-compliance.org/gmp-news/cleaning-in-6th-place-in-the-fda-warning-letter-statistics)
- Pharmaceutical Cleaning Validation Market | Global Market Analysis Report – 2035 (https://futuremarketinsights.com/reports/pharmaceutical-cleaning-validation-market)
- Cleaning Validation: 8 Steps for FDA Compliance Success – Zamann Pharma Support GmbH (https://zamann-pharma.com/2024/07/17/cleaning-validation-7-steps-for-fda-compliance-success)
- Cleaning Validation: What Warning Letters Reveal About FDA Priorities (https://linkedin.com/pulse/cleaning-validation-what-warning-letters-reveal-fda-uday-shetty-zmyac)
- Implement Effective Cleaning Validation Methodologies
- 8 Essential Steps for Effective Cleaning and Process Validation (https://avslifesciences.com/blog-post/8-essential-steps-for-effective-cleaning-and-process-validation)
- Statistics in Cleaning Validation? (https://cleaningvalidation.com/memos/statistics-for-cleaning-validation-january-2021)
- Cleaning Validation: 8 Steps for FDA Compliance Success (https://linkedin.com/pulse/cleaning-validation-8-steps-fda-compliance-success-sagar-pawar-7sotf)
- The Ultimate Guide to Cleaning Validation (https://pritchardindustries.com/cleaning-validation)
- Choosing the Right Analytical Methods for Cleaning Validation (https://pharmallies.com/choosing-the-right-analytical-methods-for-cleaning-validation)
- Address Challenges in Cleaning Validation and Solutions
- Medical Device Cleaning Market Size, Growth Trends & Industry Analysis, 2031 (https://mordorintelligence.com/industry-reports/medical-device-cleaning-market)
- Pharmaceutical Cleaning Validation Market Size & Competitors (https://researchandmarkets.com/report/cleaning-validation-market?srsltid=AfmBOor6o5DVA4q6fxg-9PnU556HkxwJwIJLGQtujUixtX062kh82VGI)
- Global Medical Device Cleaning Validation Market Outlook, In‑Depth Analysis & Forecast to 2031 (https://qyresearch.com/reports/5136798/medical-device-cleaning-validation)
- Medical Device Cleaning Validation Market’s Drivers and Challenges: Strategic Overview 2026-2034 (https://datainsightsmarket.com/reports/medical-device-cleaning-validation-1988137)
- Medical Device Cleaning Validation Market Size, Emerging Trends, Smart Automation & Regions 2026-33 (https://linkedin.com/pulse/medical-device-cleaning-validation-market-size-r9clf)

Leave a Reply