Introduction
Innovations in healthcare are rapidly transforming the landscape of patient care, especially through the integration of drug-device combination products. These groundbreaking solutions not only enhance treatment efficacy but also streamline the management of chronic conditions, offering patients a more cohesive approach to their health. However, as these technologies evolve, it’s crucial to consider the challenges and complexities that arise in their implementation and effectiveness.
What are the most innovative drug-device combinations currently shaping the future of healthcare? How can they address the pressing needs of patients and healthcare providers alike? These questions are essential as we navigate the evolving Medtech landscape, where collaboration and innovation are key to overcoming obstacles and improving patient outcomes.
bioaccess®: Accelerating Drug-Device Combination Innovations in Healthcare
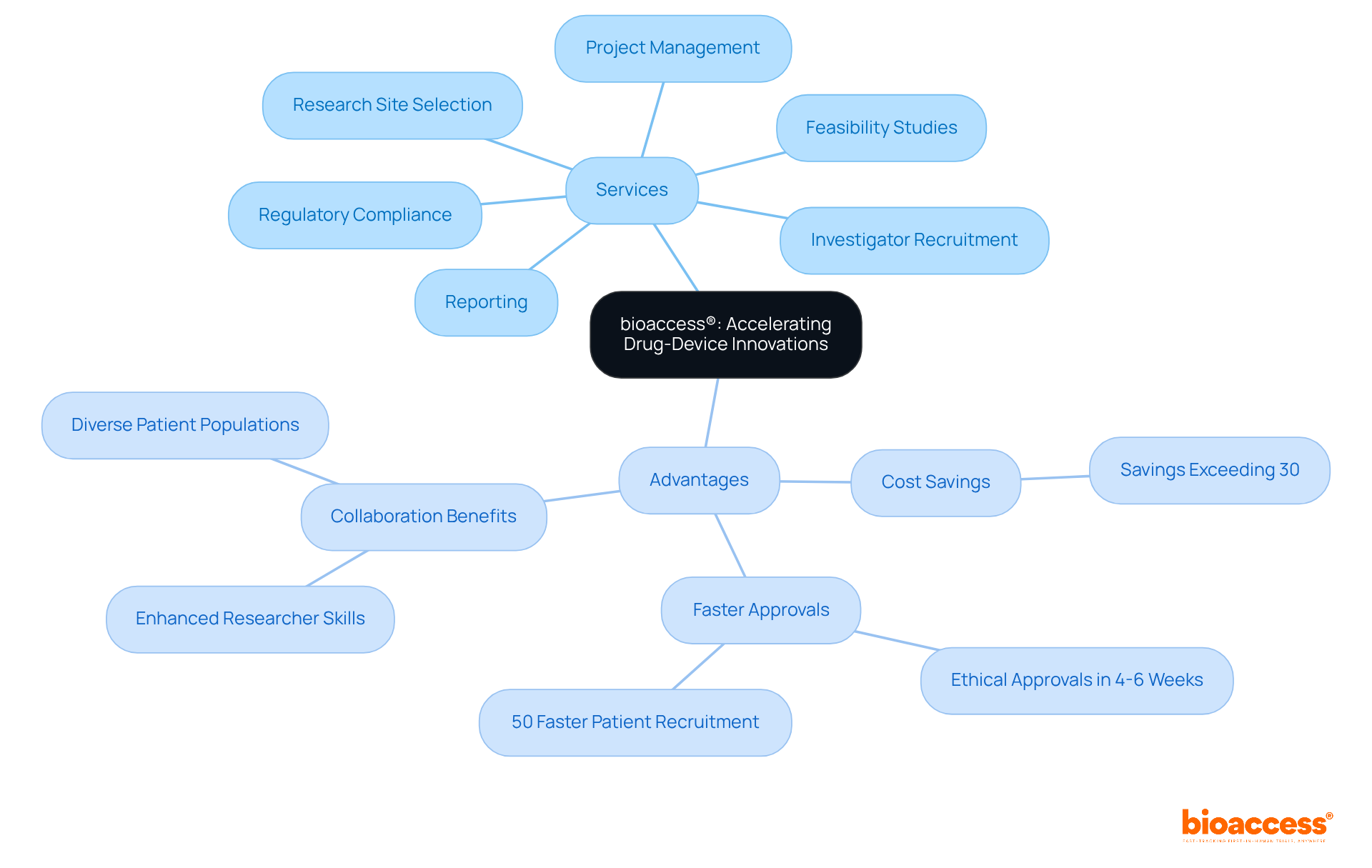
bioaccess® leverages over 15 years of to accelerate the development of drug-device combination products. With a keen focus on Latin America’s —especially in Colombia, where the total IRB/EC and MoH (INVIMA) review takes just 90-120 days—bioaccess® is committed to ensuring that innovative healthcare solutions reach the market more quickly. Our extensive services encompass:
- Research site selection
- Thorough reporting on study status and adverse events
In a landscape where and enrollment is expedited by 50%, bioaccess® is at the forefront of driving significant advancements in medical technology. This strategic approach not only enhances the speed of introducing new therapies to patients but also highlights the vital role of and —offering compared to trials in North America or Western Europe. Such advantages are crucial for Directors of Clinical Research as they design studies and aim for improved patient outcomes.
As we navigate the complexities of clinical research, collaboration becomes essential. By partnering with bioaccess®, stakeholders can harness our expertise to overcome key challenges in the Medtech landscape, ultimately leading to more effective and timely healthcare solutions.
Medtronic MiniMed 770G: Pioneering Insulin Delivery Systems
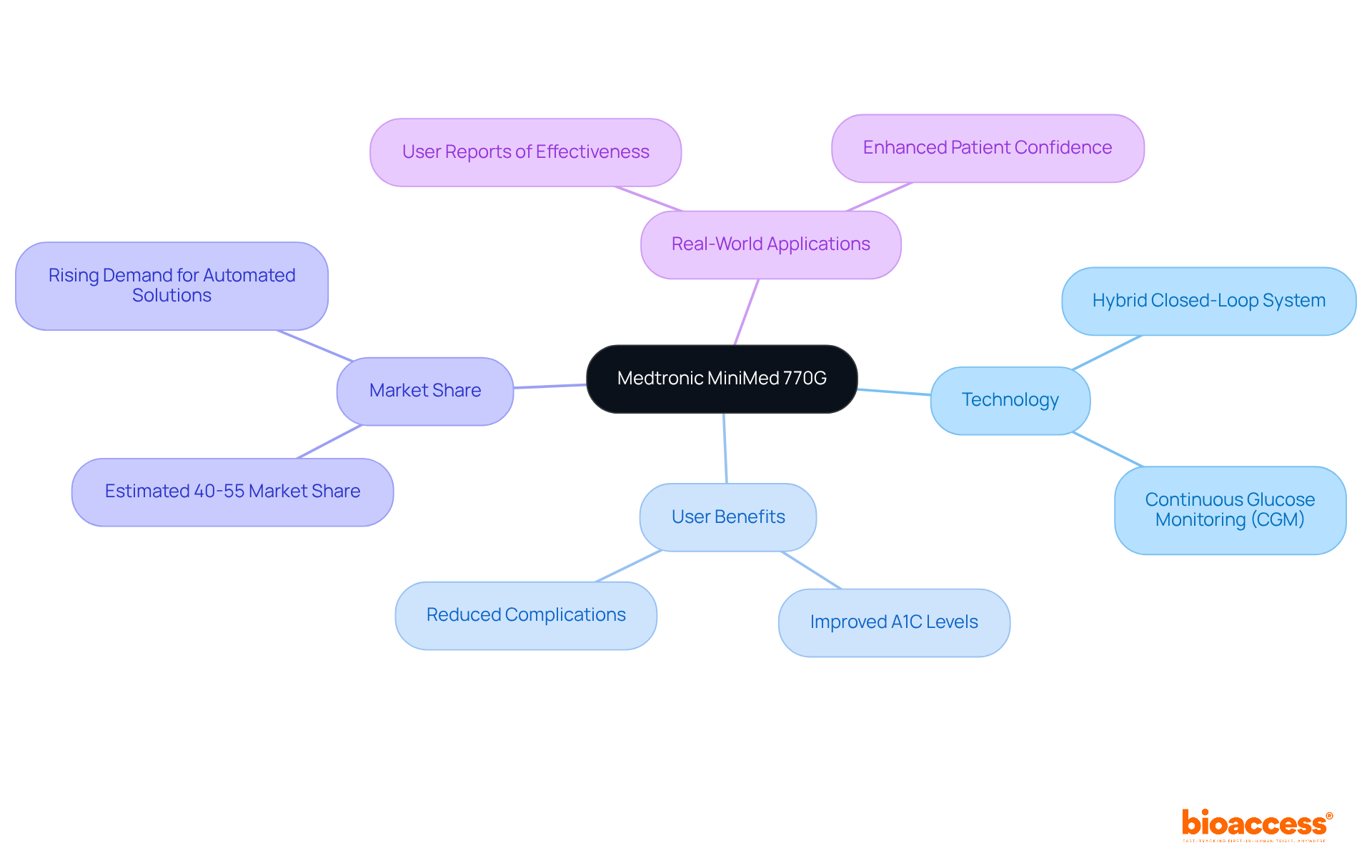
The Medtronic MiniMed 770G device represents a significant advancement in . As a hybrid closed-loop system, it autonomously adjusts insulin delivery based on real-time glucose readings, providing users with a tailored . This integration of greatly enhances glycemic control, effectively reducing the risks associated with both hyperglycemia and hypoglycemia. With an estimated market share of around 40-55% among leading , the MiniMed 770G illustrates the increasing trend towards .
Real-world applications underscore its effectiveness; users have reported and a decrease in diabetes-related complications. The innovative design of this setup not only simplifies insulin administration but also empowers patients to manage their condition with greater confidence and ease. As we navigate the evolving , the MiniMed 770G stands out as a beacon of progress, addressing key challenges in and enhancing .
Abbott FreeStyle Libre: Revolutionizing Glucose Monitoring
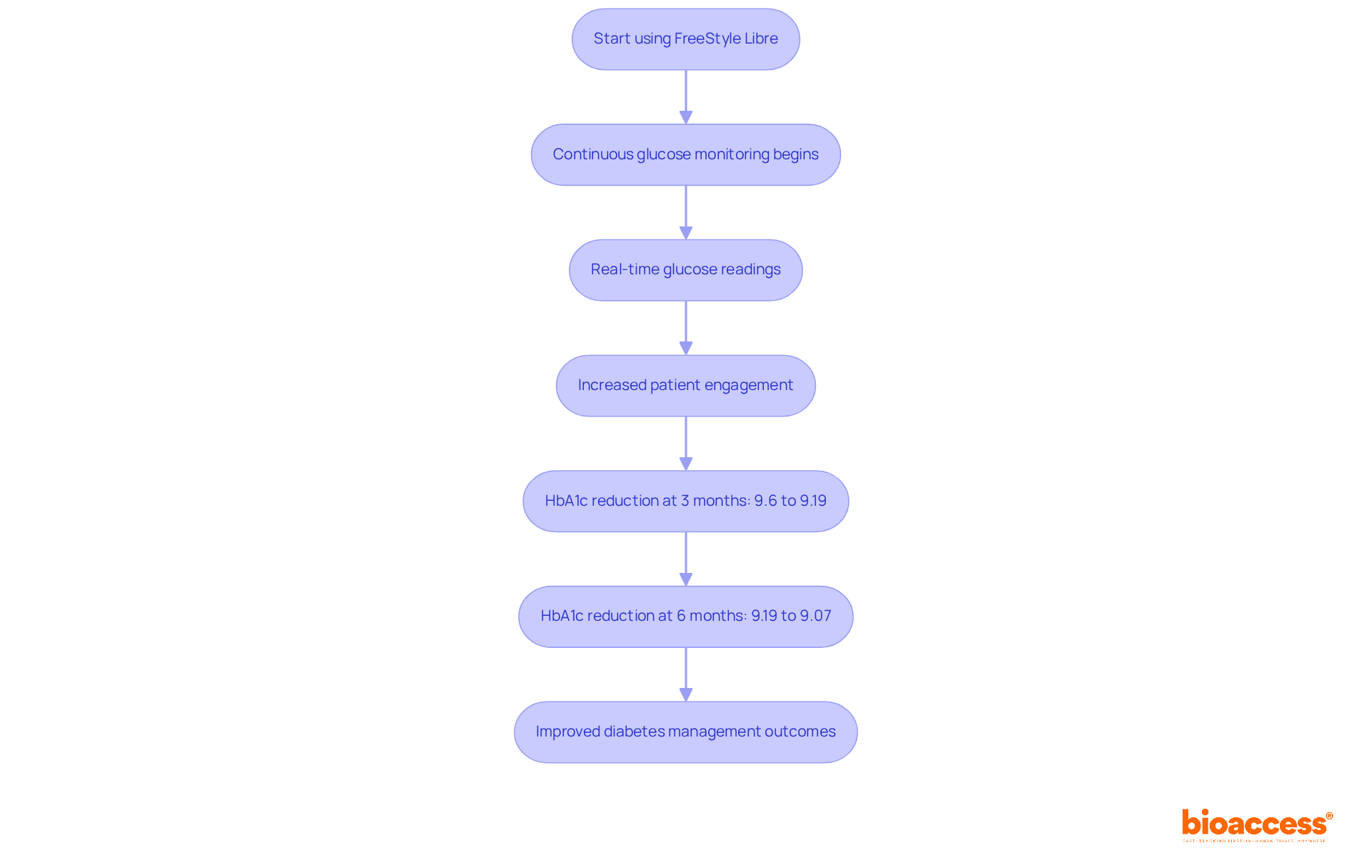
Abbott’s FreeStyle Libre device has revolutionized glucose monitoring, offering a that eliminates the need for routine fingersticks. Patients now enjoy through a small sensor worn on the skin, significantly enhancing their capabilities. The platform’s intuitive interface and seamless mobile app integration foster greater patient engagement and adherence to treatment plans.
Studies reveal that users of the experienced a median HbA1c reduction from 9.6% to 9.19% at three months, further decreasing to 9.07% at six months. This showcases the device’s effectiveness in . Moreover, the increased awareness of glucose levels has led to more frequent , with a substantial rise observed in the FreeStyle Libre group (p<0.001). This indicates a proactive strategy for managing diabetes.
This not only streamlines monitoring but also empowers individuals to take charge of their health, ultimately leading to improved outcomes in . Furthermore, with the anticipated launch of the FreeStyle Libre 3 system in February 2025, individuals can look forward to even greater advancements in glucose monitoring technology.
Boston Scientific’s Watchman: A Dual Approach to Stroke Prevention
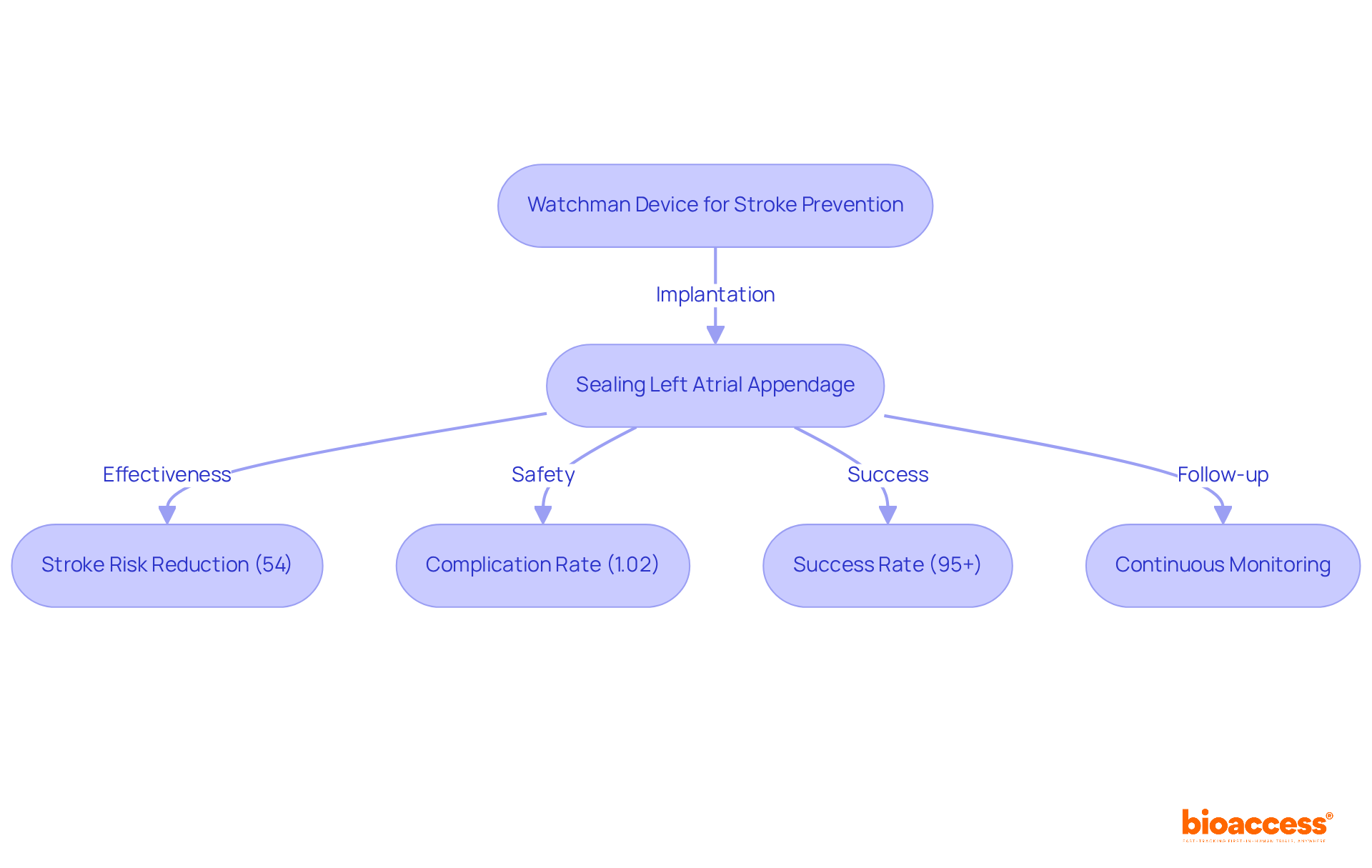
Boston Scientific’s Watchman product represents a significant advancement for individuals with at risk of stroke. By effectively sealing the left atrial appendage, where blood clots are likely to form, this implant reduces the risk of stroke substantially, offering a compelling alternative to . consistently demonstrate its efficacy, revealing a , making it a vital option for many patients.
Cardiologists have noted the device’s , with a procedural complication rate of just 1.02% for pericardial tamponade, underscoring its . However, potential complications such as bleeding and infection must also be taken into account.
The implantation procedure boasts a , highlighting both its effectiveness and safety. As innovations in atrial fibrillation treatment continue to evolve in 2025, the Watchman system stands out as a transformative solution, bolstered by robust that confirm its effectiveness in . Continuous monitoring following implantation is crucial to ensure the device functions correctly and to address any potential concerns.
As Dr. Chatani, a clinical proctor for Boston Scientific, states, ‘The Watchman system not only decreases the risk of stroke but also provides a for patients who cannot endure .
Stryker’s Neuroform Atlas: Enhancing Aneurysm Treatment
Stryker’s system is meticulously engineered for the , representing a significant advancement in . This sophisticated tool enhances support and wall apposition during , which is essential for achieving . Its unique design allows for superior navigation through complex vascular anatomy, making it a preferred choice among .
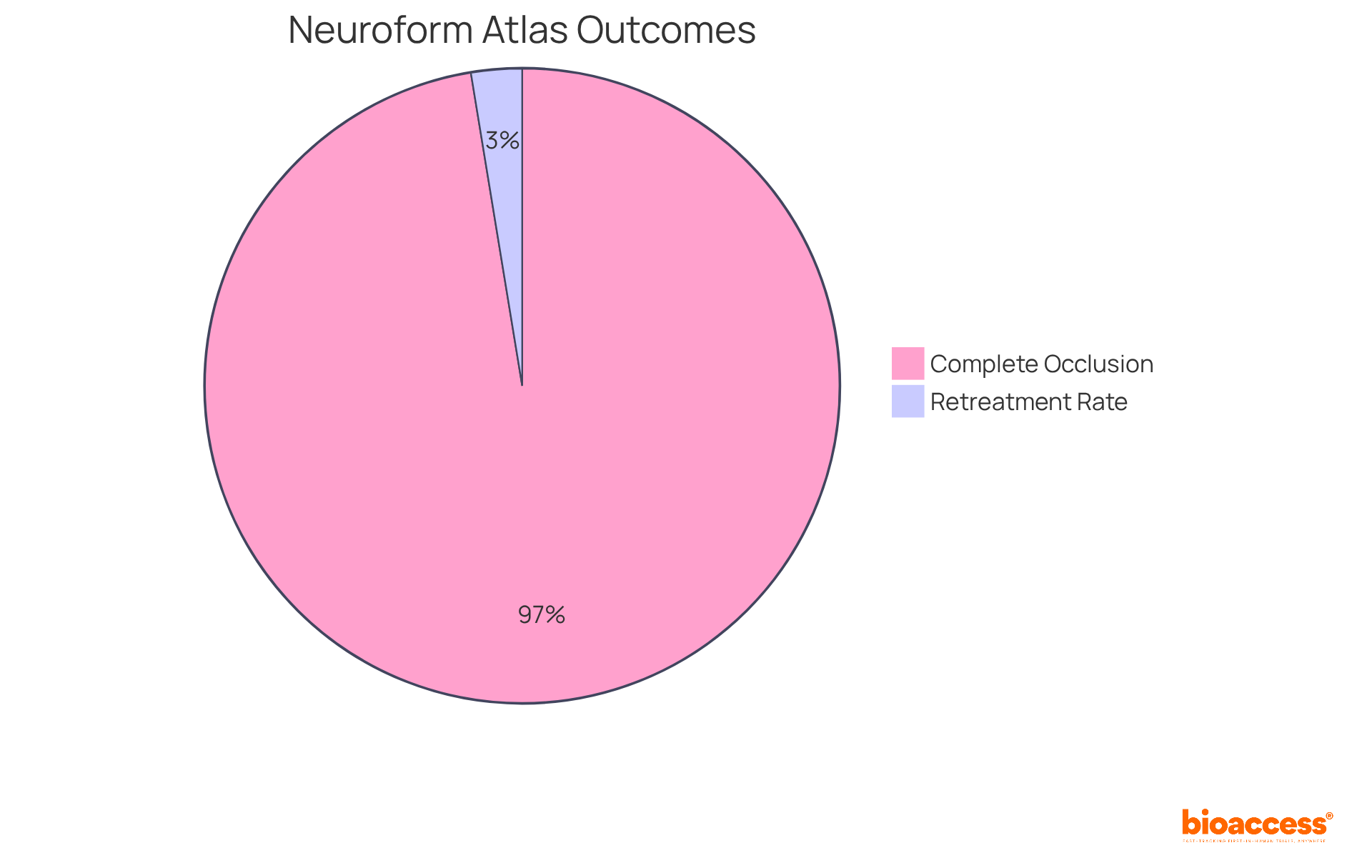
Recent studies reveal that the boasts an impressive complete occlusion rate of 90.3% at the 12-month follow-up, with 92.6% of individuals who completed the follow-up achieving the . Furthermore, the low retreatment rate of just 2.4% significantly outperforms traditional devices like the LVIS and WEB, underscoring its effectiveness in managing challenging aneurysm cases. As the demand for innovative neurovascular solutions continues to rise, the Neuroform Atlas emerges as a pivotal instrument in enhancing outcomes for patients and improving procedural success in the .
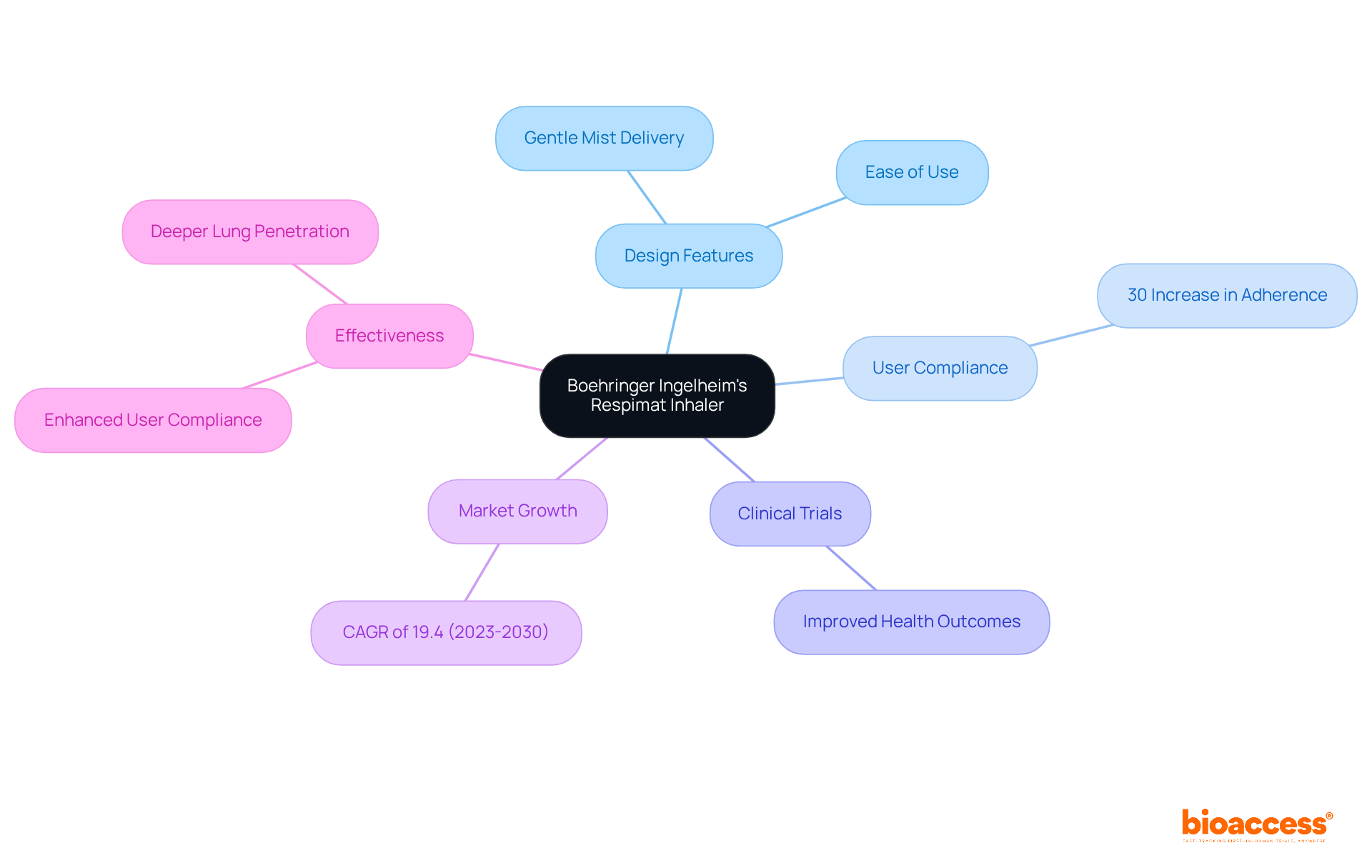
Boehringer Ingelheim’s Respimat: Transforming Inhalation Therapy
Boehringer Ingelheim’s Respimat inhaler represents a in , particularly for those dealing with like asthma and COPD. This gentle mist inhaler delivers medication in a fine mist, allowing for and enhancing user compliance. Its reduces the effort needed for inhalation, making it especially beneficial for individuals who may find traditional inhalers challenging to use.
have shown that patients utilizing the Respimat inhaler demonstrate , which is vital for effectively managing . Recent statistics reveal that the is projected to grow at a CAGR of 19.4% from 2023 to 2030, reflecting the increasing demand for cutting-edge inhalation devices. Respiratory specialists commend the device for its and its capacity to deliver consistent dosages, further establishing its significance in the realm of modern innovations.
For instance, a recent study indicated that individuals using the Respimat inhaler experienced a 30% increase in adherence compared to those using traditional inhalers. This underscores the inhaler’s effectiveness in improving outcomes for users, making it a compelling choice for those seeking reliable respiratory management solutions.
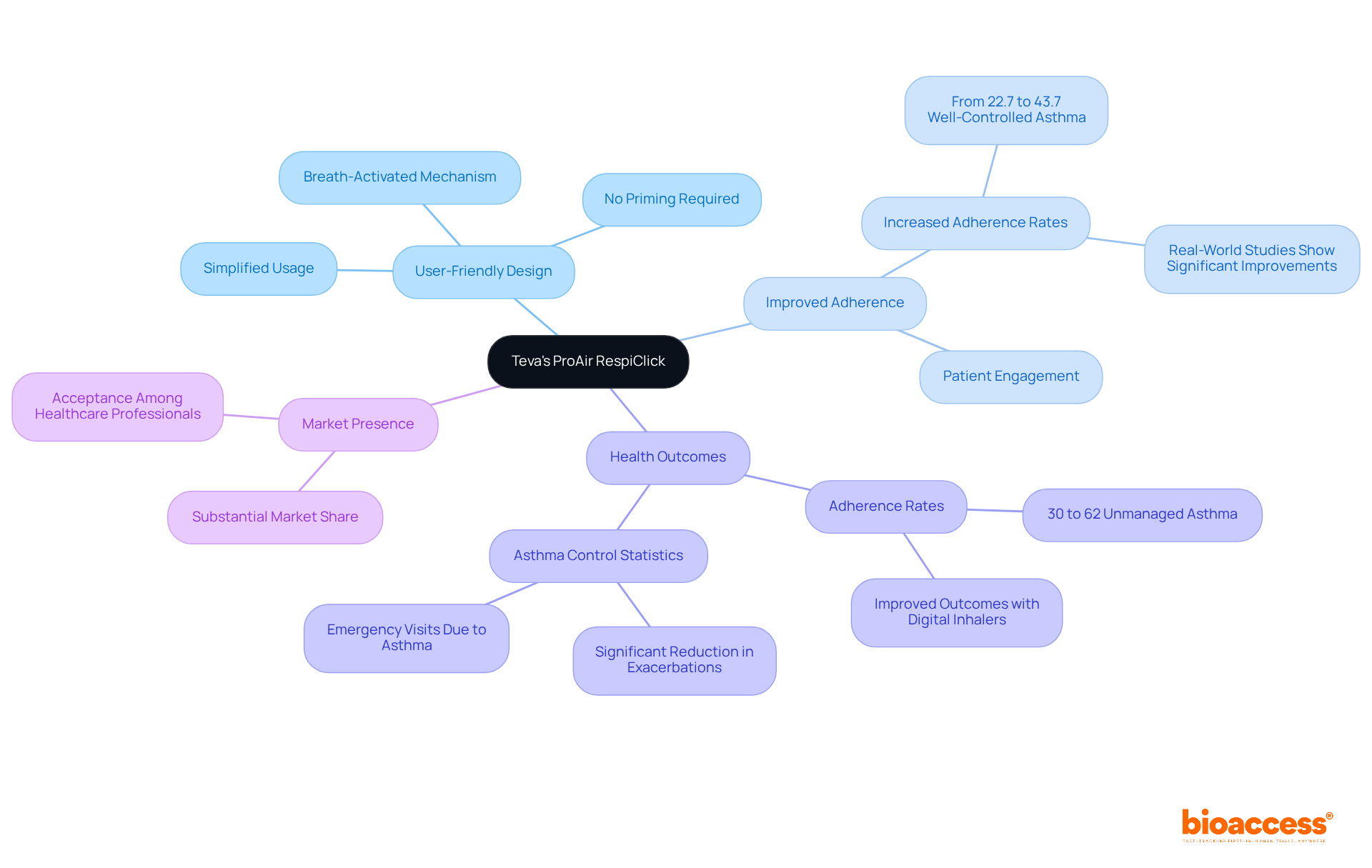
Teva’s ProAir RespiClick: Simplifying Asthma Management
Teva’s ProAir RespiClick is a groundbreaking breath-activated inhaler that significantly simplifies for individuals. By enabling users to deliver medication with a simple inhalation, it eliminates the complexities associated with , such as priming and coordination. This not only enhances usability but also promotes adherence to prescribed treatment regimens, leading to .
According to David I. Bernstein, MD, ” is significant as it eliminates the need for hand-breath coordination during inhalation,” which underscores a key benefit of this innovative device. Real-world studies have demonstrated that can lead to better outcomes for individuals, with adherence rates improving significantly. A systematic review indicated that the proportion of individuals achieving well-controlled asthma increased from 22.7% to 43.7% after using digital inhalers, highlighting the effectiveness of such innovations in .
Currently, between 30% and 62% of individuals in Europe and North America have unmanaged asthma, emphasizing the urgent need for effective management solutions like ProAir RespiClick. Teva’s ProAir RespiClick commands a substantial market share, reflecting its acceptance and effectiveness among and patients alike. As the demand for continues to rise, ProAir RespiClick emerges as a vital tool in transforming .
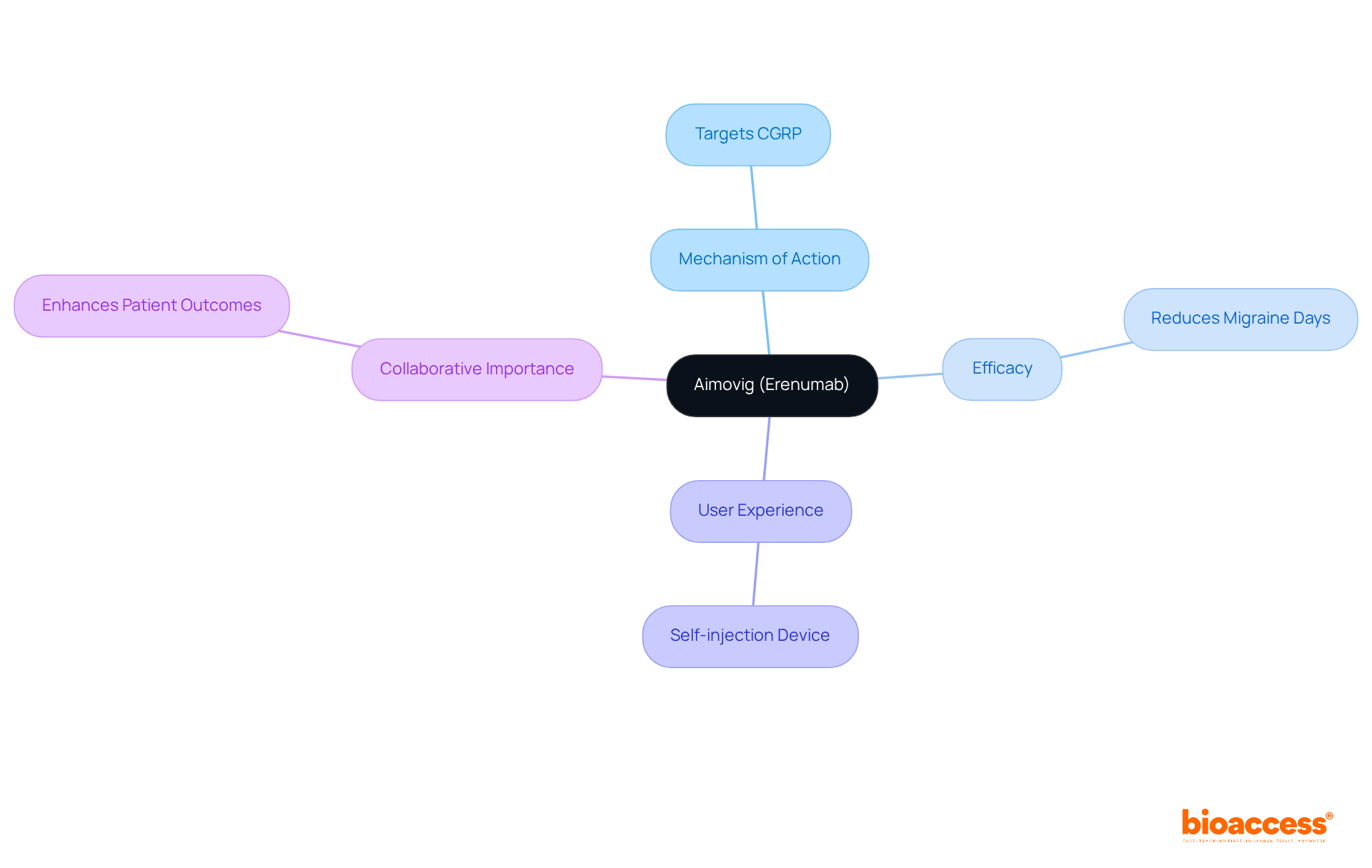
Novartis’ Aimovig: A New Era in Migraine Prevention
Novartis’ Aimovig (erenumab) stands as a groundbreaking treatment for , employing a novel mechanism of action that specifically targets the calcitonin gene-related peptide (CGRP). This self-injection device has shown remarkable efficacy in significantly reducing the number of migraine days for individuals, offering renewed hope for those grappling with . Its and proven effectiveness position Aimovig as an .
As we delve deeper into the , it’s crucial to recognize the transformative role of innovative treatments like Aimovig in addressing the pressing challenges faced by patients. The integration of such advancements not only enhances but also underscores the importance of collaboration among healthcare professionals to optimize treatment approaches.
In conclusion, Aimovig exemplifies the potential of to revolutionize migraine care. By embracing these innovations, we can pave the way for more effective management strategies, ultimately improving the quality of life for countless individuals suffering from migraines.
Eli Lilly’s Trulicity: Streamlining Diabetes Management
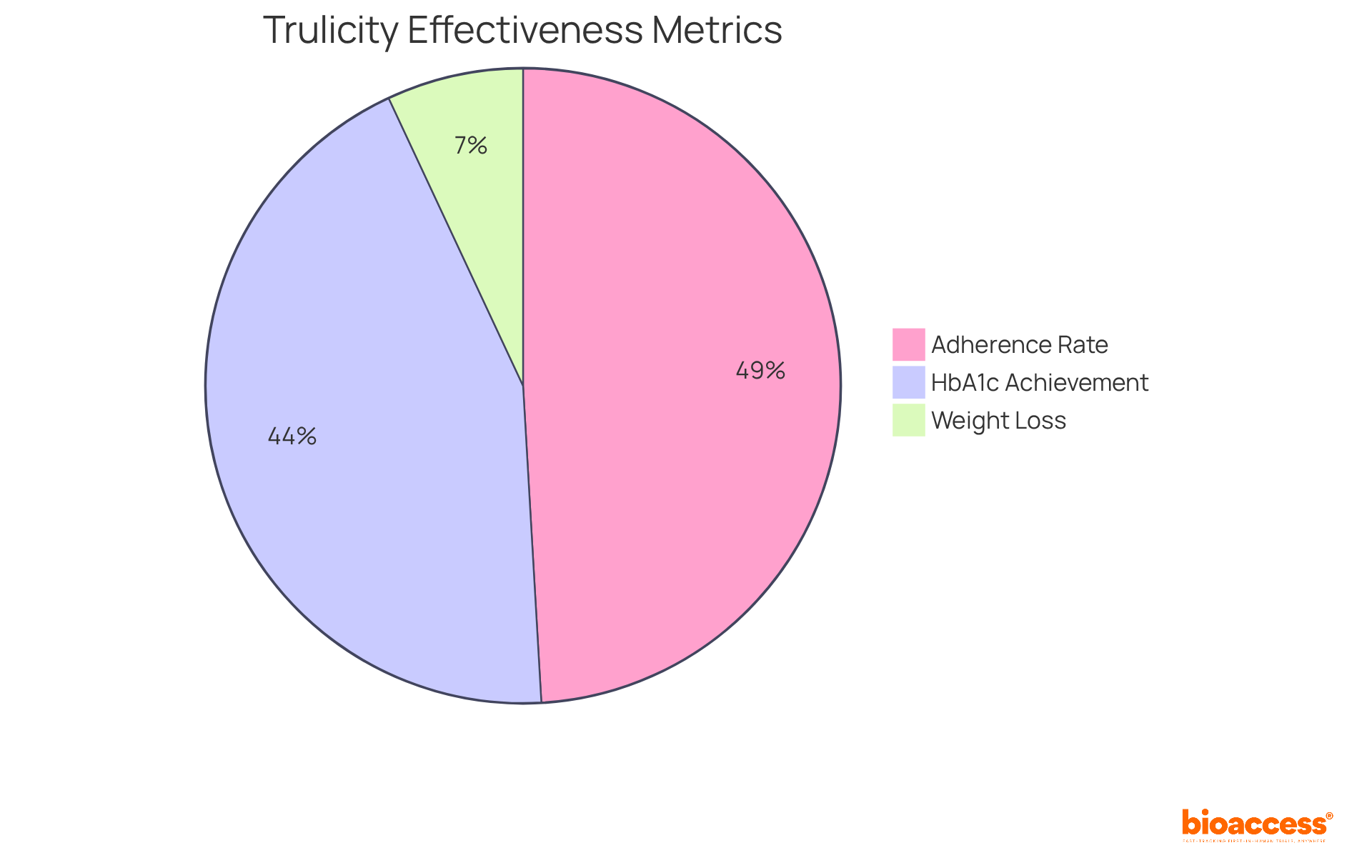
Eli Lilly’s Trulicity (dulaglutide) stands out as a once-weekly injectable medication specifically designed to manage blood sugar levels in adults with type 2 diabetes, which constitutes 90 to 95 percent of all diabetes cases in the U.S. As a GLP-1 receptor agonist, it not only aids in glycemic control but also promotes weight loss, making it a .
reveal that higher doses of dulaglutide can lead to , with decreases ranging from 0.5% to 2.2% over a period of 3 to 24 months. Notably, 23.4-55.7% of individuals achieved HbA1c levels below 7.0%, showcasing the medication’s effectiveness in meeting .
Moreover, have been documented, enhancing the overall health profile of users. The convenience of , which range from 27.2% to 61.0%. This improved compliance is crucial, as it correlates with and sustained management of the condition.
However, it is essential to acknowledge that discontinuation rates for dulaglutide fall between 26.2% and 37.0%, indicating some challenges in user experiences. Overall, Trulicity exemplifies how can transform care for individuals facing blood sugar challenges by integrating effective pharmacotherapy with improved user compliance.
Johnson & Johnson’s OneTouch Verio: Integrating Diabetes Care Solutions
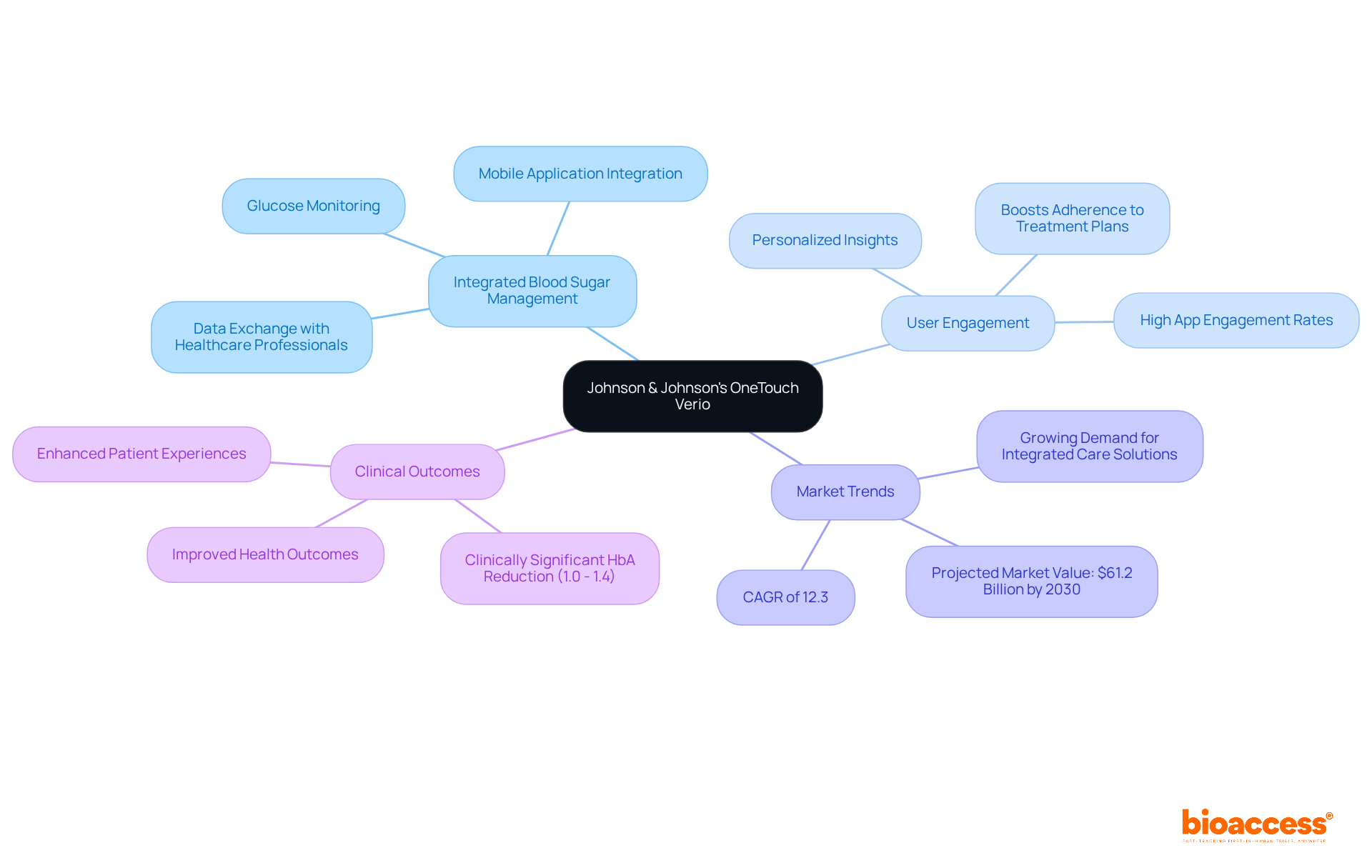
Johnson & Johnson’s represents a significant advancement in integrated , seamlessly combining with a user-friendly mobile application. This innovative framework allows patients to effectively track their glucose levels while providing that enhance self-management. By facilitating data exchange with healthcare professionals, the fosters a collaborative approach to managing blood sugar conditions. The focus on connectivity and user engagement not only boosts adherence to treatment plans but also leads to .
Current market trends reveal a growing demand for , particularly , for individuals facing blood sugar challenges, with the related devices market projected to reach USD 61.2 billion by 2030, expanding at a CAGR of 12.3%. Experts in recognize the OneTouch Verio device as a prime example of that can elevate patient experiences and outcomes in glucose care. A recent study highlighted clinically significant HbA reductions of between 1.0% and 1.4% among users, underscoring the effectiveness of this approach.
As the competitive landscape evolves, the distinguishes itself among key players in the diabetes care devices market, emphasizing the significance of in diabetes management. For directors, leveraging such integrated systems can significantly and improve overall treatment efficacy. What challenges do you face in your efforts? Consider how solutions like OneTouch Verio could address those needs and drive better outcomes.
Conclusion
Innovative drug-device combination products are reshaping the future of healthcare, offering integrated solutions that enhance treatment effectiveness and patient management. These advancements streamline chronic condition care and underscore the importance of collaboration among stakeholders to tackle the complexities of implementation. This article showcases various groundbreaking products, from bioaccess®’s accelerated development processes to Medtronic’s MiniMed 770G insulin delivery system, each playing a pivotal role in improving patient outcomes.
Key insights from the discussion reveal the transformative potential of these innovations. For instance, the Abbott FreeStyle Libre has revolutionized glucose monitoring, while Boston Scientific’s Watchman device significantly reduces stroke risk for patients with atrial fibrillation. Each product emphasizes the necessity for regulatory agility and user-friendly designs, contributing to enhanced adherence and overall health management. Moreover, the rise of integrated solutions, such as Johnson & Johnson’s OneTouch Verio, underscores the growing demand for cohesive care strategies in diabetes management.
As the healthcare landscape continues to evolve, embracing these innovative drug-device combinations is crucial for improving patient care and outcomes. Stakeholders are encouraged to explore how such technologies can address existing challenges in clinical research and patient management. The future of healthcare lies in the synergy between technology and patient-centric solutions, paving the way for more effective and accessible treatment options for all.
Frequently Asked Questions
What is bioaccess® and what does it focus on?
bioaccess® is a company that leverages over 15 years of clinical research expertise to accelerate the development of drug-device combination products, with a particular focus on Latin America’s regulatory processes, especially in Colombia.
How long does the regulatory review process take with bioaccess® in Colombia?
The total IRB/EC and MoH (INVIMA) review process in Colombia takes just 90-120 days.
What services does bioaccess® provide?
bioaccess® offers services including feasibility studies, research site selection, investigator recruitment, regulatory compliance, project management, and thorough reporting on study status and adverse events.
What advantages does bioaccess® provide in clinical research?
bioaccess® enhances the speed of introducing new therapies to patients, highlights the importance of regulatory agility, and offers cost efficiency, with savings of over 30% compared to trials in North America or Western Europe.
What is the Medtronic MiniMed 770G?
The Medtronic MiniMed 770G is a hybrid closed-loop insulin delivery system that autonomously adjusts insulin delivery based on real-time glucose readings, improving diabetes management.
How does the MiniMed 770G impact diabetes management?
The device enhances glycemic control, effectively reducing risks associated with hyperglycemia and hypoglycemia, and has been associated with improved A1C levels and a decrease in diabetes-related complications.
What is the Abbott FreeStyle Libre and how does it function?
The Abbott FreeStyle Libre is a continuous glucose monitoring (CGM) device that provides real-time glucose readings through a small sensor worn on the skin, eliminating the need for routine fingersticks.
What are the benefits of using the FreeStyle Libre system?
Users have experienced significant reductions in HbA1c levels, increased awareness of glucose levels, and more frequent healthcare provider visits, leading to improved diabetes management outcomes.
What upcoming advancements can users expect from Abbott’s FreeStyle Libre?
The anticipated launch of the FreeStyle Libre 3 system in February 2025 promises even greater advancements in glucose monitoring technology.
List of Sources
- bioaccess®: Accelerating Drug-Device Combination Innovations in Healthcare
- bioaccessla.com (https://bioaccessla.com/blog/10-essential-market-access-strategies-for-pharmaceuticals)
- bioaccessla.com (https://bioaccessla.com/blog/9-benefits-of-investigator-initiated-studies-for-clinical-research)
- bioaccessla.com (https://bioaccessla.com/blog/8-key-insights-on-class-2-medical-device-development)
- 30 Quotes About the Future of Healthcare: Expert Takes (https://deliberatedirections.com/quotes-future-of-healthcare)
- Latin American patients must wait an average of 4.7 years to access innovative or cutting-edge treatments (https://fifarma.org/en/waitindicator2024)
- Medtronic MiniMed 770G: Pioneering Insulin Delivery Systems
- Insulin Pumps Market Size, Trends & Statistics 2035 (https://factmr.com/report/insulin-pumps-market)
- Insulin Pump Utilization in 2017–2021 for More Than 22,000 Children and Adults With Type 1 Diabetes: A Multicenter Observational Study – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10788665)
- Breakthrough Studies on Automated Insulin Delivery and CGM for Type 2 Diabetes Unveiled at ADA Scientific Sessions | American Diabetes Association (https://diabetes.org/newsroom/press-releases/breakthrough-studies-automated-insulin-delivery-and-cgm-type-2-diabetes)
- U.S. Insulin Pump Market Size | Growth Analysis Report [2030] (https://fortunebusinessinsights.com/u-s-insulin-pump-market-107534)
- Abbott FreeStyle Libre: Revolutionizing Glucose Monitoring
- (PDF) The Impact of the FreeStyle LibreTM Flash Glucose Monitoring System on Glycemic Control in Patients with Diabetes; Observational Multicenter 15-Months Study (https://researchgate.net/publication/362622379_The_Impact_of_the_FreeStyle_LibreTM_Flash_Glucose_Monitoring_System_on_Glycemic_Control_in_Patients_with_Diabetes_Observational_Multicenter_15-Months_Study)
- The Use of Freestyle Libre Glucose Monitoring System and Diabetes Treatment Progression in Type 2 Diabetes Mellitus: A Retrospective Cohort Study in Saudi Arabia – PubMed (https://pubmed.ncbi.nlm.nih.gov/40161060)
- Impact of the FreeStyle Libre 2® System on Glycaemic Outcomes in Patients with Type 1 Diabetes—Preliminary Study – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11354050)
- Continuous glucose monitoring among adults with type 2 diabetes receiving noninsulin or basal insulin therapy in primary care – Scientific Reports (https://nature.com/articles/s41598-024-83548-4)
- How one teacher kicked diabetes stigma to the curb | Abbott Newsroom (https://abbott.com/corpnewsroom/diabetes-care/how-one-teacher-kicked-diabetes-stigma-to-the-curb.html)
- Boston Scientific’s Watchman: A Dual Approach to Stroke Prevention
- Pros & Cons of the Watchman Device: An In-depth Guide | CVG Cares Cardiology – CVG (https://cvgcares.com/conditions/pros-and-cons-of-the-watchman-device)
- Percutaneous left atrial appendage closure for managing thromboembolic risk in atrial fibrillation – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC6188949)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC10593162)
- Potential of a Novel Large Left Atrial Appendage Closure Device: Short‐Term Outcomes of the WATCHMAN FLX Pro 40‐mm Versus 35‐mm (https://onlinelibrary.wiley.com/doi/10.1002/ccd.70282?af=R)
- Cardiologist questions use of Boston Scientific Watchman device (https://massdevice.com/cardiologist-questions-use-of-boston-scientific-watchman-device)
- Stryker’s Neuroform Atlas: Enhancing Aneurysm Treatment
- Comparative analysis of long term effectiveness of Neuroform Atlas stent versus low profile visualized intraluminal stent/Woven EndoBridge devices in treatment of wide necked intracranial aneurysms – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11503084)
- 10 Innovative Medical Devices by Stryker Corporation Transforming Healthcare – Medico (https://medicogrp.com/stryker-corporation-medical-device)
- Neuroform Atlas Stent System for the treatment of intracranial aneurysm: primary results of the Atlas Humanitarian Device Exemption cohort – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC6703120)
- Neuroform Atlas Stent System for the treatment of intracranial aneurysm: primary results of the Atlas Humanitarian Device Exemption cohort (https://jnis.bmj.com/content/11/8/801)
- Boehringer Ingelheim’s Respimat: Transforming Inhalation Therapy
- 50 Inspiring Breathing Quotes (https://balancingelephants.com/50-breathing-quotes)
- Digital Dose Inhaler Market Size, Share Analysis Report 2030 (https://grandviewresearch.com/industry-analysis/digital-dose-inhaler-market)
- 20 Inspirational Quotes About Breathing for a Calmer Day (https://pepbuddy.com/blogs/news/20-inspirational-quotes-about-breathing-for-a-calmer-day?srsltid=AfmBOop5jAnCR_ShxmVPzTZPtwp__vOadM2tIr20-n_QKK6Ev9RuM7LH)
- Respiratory Inhalers Market – Global Market Size, Share, and Trends Analysis Report – Industry Overview and Forecast to 2032 | Data Bridge Market Research (https://databridgemarketresearch.com/reports/global-respiratory-inhalers-market?srsltid=AfmBOoporM31jKlcB27QSYn2L-ymlYCJ7lIZoF7IUrWlPiznODawP0ov)
- Teva’s ProAir RespiClick: Simplifying Asthma Management
- ProAir RespiClick (Albuterol Sulfate): First Breath-Actuated Inhaler Approved for the Treatment or Prevention of Bronchospasm (https://ahdbonline.com/articles/2016-proair-respiclick-albuterol-sulfate-first-breath-actuated-inhaler-approved-for-the-treatment-or-prevention-of-bronchospasm)
- When will the PROAIR RESPICLICK patents expire, and when will generic PROAIR RESPICLICK be available? (https://drugpatentwatch.com/p/tradename/PROAIR+RESPICLICK?srsltid=AfmBOoqC_twvRIzWxdXflm9zeEBOqhTbK0z9QzLgAUrBxQk4mLjV0XXE)
- The emerging role of digital health in the management of asthma – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10657529)
- When do the PROAIR RESPICLICK patents expire, and when will generic PROAIR RESPICLICK be available? (https://drugpatentwatch.com/p/tradename/PROAIR+RESPICLICK?srsltid=AfmBOoq6X-1OA8K2kE-hoLGj-itP1rQIuODclzMHETKsLgDjbSwlAyK3)
- Form 10-K (https://sec.gov/Archives/edgar/data/818686/000119312518039076/d529462d10k.htm)
- Eli Lilly’s Trulicity: Streamlining Diabetes Management
- nutrition.org (https://nutrition.org/nutritional-priorities-to-support-glp-1-therapy-for-obesity)
- Real-World Effectiveness of Dulaglutide in Patients with Type 2 Diabetes Mellitus: A Literature Review – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC7324465)
- Higher investigational doses of Trulicity® (dulaglutide) meaningfully reduced A1C and body weight in people with type 2 diabetes (https://biospace.com/higher-investigational-doses-of-trulicity-dulaglutide-meaningfully-reduced-a1c-and-body-weight-in-people-with-type-2-diabetes)
- New Data Show Lilly’s Once-Weekly Trulicity® (dulaglutide) in Combination with Insulin Glargine Improves Glycemic Control in People with Type 2 Diabetes | Eli Lilly and Company (https://investor.lilly.com/news-releases/news-release-details/new-data-show-lillys-once-weekly-trulicityr-dulaglutide-0)
- Johnson & Johnson’s OneTouch Verio: Integrating Diabetes Care Solutions
- Diabetes Care Devices Market Outlook 2025-2032 (https://intelmarketresearch.com/diabetes-care-devices-market-market-17142)
- Diabetes Care Devices – Worldwide | Market Forecast (https://statista.com/outlook/hmo/medical-technology/medical-devices/diabetes-care-devices/worldwide?srsltid=AfmBOooqBg7QxkTPzqsPGJvuxDBB9amK3ucfinZSZo5nlFaW4jpKDQwb)
- quotes.lifehack.org (https://quotes.lifehack.org/collections/inspirational-quotes-for-diabetes)
- diabetes.jmir.org (https://diabetes.jmir.org/2022/3/e35039)
- Diabetes Care Devices Market Size & Growth Forecast to 2030 (https://marketsandmarkets.com/Market-Reports/diabetes-care-375.html)