Introduction
In the rapidly evolving realm of medical research, the success of informed consent form (ICF) clinical trials relies on a delicate equilibrium of ethical standards, regulatory compliance, and participant engagement. As the demand for innovative medical solutions escalates, grasping the key elements that drive effective ICF trials is crucial for researchers and sponsors alike.
What strategies can be employed not only to streamline the process but also to ensure that participants are well-informed and actively engaged throughout the trial? This article explores eight essential elements that can revolutionize ICF clinical trials, enhancing both efficiency and participant satisfaction.
bioaccess®: Accelerating ICF Clinical Trials with Global Agility
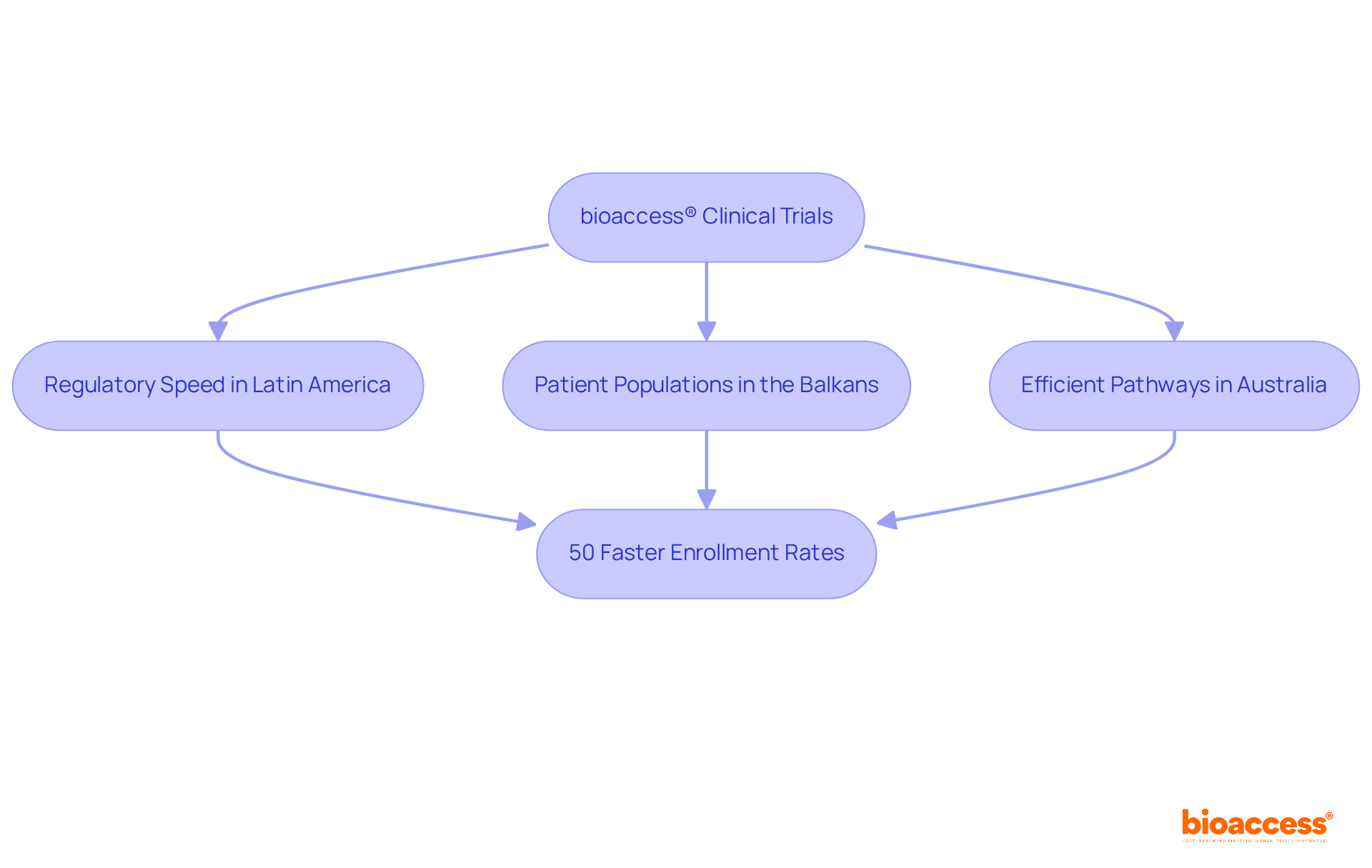
bioaccess® strategically leverages the , the diverse patient populations of the Balkans, and Australia’s efficient pathways to achieve ethical approvals in a remarkable timeframe of just 4 to 6 weeks. This powerful combination results in than those found in conventional markets, thereby positioning bioaccess® as a leader in the (ICF ) studies.
By , bioaccess® not only facilitates rapid advancements in but also empowers innovators to bring their products to market more swiftly. This ultimately enhances to , underscoring the importance of collaboration in addressing key challenges within the Medtech landscape.
Informed Consent Forms: Ensuring Participant Understanding and Compliance
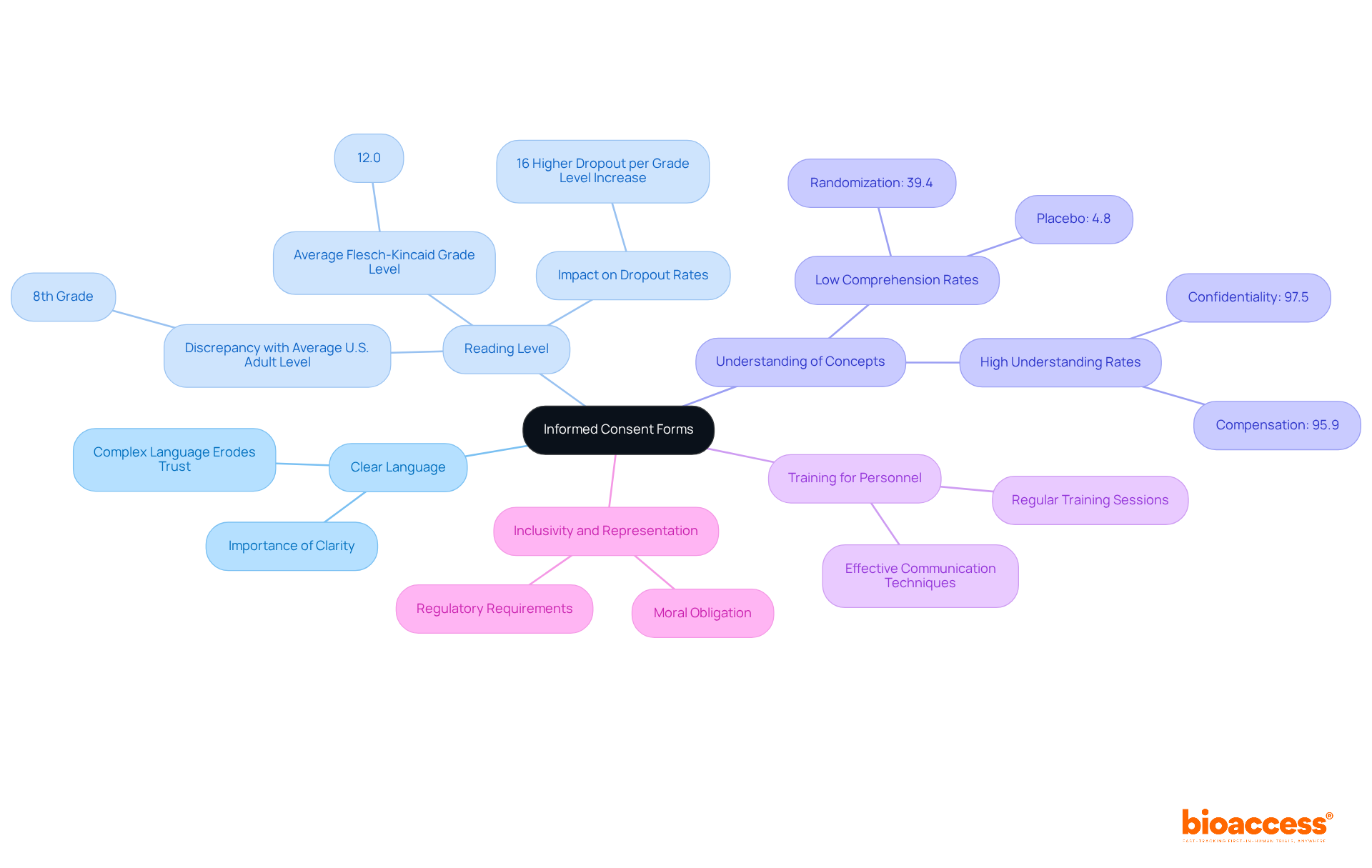
(ICFs) are critical for ensuring that individuals fully understand the nature of the ICF , the associated risks, and their rights. To achieve this, ICFs must be crafted in , providing , procedures, and potential side effects.
Research indicates that the typical Flesch-Kincaid Grade Level of consent forms is 12.0, significantly above the average reading level of 8th grade for most U.S. adults. This discrepancy can hinder retention and engagement. Alarmingly, , such as randomization and placebo, are low, with only 4.8% of individuals comprehending the concept of placebo.
Regular training sessions for research personnel on can greatly enhance understanding and compliance. This proactive approach not only fosters trust but also contributes to more . Participants who are well-informed are more likely to remain engaged throughout the research process.
Furthermore, to ensure they are inclusive and representative is not merely a moral obligation; it is increasingly becoming a regulatory requirement in medical research.
Regulatory Compliance: Upholding Ethical Standards in Clinical Trials
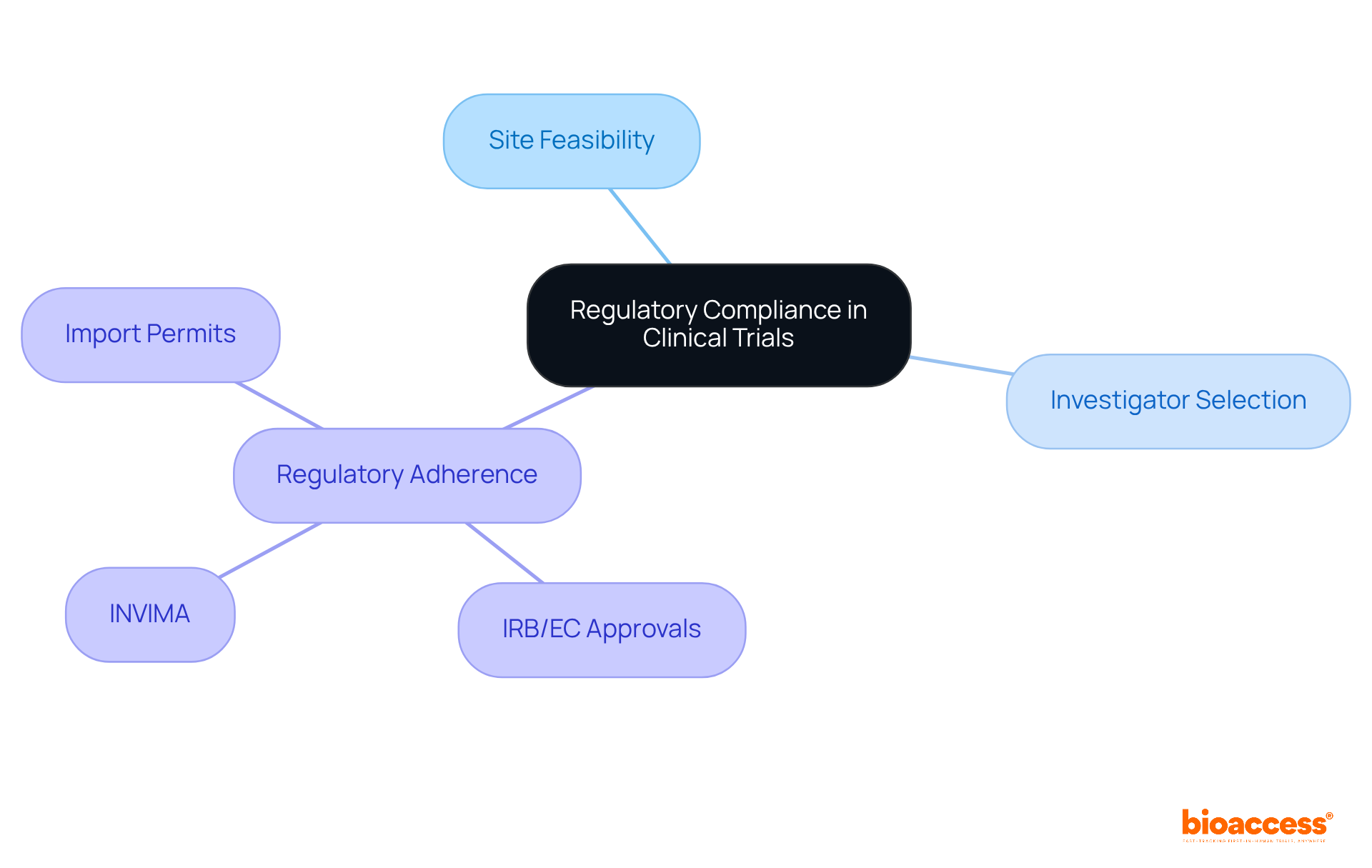
Maintaining is crucial in to ensure that ethical standards are met. This necessitates , such as the FDA or EMA, while meticulously following all .
At bioaccess®, we are dedicated to supporting in . Our comprehensive approach includes:
- Site feasibility
- Strict
Our expertise in securing necessary approvals, including , along with managing import permits, guarantees that all aspects of the study comply with local regulations. Regular audits and training play a vital role in , thereby safeguarding participants and ensuring the integrity of the research data.
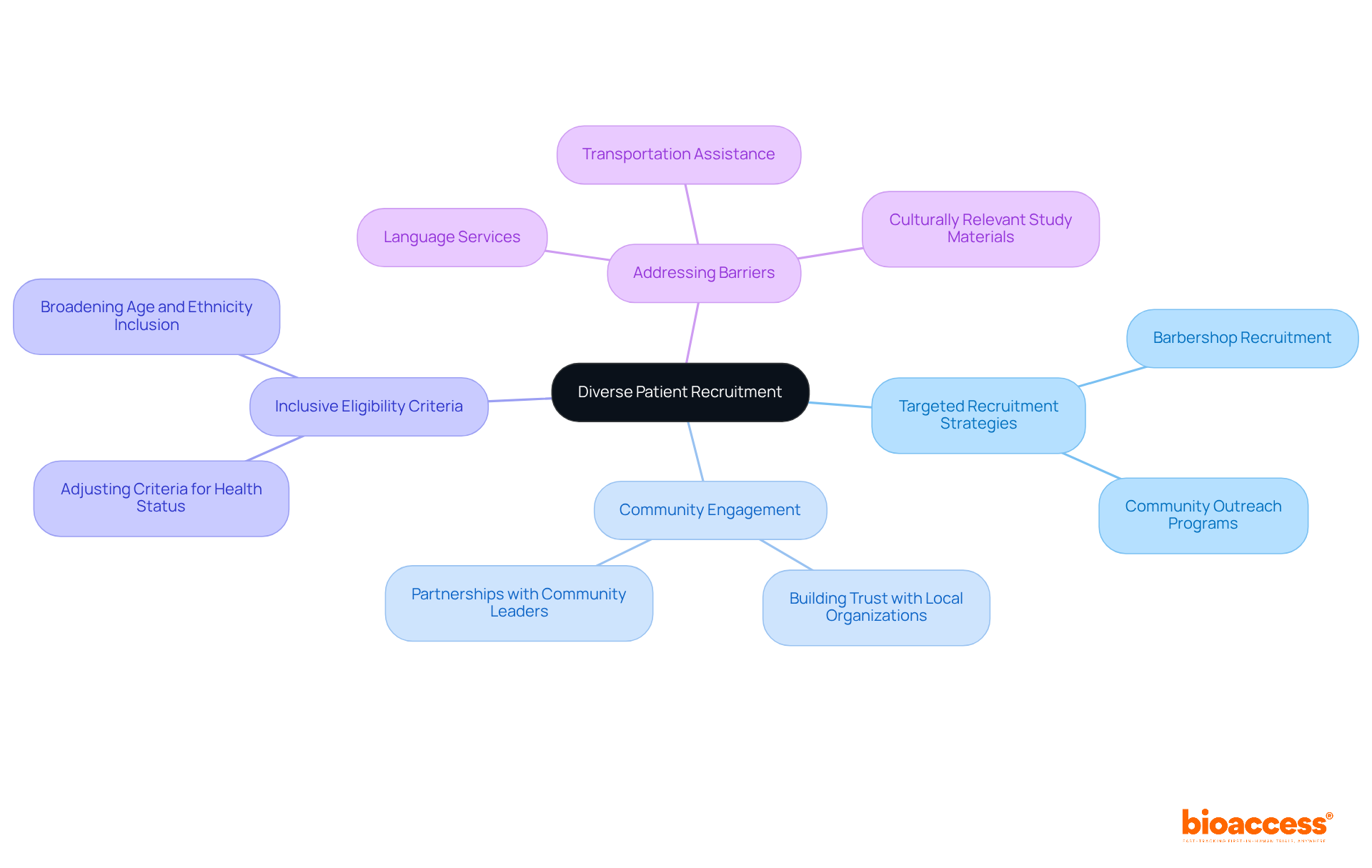
Diverse Patient Recruitment: Enhancing Data Representativeness in Trials
Recruiting a is essential for . Implementing effectively engages various demographic groups, including different age ranges, ethnicities, and health statuses.
For instance, , such as utilizing barbershops to recruit Black men, has proven successful in enhancing recruitment from underrepresented populations by fostering trust and collaboration with local organizations.
Moreover, to be more inclusive creates opportunities for diverse participation, allowing researchers to capture a broader spectrum of responses to treatments. By ensuring that the research population reflects the broader community, researchers can enhance the relevance of their findings and gain valuable insights into how different populations respond to medical interventions.
This method not only encourages but also enhances the overall reliability of research outcomes. Furthermore, addressing , such as providing , is essential for fostering inclusivity in recruitment strategies.
Data Management Systems: Ensuring Integrity and Facilitating Analysis
Establishing robust is crucial for upholding the integrity of research data. These systems must enable precise , storage, and analysis, all while . At bioaccess, we provide comprehensive for , which include:
- Feasibility studies
- Adherence reviews
- Study setup
- Import permits
- Reporting
Routine are essential; they can reveal discrepancies early, allowing for swift corrections and ensuring the reliability of results. By integrating these services, we ensure that our are not only effective but also aligned with the highest regulatory standards.
Staff Training: Equipping Teams for ICF Protocol Compliance
Continuous employee education is essential for ensuring adherence to the guidelines of the ICF in . must encompass:
- Ethical considerations surrounding
- The
A randomized controlled study evaluating training formats revealed that merely 46% of participants completed the training within the shortest timeframe, highlighting the challenges faced in achieving effective training compliance. By equipping research teams with the requisite knowledge and skills, organizations can significantly enhance , thereby ensuring that all consent procedures are executed ethically and efficiently.
As John F. Kennedy aptly stated, ‘Leadership and learning are indispensable to each other,’ emphasizing the vital link between training and effective leadership in ICF s. This dedication to not only but also empowers staff to adeptly navigate the complexities of with confidence and integrity.
Technology Integration: Streamlining ICF Processes for Efficiency
Incorporating technology into the significantly streamlines operations and . Electronic consent (eConsent) platforms provide engaging, user-friendly experiences, ensuring individuals fully comprehend study details. These platforms facilitate and monitoring, improving overall and bolstering compliance with regulatory standards.
By and minimizing manual errors, can reduce by up to 57%, leading to more reliable study outcomes. Furthermore, eConsent enhances and engagement, achieving retention rates that are 30% higher compared to traditional paper-based methods.
As the adoption of eConsent continues to rise, it is poised to become a lasting component in research, offering significant advantages to all stakeholders involved.
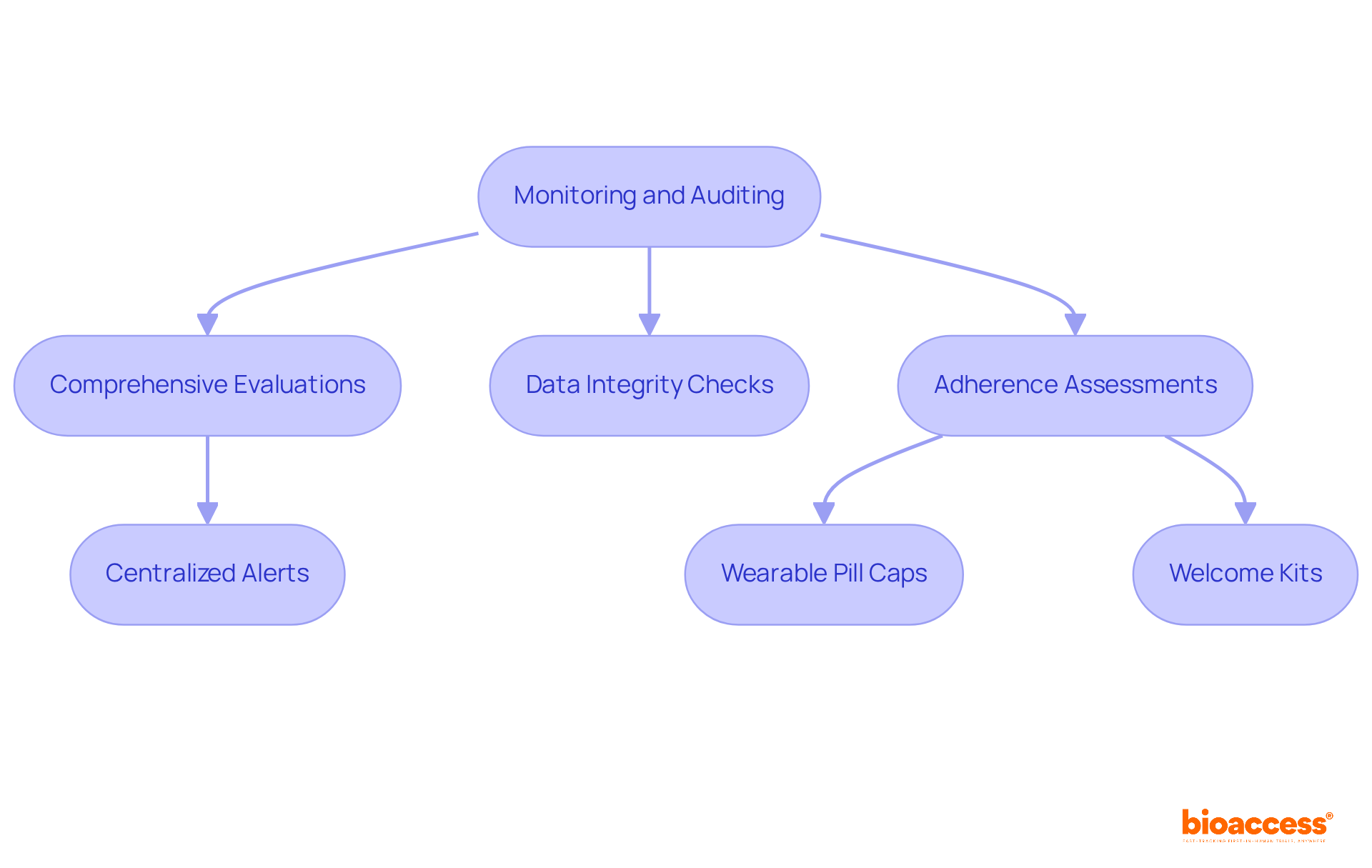
Monitoring and Auditing: Ensuring Protocol Adherence and Continuous Improvement
Regular in ensuring within . This systematic approach encompasses comprehensive evaluations of experimental activities, stringent , and meticulous adherence assessments. By establishing a robust monitoring system, organizations like bioaccess can identify areas for improvement and ensure that the ICF clinical trial studies are conducted in accordance with established guidelines.
For instance, a collaboration between a sponsor and a (CRO) successfully implemented centralized alerts across 22 global sites, leading to a 50% reduction in protocol deviations. Furthermore, the use of wearable pill caps in a neurology study resulted in a 40% increase in medication adherence, underscoring the impact of monitoring on subject compliance.
and insufficient communication from site staff is essential for sustaining adherence. Additionally, bioaccess’s —including feasibility studies, site selection, compliance assessments, and —facilitate the implementation of clear onboarding and educational strategies.
Initiatives such as welcome kits and visual timelines can significantly enhance participant understanding and engagement. These efforts not only improve the but also foster a culture of continuous improvement, ultimately leading to more .
Stakeholder Communication: Fostering Transparency and Collaboration
is essential for the success of clinical studies. Regular updates and open lines of communication with investigators, sponsors, regulatory bodies, and ethics committees foster collaboration and build trust. ensures that they remain informed and engaged, resulting in more efficient operations and swift resolution of any issues. This proactive strategy not only enhances the overall experience but also contributes to achieving by aligning expectations and enabling .
For instance, organizations like that implement organized communication strategies—such as regular stakeholder meetings and clear reporting protocols—often report improved , alongside greater adherence to .
Furthermore, ‘s , including feasibility assessments, site selection, compliance evaluations, setup, and project oversight, underscore the importance of transparency in research. Such transparency bolsters public perception and trust in study sponsors and the pharmaceutical sector, reinforcing the ethical commitment to openness as a regulatory responsibility.
Ultimately, transcends regulatory necessity; it embodies a commitment to ethical principles that can profoundly influence study outcomes.
Post-Trial Follow-Up: Gathering Feedback and Ensuring Ongoing Compliance
is an essential component of the , enabling researchers to gather critical feedback from subjects and evaluate the . This stage not only ensures compliance with but also fosters a sense of community and trust among contributors. Engaging with individuals after the trial significantly enhances their willingness to participate in future studies, thereby supporting the advancement of medical knowledge.
Research indicates that effective follow-up strategies, such as and retention efforts, lead to higher response rates and increased satisfaction among participants. Additionally, collecting feedback allows researchers to identify areas for improvement in study design and execution, ultimately resulting in more reliable and ethically sound medical research. Insights from contributors can also inform the development of tailored interventions that address the unique needs of diverse populations, including marginalized groups.
By prioritizing feedback from participants, research studies can evolve to better meet the needs of both researchers and contributors, ensuring equitable sharing of research benefits. As ethical discussions underscore, the rights, safety, and well-being of study participants must extend beyond the research duration, highlighting the importance of .
Furthermore, bioaccess® emphasizes comprehensive trial management services, including , , and , ensuring that and feedback are integral to the trial process. This commitment to ongoing compliance and the welfare of participants is vital for building trust and enhancing the overall success of clinical research.
A notable example of this is the collaboration with Welwaze Medical Inc. for the Celbrea® medical device launch, which underscores the importance of regulatory access and participant engagement in achieving successful outcomes.
Conclusion
The success of informed consent form (ICF) clinical trials is fundamentally rooted in several key elements that collectively enhance efficiency, uphold ethical standards, and boost participant engagement throughout the research process. By emphasizing strategic approaches such as:
- Regulatory compliance
- Diverse patient recruitment
- The integration of technology
Organizations can significantly accelerate the development and implementation of clinical trials. This ultimately leads to improved patient access to innovative therapies.
Throughout this discussion, the importance of:
- Crafting clear ICFs
- Maintaining ethical standards
- Ensuring diverse representation in trials
has been underscored. Effectively training staff, employing robust data management systems, and fostering open communication among stakeholders are critical to achieving compliance and enhancing the overall quality of research. Moreover, the role of post-trial follow-up in gathering feedback and ensuring ongoing adherence to ethical standards is vital, as it contributes to building trust and improving future studies.
In conclusion, the landscape of ICF clinical trials is evolving, and the adoption of best practices is essential for success. Organizations are strongly encouraged to prioritize these elements to not only meet regulatory requirements but also to enhance the integrity and reliability of research outcomes. By embracing these strategies, stakeholders can collaborate effectively to foster a culture of transparency and continuous improvement in clinical research, ultimately benefiting participants and advancing medical knowledge.
Frequently Asked Questions
What is bioaccess® and what role does it play in clinical trials?
bioaccess® accelerates informed consent form (ICF) clinical trials by leveraging the regulatory speed of Latin America, diverse patient populations in the Balkans, and efficient pathways in Australia, achieving ethical approvals in just 4 to 6 weeks.
How does bioaccess® improve enrollment rates in clinical trials?
By streamlining the clinical research process, bioaccess® achieves enrollment rates that are 50% faster than conventional markets, positioning itself as a leader in expedited ICF clinical trial studies.
Why are Informed Consent Forms (ICFs) important in clinical trials?
ICFs ensure that participants fully understand the nature of the trial, associated risks, and their rights. They must be written in clear, accessible language to facilitate comprehension and engagement.
What challenges exist regarding the readability of Informed Consent Forms?
The typical Flesch-Kincaid Grade Level of consent forms is 12.0, which is significantly higher than the average reading level of 8th grade for most U.S. adults, potentially hindering understanding and retention.
How can understanding and compliance with ICFs be improved?
Regular training sessions for research personnel on effective communication techniques can enhance understanding and compliance, fostering trust and contributing to more successful outcomes in clinical trials.
What is the importance of inclusivity in consent forms?
Streamlining consent forms to be inclusive and representative is not only a moral obligation but is also increasingly becoming a regulatory requirement in medical research.
Why is regulatory compliance crucial in clinical trials?
Maintaining regulatory compliance ensures that ethical standards are met, safeguarding participants and ensuring the integrity of the research data.
What services does bioaccess® provide to support regulatory compliance in clinical studies?
bioaccess® offers site feasibility, investigator selection, and strict regulatory adherence, including securing necessary approvals and managing import permits to comply with local regulations. Regular audits and training are also part of their comprehensive approach.
List of Sources
- bioaccess®: Accelerating ICF Clinical Trials with Global Agility
- quotlr.com (https://quotlr.com/quotes-about-clinical-research)
- bioaccess® | MedTech, Biopharma & Radiopharma CRO in Latin America (https://bioaccessla.com/blog/the-latin-american-advantage-why-clinical-trial-recruitment-and-retention-outpace-the-us-and-europe)
- TOP 10 CLINICAL TRIALS QUOTES | A-Z Quotes (https://azquotes.com/quotes/topics/clinical-trials.html)
- azquotes.com (https://azquotes.com/quotes/topics/medical-research.html)
- statista.com (https://statista.com/statistics/1013599/latin-america-clinical-trials)
- Informed Consent Forms: Ensuring Participant Understanding and Compliance
- azquotes.com (https://azquotes.com/quotes/topics/informed-consent.html)
- Participants’ understanding of informed consent in clinical trials: A systematic review and updated meta-analysis – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10760836)
- thelancet.com (https://thelancet.com/journals/eclinm/article/PIIS2589-5370(24)00393-6/fulltext)
- Informed Consent—We Can and Should Do Better (https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2779253)
- Consent Quotes | Art of Consent (https://artofconsent.co.uk/consent-quotes)
- Regulatory Compliance: Upholding Ethical Standards in Clinical Trials
- numberanalytics.com (https://numberanalytics.com/blog/regulatory-compliance-in-biostatistics-ultimate-guide)
- iddi.com (https://iddi.com/services/strategic-consulting/regulatory-statistics)
- statista.com (https://statista.com/topics/6756/clinical-trials)
- TOP 10 CLINICAL TRIALS QUOTES | A-Z Quotes (https://azquotes.com/quotes/topics/clinical-trials.html)
- azquotes.com (https://azquotes.com/quotes/topics/medical-research.html)
- Diverse Patient Recruitment: Enhancing Data Representativeness in Trials
- improve-medical.net (https://improve-medical.net/resources-34/The-Importance-of-Diversity-in-Patient-Recruitment-for-Clinical-Trials:-Strategies-and-Solutions-for-Healthcare-Professionals)
- pharmaphorum.com (https://pharmaphorum.com/patients/how-advance-diversity-equity-and-inclusion-clinical-trial-recruitment)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC9133187)
- wcgclinical.com (https://wcgclinical.com/insights/diverse-recruitment-strategies-and-the-future-of-inclusive-clinical-trials)
- Diversity data in the US: Understanding the FDA’s push for more representation in clinical trials (https://clinicaltrialsarena.com/sponsored/diversity-data-in-the-us-understanding-the-fdas-push-for-more-representation-in-clinical-trials)
- Data Management Systems: Ensuring Integrity and Facilitating Analysis
- Data Integrity – Clinical Research Explained | VIARES (https://viares.com/blog/clinical-research-explained/data-integrity)
- 2025 Clinical Data Trend Report | Veeva (https://veeva.com/2025-clinical-data-trend-report)
- bioaccessla.com (https://bioaccessla.com/blog/how-to-ensure-data-integrity-in-clinical-research-a-step-by-step-guide)
- raptimresearch.com (https://raptimresearch.com/blog/ensuring-data-integrity-quality-assurance-and-compliance-in-modern-cros)
- jafconsulting.com (https://jafconsulting.com/blog/ensuring-data-integrity-in-clinical-trials-key-considerations)
- Staff Training: Equipping Teams for ICF Protocol Compliance
- skyprep.com (https://skyprep.com/2013/07/29/15-inspiration-learning-and-training-quotes)
- A training protocol compliance of 13% was observed in a research study of clinical research professionals – PubMed (https://pubmed.ncbi.nlm.nih.gov/39090704)
- 42 Training Quotes: Inspirational Words for Learning | ITD World (https://itdworld.com/blog/human-resources/training-quotes-inspirational)
- 25 Brilliant Employee Training Quotes For 2026 | Vouch (https://vouchfor.com/blog/employee-training-quotes)
- 18 of Our Favorite Quotes About the Power of Training & Development – Abilitie (https://abilitie.com/blog/2018-7-6-18-of-our-favorite-quotes-about-the-power-of-training-development)
- Technology Integration: Streamlining ICF Processes for Efficiency
- ascopubs.org (https://ascopubs.org/doi/10.1200/CCI.22.00122)
- pharmoutsourcing.com (https://pharmoutsourcing.com/Featured-Articles/611075-Why-eConsent-is-a-Win-Win-for-Patients-Clinical-Trial-Sponsors-and-Staf)
- azquotes.com (https://azquotes.com/quotes/topics/informed-consent.html)
- ccrps.org (https://ccrps.org/clinical-research-blog/benefits-of-econsent-in-clinical-trials)
- clinicalresearchnewsonline.com (https://clinicalresearchnewsonline.com/news/2024/05/03/econsent-in-decentralized-clinical-trials—a-path-to-participant-empowerment-and-transparency)
- Monitoring and Auditing: Ensuring Protocol Adherence and Continuous Improvement
- quotlr.com (https://quotlr.com/quotes-about-clinical-research)
- mahalo.health (https://mahalo.health/insights/how-to-increase-adherence-and-protocol-compliance-for-clinical-trials)
- Quotes About Medical Research (https://changingthepresent.org/pages/quotes-about-medical-research)
- Stakeholder Communication: Fostering Transparency and Collaboration
- Stakeholder engagement (https://who.int/our-work/science-division/research-for-health/implementation-of-the-resolution-on-clinical-trials/stakeholder-engagement)
- who.int (https://who.int/news-room/events/detail/2025/03/28/default-calendar/shaping-the-future-of-clinical-trials–building-an-ethical–efficient–and-equitable-ecosystem)
- grantthornton.com (https://grantthornton.com/insights/articles/advisory/2023/challenges-opportunities-of-clinical-trial-transparency)
- clinicalresearchtech.com (https://clinicalresearchtech.com/fda-and-clinical-trials-advancing-transparency-and-participation)
- Post-Trial Follow-Up: Gathering Feedback and Ensuring Ongoing Compliance
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC4394585)
- cambridge.org (https://cambridge.org/core/journals/journal-of-clinical-and-translational-science/article/case-example-of-a-jailbased-cancer-prevention-clinical-trial-social-determinants-of-health-framework-novel-experimental-design-and-retention-strategies-to-facilitate-longterm-followup-of-clinical-trial-participants/80232CCE45047025675A6699F8A2B7F1)
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S1551714424001629)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC5159967)

Leave a Reply