Introduction
Navigating the intricate landscape of clinical trials demands precise tools that can adapt to shifting data and evolving research needs. Adaptive design statistical packages have become indispensable assets for researchers, providing the flexibility to modify trial parameters based on interim results. Yet, with a multitude of options at their disposal, how can researchers effectively select and purchase the software that best aligns with their specific project requirements? This guide presents a systematic approach to help researchers identify, evaluate, and acquire the optimal adaptive design statistical package, ensuring that their clinical trials are not only efficient but also compliant.
Understand Adaptive Design Statistical Packages
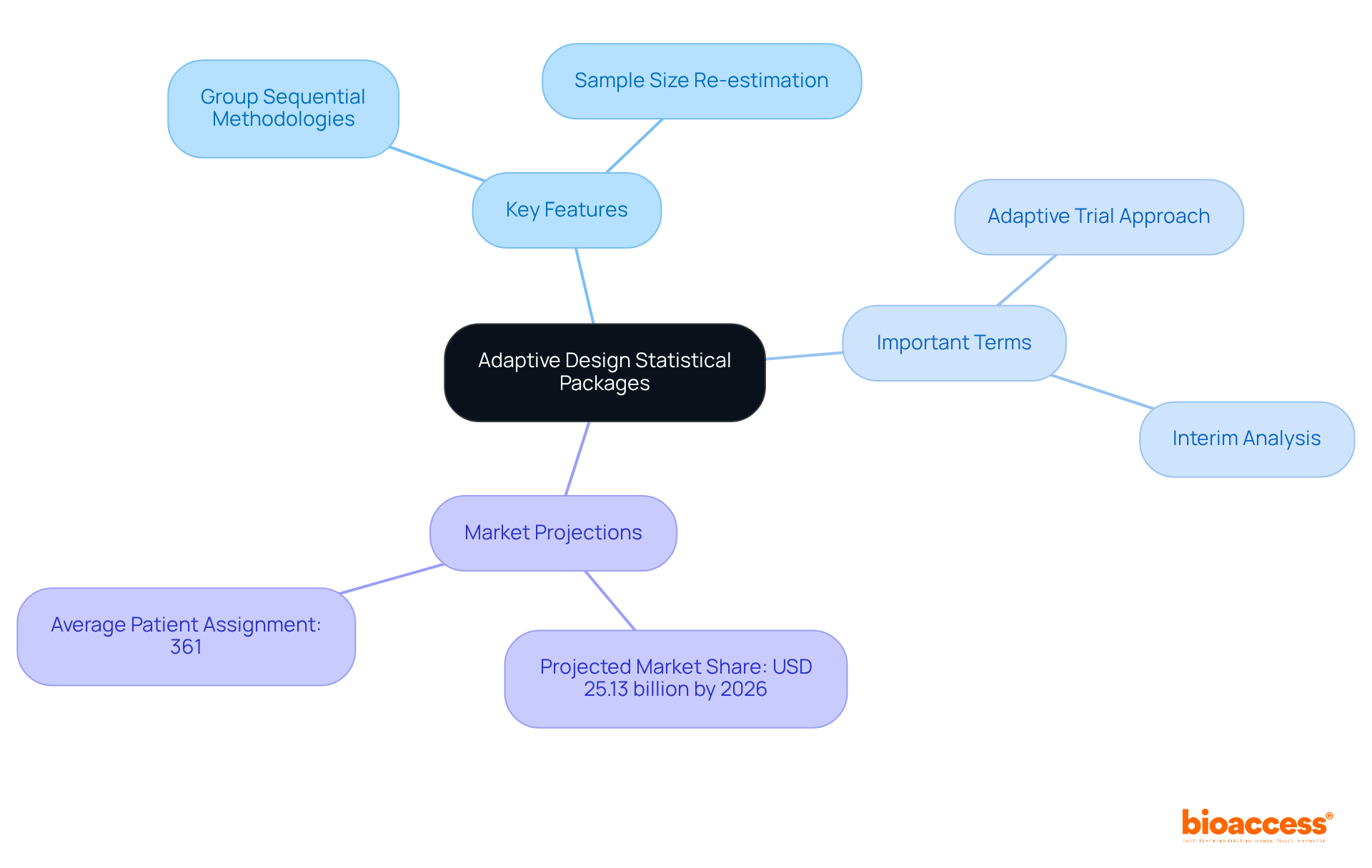
are specialized applications that significantly enhance the planning and analysis of , adapting seamlessly based on interim results. These innovative packages empower researchers to modify critical trial parameters – like sample size or treatment allocation – responding dynamically to data collected throughout the trial. Key features include the ability to implement group sequential methodologies and conduct sample size re-estimation, both essential for improving trial efficiency and addressing ethical considerations.
Understanding terms such as ” and ” is crucial for navigating the diverse options available in the market. As the demand for grows, these tools are becoming indispensable, with projections indicating a market share of USD 25.13 billion by 2026. Notably, the average number of patients assigned in flexible approaches is 361, underscoring their effectiveness in achieving better outcomes.
Experts emphasize the importance of these tools, asserting that flexible frameworks can lead to and more efficient trials. Practical applications of flexible trial systems demonstrate their capability to enhance clinical research processes, ultimately driving better patient outcomes and advancing the field.
Identify Your Project Requirements
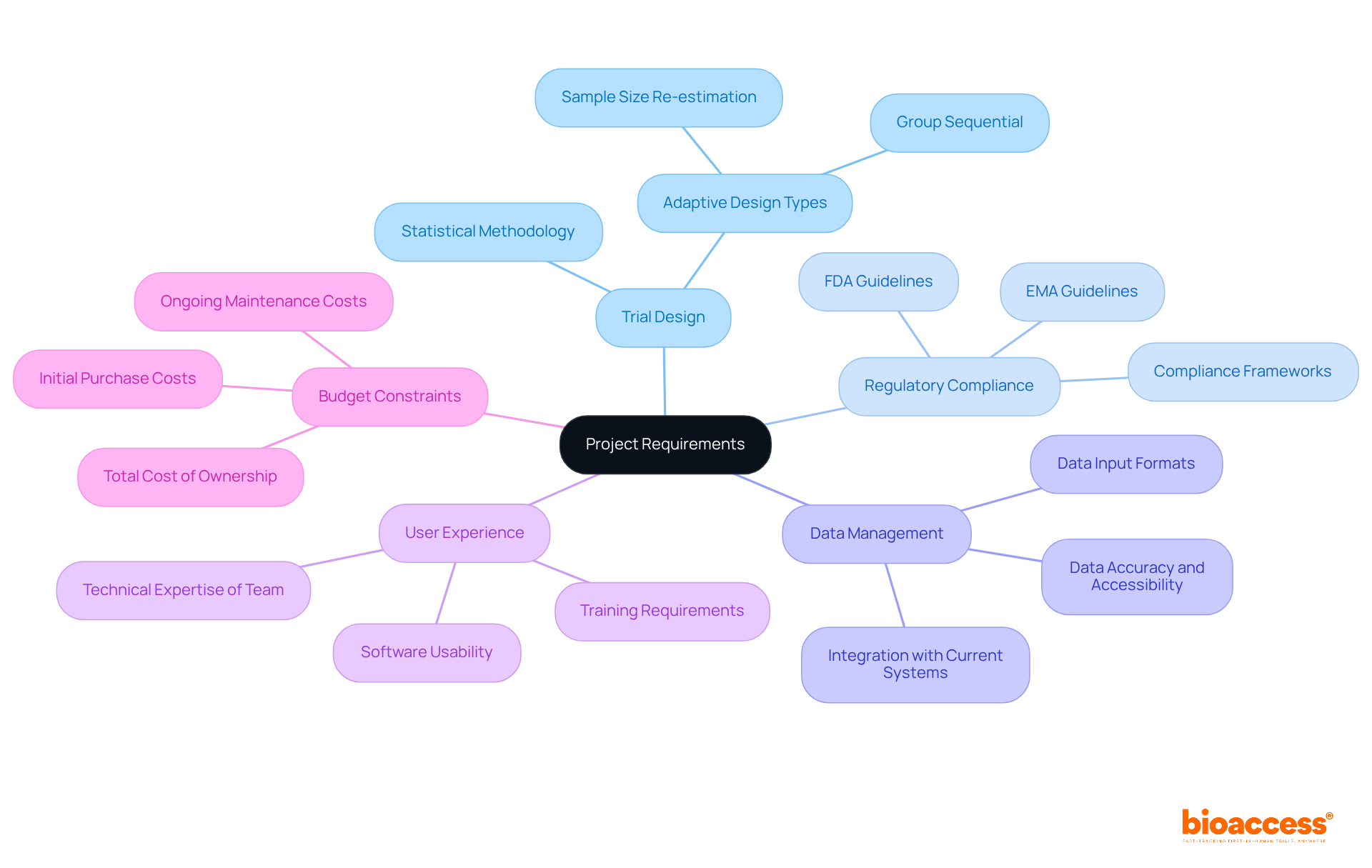
Before deciding to purchase an , it’s crucial to define your project needs clearly. Consider these key factors to guide your decision-making process:
- : Identify the specific type of adaptive design you plan to implement, such as group sequential or sample size re-estimation. This ensures the system aligns with your study’s objectives.
- : Confirm that the application adheres to relevant , including those set by the FDA or EMA. This step is vital for facilitating smooth approval processes and maintaining compliance. For example, ensuring that the system supports the latest regulatory frameworks can prevent delays in trial approvals.
- : Evaluate your requirements, focusing on data input formats and the system’s ability to integrate seamlessly with your current setups. A well-integrated system is essential for effective , significantly enhancing data accuracy and accessibility.
- : Consider the technical expertise of your team alongside the software’s user-friendliness. An intuitive solution can greatly boost productivity and reduce training time. Clinical research directors emphasize that user-friendly interfaces are critical for minimizing the learning curve.
- : Develop a comprehensive budget that includes not only the initial purchase but also ongoing costs related to maintenance and support. This ensures that your investment remains sustainable. Understanding the is vital for long-term planning.
By meticulously defining these requirements, you can streamline the evaluation process and make the decision to purchase an that best meets the needs of your .
Evaluate Available Statistical Packages
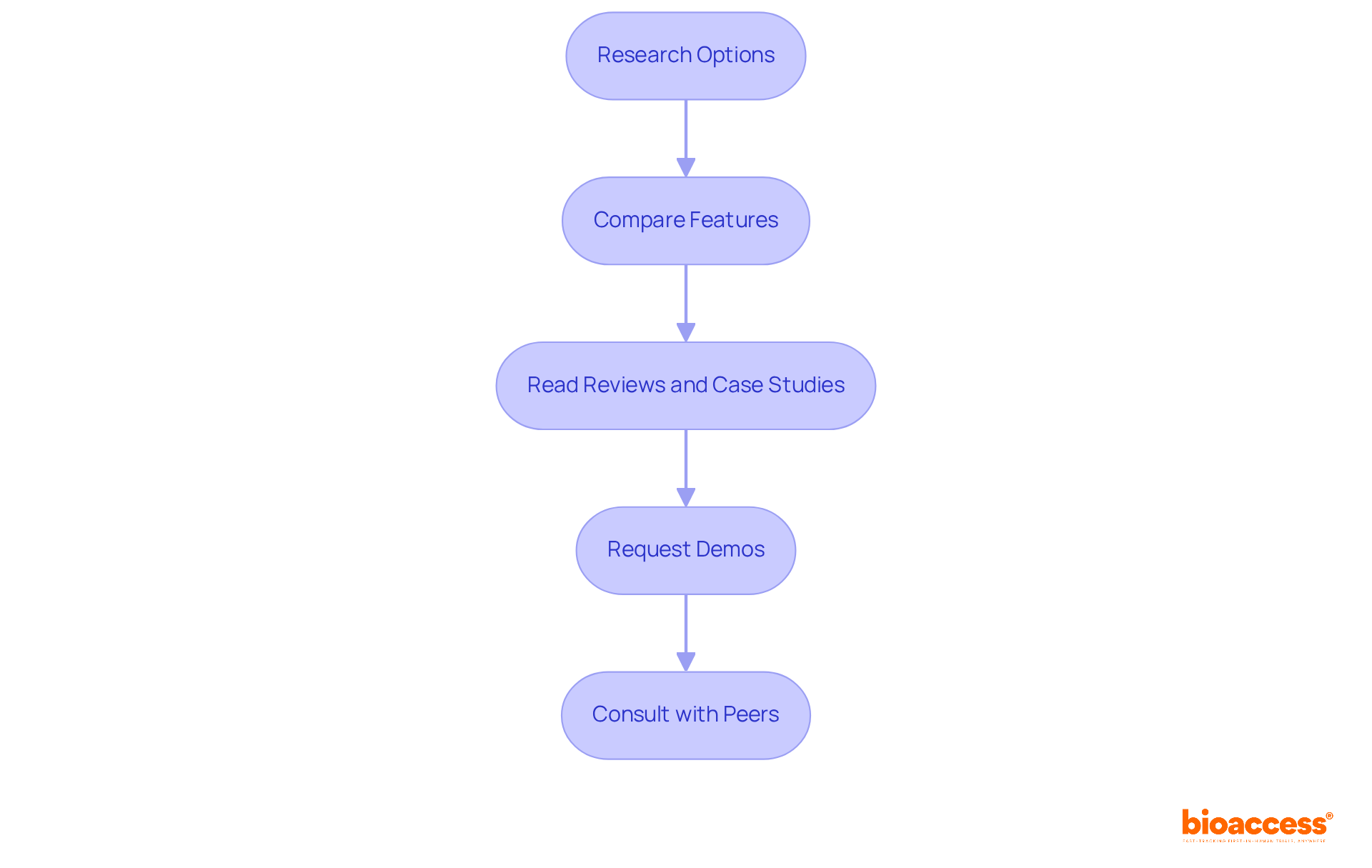
To effectively evaluate for your project, follow these essential steps:
-
Research Options: Begin by compiling a list of reputable tools such as nQuery, rpact, and ADDPLAN, all recognized for their capabilities in frameworks.
-
Compare Features: , including:
- Available statistical methods
- User interface and overall ease of use
-
Read Reviews and Case Studies: Delve into to uncover insights from researchers who have utilized these programs. Look for satisfaction ratings and specific comments on performance and usability. For example, and robust statistical procedures, while rpact receives praise for its flexibility in design options.
-
Request Demos: Contact vendors to . Observing the interface and functionalities firsthand will help you assess how well it meets your needs.
-
Consult with Peers: to gather insights on their experiences with different tools. Peer recommendations can provide valuable context and assist you in making an informed decision.
By systematically evaluating these factors, including user satisfaction ratings and real-world case studies, you can determine the best way to purchase an adaptive design statistical package for your . This ensures alignment with your project requirements and enhances your .
Purchase the Selected Statistical Package
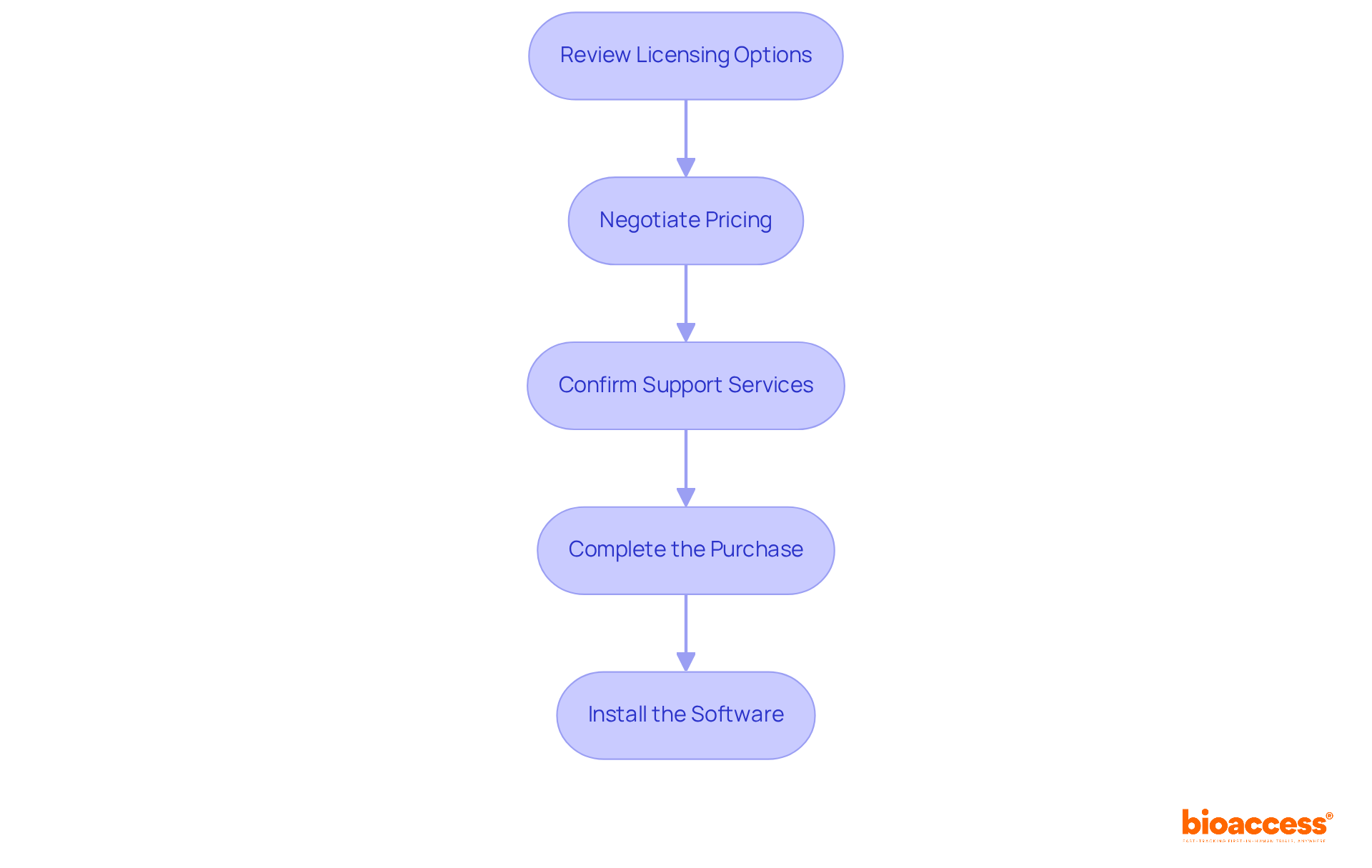
After assessing and selecting the most suitable adaptive design statistical tool, it’s time to move forward with the purchase of the . Here’s how to navigate this crucial process:
- Review : Understand the various licensing models available – whether it’s a one-time purchase or a subscription-based model – and select the one that best fits your budget and usage needs.
- Negotiate Pricing: Don’t shy away from negotiating with the vendor for better pricing or additional features, especially if you’re acquiring multiple licenses.
- : Ensure that your purchase includes adequate , such as technical assistance and training resources, to facilitate a smooth experience.
- Complete the Purchase: Follow the vendor’s purchasing process, which may involve filling out forms and providing payment information.
- Install the Software: Once the purchase is complete, adhere to the provided by the vendor to set up the software on your systems.
By carefully navigating this purchasing process, you can ensure a seamless acquisition when you choose to purchase the , setting the stage for successful .
Access Training and Support Resources
To maximize the effectiveness of your purchase adaptive design statistical package, accessing is crucial. Here’s how you can do it:
- : Investigate whether the vendor offers , webinars, or tutorials that can help you and your team become proficient in using the application.
- : Thoroughly review the provided with the program for comprehensive instructions on its features and functionalities.
- : Engage with online forums or user groups related to the software, where you can ask questions, share experiences, and learn from fellow users.
- : Understand the options available, including how to reach out for assistance and the hours during which support is offered.
- : Keep abreast of by subscribing to newsletters or following the vendor’s blog.
By actively engaging with these resources, you can enhance your skills and ensure you’re fully leveraging your purchase adaptive design statistical package.
Conclusion
Investing in an adaptive design statistical package is a pivotal move for enhancing the efficiency and effectiveness of clinical trials. Understanding the unique capabilities of these tools, alongside the specific needs of a project, empowers researchers to make informed decisions that ultimately lead to improved patient outcomes and streamlined trial processes.
Key considerations in this process include:
- Clearly defining project requirements
- Evaluating various software options
- Ensuring compliance with regulatory standards
This involves thorough research, feature comparisons, and engaging with industry peers to gather valuable insights. With a strategic approach, selecting and acquiring a statistical package that aligns with trial objectives becomes a manageable and straightforward task.
Ultimately, the investment in an adaptive design statistical package transcends mere software acquisition; it embodies a commitment to a transformative approach in clinical research. By leveraging these innovative tools, researchers can adapt to real-time data, refine trial designs, and pave the way for significant advancements in healthcare. Taking decisive action now can profoundly impact the future of clinical trials and enhance patient care.
Frequently Asked Questions
What is an adaptive design statistical package?
An adaptive design statistical package is a specialized software tool that enhances the planning and analysis of clinical trials by allowing modifications to trial parameters, such as sample size or treatment allocation, based on interim results.
What are the key features of adaptive statistical software tools?
Key features include the ability to implement group sequential methodologies and conduct sample size re-estimation, which improve trial efficiency and address ethical considerations.
Why is it important to understand terms like ‘adaptive trial approach’ and ‘interim analysis’?
Understanding these terms is crucial for navigating the various options available in the market and effectively utilizing adaptive design statistical packages in clinical research.
What is the projected market share for adaptive design statistical packages by 2026?
The projected market share is USD 25.13 billion by 2026.
What are some factors to consider before purchasing an adaptive design statistical package?
Key factors include trial design, regulatory compliance, data management, user experience, and budget constraints.
How can trial design impact the choice of an adaptive design statistical package?
Identifying the specific type of adaptive design, such as group sequential or sample size re-estimation, ensures the software aligns with the study’s objectives.
Why is regulatory compliance important when selecting an adaptive design statistical package?
Ensuring the application adheres to relevant regulatory guidelines helps facilitate smooth approval processes and maintain compliance, preventing delays in trial approvals.
What should be evaluated regarding data management when choosing an adaptive design statistical package?
It’s important to assess data input formats and the system’s ability to integrate with existing setups, as effective data handling enhances accuracy and accessibility.
How does user experience affect the selection of adaptive design statistical software?
Considering the technical expertise of the team and the software’s user-friendliness can boost productivity and reduce training time, making intuitive solutions critical.
What should be included in the budget when planning to purchase an adaptive design statistical package?
The budget should encompass the initial purchase cost as well as ongoing maintenance and support expenses to ensure sustainable investment and long-term planning.
List of Sources
- Understand Adaptive Design Statistical Packages
- Clinical Trial Design Trends for 2025 (https://statsols.com/guides/clinical-trial-design-trends-2025)
- Adaptive Design of Confirmatory Trials: Advances and Challenges – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC4639447)
- Evolution of clinical trials: An overview on adaptive… : Journal of Current Research in Scientific Medicine (https://journals.lww.com/jcsm/fulltext/2025/07000/evolution_of_clinical_trials__an_overview_on.3.aspx)
- Adaptive design methods in clinical trials – a review – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC2422839)
- Europe Clinical Trials Market Size, Growth and Trends Outlook 2031 (https://mordorintelligence.com/industry-reports/europe-clinical-trials-market)
- Identify Your Project Requirements
- How Should I Document My Solution Design Software Requirements? (https://soltech.net/how-should-i-document-my-software-requirements)
- Adaptive Software Development Explained: Guide & Key Insights (https://devtrust.biz/resources/blog/adaptive-software-development)
- Statistical considerations for outcomes in clinical research: A review of common data types and methodology – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC9134761)
- 7 Key Findings from the 2022 Clinical Trial Operations Technology Survey (https://florencehc.com/blog-post/7-key-findings-from-the-2022-clinical-trial-operations-technology-survey)
- In Focus: The Top Needs in a Clinical Trials Software Package | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/in-focus-the-top-needs-in-a-clinical-trials-software-package)
- Evaluate Available Statistical Packages
- Adaptive Clinical Trial Design with nQuery (https://statsols.com/nquery/adapt)
- An overview of methodological considerations regarding adaptive stopping, arm dropping, and randomization in clinical trials (https://jclinepi.com/article/S0895-4356(22)00274-8/fulltext)
- QUOTES | Quantification and Optimization of Trial Expectations Simulator by Berry (https://berryconsultants.com/software/quotes)
- Adaptive Trial Design: A Guide to Flexible Clinical Trials | IntuitionLabs (https://intuitionlabs.ai/articles/adaptive-clinical-trial-design)
- Access Training and Support Resources
- Statistics in Clinical Trials (https://udemy.com/course/statistics-in-clinical-trials?srsltid=AfmBOorYlPbxgnF2H_S_ezCYhon4CldFvElMR11KZCM6fBbkRcz-bJBW)
- Webinars (https://statsols.com/webinars)
- Statistical Analysis Training For Clinical Trials (https://evoclinical.com/services/statistical-training)
- 40 Statistics You Should Know About Online Communities (https://social.plus/blog/40-statistics-you-should-know-about-online-communities)
- Online Community Stats for 2026 (https://bettermode.com/blog/online-community-stats)

Leave a Reply