Introduction
The landscape of Class 1 medical devices is evolving at a rapid pace, driven by stringent regulatory frameworks and an urgent need for compliance. As manufacturers navigate this complex terrain, grasping the key compliance insights for 2025 is essential for ensuring patient safety and achieving market success.
What critical regulations and best practices can empower producers to not only meet compliance requirements but also enhance their operational efficiency? This article explores ten vital insights designed to equip manufacturers with the knowledge necessary to thrive in the competitive medical device sector.
bioaccess®: Accelerating Compliance for Class 1 Medical Devices
bioaccess® leverages its extensive expertise in , the Balkans, and Australia to streamline compliance for . By harnessing local knowledge and established networks, bioaccess® empowers manufacturers to secure – significantly faster than traditional timelines. This rapid turnaround is crucial for innovators eager to seize in the medical equipment sector.
Recent , including the alignment of , further bolster the efficiency of market entry for . For example, the Therapeutic Goods Administration (TGA) has underscored the importance of , ensuring that manufacturers not only fulfill compliance requirements but also prioritize patient safety. Successful case studies illustrate that companies utilizing bioaccess®’s services have adeptly navigated these regulatory landscapes, achieving and enhance patient outcomes.
Understanding Regulatory Frameworks for Class 1 Devices
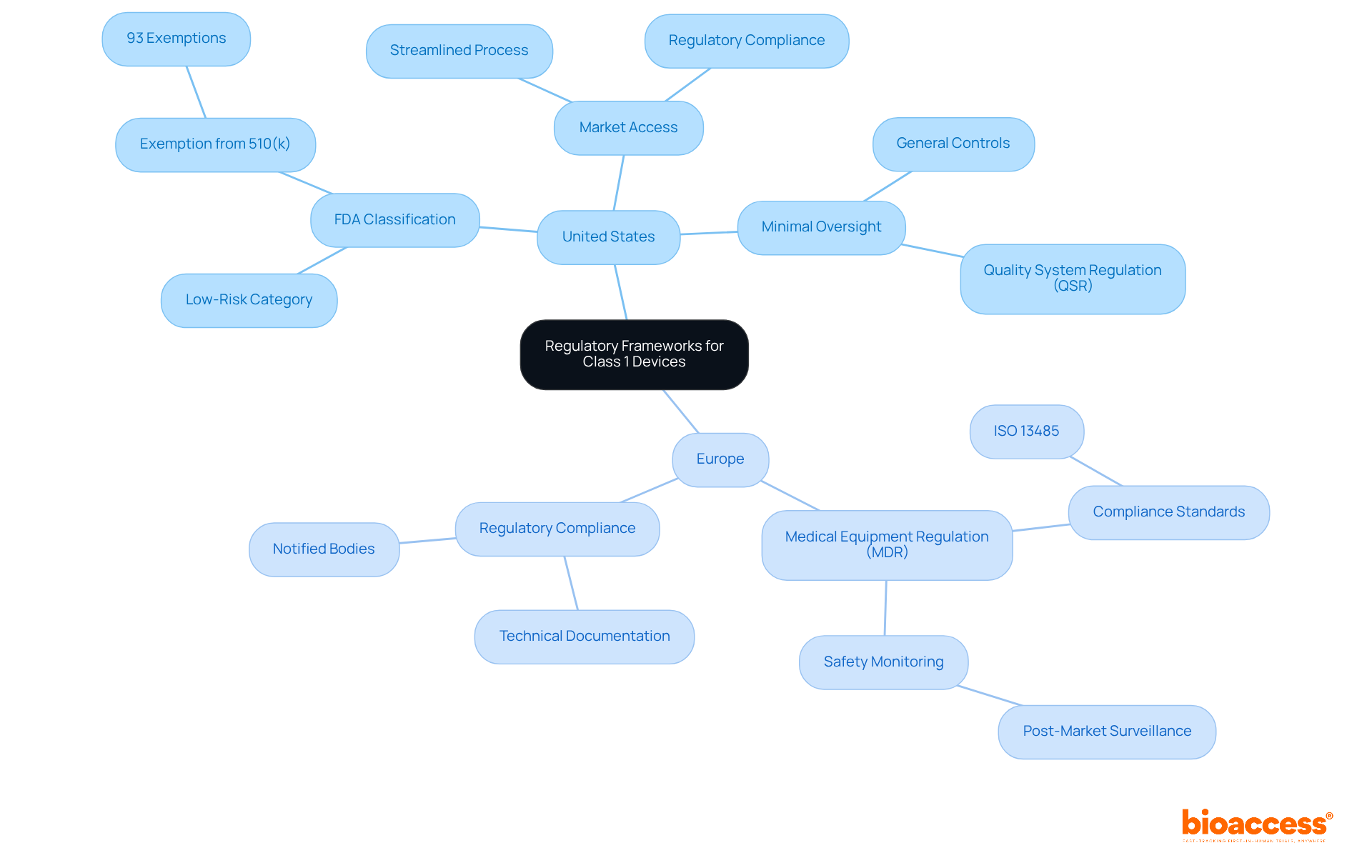
represent a crucial segment of the healthcare landscape, categorized as low-risk products with minimal . In the United States, the FDA classifies these products as a class 1 device under the , allowing approximately 93% of them to enter the market without the need for (510(k)). This streamlined process not only facilitates but is vital for manufacturers eager to introduce their innovations efficiently.
In Europe, the outlines specific provisions for , mandating . Adhering to these regulations is not just a formality; it ensures that equipment meets and is monitored effectively throughout its lifecycle. Manufacturers must navigate these frameworks diligently to maintain compliance and protect public health. Notably, nearly half of all , which are considered , fall into this category, underscoring their significance in healthcare and the necessity for rigorous oversight.
Quality Management System (QMS) Requirements for Class 1 Devices
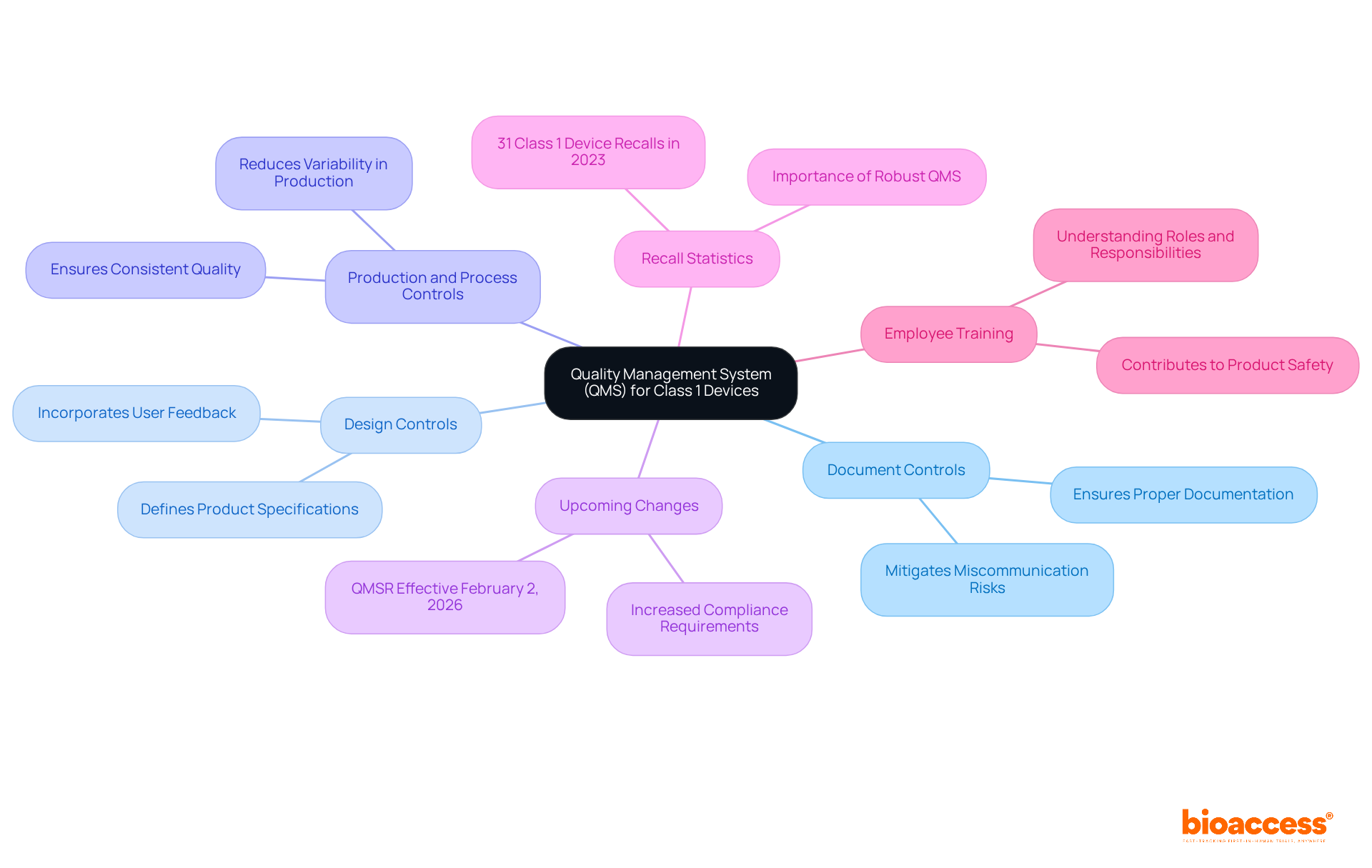
Producers of must implement a (QMS) that adheres to specified in 21 CFR Part 820. Establishing a QMS is , even though numerous Category 1 items are exempt from certain QMS requirements. Key components of a QMS include:
- Document controls
- Design controls
- Production and process controls
These components help mitigate risks and enhance product reliability.
The upcoming changes to the (QMSR), effective February 2, 2026, will further emphasize the need for compliance. In 2023, there were 31 recalls of class 1 devices, highlighting the importance of a in preventing such issues. The serves as the foundation for a QMS, guiding producers in upholding quality and compliance.
Moreover, to ensure that all personnel understand their roles and responsibilities, contributing to overall . Remarkably, roughly 80% of producers successfully navigate the regulatory environment for , highlighting the significance of a well-organized QMS in attaining compliance and promoting innovation.
As Peter Drucker wisely noted, efficiency alone does not equate to value; thus, a focus on quality management is vital for delivering meaningful improvements in product performance and customer satisfaction.
Labeling and Documentation Standards for Class 1 Medical Devices
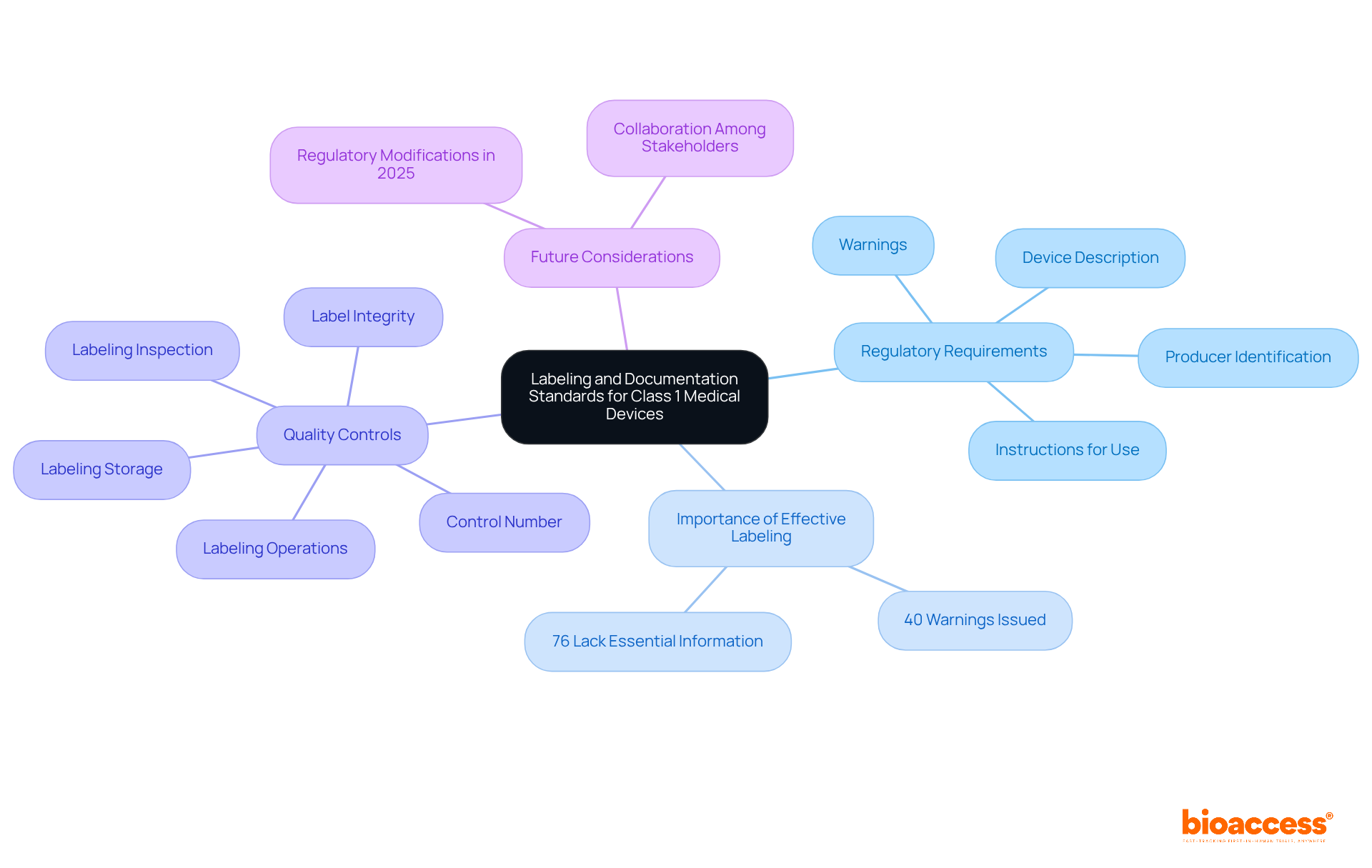
is not only a but also a critical component of . According to FDA regulations specified in 21 CFR Part 801, manufacturers must provide , a detailed device description, and any necessary warnings or precautions. This documentation must include easily understandable instructions for use (IFU) to ensure safe operation.
Effective labeling is crucial for compliance, as nearly 40 warnings have been issued to manufacturers for inadequate documentation of training activities. This statistic underscores the importance of thoroughness in this area. Alarmingly, approximately 76% of healthcare instruments lack essential labeling information, indicating a significant need for improvement. Adhering to documentation standards not only enhances patient safety but also mitigates the risk of adverse events related to labeling issues.
Manufacturers must maintain clear labeling procedures that focus on five quality controls:
- Label integrity
- Labeling inspection
- Labeling storage
- Labeling operations
- Control number
is essential, especially with expected regulatory modifications in 2025. This ensures that all labeling reflects any alterations in the product’s intended use and adheres to .
In conclusion, , and collaboration among stakeholders is vital. By prioritizing compliance and thorough documentation, manufacturers can significantly improve patient safety and operational efficiency.
Post-Market Surveillance and Vigilance for Class 1 Devices
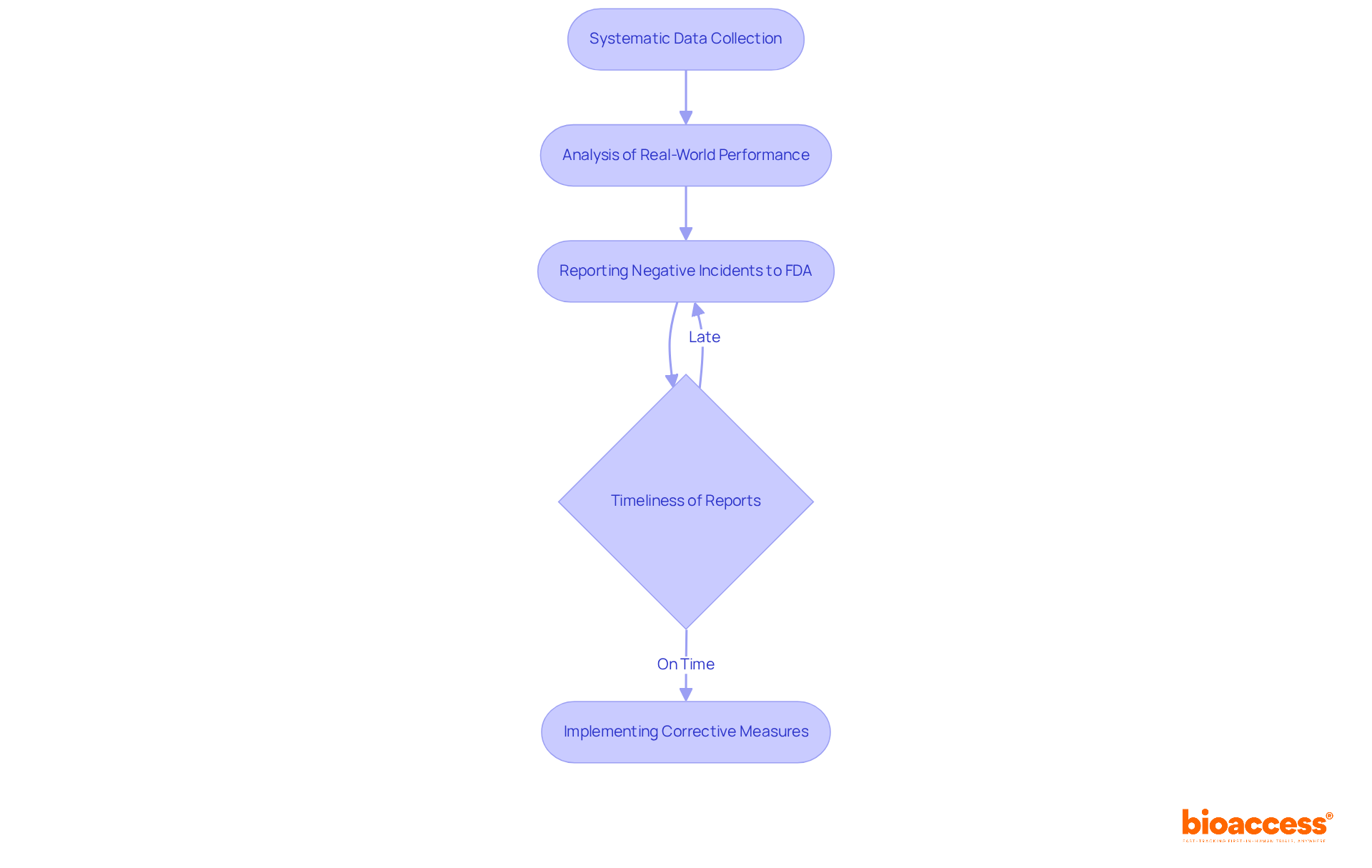
for a class 1 device is essential, involving the systematic collection and analysis of data regarding its . Manufacturers must report any negative incidents or product malfunctions to the FDA within specific timeframes. Alarmingly, nearly 33% of from manufacturers to the FDA are submitted late, often exceeding six months after the manufacturer was informed. This delay can hinder timely interventions and .
Awareness in overseeing is crucial for recognizing potential concerns and ensuring that . Advocates for protection highlight that a not only shields patients but also enhances the overall . Regular reviews and updates to are vital to adapt to current trends and maintain compliance, ultimately fostering a safer healthcare environment.
As John C. Maxwell aptly states, ‘Tend to the people, and they will tend to the business,’ underscoring the significance of prioritizing security in all operations. This commitment to vigilance is not just a regulatory requirement; it is a moral imperative that safeguards patient health and builds trust in the healthcare system.
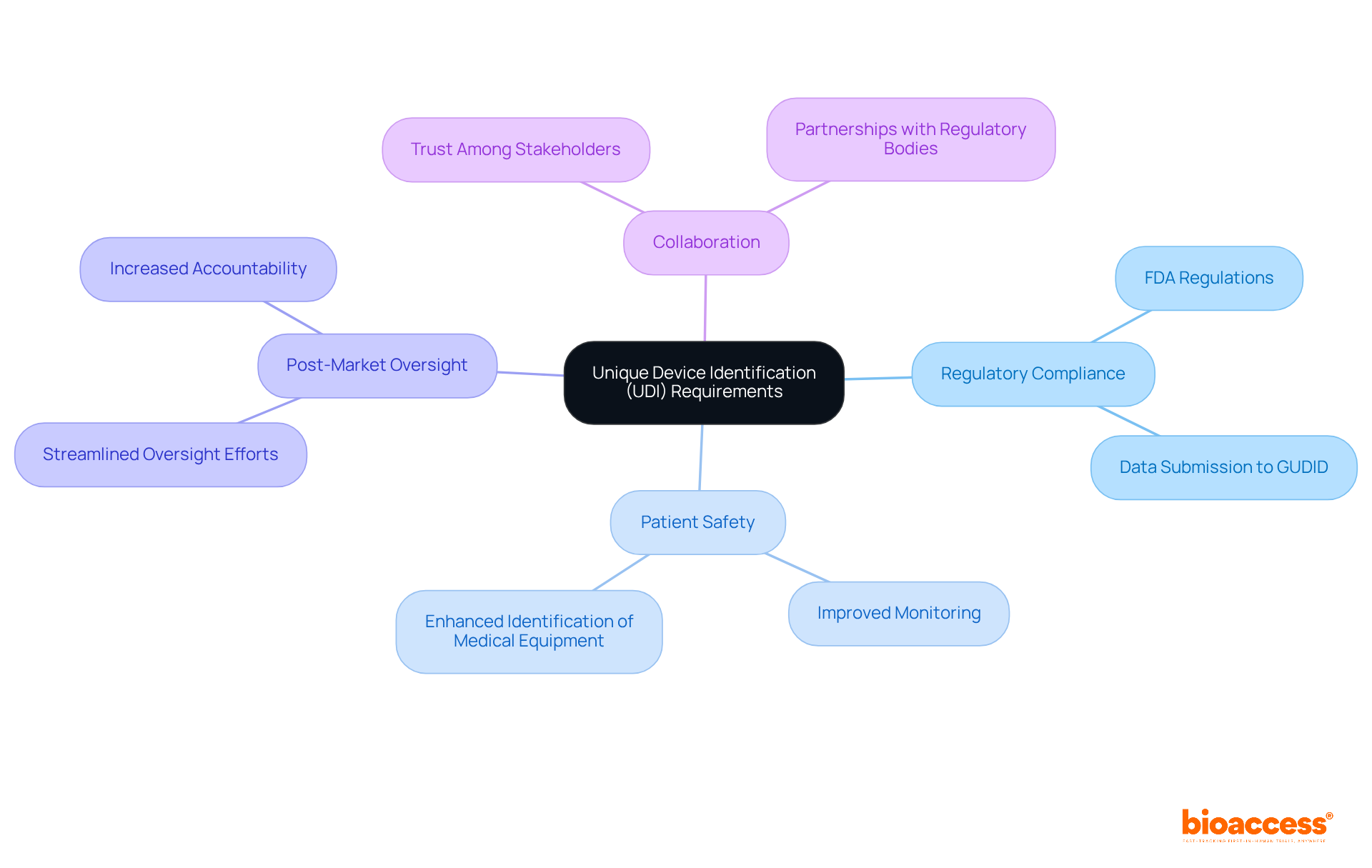
Unique Device Identification (UDI) Requirements for Class 1 Devices
The is crucial for ensuring that Category 1 items prominently display a UDI on their labels and packaging. This requirement not only facilitates improved monitoring and identification of but also significantly boosts and enhances . In the evolving Medtech landscape, manufacturers must ensure their UDI complies with , which encompass specific formatting and data submission requirements to the Global Unique Device Identification Database (GUDID).
As the healthcare industry continues to advance, the importance of adhering to these regulations cannot be overstated. By implementing the UDI system effectively, manufacturers can address key challenges in and contribute to a safer healthcare environment. This commitment to compliance not only fosters trust among stakeholders but also reinforces the credibility of the Medtech sector as a whole.
In conclusion, collaboration among manufacturers, regulatory bodies, and healthcare providers is essential for navigating the complexities of the UDI system. By taking proactive steps to ensure compliance, stakeholders can enhance and streamline .
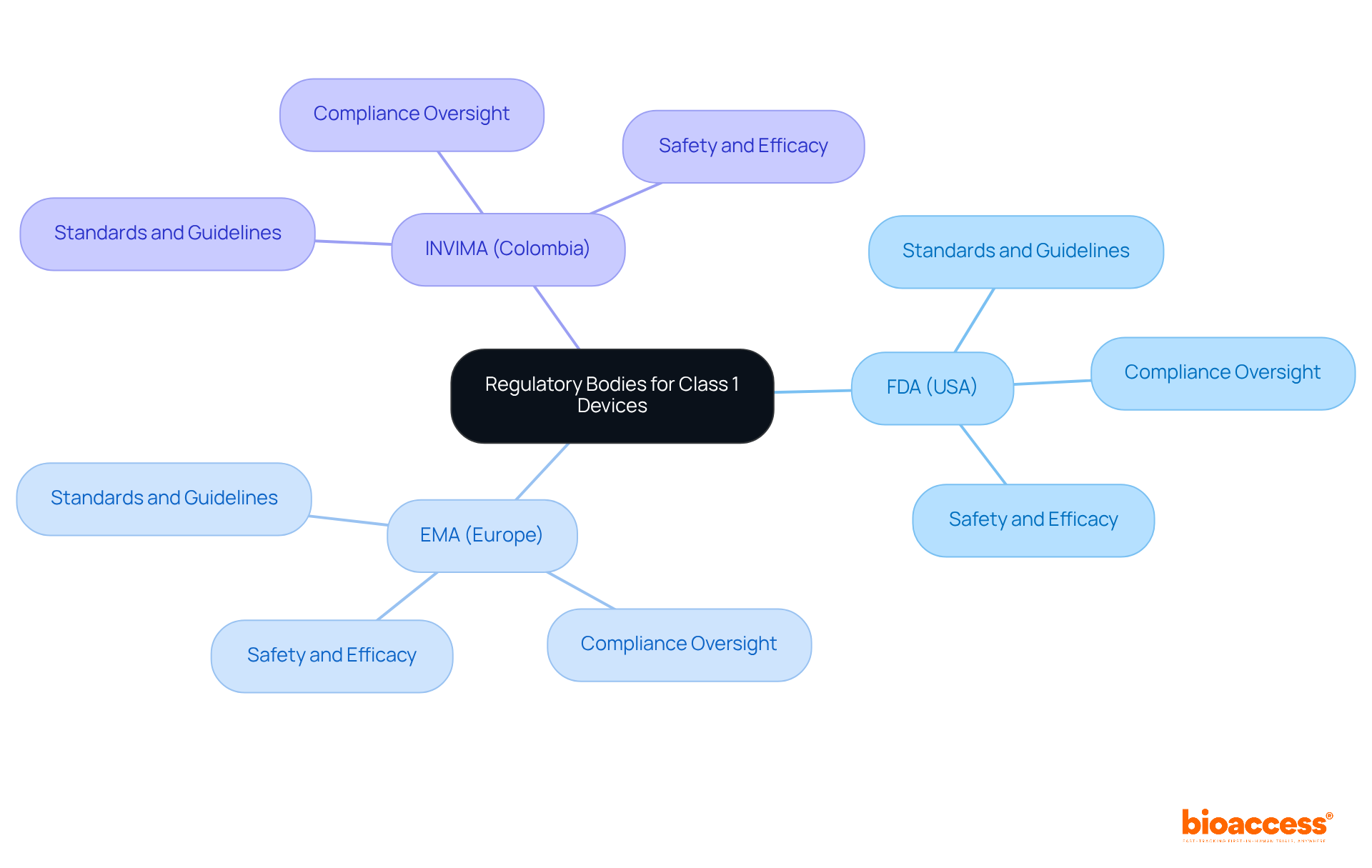
Regulatory Bodies and Standards Governing Class 1 Devices
are primarily regulated by the FDA in the United States and the (EMA) in Europe. These organizations set the standards and guidelines that manufacturers must adhere to for compliance. In Colombia, the INVIMA (Colombia National Food and Drug Surveillance Institute) plays a crucial role in overseeing medical equipment, ensuring they meet . Recognized as a Level 4 health authority by the Pan American Health Organization/World Health Organization, INVIMA demonstrates its competence in health regulation functions.
Furthermore, specialists like Ana Criado, Director of Regulatory Affairs at bioaccess, bring invaluable expertise from INVIMA and academia, offering critical insights into the regulatory landscape for . Understanding the roles of these is essential for producers aiming to maintain compliance and secure market access. As the Medtech landscape evolves, collaboration with these authorities becomes increasingly vital for navigating the complexities of .
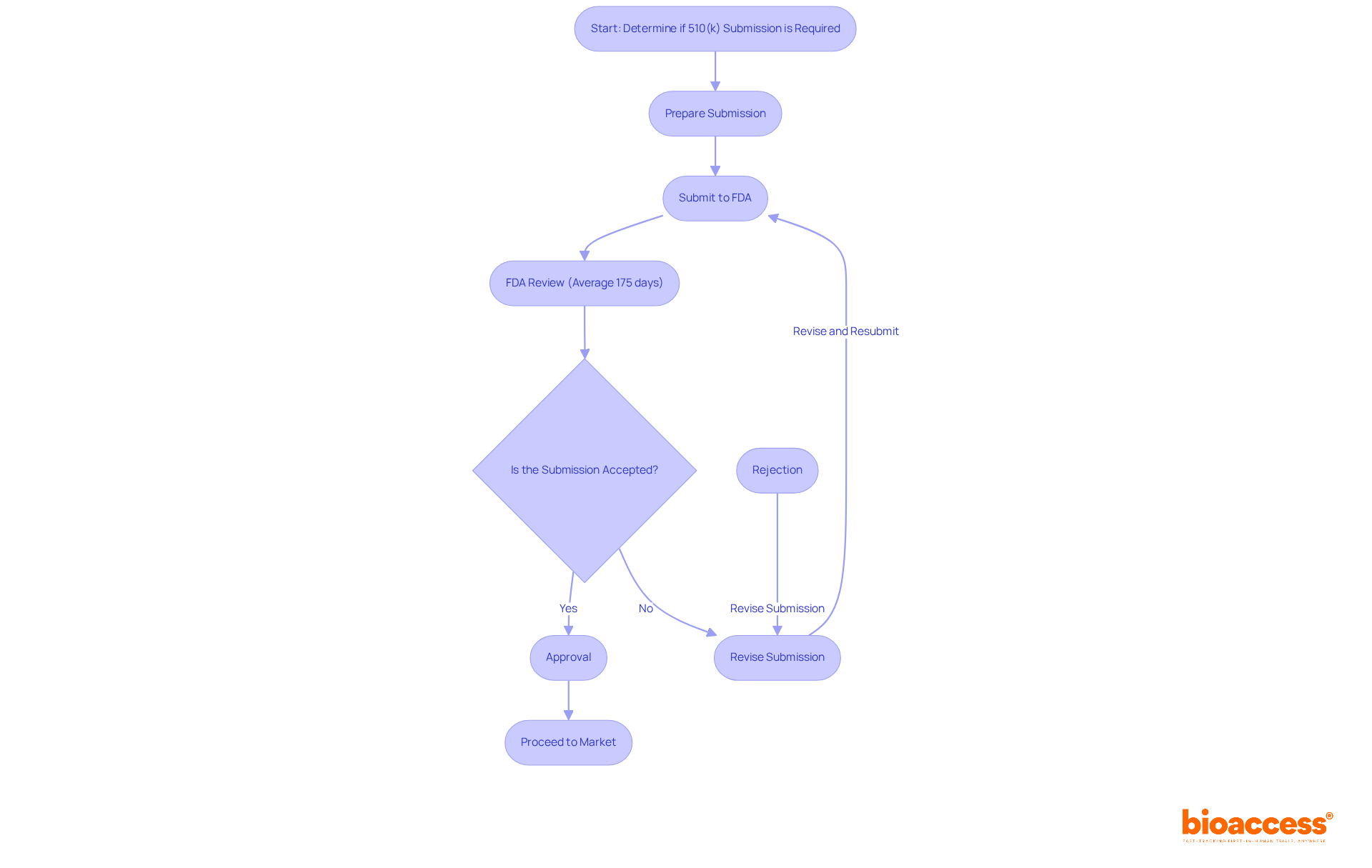
Navigating the FDA 510(k) Process for Class 1 Devices
While most products classified as a are exempt from the , certain items may still necessitate a submission if they do not meet specific criteria. The , requiring manufacturers to prove that their product is to a legally marketed item. This entails preparing a thorough submission that contains detailed information on security and effectiveness. In fact, approximately 30% of 510(k) submissions were not accepted for initial review in 2022, underscoring the importance of .
Successful instances of often encompass items like bandages and portable surgical tools, recognized as s, which have maneuvered through the process efficiently. Recent alterations in 510(k) requirements have clarified that even minor adjustments to a may require a if they affect safety or efficacy. Grasping this process is crucial for producers to guarantee compliance and enable prompt market entry. The , with an average approval duration of 175 days and a median approval duration of 85 days.
Engaging with the FDA early and maintaining clear communication can further enhance the likelihood of a successful submission. This proactive approach not only streamlines the process but also fosters a collaborative relationship with regulatory bodies, ultimately benefiting the entire .
Understanding the EU CE Marking Process for Class 1 Devices
To effectively promote Category 1 , producers must secure , a crucial verification of compliance with EU regulations. This process involves:
- Preparing a
- Conducting a thorough risk assessment
- Ensuring adherence to the (GSPRs) outlined in the Medical Device Regulation (MDR)
While many can be self-certified, certain – such as sterile products, those with a measuring function, and reusable surgical instruments – require Notified Body involvement. Understanding these limitations is essential for navigating the .
Recent trends indicate a growing reliance on self-certification, particularly as producers strive to expedite their entry into the competitive European market. For example, devices that are non-sterile, non-measuring, and not reusable often follow this streamlined route, facilitating quicker compliance and reducing costs. Successful cases of self-certification in Europe demonstrate that with proper documentation and adherence to , companies can effectively navigate the complexities of and achieve their market objectives.
The significance of cannot be overstated; it not only paves the way for entry into the European market but also enhances a producer’s reputation, assuring customers of product quality, safety, and efficacy. As Maven points out, ‘Obtaining CE certification in Europe enables producers to access the entire European market, significantly expanding their business reach.’ Furthermore, producers must retain following the last product produced. It’s crucial to note that the final deadline for acceptance of CE marked medical equipment in Great Britain is 30 June 2030.
. Each box represents a key action, and the decision point shows whether self-certification or Notified Body involvement is required based on the type of device. Follow the arrows to see the steps needed for CE marking. Each box represents a key action, and the decision point shows whether self-certification or Notified Body involvement is required based on the type of device.](https://images.tely.ai/telyai/jmpvayoq-follow-the-arrows-to-see-the-steps-needed-for-ce-marking-each-box-represents-a-key-action-and-the-decision-point-shows-whether-self-certification-or-notified-body-involvement-is-required-based-on-the-type-of-device.webp)
Implementing Corrective and Preventive Actions (CAPA) for Class 1 Devices
Implementing is crucial for producers of , as it effectively addresses non-conformities and prevents future issues. A robust involves:
- Identifying problems
- Thoroughly investigating root causes
- Executing corrective actions aimed at mitigating risks
For example, a catheter producer tackled variations in material hardness by requalifying suppliers and enhancing raw material inspection protocols. This case illustrates the real-world effectiveness of CAPA.
Regular reviews and updates to the are vital for producers to maintain compliance with , such as the . This but also promotes in product quality. , including the CAPA closure rate and the number of overdue CAPAs, serve as .
As regulatory expectations increase, the becomes a cornerstone of quality assurance, enabling manufacturers to sustain market access and build trust in their products. By prioritizing CAPA, companies can adeptly navigate the complexities of compliance while enhancing the overall safety and efficacy of their .
Conclusion
The compliance landscape for Class 1 medical devices is evolving, driven by regulatory updates and the imperative for manufacturers to prioritize patient safety and product quality. Understanding the frameworks and requirements is essential for navigating the complexities of bringing these low-risk devices to market efficiently. By leveraging expertise and resources like those offered by bioaccess®, manufacturers can significantly enhance their compliance processes, ensuring timely approvals and robust market entry.
Key insights discussed throughout the article underscore the importance of:
- A comprehensive Quality Management System (QMS)
- Adherence to labeling standards
- Effective post-market surveillance
The implementation of Unique Device Identification (UDI) requirements and a thorough understanding of regulatory bodies further reinforce the necessity for manufacturers to stay informed and compliant. Moreover, proactive engagement with the FDA and the EU CE marking process is vital for maintaining a competitive advantage in the ever-evolving Medtech landscape.
Ultimately, the commitment to compliance transcends mere regulatory obligation; it embodies a moral imperative that safeguards patient health and fosters trust within the healthcare system. Manufacturers are encouraged to embrace these insights and best practices, ensuring their Class 1 devices meet the highest standards of safety and efficacy. By prioritizing compliance, the industry can pave the way for innovation while upholding the integrity of medical device manufacturing.
Frequently Asked Questions
What is bioaccess® and how does it help with compliance for Class 1 medical devices?
bioaccess® is a regulatory consulting service that leverages expertise in regulatory frameworks across Latin America, the Balkans, and Australia to streamline compliance for Class 1 medical devices. It enables manufacturers to secure ethical approvals in 4-6 weeks, significantly faster than traditional timelines.
What recent updates have been made to the regulatory processes for Class 1 devices?
As of 2025, regulatory processes have been aligned with international standards, enhancing the efficiency of market entry for Class 1 medical devices. Notably, the Therapeutic Goods Administration (TGA) has emphasized the importance of cybersecurity throughout the product lifecycle.
What are Class 1 devices and what is their significance?
Class 1 devices are categorized as low-risk medical products with minimal regulatory oversight. In the U.S., approximately 93% of these devices can enter the market without premarket notification, facilitating quicker access for manufacturers and highlighting their importance in the healthcare landscape.
What are the Quality Management System (QMS) requirements for Class 1 devices?
Producers of Class 1 devices must implement a QMS that adheres to FDA regulations specified in 21 CFR Part 820. Key components of a QMS include document controls, design controls, and production and process controls, which help ensure consistent product quality and mitigate risks.
What changes are expected in the Quality Management System Regulation (QMSR) in 2026?
Effective February 2, 2026, there will be changes to the QMSR that will further emphasize the need for compliance among producers of Class 1 devices.
How important is employee training in the QMS for Class 1 devices?
Employee training is crucial within the QMS to ensure that all personnel understand their roles and responsibilities, which contributes to overall product safety and efficacy.
What percentage of producers successfully navigate the regulatory environment for Class 1 products?
Approximately 80% of producers successfully navigate the regulatory environment for Class 1 products, indicating the importance of a well-organized QMS in achieving compliance and fostering innovation.
List of Sources
- bioaccess®: Accelerating Compliance for Class 1 Medical Devices
- Medical Devices Archives (https://regask.com/category/news/medical-devices-insights)
- bioaccessla.com (https://bioaccessla.com/blog/10-key-fda-insights-on-class-1-medical-devices-you-need-to-know)
- Medical Devices Compliance & Regulatory News – Newsletter Oct 2025 | Sushvin Consulting (https://sushvin.com/medical-devices-compliance-updates-newsletter-Oct-2025.html)
- Latin American patients must wait an average of 4.7 years to access innovative or cutting-edge treatments (https://fifarma.org/en/waitindicator2024)
- Understanding Regulatory Frameworks for Class 1 Devices
- bioaccessla.com (https://bioaccessla.com/blog/10-key-fda-insights-on-class-1-medical-devices-you-need-to-know)
- Regulatory Requirements for Class 1 Medical Devices | DeviceLab (https://devicelab.com/blog/regulatory-requirements-for-class-1-medical-devices)
- FDA Class I Medical Device Regulations and Compliance – Ketryx Compliance Framework (https://ketryx.com/blog/fda-class-i-medical-device-regulations-and-compliance)
- Quality Management System (QMS) Requirements for Class 1 Devices
- bioaccessla.com (https://bioaccessla.com/blog/10-key-fda-insights-on-class-1-medical-devices-you-need-to-know)
- 5 Important Things for Life Sciences Companies to Consider | MasterControl (https://mastercontrol.com/gxp-lifeline/quality-inspiration)
- QMSR: Understanding Part 820.10 “Requirements for a Quality Management System” and the “Applicable Regulatory Requirements” | AAMI (https://aami.org/news/qmsr-understanding-part-820-10-requirements-for-a-quality-management-system-and-the-applicable-regulatory-requirements)
- Achieving Medical Device Quality Management System Excellence (https://mddionline.com/manufacturing/achieving-medical-device-quality-management-system-excellence)
- 25 Quotes to Inspire Quality And Success – Pro QC Blog (https://proqc.com/blog/25-quotes-to-inspire-quality-success)
- Labeling and Documentation Standards for Class 1 Medical Devices
- bioaccessla.com (https://bioaccessla.com/blog/10-key-fda-insights-on-class-1-medical-devices-you-need-to-know)
- bioaccessla.com (https://bioaccessla.com/blog/4-key-insights-on-fda-medical-device-class-1-regulations)
- FDA Class I Medical Device Regulations and Compliance – Ketryx Compliance Framework (https://ketryx.com/blog/fda-class-i-medical-device-regulations-and-compliance)
- highpowervtls.com (https://highpowervtls.com/2025/04/why-medical-device-labeling-is-important)
- A Comprehensive Analysis of Class I Medical Device Recalls: Unveiling Patterns, Causes and Global Impacts – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11416579)
- Post-Market Surveillance and Vigilance for Class 1 Devices
- Medical Device Safety: The Rise of Active Postmarket Surveillance (https://postmarketmonitor.com/blog-posts/medical-device-safety-the-rise-of-active-postmarket-surveillance)
- rheumatologyadvisor.com (https://rheumatologyadvisor.com/news/considerable-proportion-of-medical-device-adverse-events-reported-late)
- Top 20 Safety Quotes to Educate and Inspire Your Team (https://fluix.io/blog/safety-quotes)
- 22 Workplace Safety Quotes From Leaders and Experts – AlertMedia (https://alertmedia.com/blog/workplace-safety-quotes)
- Navigating the FDA 510(k) Process for Class 1 Devices
- Understanding the New FDA Guidance on Changes to a 510(k) (https://greenlight.guru/blog/fda-guidance-changes-to-510k)
- Everything you need to know about the FDA 510(k) submission (https://qualio.com/blog/fda-510k-submission)
- Navigating the FDA 510k Submission: A Complete Guide (https://matrixone.health/blog/navigating-the-fda-510k-submission-complete-guide)
- thefdagroup.com (https://thefdagroup.com/blog/510k-explained)
- Understanding the EU CE Marking Process for Class 1 Devices
- Guidance on Class I medical devices (https://gov.uk/government/collections/guidance-on-class-1-medical-devices)
- How to achieve MDR Compliance for Class I medical devices (https://mantrasystems.com/articles/how-to-achieve-mdr-compliance-for-class-i-medical-devices)
- CE marking for Class I medical devices: Can I self-certify my product? | CMC Medical Devices (https://cmcmedicaldevices.com/ce-marking-for-class-i-medical-devices-can-i-self-certify-my-product)
- Guide to European CE Marking for Medical Devices (https://mavenprofserv.com/european-ce-marking-for-medical-devices)
- scilife.io (https://scilife.io/blog/ce_certification_mark_medical_device)
- Implementing Corrective and Preventive Actions (CAPA) for Class 1 Devices
- CAPA for Medical Devices: Ensure Compliance and Quality (https://qualityze.com/blogs/capa-medical-devices-industry)
- Thermax’s ROSS simplifies machine monitoring and corrective maintenance at Adam Afrique – Thermax | Trusted Partner in Energy Transition (https://thermaxglobal.com/case_studies/thermaxs-ross-simplifies-machine-monitoring-and-corrective-maintenance-at-adam-afrique)
- Essential CAPA KPIs – The Learning Reservoir (https://thelearningreservoir.com/essential-capa-kpis-for-medical-device-companies)

Leave a Reply