Introduction
In Brazil, the landscape of post-market vigilance for medical devices is undergoing a significant transformation, driven by evolving regulations and the introduction of the Unique Device Identification (UDI) system set for 2025. This shift presents a critical opportunity for manufacturers to enhance compliance and operational efficiency through effective outsourcing strategies. However, as companies navigate these complex requirements, they face the challenge of selecting the right outsourcing model that aligns with their unique capabilities and goals.
How can organizations ensure they not only meet regulatory expectations but also foster a culture of safety and trust within the healthcare system? This question is pivotal as it underscores the importance of strategic decision-making in a rapidly changing environment.
Understand Post-Market Vigilance Requirements in Brazil
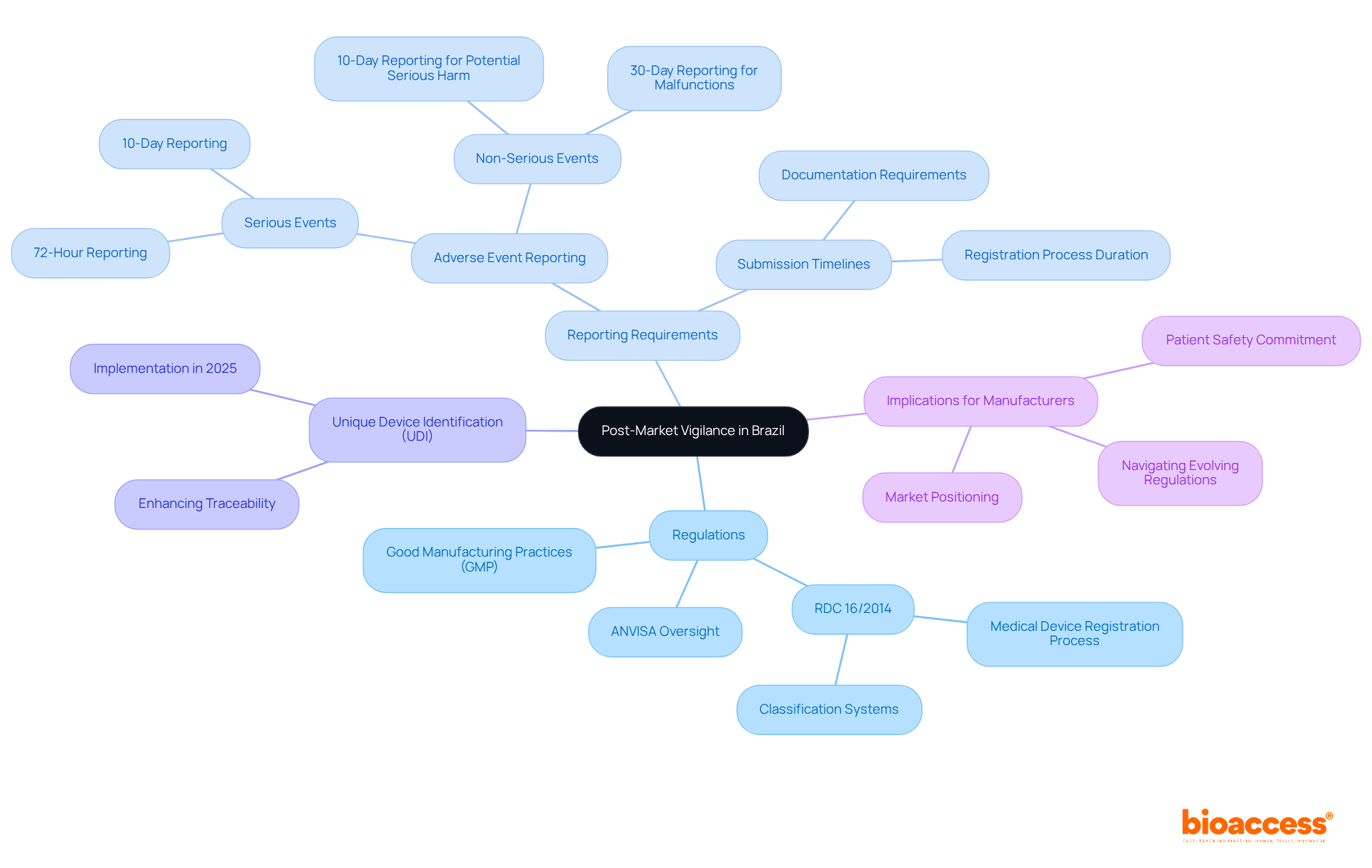
In Brazil, the for devices is crucial for ensuring their safety and efficacy. The National Health Surveillance Agency (ANVISA) oversees this process, requiring manufacturers to establish . These regulations mandate that manufacturers report within specific timeframes, underscoring the importance of .
Starting in 2025, the introduction of the will significantly enhance traceability and accountability in . This development not only aligns with but also fosters trust and safety within the healthcare landscape. Companies must be well-versed in , documentation requirements, and the types of incidents that necessitate notification. Are you prepared to navigate these evolving regulations?
This proactive approach mitigates the risk of penalties and reinforces the . By understanding and adhering to these guidelines, manufacturers can position themselves as leaders in the Medtech landscape, ultimately contributing to a more reliable healthcare system.
Evaluate Outsourcing Models for Post-Market Vigilance
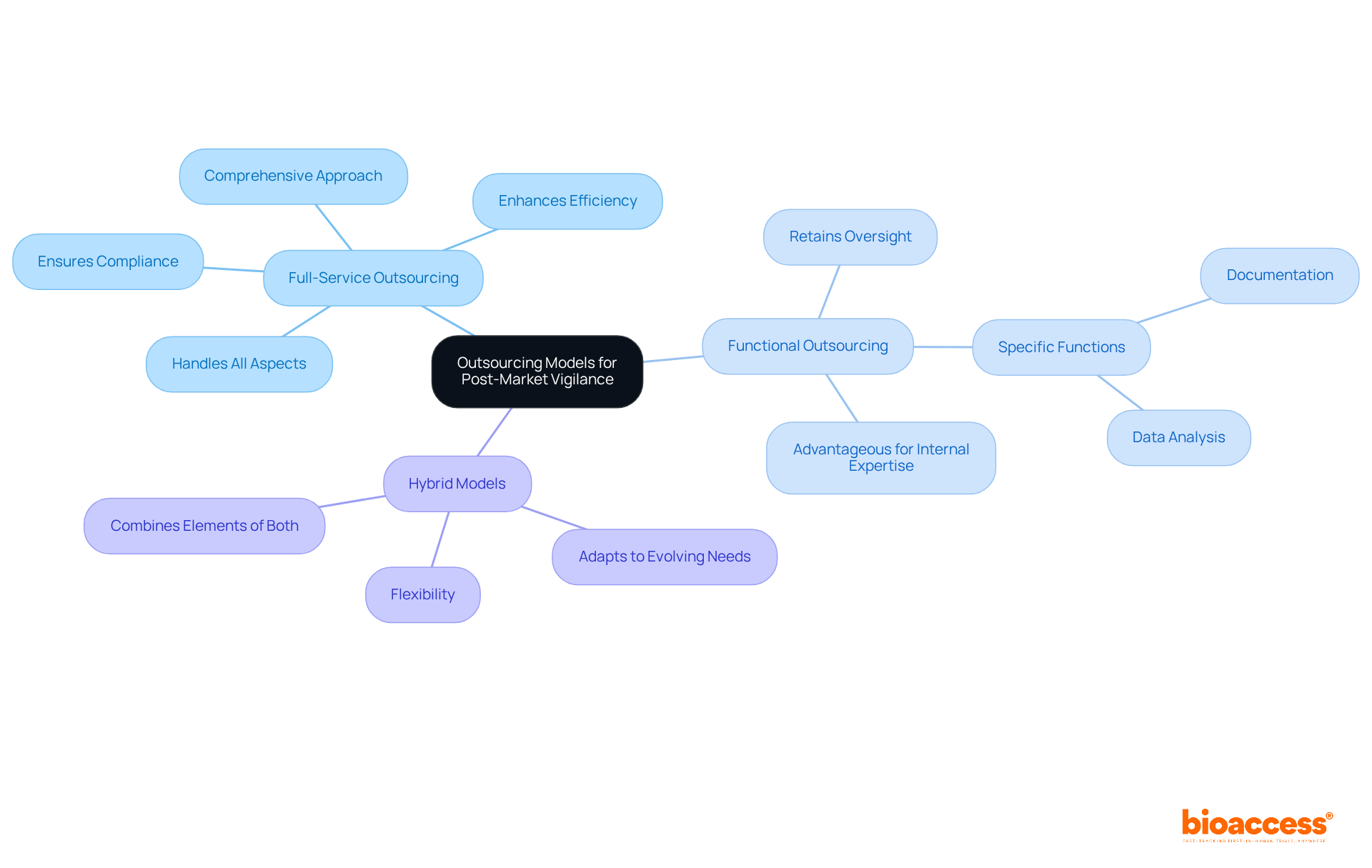
When considering , companies must evaluate several models:
Full-service outsourcing involves partnering with a specialized company like , which handles all aspects of in Brazil for devices. This includes , site selection, regulatory reviews, trial setup, import permits, and project management. Such a comprehensive approach not only enhances efficiency but also ensures compliance with regulations related to .
On the other hand, functional outsourcing allows firms to retain oversight of specific functions, such as data analysis or documentation. This can be particularly advantageous for companies with existing internal expertise. Meanwhile, a hybrid model merges elements of both approaches, providing the flexibility to adapt to evolving needs. Each model presents its own advantages and challenges, prompting companies to assess their internal capabilities, budget constraints, and the complexity of their product portfolio to identify the most suitable option.
Engaging with local specialists, like those at , can significantly aid in navigating Brazilian regulations related to Brazil devices. This collaboration ultimately enhances the effectiveness of and positively impacts local economies. As you consider your options, reflect on how these outsourcing models can address your unique challenges in clinical research.
Implement Effective Monitoring Systems for Compliance
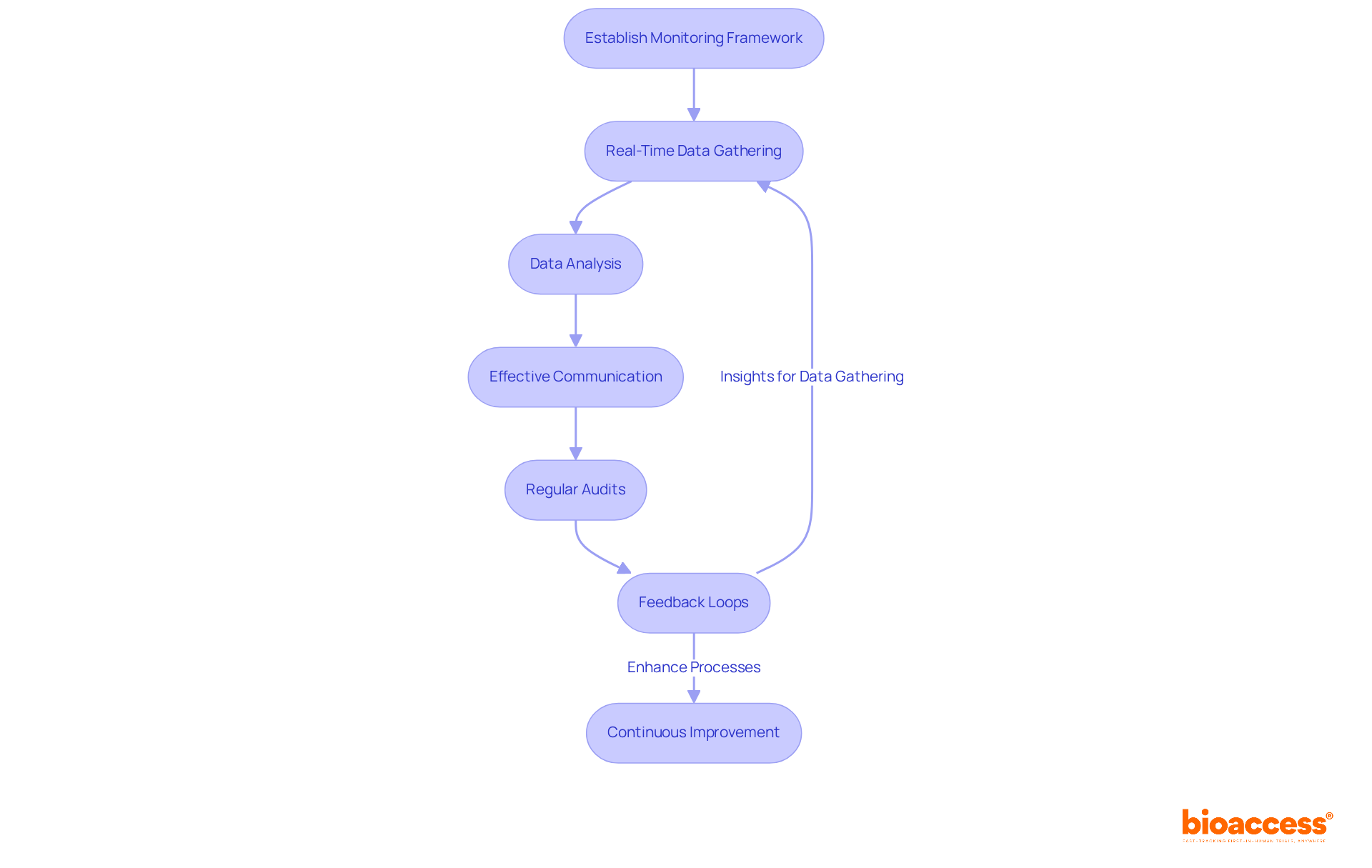
To fulfill in Brazil for devices, companies must establish robust , analysis, and effective communication methods. By leveraging technology – such as – organizations can significantly streamline the process of capturing . of these are crucial for identifying gaps and enhancing processes. Moreover, fostering and patients can greatly bolster . Integrating feedback loops into the monitoring systems facilitates continuous improvement, ensuring that these systems evolve based on insights derived from the data.
Enhance Team Competence through Continuous Training
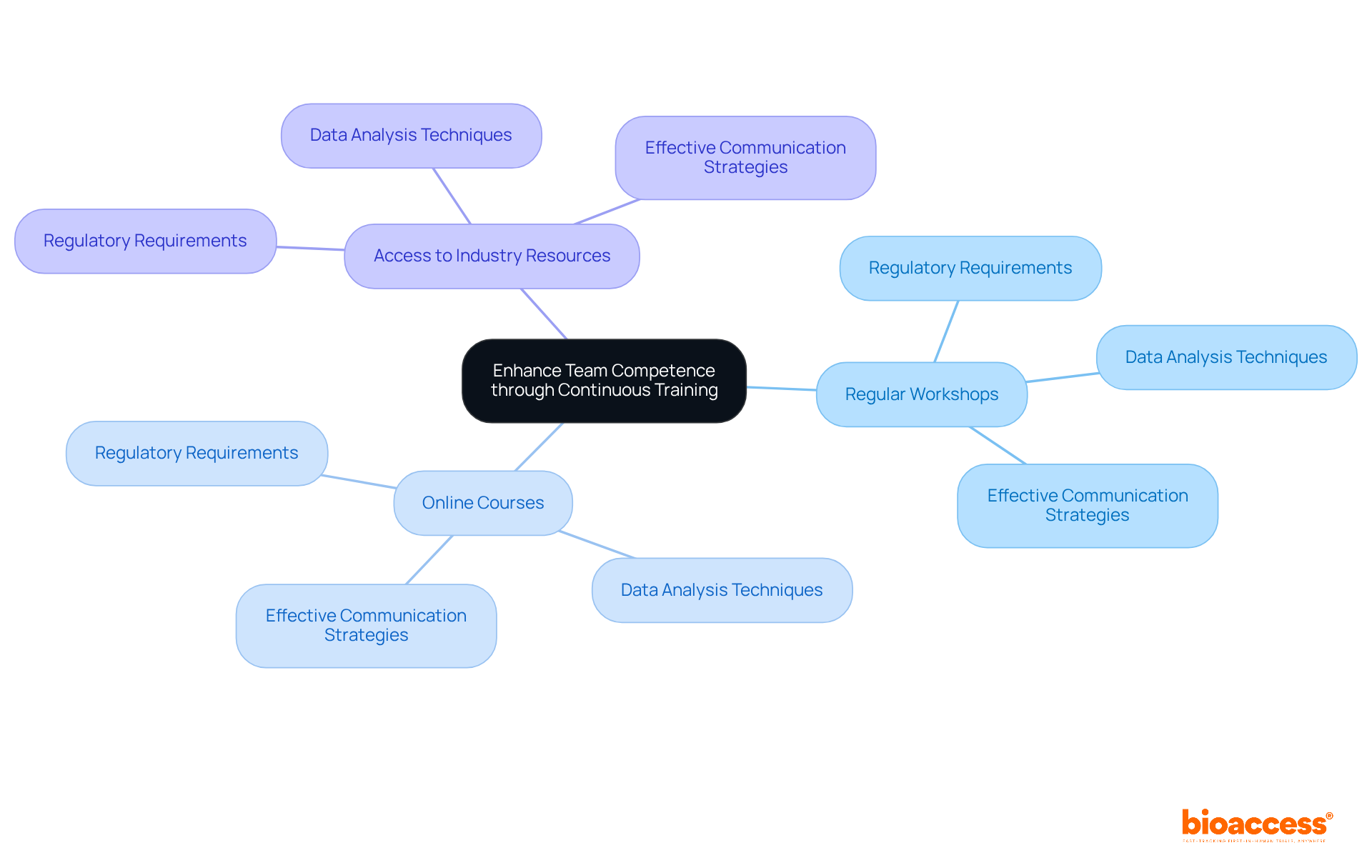
is vital for teams engaged in , ensuring they remain informed about and best practices. Companies must establish a that includes:
- Regular workshops
- Online courses
- Access to industry resources
This program should address such as:
- Data analysis techniques
Encouraging team members to participate in can provide valuable insights into emerging trends and challenges in . By cultivating a culture of ongoing education, organizations can significantly enhance their team’s proficiency and ensure compliance with ANVISA’s evolving regulations. Statistics reveal that only 10% of employees feel that has impacted their work practices, underscoring the need for engaging and effective strategies.
Incorporating microlearning modules and interactive content can improve knowledge retention and application, while regular assessments can measure effectiveness and pinpoint areas for improvement. Furthermore, structured training programs that align with regulatory standards not only promote an ethical workplace culture but also build market trust, ultimately supporting the organization’s long-term success in outsourcing Brazil devices.
Conclusion
Navigating the complexities of post-market vigilance outsourcing for medical devices in Brazil is not just essential; it’s a critical component for ensuring safety and compliance. Understanding the evolving regulatory landscape, especially with the upcoming implementation of the Unique Device Identification (UDI) system in 2025, is paramount. By adopting a proactive approach to compliance, manufacturers can not only meet regulations but also build trust within the healthcare sector.
Key insights reveal the evaluation of various outsourcing models – full-service, functional, and hybrid approaches – each presenting unique advantages tailored to specific organizational needs. Establishing effective monitoring systems through real-time data collection and continuous training equips teams to manage compliance effectively and enhance overall device safety. Moreover, collaborating with local experts can significantly boost the effectiveness of post-market vigilance strategies.
Ultimately, the onus is on manufacturers to embrace these best practices and adapt to the shifting regulatory requirements. Investing in robust monitoring frameworks and ongoing team education allows organizations to mitigate risks while contributing to a safer healthcare environment. Engaging with these strategies empowers companies to lead in the Medtech landscape, upholding the highest standards of patient safety in Brazil.
Frequently Asked Questions
What is the role of post-market vigilance in Brazil for medical devices?
Post-market vigilance in Brazil is crucial for ensuring the safety and efficacy of medical devices, overseen by the National Health Surveillance Agency (ANVISA).
What are manufacturers required to do under ANVISA’s regulations?
Manufacturers are required to establish comprehensive monitoring frameworks, report adverse events and device malfunctions within specific timeframes, and ensure timely communication to maintain product integrity.
What significant change will occur in Brazil in 2025 regarding medical devices?
In 2025, the introduction of the Unique Device Identification (UDI) system will enhance traceability and accountability in post-market vigilance for medical devices.
How does the UDI system benefit the healthcare landscape in Brazil?
The UDI system aligns with regulatory expectations and fosters trust and safety within the healthcare landscape by improving the traceability of medical devices.
What should companies be knowledgeable about regarding post-market vigilance?
Companies must be well-versed in submission timelines, documentation requirements, and the types of incidents that necessitate notification.
What are the benefits of adhering to post-market vigilance guidelines?
Adhering to these guidelines mitigates the risk of penalties and reinforces the commitment to patient safety, allowing manufacturers to position themselves as leaders in the Medtech landscape.
List of Sources
- Understand Post-Market Vigilance Requirements in Brazil
- Brazil Medical Device Post Market Surveillance and Vigilance | Pure Global (https://pureglobal.com/markets/brazil/anvisa-medical-device-postmarket-surveillance)
- bioaccessla.com (https://bioaccessla.com/blog/7-key-anvisa-regulations-every-medtech-innovator-must-know)
- Brazilian Medical Device Regulatory System | QMS Templates (https://qmsdoc.com/2026/01/14/brazilian-medical-device-regulatory-system)
- 7 Key Insights on Post-Market Surveillance under ANVISA | bioaccess® (https://bioaccessla.com/blog/7-key-insights-on-post-market-surveillance-under-anvisa)
- Evaluate Outsourcing Models for Post-Market Vigilance
- U.S. Medical Device Outsourcing Market Size Report, 2030 (https://grandviewresearch.com/industry-analysis/us-medical-device-outsourcing-market)
- Medical Device Vigilance Market Size and Forecast] 2025-2032 (https://coherentmarketinsights.com/market-insight/medical-device-vigilance-market-2830)
- Outsourcing Post Market Surveillance and Support – Regulatory Compliance Associates (https://rcainc.com/outsourcing-post-market-surveillance-and-support)
- Medical Devices Vigilance Market Size, Trends, Growth Report 2033 (https://databridgemarketresearch.com/reports/global-medical-devices-vigilance-market?srsltid=AfmBOoqWO78FV8CZFsU2Qd6FfAWMe9WkYfSxKFif7ZYvjLqbof2VbZYd)
- Implement Effective Monitoring Systems for Compliance
- 7 Key Insights on Post-Market Surveillance under ANVISA | bioaccess® (https://bioaccessla.com/blog/7-key-insights-on-post-market-surveillance-under-anvisa)
- Post Market Surveillance: What You Need to Know to Ensure Patient… (https://nsf.org/knowledge-library/post-market-surveillance-what-you-need-to-know-to-ensure-patient-safety)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC12624750)
- Medical Devices: FDA Has Begun Building an Active Postmarket Surveillance System (https://gao.gov/products/gao-24-106699)
- Global trends in post-market surveillance of high-risk medical devices: An empirical analysis based on regulatory data (https://ijmr.org.in/global-trends-in-post-market-surveillance-of-high-risk-medical-devices-an-empirical-analysis-based-on-regulatory-data)
- Enhance Team Competence through Continuous Training
- trainingindustry.com (https://trainingindustry.com/articles/compliance/10-metrics-to-measure-compliance-training)
- Understanding post-market surveillance for medical devices (https://qualio.com/blog/post-market-surveillance)
- The Vital Role of Training and Competency in Medical Device Companies (https://mbcaconsulting.com/post/the-vital-role-of-training-and-competency-in-medical-device-companies)
- 7 Key Insights on Post-Market Surveillance under ANVISA | bioaccess® (https://bioaccessla.com/blog/7-key-insights-on-post-market-surveillance-under-anvisa)

Leave a Reply