Introduction
The landscape of medical research is increasingly shaped by the complex processes of clinical trials, which serve as the backbone for validating new therapies. These trials not only ensure the safety and efficacy of medical interventions but also drive innovation in healthcare. However, with the challenges of recruiting participants and navigating regulatory landscapes, researchers must consider: how can they optimize their strategies to enhance trial outcomes? This article explores the phases of clinical trials, effective recruitment techniques, and the advantages of conducting studies in regions like Latin America, the Balkans, and Australia. By offering insights into these areas, we aim to illuminate pathways that could transform the future of medical research.
Define Clinical Trials and Their Importance in Medical Research
Imp serve as organized inquiries aimed at evaluating the safety and effectiveness of , such as medications, devices, and treatment protocols. These experiments are vital for translating scientific findings into practical medical applications, particularly through an to ensure that new therapies are both effective and safe for human use. By adhering to strict protocols and regulatory standards, research studies enrich the medical knowledge base, ultimately improving outcomes for individuals and fostering .
Consider the role of bioaccess in this landscape: it accelerates by achieving participant enrollment 50% faster than Western locations. This efficiency facilitates the timely inclusion of cardiology or neurology groups, enhancing the recruitment process significantly. Moreover, it leads to substantial cost savings of $25K per patient with , eliminating the need for rework and delays.
The incorporation of (PROs) into early-phase studies is increasingly essential, as highlighted by FDA initiatives like Project Optimus, which aim to provide comprehensive safety profiles. Without imp , the medical community would lack the necessary to support new treatments, highlighting their indispensable role in medical research and development.
In summary, collaboration in is paramount. As we navigate the complexities of the Medtech landscape, embracing innovative approaches like those offered by bioaccess can pave the way for more efficient and effective s.
Explore the Phases of Clinical Trials: From First-in-Human to Market Approval
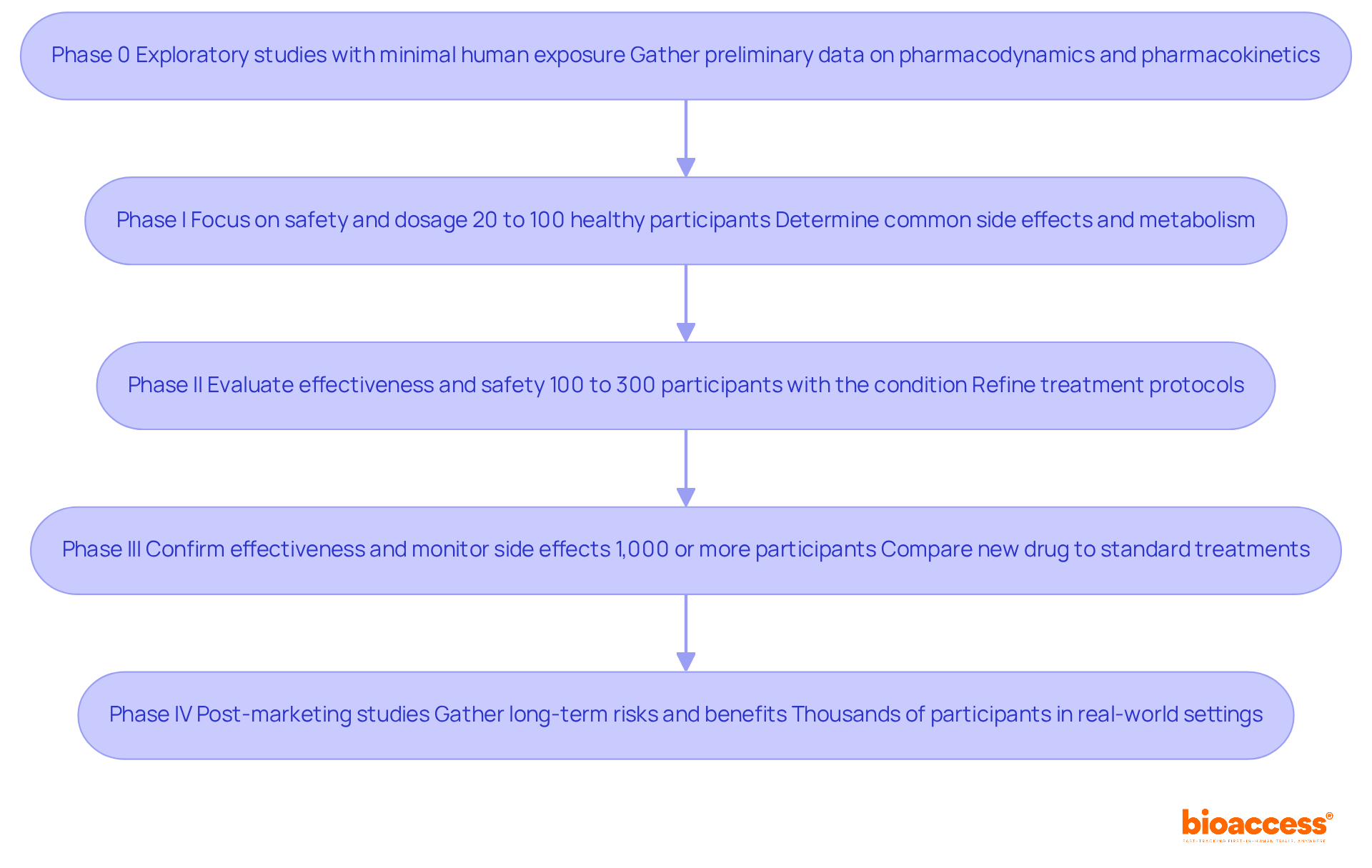
s are systematically structured into several phases, each serving a specific purpose in the development of new therapies:
- Phase 0: These exploratory studies involve minimal human exposure to the drug, primarily aimed at gathering preliminary data on pharmacodynamics and pharmacokinetics.
- Phase I: This phase focuses on safety, determining the drug’s most common side effects and its metabolism and excretion. Typically involving 20 to 100 healthy participants, Phase I studies are essential for determining safe dosage ranges and observing side effects. Successful examples from this phase often set the stage for further development.
- Phase II: Focused on evaluating the drug’s effectiveness and further examining its safety, Phase II studies engage 100 to 300 individuals who have the condition the drug aims to address. This phase is where researchers refine treatment protocols and determine if the new therapy shows real promise, with a focus on both effectiveness and short-term side effects.
- Phase III: Conducted on large populations, , monitor side effects, and compare the new drug to standard treatments. These studies usually include 1,000 or more participants and offer the most convincing evidence for FDA authorization. Successful completion of this phase is often a prerequisite for regulatory approval, making it a critical step in the process. With , organizations can , significantly reducing the time to market and enhancing the likelihood of regulatory success.
- Phase IV: After a drug has received market approval, about its long-term risks, benefits, and optimal use in the general population. These post-marketing studies involve thousands of individuals and focus on real-world applications, ensuring ongoing monitoring of the treatment’s safety and effectiveness.
Each phase is crucial for guaranteeing that only safe and effective treatments reach the market, which is vital for conducting an , thereby safeguarding health and advancing medical science. Recent highlight the significance of data integrity and participant diversity, which are anticipated to influence the future of research studies considerably. By utilizing bioaccess®’s , researchers can tackle recruitment challenges and simplify the process, ultimately resulting in significant savings of $25K per patient. Understanding these phases helps stakeholders recognize the journey of new treatments from initial research to market availability.
Implement Effective Recruitment and Compliance Strategies in Clinical Trials
Effective recruitment and compliance strategies are essential for the success of the imp . By focusing on these key approaches, researchers can significantly enhance their initiatives:
- Develop : Clearly defining participant eligibility ensures that the right population is targeted, enhancing the validity of trial results. This specificity can lead to , as individuals are more likely to meet the criteria and understand their role in the study.
- : Leveraging diverse platforms such as social media, healthcare providers, and patient advocacy groups broadens outreach to potential recruits. Recent trends indicate that multi-channel recruitment can significantly boost participant engagement and enrollment rates.
- : Building relationships with local healthcare providers and community organizations fosters trust and encourages participation. Involving community leaders can also enhance awareness and acceptance of clinical studies, resulting in higher .
- : Providing comprehensive information about the trial is crucial for ethical compliance. Ensuring that individuals fully comprehend the risks and benefits not only satisfies regulatory requirements but also fosters a culture of transparency, which can enhance retention rates throughout the study.
- : Implementing systems to track subject adherence to the study protocol is essential. Regular follow-ups and reminders help maintain data integrity and ensure that participants remain engaged. Organizations that utilize technology for real-time monitoring often report a reduction in data errors and improved compliance outcomes.
By concentrating on these strategies, researchers can improve and guarantee adherence in the imp , ultimately resulting in more dependable and influential study outcomes.
Leverage Regional Advantages: Conducting Clinical Trials in Latin America, the Balkans, and Australia
Conducting in regions such as , the Balkans, and Australia offers strategic advantages that can significantly enhance research outcomes:
- Regulatory Speed: is known for its , allowing quicker approvals and faster initiation of trials compared to traditional markets. This efficiency is crucial for innovators eager to .
- : The Balkans provide access to a , improving the generalizability of study outcomes and aiding in recruitment. This diversity is particularly beneficial for studies requiring varied demographic representation.
- : Australia’s regulatory framework supports , offering clear guidelines that assist researchers in navigating the approval process effectively. This clarity reduces delays and fosters a smoother operational flow.
- Cost-Effectiveness: Conducting experiments in these regions can be more economical, allowing companies to allocate resources more efficiently while maintaining high-quality standards. The potential for significant is a compelling reason for many organizations to consider these locations.
By leveraging these regional benefits, innovators can accelerate their research efforts, ultimately introducing new therapies to market more rapidly and efficiently. Recent trends indicate that the Balkans are becoming increasingly attractive for , with a growing number of studies being conducted, reflecting the region’s commitment to enhancing its research capabilities.
Conclusion
Understanding the complexities and significance of clinical trials is crucial for advancing medical research and ensuring that new therapies are both effective and safe for patient use. These trials provide essential evidence for the approval of innovative treatments and foster collaboration and efficiency within the healthcare landscape. By embracing modern strategies and regional advantages, researchers can enhance the trial process, ultimately leading to improved patient outcomes.
The article explores the structured phases of clinical trials, from initial exploratory studies to post-marketing evaluations, highlighting the importance of each stage in ensuring the safety and efficacy of new medical interventions. Key insights include the role of effective recruitment strategies, compliance monitoring, and the benefits of conducting trials in regions like Latin America, the Balkans, and Australia. These areas offer unique advantages, such as faster regulatory approvals and diverse patient populations, which can significantly impact trial success.
In light of these insights, stakeholders in the healthcare sector must prioritize innovative recruitment and compliance strategies while considering the strategic advantages offered by various global regions. By doing so, the medical community can accelerate the delivery of groundbreaking therapies to patients, reinforcing the vital role of clinical trials in enhancing healthcare and improving lives. The time to act is now—collaboration and strategic planning are essential for overcoming challenges and advancing the future of medical research.
Frequently Asked Questions
What are clinical trials?
Clinical trials are organized inquiries aimed at evaluating the safety and effectiveness of medical interventions, such as medications, devices, and treatment protocols.
Why are clinical trials important in medical research?
Clinical trials are vital for translating scientific findings into practical medical applications, ensuring that new therapies are both effective and safe for human use. They enrich the medical knowledge base, improve outcomes for individuals, and foster healthcare innovation.
How does bioaccess contribute to clinical trials?
Bioaccess accelerates clinical studies by achieving participant enrollment 50% faster than Western locations, facilitating the timely inclusion of treatment-naive cardiology or neurology groups, and leading to substantial cost savings of $25K per patient with FDA-ready data.
What are patient-reported outcomes (PROs) and why are they important?
Patient-reported outcomes (PROs) are increasingly essential in early-phase studies as they provide comprehensive safety profiles. Initiatives like the FDA’s Project Optimus highlight the importance of incorporating PROs into clinical trials.
What would happen without clinical trials?
Without clinical trials, the medical community would lack critical evidence necessary to support new treatments, underscoring their indispensable role in medical research and development.
What is the significance of collaboration in clinical research?
Collaboration in clinical research is paramount for navigating the complexities of the Medtech landscape and embracing innovative approaches to enhance the efficiency and effectiveness of clinical trials.
List of Sources
- Define Clinical Trials and Their Importance in Medical Research
- The Future of Clinical Trials: 2025 Outlook | Signant Health (https://signanthealth.com/resources/blog/the-future-of-clinical-trials-2025-trends)
- recoverytrial.net (https://recoverytrial.net/case_studies/from-buckinghamshire-to-worldwide-impact-dr-raha-west2019s-journey-with-the-recovery-trial)
- Core funders of medical research commit to strengthening clinical trials worldwide (https://who.int/news/item/25-09-2025-core-funders-of-medical-research-commit-to-strengthening-clinical-trials-worldwide)
- Explore the Phases of Clinical Trials: From First-in-Human to Market Approval
- primeclinicalresearchinc.com (https://primeclinicalresearchinc.com/phases-of-clinical-trials-explained)
- 10 Clinical Trial Regulatory Changes Coming in 2025 (https://en.ennov.com/blog/clinical-blog/trial-regulatory-changes-coming-2025)
- clinicaltrialshub.htq.org.au (https://clinicaltrialshub.htq.org.au/blog/understanding-the-clinical-trial-phases)
- TOP 10 CLINICAL TRIALS QUOTES | A-Z Quotes (https://azquotes.com/quotes/topics/clinical-trials.html)
- Implement Effective Recruitment and Compliance Strategies in Clinical Trials
- 10 inspirational quotes for the pharma sector (https://pharmaceuticalmanufacturer.media/pharmaceutical-industry-insights/10-inspirational-quotes-for-the-pharma-sector)
- Clinical Research Compliance: 5 Critical Factors for 2024 Success (https://infonetica.net/articles/clinical-research-compliance)
- bioaccessla.com (https://bioaccessla.com/blog/10-essential-capa-strategies-for-clinical-research-compliance)
- Leverage Regional Advantages: Conducting Clinical Trials in Latin America, the Balkans, and Australia
- cordmagazine.com (https://cordmagazine.com/business/success-stories/aleksandar-tripkovic-astra-zeneca-science-and-partnership-for-better-health)
- Latin America Clinical Trials Market to Apex USD 9.17 Bn by 2035 (https://towardshealthcare.com/insights/latin-america-clinical-trials-market-sizing)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)

Leave a Reply