Introduction
Understanding the landscape of medical continuing education (CE) is crucial for clinical research directors who strive to remain at the forefront of healthcare advancements. This guide explores the vital role of CE, highlighting how it not only enriches knowledge but also has a direct impact on patient care and safety. As the demands of clinical research evolve, how can directors effectively navigate their CE requirements while ensuring they choose the most beneficial programs?
In an era where healthcare is rapidly advancing, staying informed through CE is not just beneficial; it’s essential. The right educational programs can empower directors to tackle emerging challenges and enhance their contributions to clinical research. By prioritizing CE, directors can ensure they are equipped with the latest insights and skills necessary for optimal patient outcomes.
Understand the Importance of Medical Continuing Education
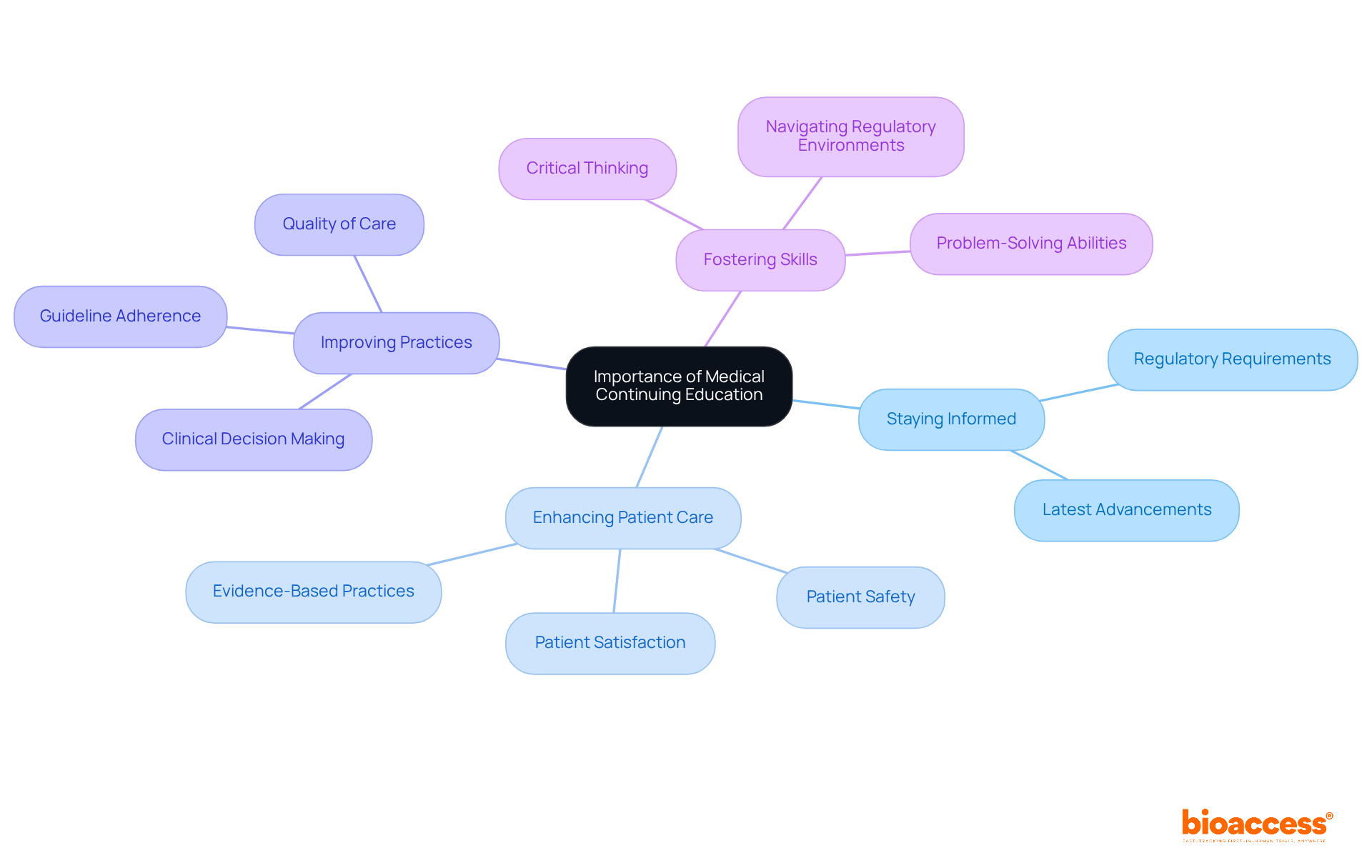
is crucial for healthcare directors, ensuring they stay informed about the . This commitment to not only enhances knowledge but also significantly elevates patient care and supports the maintenance of licensure. By embracing , healthcare professionals can improve medical practices and outcomes.
Engaging in equips trial directors with the essential skills to effectively navigate the complexities of studies and regulatory environments. Moreover, continuous education fosters , which are vital for managing health-related projects successfully. The impact of on trial outcomes is substantial; it empowers professionals to implement , ultimately enhancing patient safety and satisfaction.
The success derived from underscores their significance. Well-informed are positioned to and . As you consider your own challenges in clinical research, reflect on how investing in CE can transform your practice and lead to better .
Identify Your CE Requirements and Options
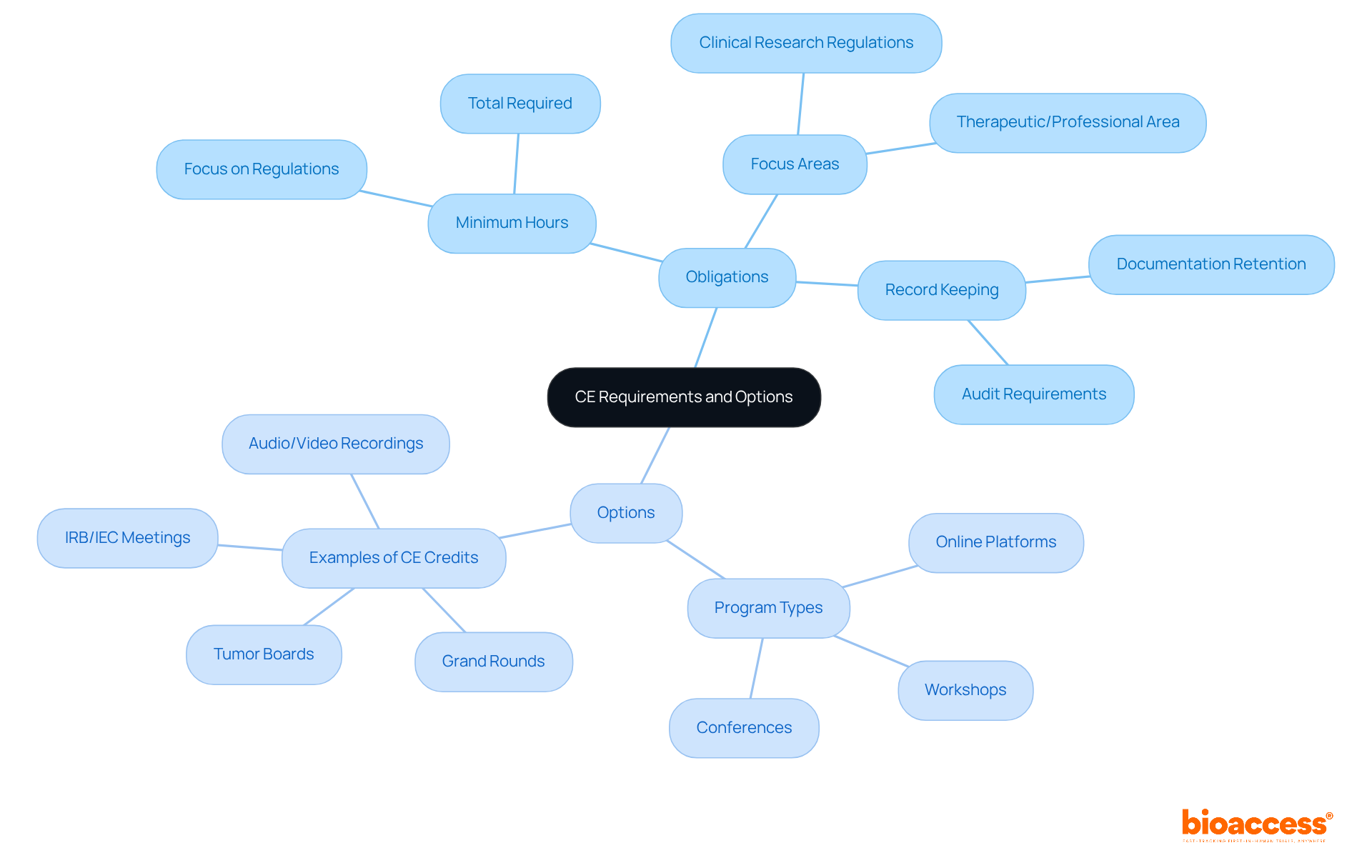
To effectively determine your ongoing , it’s crucial to examine the regulations set by your licensing board or professional organization. Each state or organization may impose different requirements regarding the number of needed and the . For example, healthcare professionals typically need a minimum of during their certification period, with at least 22 hours focused on regulations and policies related to .
Moreover, maintaining accurate records of your medical ce activities is essential, as random audits of medical ce documentation occur annually for up to two years after the certification period ends. Once you grasp your specific requirements, explore various options for , including online platforms, workshops, and conferences. Organizations like ACRP and SOCRA offer extensive lists of tailored for , detailing maximum limits on , such as:
- 15 CE for audio/video recordings
- 2 CE from grand rounds, tumor boards, and IRB/IEC meetings combined per year
Furthermore, consider your learning preferences-whether you favor interactive workshops, online programs, or self-paced study-to select the most suitable options for your professional growth. By understanding your obligations and available resources, you can enhance your expertise and stay compliant in the ever-evolving field of clinical research.
Select Accredited CE Courses That Fit Your Needs
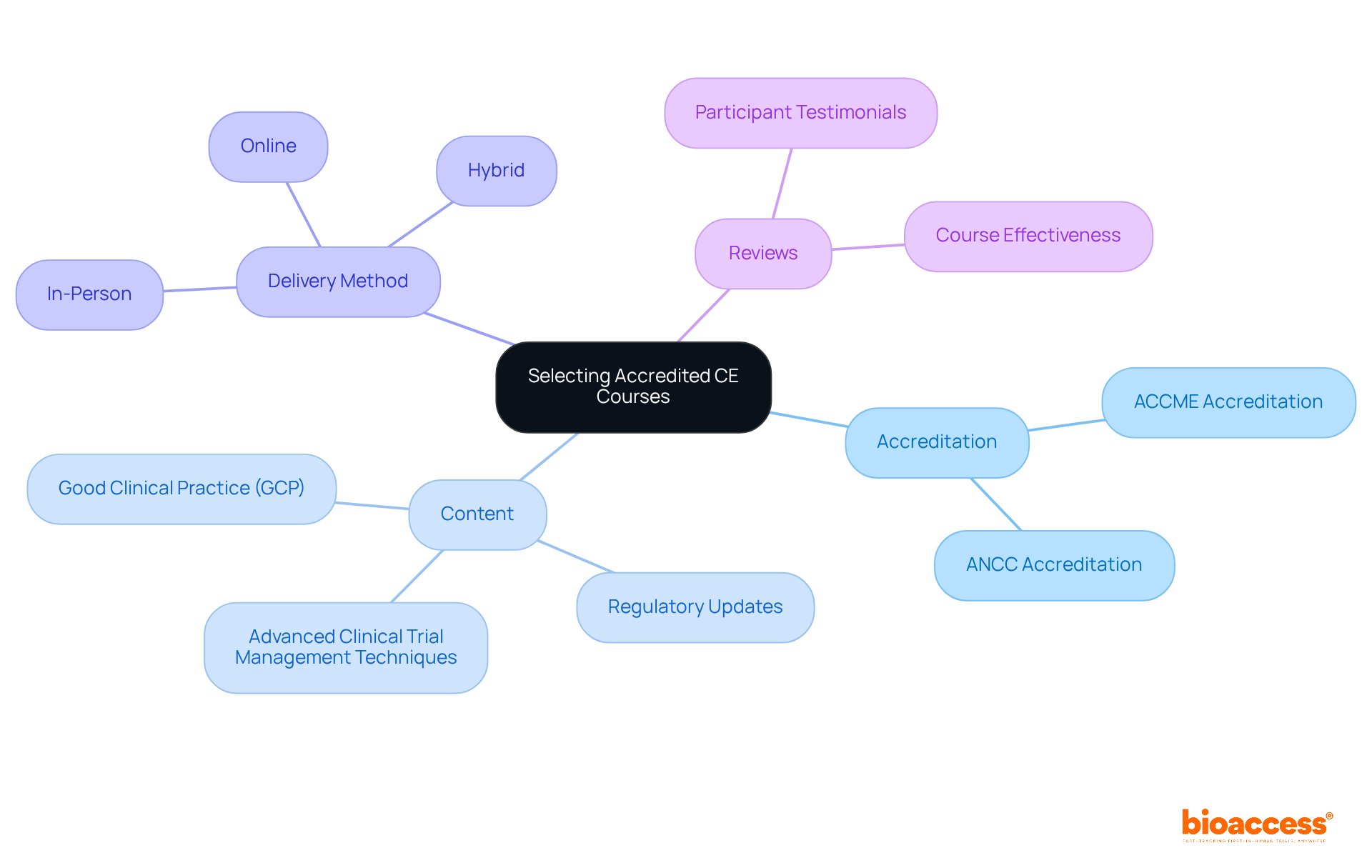
When selecting for continuing education, it’s crucial to prioritize those like the or the American Nurses Credentialing Center (ANCC). Accreditation not only guarantees adherence to established educational standards but also ensures recognition by medical ce licensing boards. Evaluate programs based on their content, structure, and relevance to your role as a research director. Focus on programs that delve into such as:
Additionally, consider the delivery method-whether in-person, online, or hybrid-to align with your preferred learning style. Reading reviews and testimonials from previous participants can offer valuable insights into the program’s effectiveness and its relevance to your professional development. By making informed choices, you can enhance your expertise and better navigate the complexities of clinical research.
Track and Document Your CE Credits Effectively
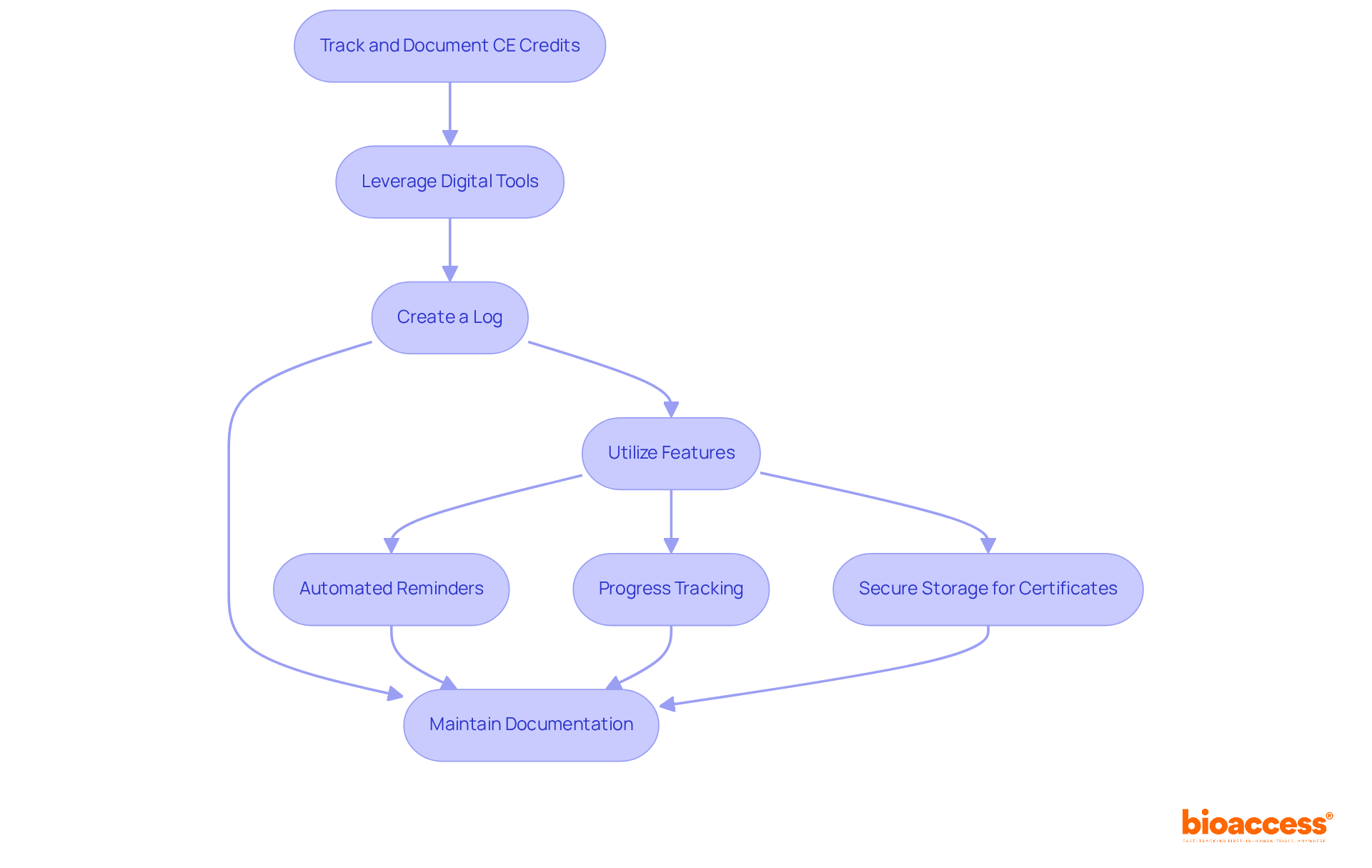
Effectively tracking and documenting your is crucial in today’s fast-paced environment. Leveraging , such as specialized or mobile applications, is essential. These platforms automate the process, allowing you to maintain an organized record of your effortlessly. For instance, consider creating a log that includes the course title, provider, date completed, and number of credits earned. This organized method not only streamlines management but also guarantees , which is vital; failing to comply can result in fines or loss of license.
Many organizations have successfully adopted for CE credit management, enhancing their ability to track progress and meet deadlines. For example, using the CE App can increase event revenue by 20.8%, demonstrating the financial benefits of efficient tracking. Features to look for in these tools include:
- Secure storage for digital certificates
As Aspira Continuing Education states, “Our user-friendly platform supports real-time updates, , and mobile access, so you can focus on learning, not paperwork.” By utilizing these resources, you can concentrate on your learning rather than administrative tasks.
Consistently examining your CE obligations is essential for keeping on schedule with renewal dates. Keeping and documentation of attendance is also vital for verification purposes. By staying organized and proactive, you can ensure that you meet all CE requirements without the stress of last-minute preparations.
Conclusion
Mastering medical continuing education (CE) is not just essential; it’s a cornerstone for clinical research directors who aim to elevate their expertise and enhance patient care. By prioritizing ongoing education, healthcare professionals can stay updated on the latest advancements and regulatory requirements, ultimately leading to improved outcomes in clinical practice. Embracing lifelong learning supports professional growth and reinforces a steadfast commitment to patient safety and satisfaction.
This guide has underscored the significance of understanding CE requirements, selecting accredited courses, and effectively tracking credits. Key insights highlight the necessity of aligning CE programs with personal learning preferences and professional obligations, along with the advantages of utilizing digital tools for credit management. Each of these steps contributes to a comprehensive approach to medical CE, ensuring that clinical research directors are well-equipped to navigate the complexities of their roles.
In conclusion, the journey of continuous education transcends mere obligation; it represents a vital opportunity for growth and innovation within the healthcare sector. As clinical research directors contemplate their CE paths, they are urged to actively seek resources and programs that resonate with their professional aspirations. Investing in medical CE is a commitment to excellence that will ultimately benefit not only the individual but also the broader healthcare community and the patients they serve.
Frequently Asked Questions
Why is medical continuing education (CE) important for healthcare directors?
Medical CE is crucial for healthcare directors as it helps them stay informed about the latest advancements in medical science and regulatory requirements, enhancing their knowledge and significantly improving patient care.
How does medical CE contribute to patient care?
By engaging in medical CE, healthcare professionals can embrace lifelong learning, which improves medical practices and outcomes, ultimately enhancing patient safety and satisfaction.
What skills does medical CE provide to trial directors?
Medical CE equips trial directors with essential skills to navigate the complexities of studies and regulatory environments, fostering critical thinking and problem-solving abilities vital for managing health-related projects successfully.
What is the impact of medical CE on trial outcomes?
The impact of medical CE on trial outcomes is substantial; it empowers professionals to implement evidence-based practices, which enhances patient safety and satisfaction.
How can investing in medical CE transform clinical research practices?
Investing in medical CE can transform clinical research practices by equipping professionals with updated knowledge and skills, driving innovation, and elevating the quality of care within their organizations.
List of Sources
- Understand the Importance of Medical Continuing Education
- Accredited Continuing Medical Education Delivers: Evidence of Value, Trust, and Impact Across the Healthcare System – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC12587789)
- Global Education Group Survey Reveals Importance of Continuing Medical Education (https://ultimatemedical.edu/newsroom/global-education-group-survey)
- Primary health care professionals’ opinions regarding continuing medical education: A cross sectional study – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11651445)
- The Vital Importance of Continuing Medical Education (https://propharmaresearch.com/en/resources/diffusion/vital-importance-continuing-medical-education)
- Identify Your CE Requirements and Options
- Continuing Education Requirements (https://socra.org/certification/maintenance-of-certification/continuing-education-requirements)
- Beginner’s Guide to Continuing Education Units (CEUs) for Clinical Research (https://advarra.com/blog/beginners-guide-to-continuing-education-units-ceus-for-clinical-research)
- Descriptions of Acceptable CE (https://socra.org/certification/maintenance-of-certification/descriptions-of-acceptable-ce)
- CE Requirements by State — Clearly Clinical® (https://clearlyclinical.com/ceu-requirements-by-state)
- Select Accredited CE Courses That Fit Your Needs
- Making Informed Decisions: Key Statistical Principles to Clinical Trial Design Program – Georgia CTSA & SC CTSI – Translational Workforce Development (https://twd.ce.emorynursingexperience.com/programs/make-informed-decisions)
- Statistical Principles for Clinical Trials: Overview of ICH E9 – ACRP (https://acrpnet.org/learning/courses/statistical-principles-for-clinical-trials-overview-of-ich-e9)
- Statistical Concepts Used in Clinical Trials – Peri.org (https://peri.org/distance-education-courses/statistical-concepts-used-in-clinical-trials)
- Statistics in Clinical Trials (https://udemy.com/course/statistics-in-clinical-trials?srsltid=AfmBOooL1EPbZTKuN3_bxgj9vf8evh7veGeEkU4_1VQdOIz369lIe4fD)
- Accredited Continuing Medical Education Delivers: Evidence of Value, Trust, and Impact Across the Healthcare System – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC12587789)
- Track and Document Your CE Credits Effectively
- UpToDate CME/CE/CPD Tracking (https://wolterskluwer.com/en/solutions/uptodate/about/continuing-medical-education/cme-ce-cpd-tracking)
- CE Tracking Software (https://jollytech.com/blog/tips-apps-tracking-ce-cpd-credits-hours-certs.php?srsltid=AfmBOopHVLxgZzbFLsXLFxYmTtxB7_XVJTujJsZ6ar4Krx_J0cuUVj6I)
- 5 Top CE tracking apps and online tools (https://myceapp.com/blog/recommend-ce-tracking-software)
- CE Tracking Tools and Apps: Simplify Your Education Goals – Online CEUs – Aspira Continuing Education (https://aspirace.com/guides/ce-tracking-tools-and-apps?srsltid=AfmBOoqbO2ggS-92aUeauLyOkDlnzjmIL0egSziiXOGy5mgQCPTRLIwr)

Leave a Reply