Introduction
Ensuring the safety and efficacy of medical devices relies heavily on effective packaging validation – a process often riddled with challenges. Manufacturers must navigate the stringent requirements of ISO 11607, balancing compliance with the imperative of safeguarding patient health. This article explores four best practices that not only streamline the validation process but also bolster product integrity and regulatory adherence.
Can manufacturers truly afford to overlook these critical steps? The repercussions of inadequate validation could be both costly and detrimental.
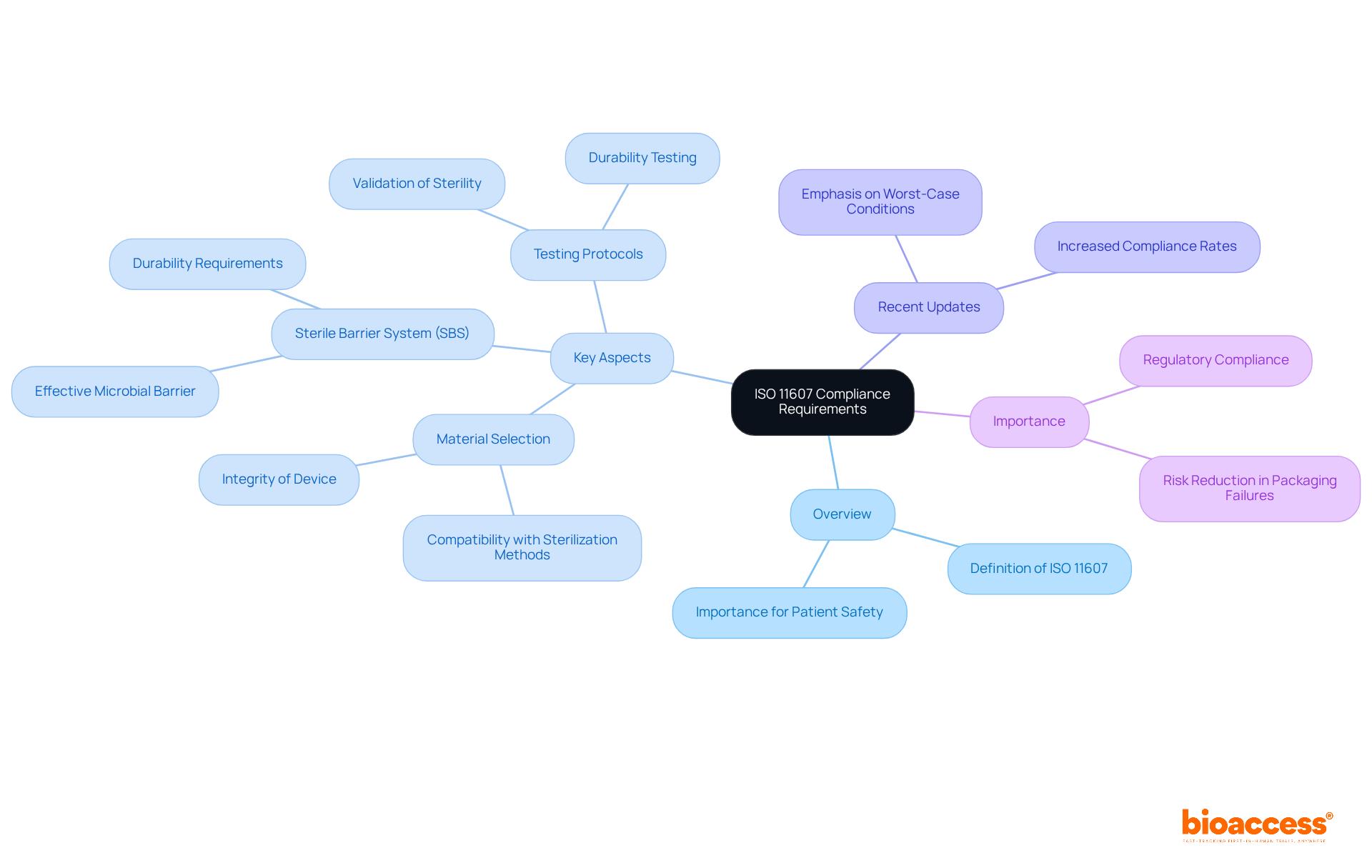
Understand ISO 11607 Compliance Requirements
is the international standard that outlines the . This standard is crucial for ensuring that the container maintains the sterility of the device until the point of use. It consists of two parts: Part 1 focuses on the requirements for materials and sterile barrier systems, while Part 2 addresses for forming, sealing, and assembly processes. Understanding these standards is essential for .
Key aspects include:
- : Ensure that materials used in packaging are compatible with the sterilization method and do not compromise the device’s integrity.
- : The container must provide an effective barrier against microbial contamination.
- : Implement rigorous to validate that the containers meet the required standards for sterility and durability.
Recent updates to emphasize the importance of when validating containers under worst-case conditions. This approach helps ensure that products remain safe and effective during distribution. Adherence to these standards is not merely a legal obligation; it is vital for protecting . Currently, a significant percentage of medical device manufacturers are compliant with , reflecting its critical role in the industry. By following these guidelines, manufacturers can effectively reduce risks related to containment failures, thereby enhancing patient safety and ensuring .
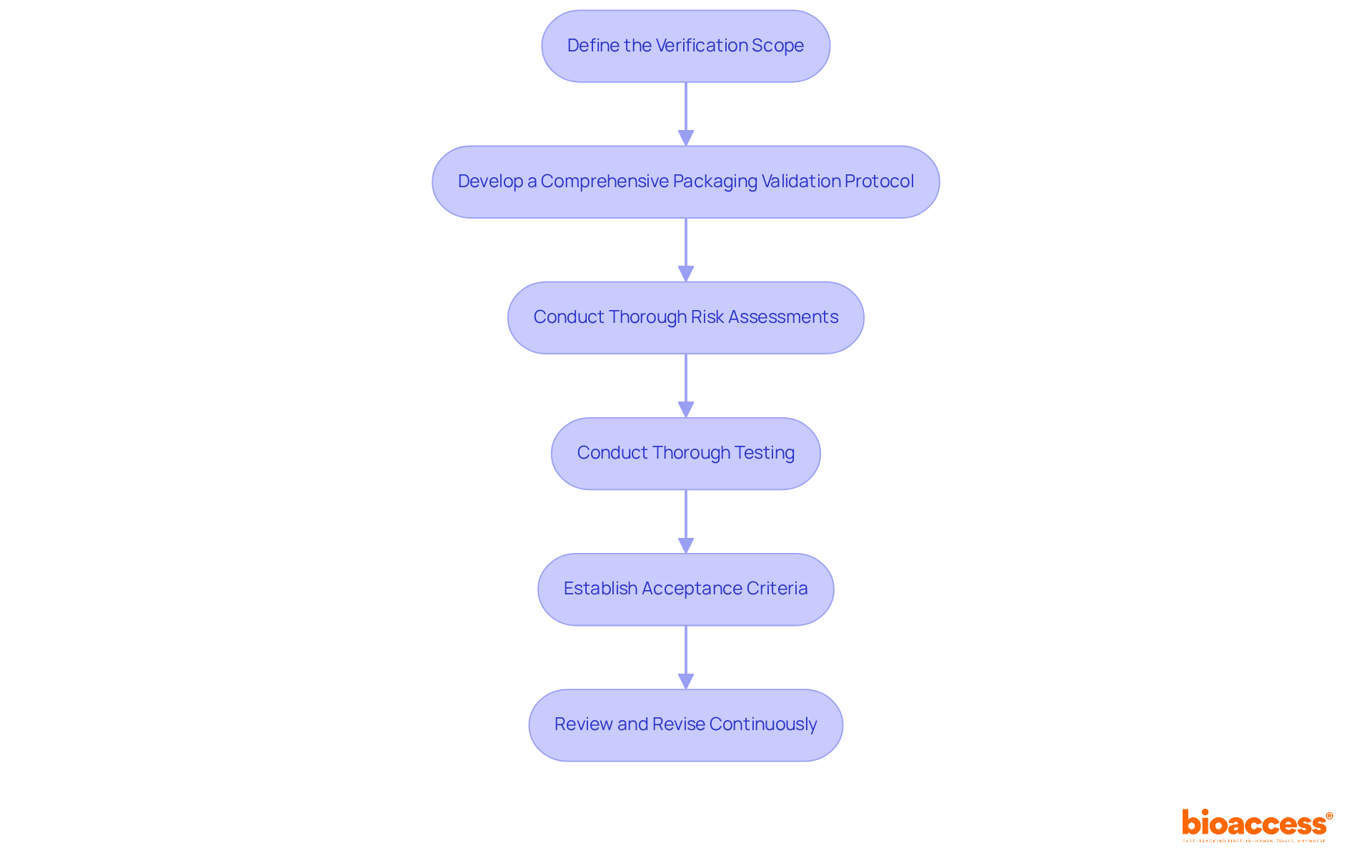
Implement Key Components of the Validation Process
Successful in hinges on several essential components:
- : Establish clear objectives, requirements, and expected outcomes for the verification process. This foundational step ensures that all stakeholders understand the goals and parameters of the validation effort.
- Develop a : Craft a detailed protocol that outlines specific testing methods, acceptance criteria, and timelines. This protocol should encompass various tests, including , bubble leak tests, and microbial barrier evaluations, to ensure compliance with regulatory standards.
- : Identify potential risks associated with container materials and processes. By comprehending these risks, manufacturers can create effective mitigation strategies, enhancing the overall reliability of the containment system.
- : Carry out a series of tests to confirm the performance of the package. Key tests include dye penetration for seal integrity, vacuum decay for leak detection, and aging studies to assess shelf-life. Significantly, more than a third of all package assessments fail laboratory examinations for transport, emphasizing the significance of these evaluations in ensuring that containers uphold their integrity under real-world conditions.
- : Define clear acceptance criteria that establish benchmarks for durability, sterility maintenance, and resistance to environmental factors. Meeting these criteria is essential for and efficient product performance.
- : Regularly assess the confirmation process and make necessary adjustments based on findings and stakeholder feedback. This iterative method aids in refining protocols and enhancing outcomes, ultimately decreasing the chances of failures in the process.
By applying these best practices, manufacturers can improve the efficiency of their container assessment procedures through medical device , ensuring product safety and adherence while reducing the risk of expensive recalls and regulatory problems.
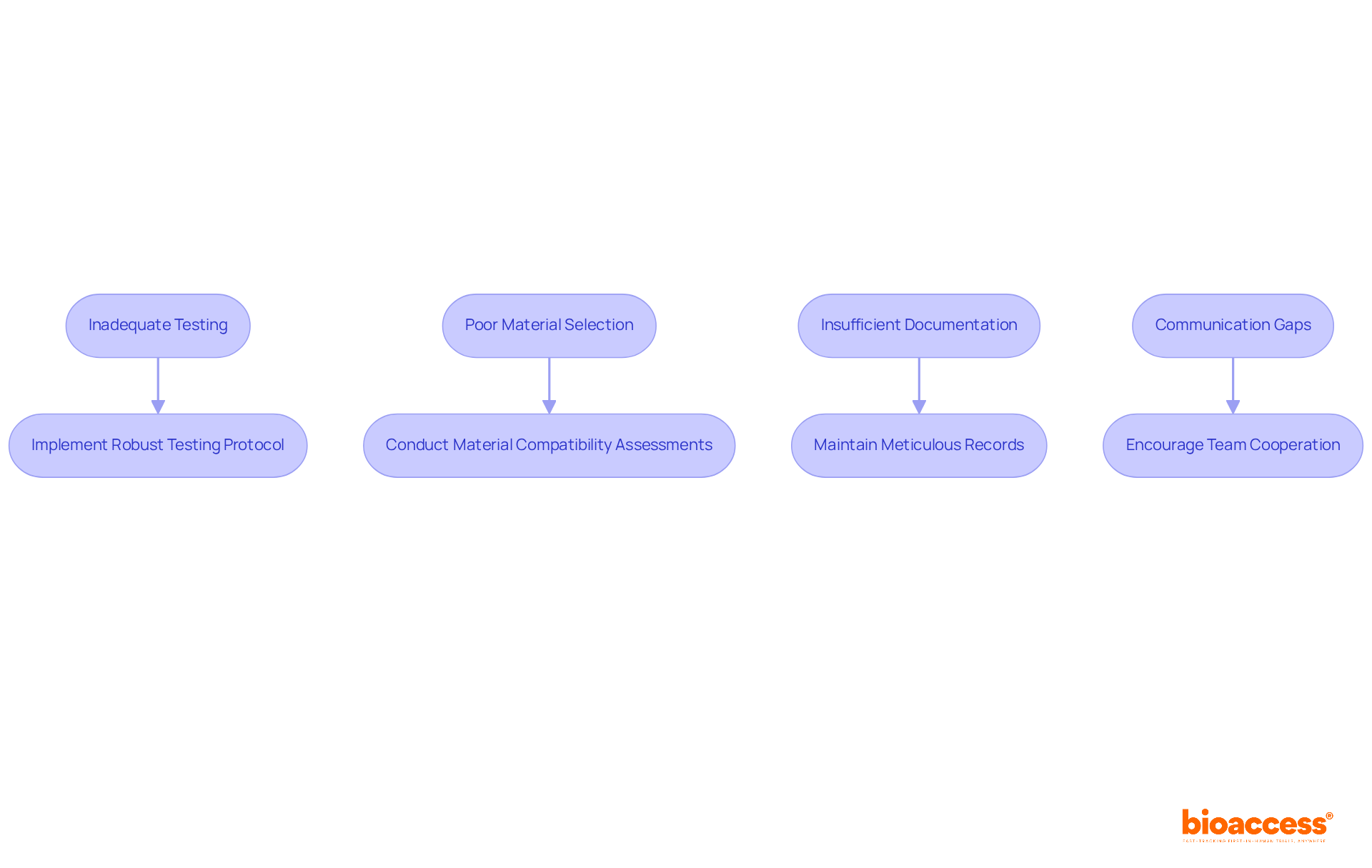
Address Common Challenges with Effective Solutions
Common challenges in are critical to address for ensuring product integrity and compliance:
- Inadequate Testing: Many manufacturers overlook , which can lead to significant failures. Solution: Implement a robust that encompasses all aspects of packaging performance. This includes understanding and their related testing requirements, ensuring that every potential issue is identified and addressed.
- Poor Material Selection: The use of inappropriate materials can jeopardize sterility. Solution: Conduct thorough that take into account the product’s weight and geometry. This ensures adherence to and maintains the integrity of the device throughout its lifecycle.
- Insufficient Documentation: A lack of proper documentation can result in . Solution: Maintain meticulous records of all . This includes detailed test results, protocol revisions, and risk-based rationales, creating a comprehensive paper trail that supports .
- Communication Gaps: Miscommunication between departments can lead to delays in the verification process. Solution: Encourage cooperation among teams involved in verification to guarantee alignment and clarity. Awareness of potential issues that arise when product development occurs too late or in isolation is crucial.
By proactively addressing these challenges, manufacturers can streamline their verification efforts and significantly enhance the quality and reliability of their systems.
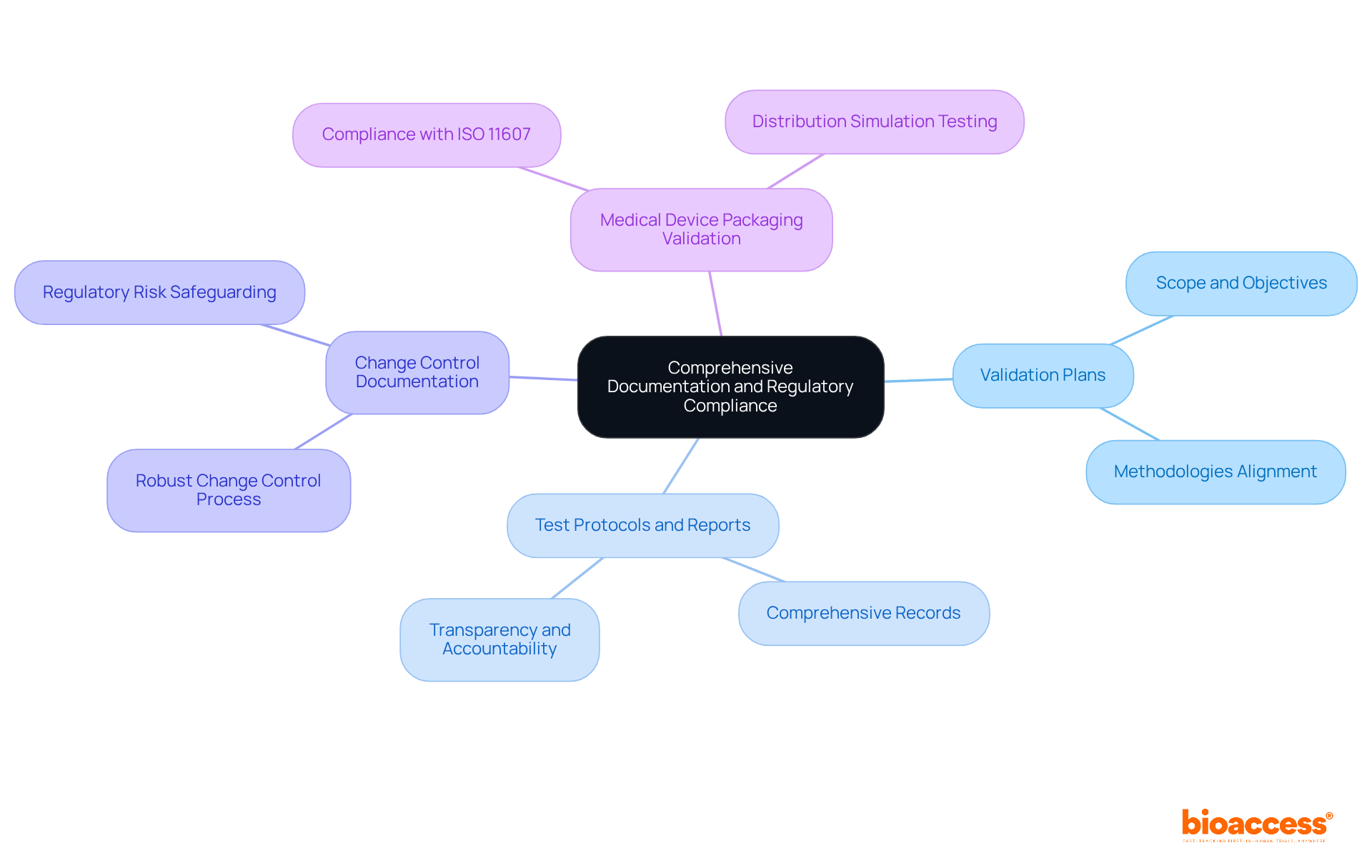
Ensure Comprehensive Documentation and Regulatory Compliance
is essential for with legal standards and ensuring the integrity of the assessment process. Key documentation practices include:
- : Develop detailed that clearly outline the scope, objectives, and methodologies for the validation process, ensuring alignment with .
- : Maintain comprehensive records of all testing protocols, results, and any deviations from planned procedures, which are vital for transparency and accountability.
- : Implement a robust change control process to document any modifications to container design or verification protocols, safeguarding against .
- : Prepare all documentation meticulously for submission to regulatory bodies, including evidence of compliance with ISO 11607, which Jeff Barrett, CEO of J-Pac Medical, describes as “the Bible for ,” along with other relevant standards, to facilitate timely approvals.
Moreover, including distribution simulation testing is crucial to ensure that containers can withstand real-world shipping and handling conditions. This aspect is vital for preserving the integrity of the container throughout its lifecycle.
By prioritizing documentation and , manufacturers can enhance their credibility and ensure that their container systems meet necessary safety and efficacy standards. Non-compliance with packaging regulations can lead to significant delays in market approvals and costly recalls, making even more critical. This approach not only mitigates risks associated with non-compliance but also fosters trust among healthcare providers and end-users, ultimately supporting successful market entry.
Conclusion
The success of medical device packaging validation is fundamentally rooted in a thorough understanding of industry standards and best practices. By prioritizing compliance with ISO 11607 and implementing structured validation processes, manufacturers can significantly enhance the safety and effectiveness of their products. This commitment not only meets regulatory requirements but also safeguards patient health – the ultimate goal of any medical device manufacturer.
Key insights from this article underscore the importance of:
- Defining verification scopes
- Developing robust testing protocols
- Addressing common challenges such as inadequate testing and poor material selection
By adhering to these best practices, manufacturers can streamline their validation processes, ensuring that packaging systems maintain sterility and integrity throughout the product lifecycle. Furthermore, thorough documentation plays a vital role in demonstrating compliance and fostering trust among stakeholders, ultimately supporting successful market entry.
In conclusion, embracing these best practices for medical device packaging validation transcends mere regulatory obligation; it is a crucial step toward ensuring patient safety and product reliability. By committing to continuous improvement and proactive risk management, manufacturers can navigate the complexities of validation with confidence, paving the way for innovation and excellence in the medical device industry.
Frequently Asked Questions
What is ISO 11607 and why is it important?
ISO 11607 is the international standard that outlines requirements for containment systems for terminally sterilized medical devices, crucial for maintaining the sterility of devices until use.
What are the two parts of ISO 11607?
Part 1 focuses on the requirements for materials and sterile barrier systems, while Part 2 addresses validation requirements for forming, sealing, and assembly processes.
What is the significance of material selection in ISO 11607 compliance?
Materials used in packaging must be compatible with the sterilization method and should not compromise the integrity of the medical device.
What is a Sterile Barrier System (SBS)?
A Sterile Barrier System is a container that provides an effective barrier against microbial contamination, ensuring the sterility of the medical device.
What are the testing protocols required by ISO 11607?
Rigorous testing protocols must be implemented to validate that the containers meet the required standards for sterility and durability.
What recent updates have been made to ISO 11607?
Recent updates emphasize the importance of validating medical device packaging under worst-case conditions to ensure products remain safe and effective during distribution.
Why is adherence to ISO 11607 standards essential for manufacturers?
Adherence is vital not only for legal compliance but also for protecting patient health and reducing risks related to containment failures.
How prevalent is compliance with ISO 11607 among medical device manufacturers?
A significant percentage of medical device manufacturers are currently compliant with ISO 11607, highlighting its critical role in the industry.
List of Sources
- Understand ISO 11607 Compliance Requirements
- Packaging Validation Best Practices for Medical Devices (https://greenlight.guru/blog/packaging-validation-best-practices)
- Medical Device Packaging Market Growth Analysis – Size and Forecast 2025-2029 | Technavio (https://technavio.com/report/medical-device-packaging-market-industry-analysis)
- EU MDR Updates on Sterile Packaging: How Guardian’s CapSure Technology Provides OEMs a Compliant, User-Friendly Solution for Usability and Aseptic Transfer – Guardian Medical (https://guardianmedicalusa.com/resources/blogs/eu-mdr-updates-on-sterile-packaging)
- ISO 11607 for Medical Device Packaging: Guide (https://flexpakinc.com/iso-11607-2)
- Implement Key Components of the Validation Process
- Packaging Validation Best Practices for Medical Devices (https://greenlight.guru/blog/packaging-validation-best-practices)
- Is Statistical Analysis Required in Packaging? | Packaging Compliance Labs (https://pkgcompliance.com/is-statistical-analysis-required-in-packaging)
- Medical Device Packaging Validation: Key Steps Explained (https://profounddevelopmentgroup.com/blog/f/medical-device-packaging-validation-key-steps-explained)
- Address Common Challenges with Effective Solutions
- Packaging Validation Best Practices for Medical Devices (https://greenlight.guru/blog/packaging-validation-best-practices)
- Packaging Validation Protocol: Common Mistakes that Can Lead to Failures (https://ddltesting.com/blog/packreview-common-mistakes-that-can-lead-to-failures-during-your-package-validation)
- Medical Device Packaging Validation: 5 Testing Best Practices for 2023 (https://namsa.com/resources/blog/medical-device-packaging-validation-5-testing-best-practices-for-2023)
- Medical Device Packaging Streamlined to Meet New Regulations (https://millstonemedical.com/streamline-medical-device-packaging-to-get-ahead-of-regulatory-demands)
- Medical Device Packaging Validation Procedure: Overview – PRO-TECH Design (https://protechdesign.com/articles/medical-device-packaging-validation-testing-compliance-guide)
- Ensure Comprehensive Documentation and Regulatory Compliance
- Importance of Documentation – Expert and Influential Leader Quotes : Erase Your Risk with Proper Documentation (https://forensicnotes.com/importance-of-documentation-expert-and-influential-leader-quotes)
- Ensuring Compliance with Medical Device Packaging Validation Regulations (https://regdesk.co/blog/ensuring-compliance-with-medical-device-packaging-validation-regulations)
- Medical Device Packaging Validation: Key Steps Explained (https://profounddevelopmentgroup.com/blog/f/medical-device-packaging-validation-key-steps-explained)
- Packaging Validation Best Practices for Medical Devices (https://greenlight.guru/blog/packaging-validation-best-practices)
- Medical Device Packaging Streamlined to Meet New Regulations (https://millstonemedical.com/streamline-medical-device-packaging-to-get-ahead-of-regulatory-demands)

Leave a Reply