Introduction
The landscape of gene therapy is rapidly evolving, presenting unprecedented opportunities for groundbreaking treatments. As clinical researchers embark on first-in-human trials, grasping the essential basics is crucial for navigating this complex field.
What challenges do these pioneers face as they strive to balance innovative approaches with stringent regulatory requirements and ethical considerations?
Exploring the key components of successful gene therapy trials illuminates pathways to overcoming these obstacles and advancing the future of personalized medicine.
bioaccess: Accelerate First-in-Human Gene Therapy Trials in Latin America
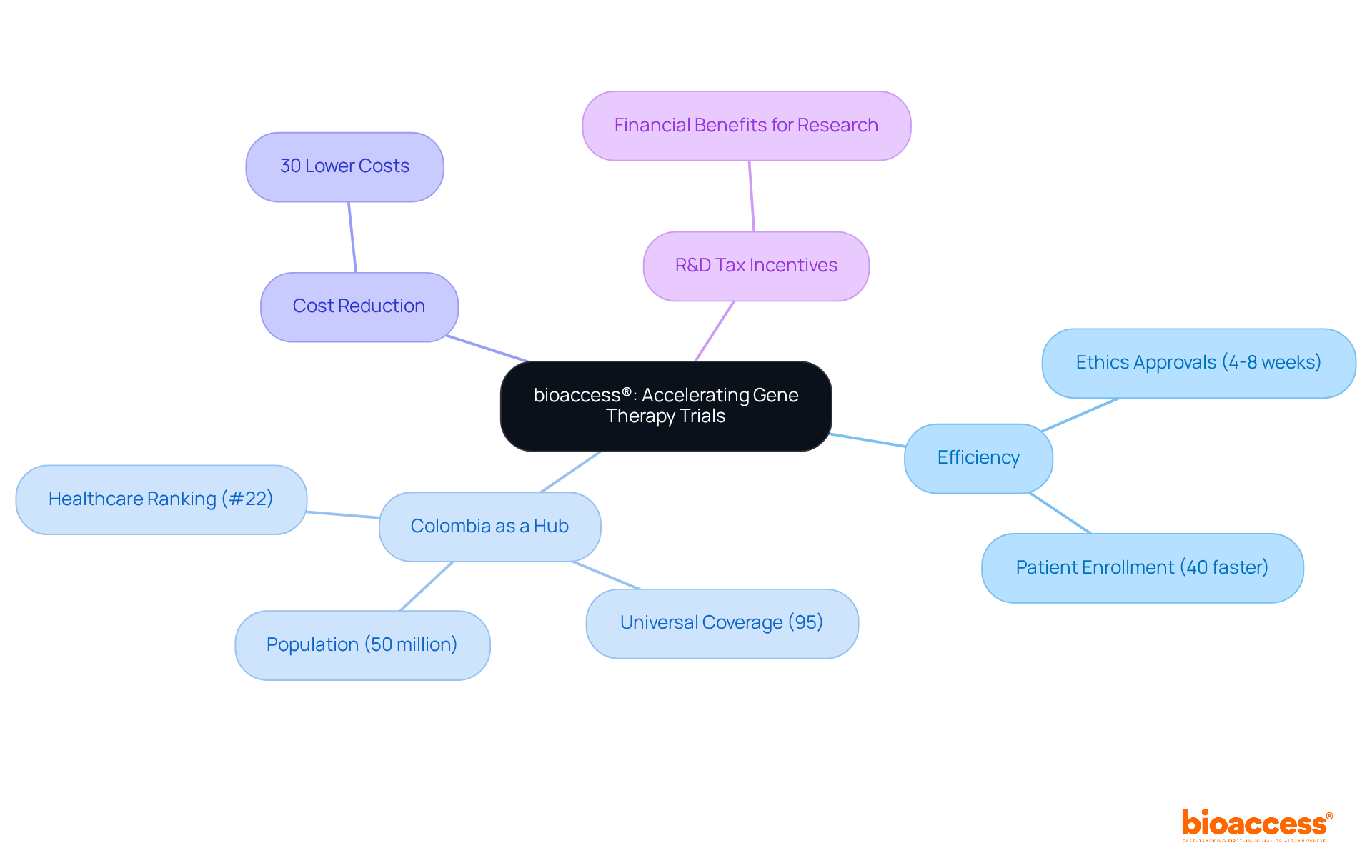
bioaccess® stands at the forefront of accelerating across Latin America, offering a within just 4 to 8 weeks. This impressive turnaround is bolstered by a than in the US and EU, significantly enhancing efficiency. Moreover, bioaccess® delivers an compared to traditional trial expenses in developed markets.
, boasting a by the World Health Organization. Its hospitals are recognized as among the best in Latin America, having successfully navigated rigorous ICH/GCP certification processes for conducting research. These factors, combined with a population exceeding 50 million and universal , position Colombia as an ideal hub for patient recruitment.
Additionally, bioaccess® leverages for clinical research. This establishes the company as a premier choice for Medtech and Biopharma firms eager to expedite their clinical research efforts in a region characterized by a . Recent advancements in gene therapies, particularly in countries like Brazil and Mexico, underscore the region’s potential, with the paving the way for broader applications in .
Understand Regulatory Requirements for First-in-Human Trials
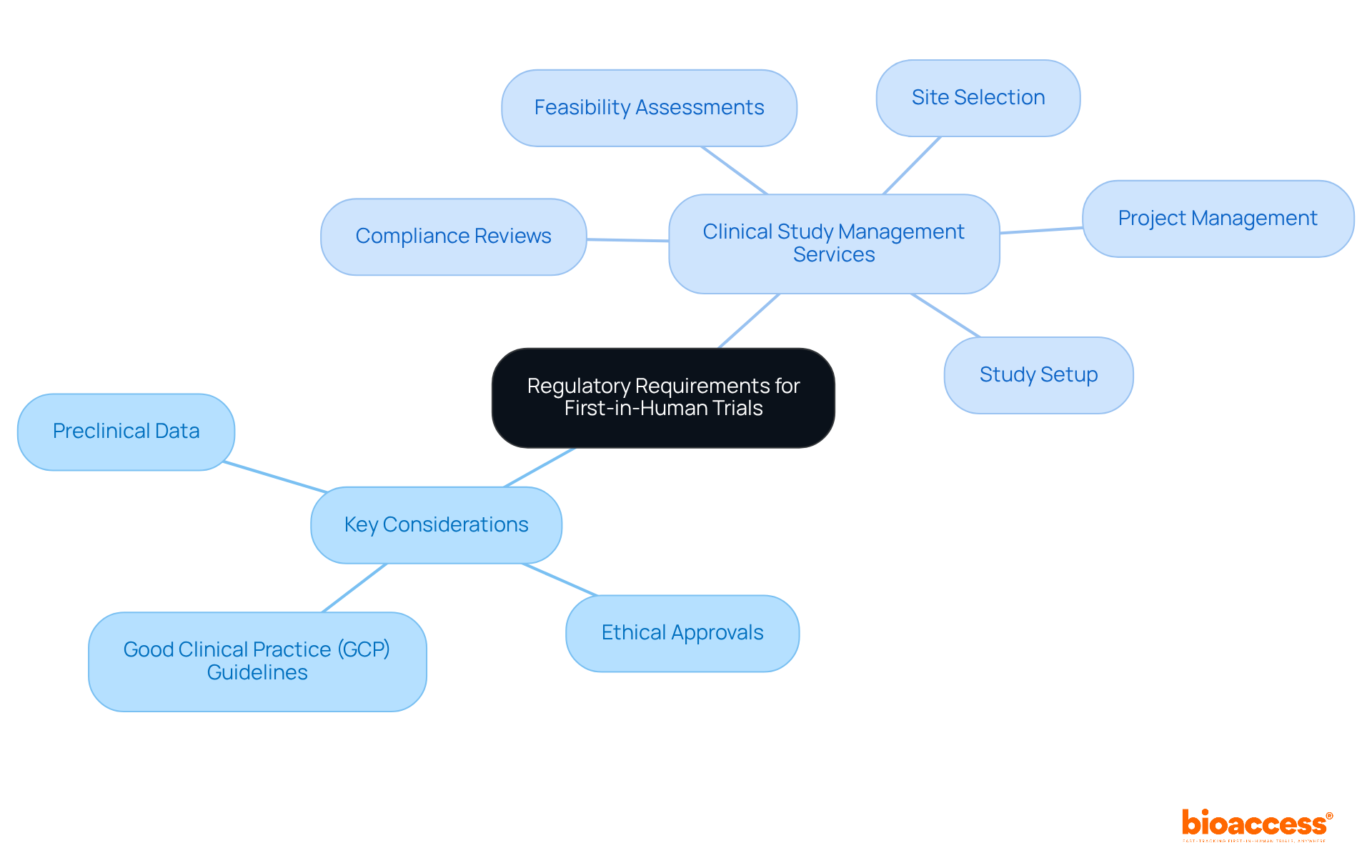
Navigating the is crucial for achieving success in . Understanding the in Brazil and COFEPRIS in Mexico is essential. Key considerations include:
- Preclinical data
- Strict compliance with
Moreover, effective play a vital role in ensuring adherence to these regulations. This includes:
- Conducting
- Selecting appropriate sites
- Performing compliance reviews
- Setting up studies
- Managing projects efficiently
Familiarity with these procedures not only simplifies the approval process but also significantly reduces risks associated with delays.
As you consider your own challenges in , reflect on how a thorough understanding of these can enhance your study’s success. Collaboration with experienced professionals can pave the way for smoother navigation through the complexities of regulatory compliance.
Implement Effective Patient Recruitment Strategies
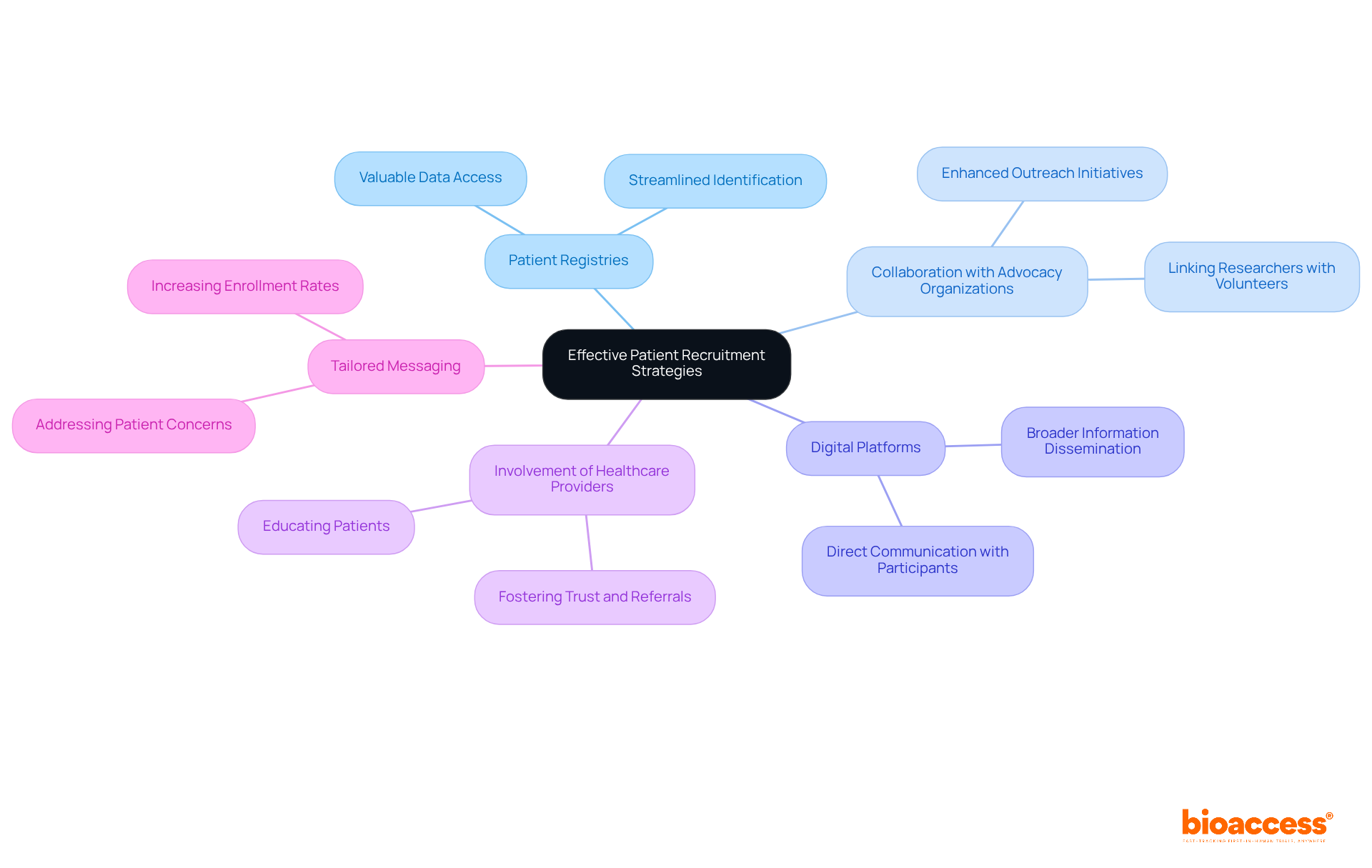
To effectively enlist individuals for the basics, implementing is essential. Leveraging patient registries streamlines the identification of eligible candidates, as these registries often contain valuable data on individuals interested in participating in . Collaborating with enhances outreach initiatives, linking researchers with potential volunteers already engaged in relevant .
Utilizing is another key trend in for 2026. These platforms enable broader dissemination of information and facilitate direct communication with potential participants. Involving healthcare providers is crucial; they play a vital role in educating patients about . By equipping practitioners with clear, transparent information about the study’s scope and eligibility criteria, researchers can foster trust and encourage referrals.
Tailoring recruitment messages to address the specific concerns and motivations of patients is vital for increasing . Understanding the distinct viewpoints of prospective individuals leads to more efficient communication approaches, ultimately enhancing the chances of successful recruitment in these innovative studies.
Design Robust Clinical Trial Protocols
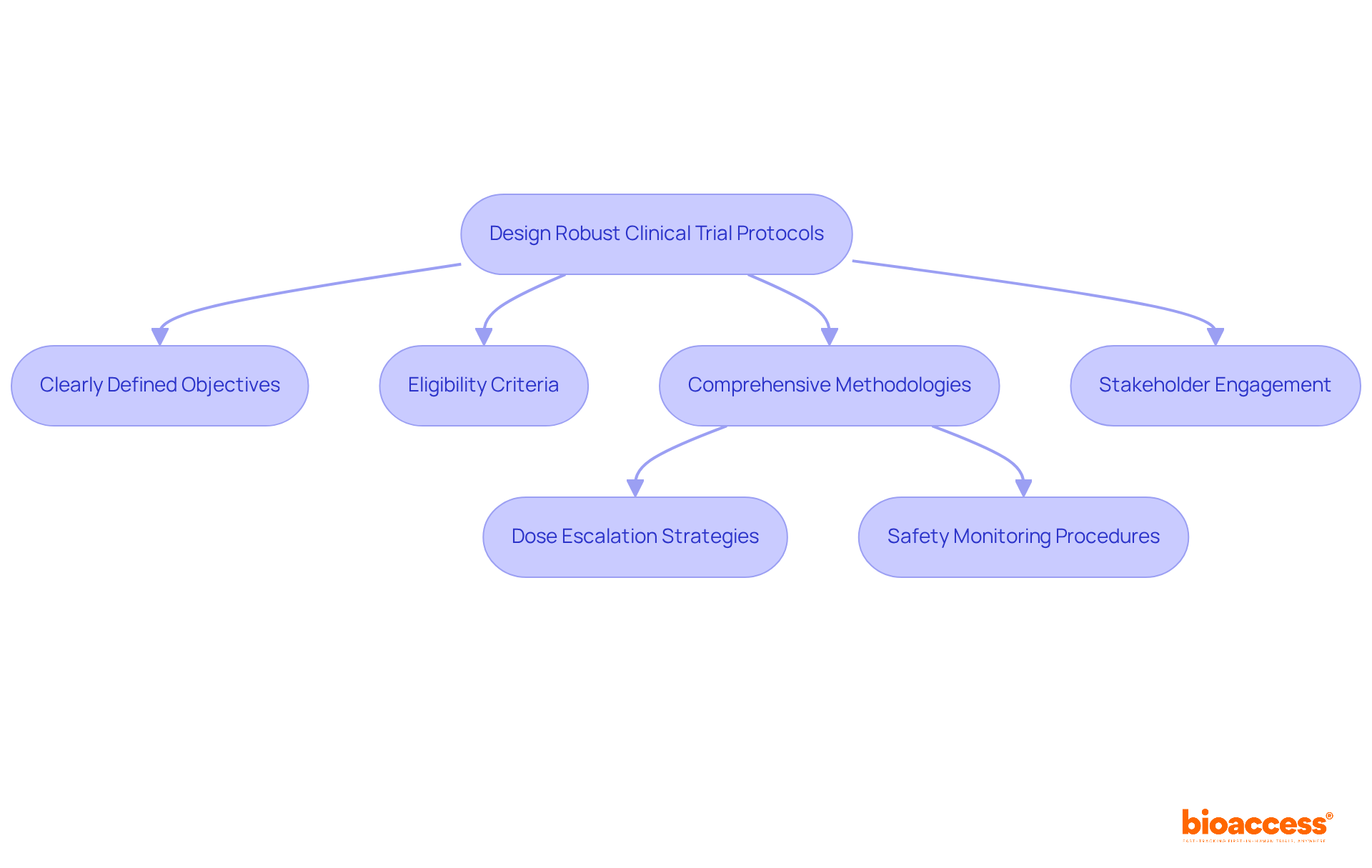
A well-organized is crucial for ensuring and data integrity. It must encompass several key elements, including:
- Clearly defined objectives
- Eligibility criteria
- Comprehensive methodologies
Special attention should be given to , alongside throughout the study. Furthermore, endpoints must be meticulously specified to accurately measure the study’s success.
- such as patient advocates and regulatory bodies – during the significantly enhances the protocol’s feasibility and acceptance. This proactive approach is essential for startups, particularly those , as it leads to more and aids in navigating the complexities of the approval process. Bioaccess also offers that empower startups to overcome , ensuring a seamless process for initiating and executing their projects.
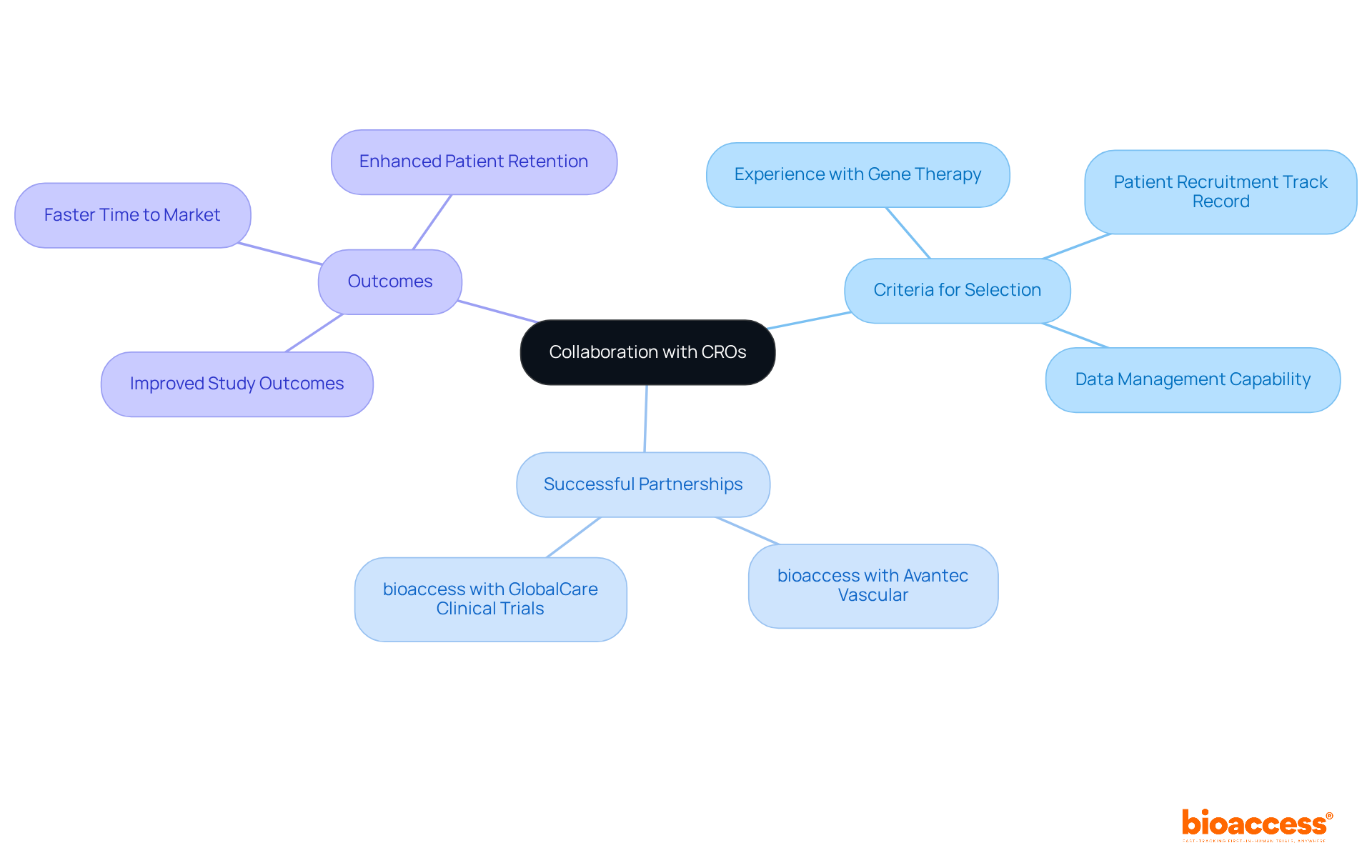
Collaborate with Experienced Clinical Research Organizations
Partnering with a reputable CRO like bioaccess can significantly streamline the management of the basics of . This collaboration provides access to , , and established networks that are crucial in today’s . For example, bioaccess is currently assisting Avantec Vascular with the first-in-human gene therapy trial basics in Latin America for an innovative vascular device. This partnership showcases bioaccess’s ability to facilitate critical processes, such as and submitting .
When selecting a CRO, it’s essential to consider:
- Their experience with gene therapy studies
- Their track record in
- Their capability to manage data effectively
A strong partnership can lead to and faster time to market. Take, for instance, bioaccess’s collaboration with GlobalCare , which achieved over a 50% reduction in recruitment time and impressive 95% retention rates. Such statistics not only highlight the effectiveness of bioaccess but also prompt you to reflect on your own challenges in clinical research.
In conclusion, the importance of collaboration in cannot be overstated. By choosing the right CRO, you can enhance your study’s success and navigate the complexities of the Medtech landscape with confidence. Consider taking the next step in your clinical research journey by partnering with a CRO that demonstrates proven expertise and a commitment to excellence.
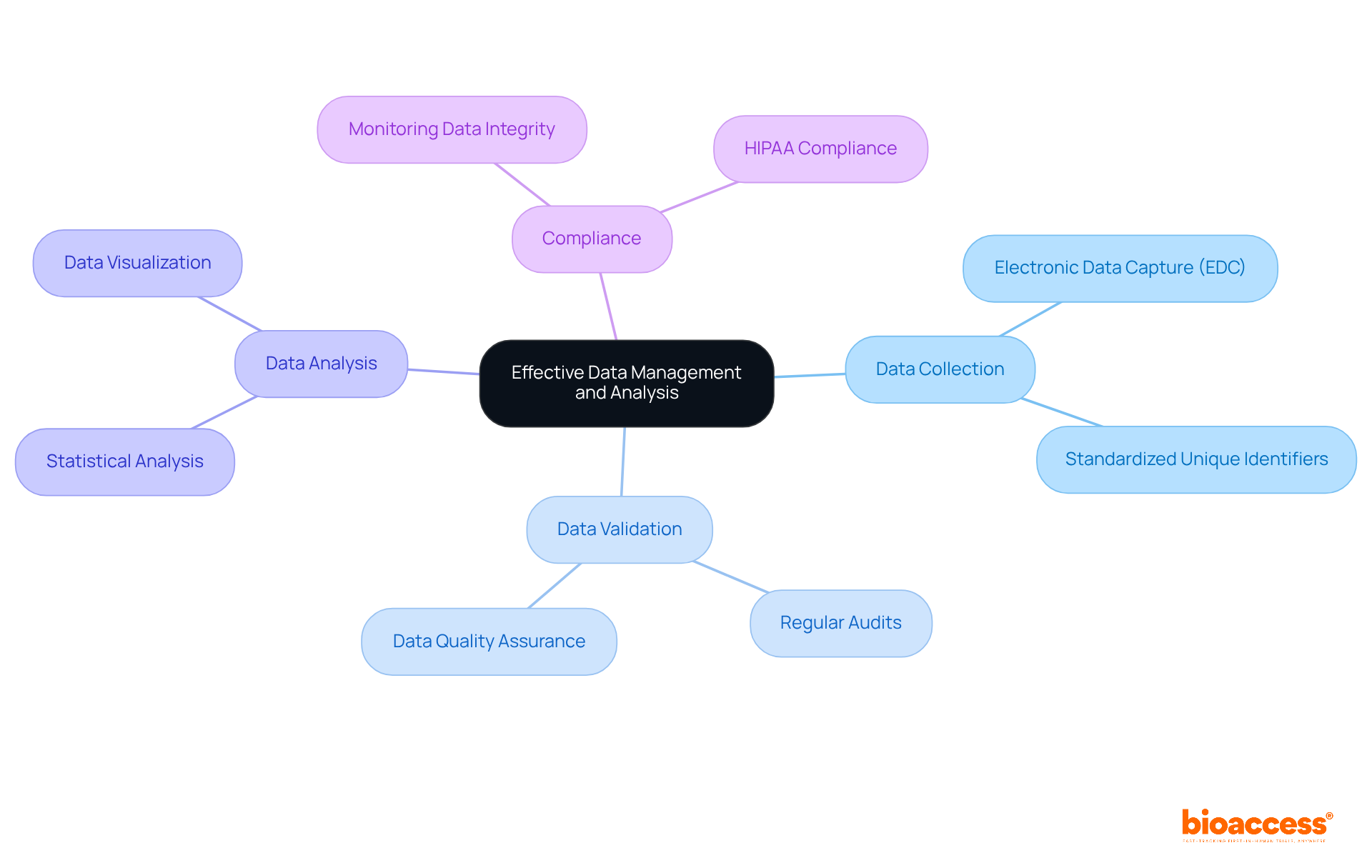
Ensure Effective Data Management and Analysis
Implementing a robust is not just essential; it’s a cornerstone for the . Establishing clear protocols for data collection, validation, and analysis is crucial. By utilizing , you can significantly enhance data accuracy and streamline the reporting process. Have you considered how regular audits and monitoring of can ensure compliance with ? This practice not only facilitates timely decision-making but also strengthens the overall research framework.
With over 20 years of expertise in overseeing , including the , bioaccess stands ready to support your effectively. Our comprehensive are designed to address the unique challenges of the Medtech landscape. Trust in our proven track record to navigate the complexities of clinical research, ensuring that your is both robust and reliable.
Prioritize Ethical Considerations in Trial Design
stands as a cornerstone of ethical considerations in the basics of gene therapy trials. It is imperative that researchers ensure individuals fully associated with their participation. not only fosters trust but also cultivates respect – both essential for engaging those involved in these trials.
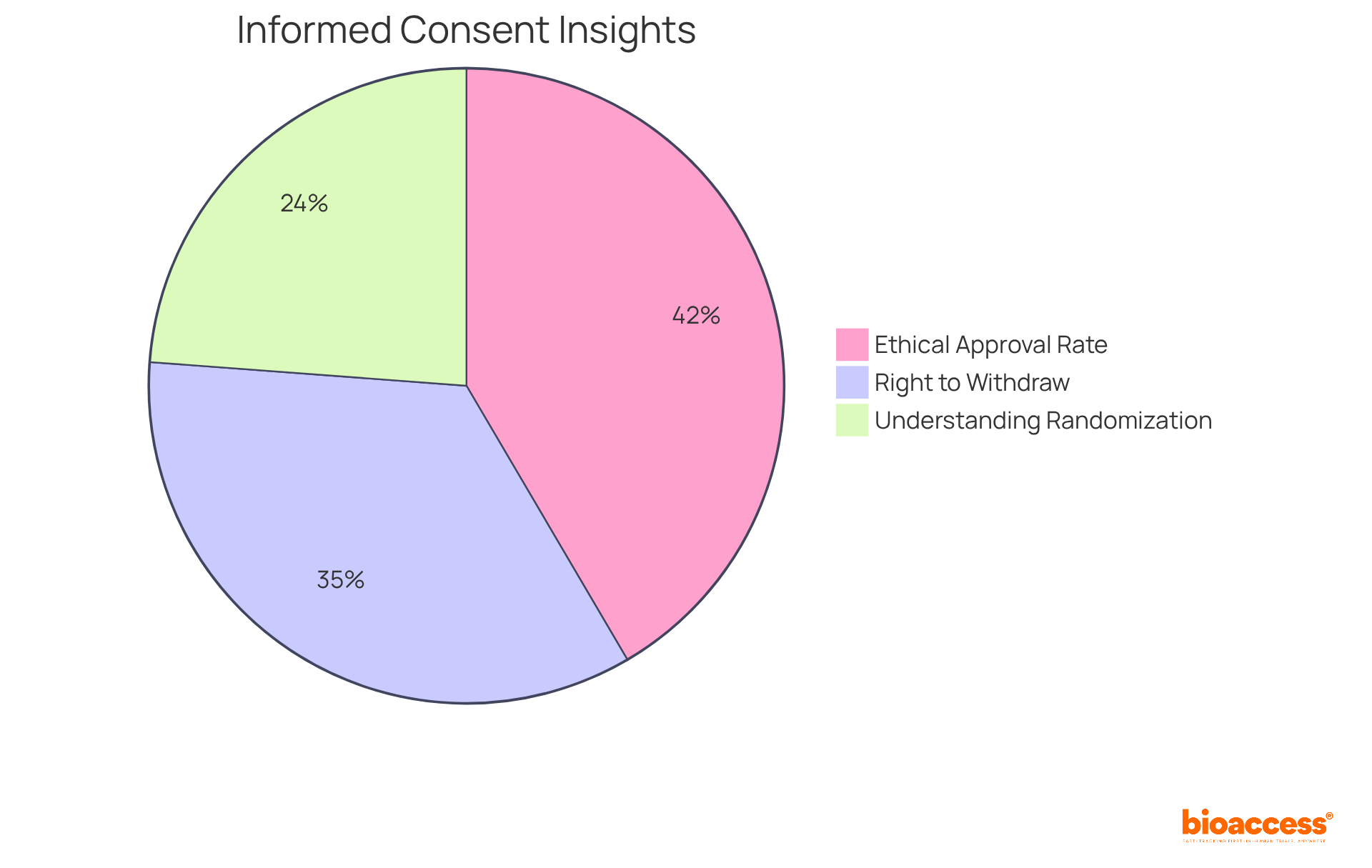
Current compliance rates with (IRBs) reveal that 90.9% of studies receive ethical approval, reflecting a strong commitment to the well-being of subjects. However, this approval hinges on , which are critical for maintaining . Alarmingly, only 52.1% of individuals grasp fundamental concepts like randomization, highlighting the urgent need for improved consent processes. Furthermore, 75.8% of individuals are aware of their right to withdraw from trials, underscoring the importance of ensuring that participants are fully informed.
Experts emphasize that obtaining is essential for . As the landscape of evolves, ongoing discussions about practices remain vital to guarantee the and safety of individuals participating in studies related to gene therapy trial basics.
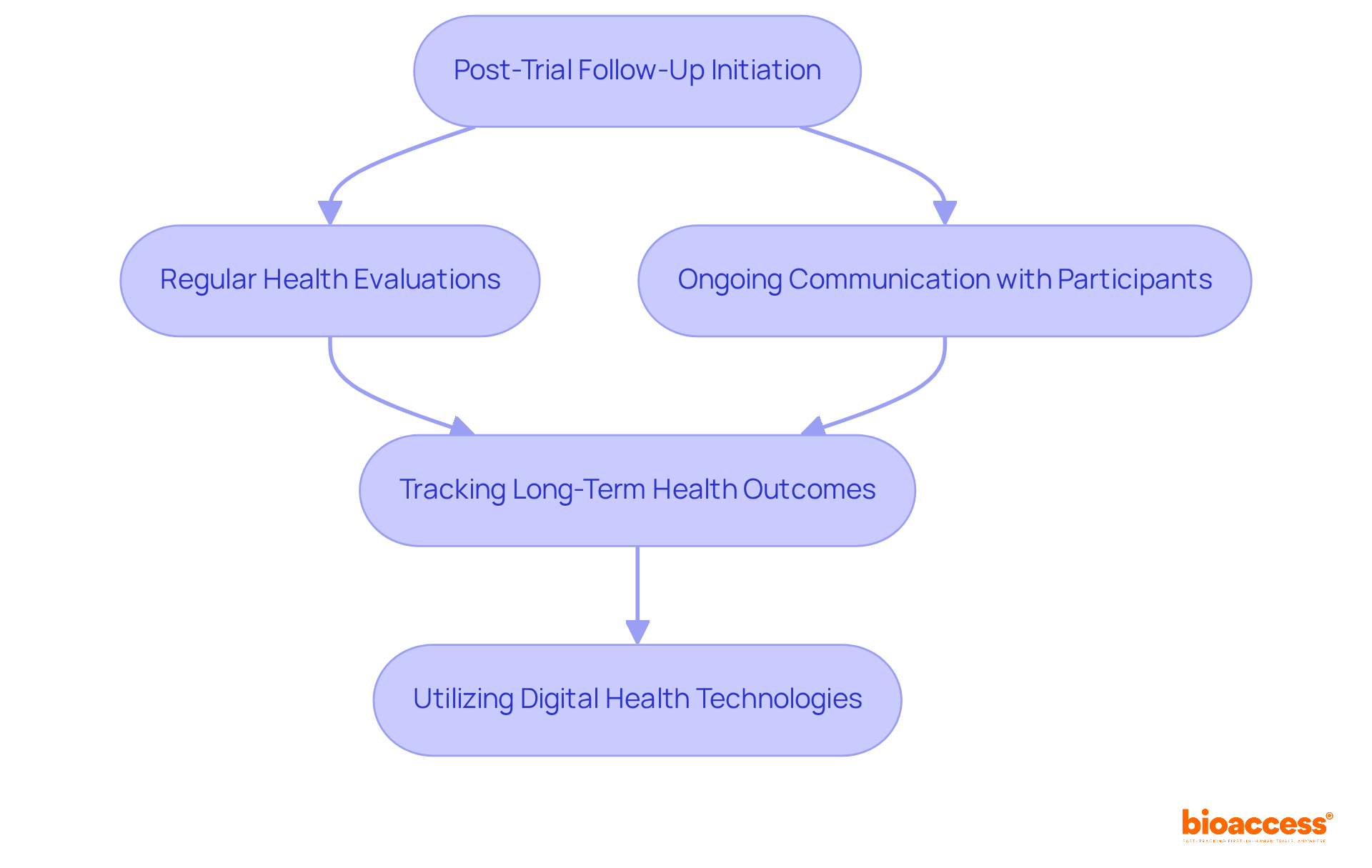
Implement Post-Trial Follow-Up and Monitoring
Post-trial follow-up is essential for tracking individuals’ and identifying any delayed associated with . Recent studies reveal that the average duration of can extend up to 15 years, facilitating a thorough assessment of treatment effects and safety. A well-structured , which includes regular health evaluations and ongoing communication with participants, not only enhances safety but also provides invaluable insights for future research.
Engaging individuals in this process fosters trust and encourages their continued involvement in research, which is critical for the success of long-term . As we look ahead to 2026, advancements in and decentralized study designs are being harnessed to boost and streamline data collection. This ensures that are effectively monitored and analyzed, paving the way for improved .
Invest in Continuous Education and Training for Research Teams
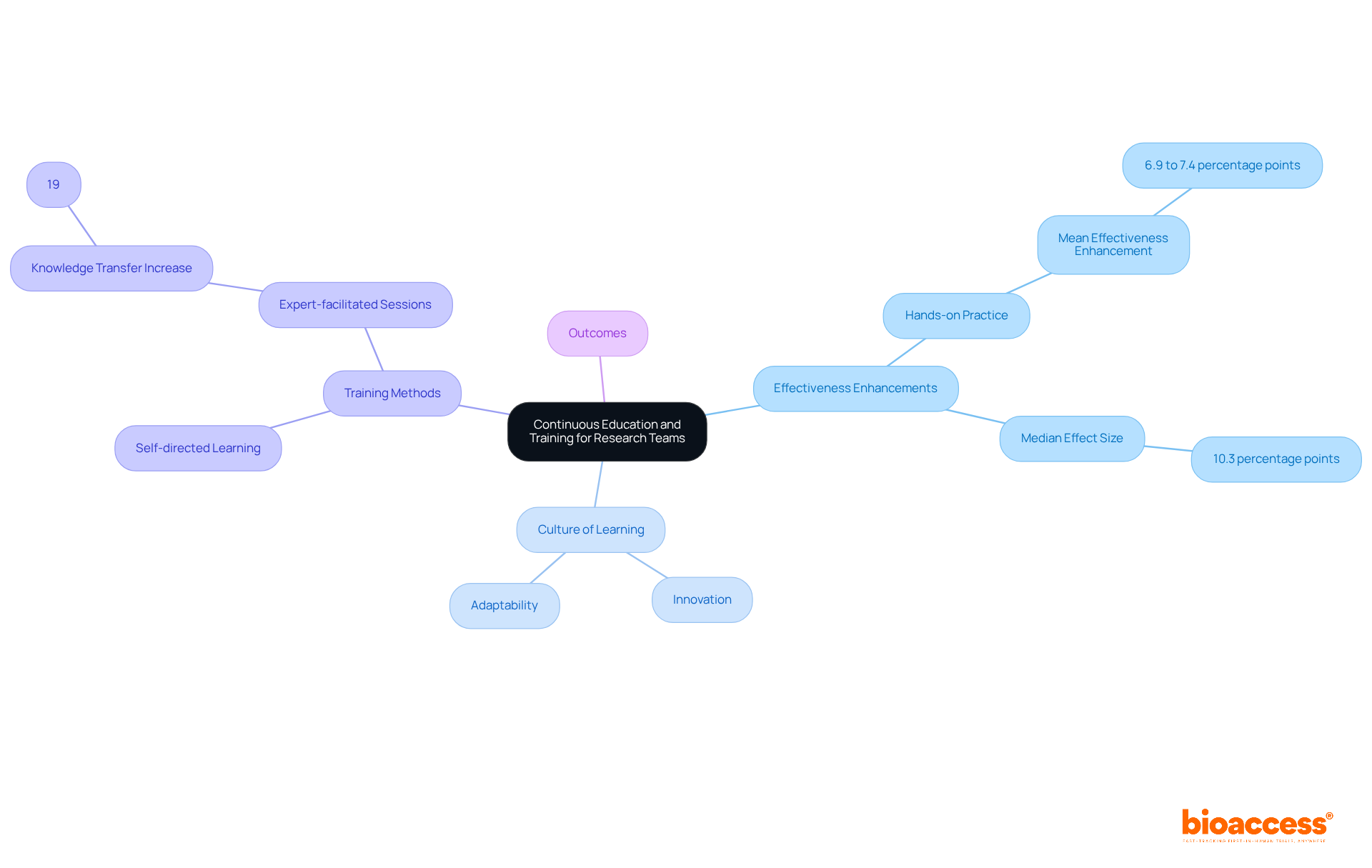
are crucial in keeping pace with the rapid advancements in and the evolving regulatory landscape. Access to workshops, online courses, and certification programs significantly boosts team competencies, leading to improved outcomes. For example, training programs that incorporate of 6.9 to 7.4 percentage points. Additionally, a systematic review reveals a median effect size of 10.3 percentage points, highlighting the importance of .
Moreover, cultivating a within research teams not only fosters innovation but also enhances adaptability, ultimately benefiting the research process. Organizations like CTTI advocate for that blend self-directed learning with expert-facilitated sessions, which can result in a 19% increase in knowledge transfer during investigator meetings. As CTTI emphasizes, to ensure that learners remain attentive and grasp the content.
As the gene treatment landscape continues to evolve, investing in comprehensive is essential for maintaining high standards in medical studies. Are your research teams equipped to meet these challenges? The time to is now.
Develop Effective Communication Strategies for Research Teams
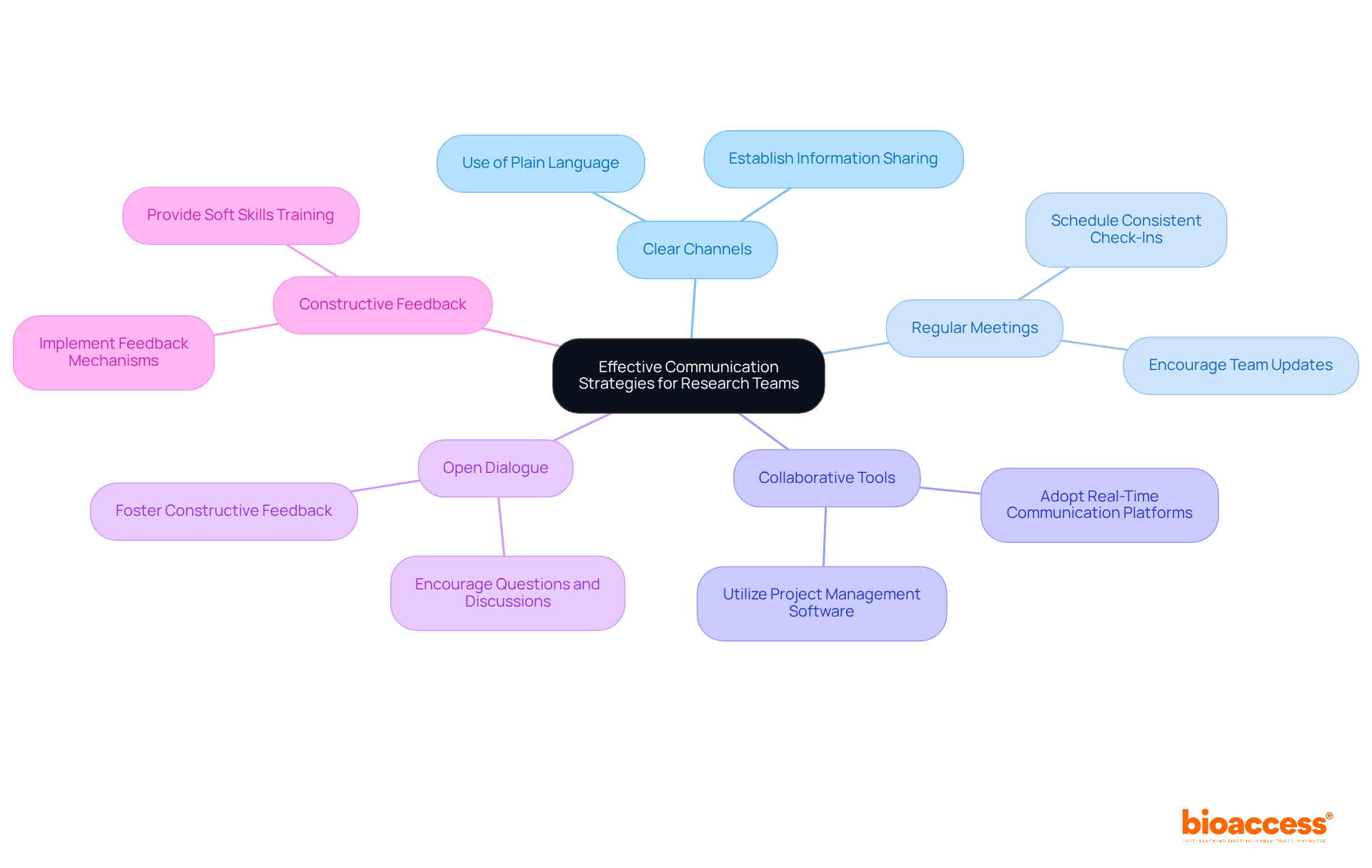
are essential for the success of research groups, especially when discussing . Establishing and scheduling regular meetings significantly enhances team coordination. , with many teams adopting platforms that facilitate real-time communication and project management. In fact, organizations that promote collaborative working are five times more likely to be high performing, underscoring the effectiveness of these tools in research environments.
Encouraging open dialogue and , empowering members to proactively tackle challenges and align their efforts toward common goals. This collaborative approach is vital in navigating the complexities of , especially when considering the , where can lead to improved outcomes and efficiency. As highlighted in a case study on the importance of communication in teams, and can prevent failures.
In conclusion, prioritizing efficient communication strategies not only enhances team performance but also drives successful outcomes in clinical research. Organizations must take action to implement these strategies, ensuring that their teams are equipped to meet the challenges of the evolving .
Conclusion
The exploration of first-in-human gene therapy trials unveils a landscape brimming with potential and complexity. Understanding foundational aspects – from regulatory requirements and patient recruitment strategies to ethical considerations and data management – is essential for success in this innovative field. By harnessing the unique advantages offered by regions like Latin America, particularly through organizations such as bioaccess®, researchers can significantly boost the efficiency and effectiveness of their clinical trials.
Key insights throughout this discussion underscore the necessity of navigating regulatory frameworks, implementing robust clinical trial protocols, and ensuring ethical integrity through informed consent. Moreover, the significance of effective patient recruitment strategies and ongoing education for research teams cannot be overstated, as these elements directly contribute to the overall success and reliability of clinical research endeavors.
As the gene therapy landscape continues to evolve, embracing collaboration with experienced clinical research organizations and investing in continuous education will be vital for overcoming challenges and optimizing outcomes. The future of gene therapy holds immense promise; by prioritizing these essential basics, researchers can pave the way for groundbreaking advancements in personalized medicine. The time to act is now – commit to these practices and contribute to the transformative potential of gene therapy.
Frequently Asked Questions
What is bioaccess® and what role does it play in gene therapy trials in Latin America?
bioaccess® accelerates first-in-human gene therapy trials in Latin America by providing a streamlined approach that guarantees ethics approvals within 4 to 8 weeks, a patient enrollment process that is 40% faster than in the US and EU, and an average cost reduction of 30% compared to traditional trial expenses.
Why is Colombia considered an ideal location for conducting gene therapy trials?
Colombia is considered ideal due to its healthcare system ranked #22 globally by the World Health Organization, its hospitals being among the best in Latin America, a population exceeding 50 million, and universal healthcare coverage for 95% of its citizens.
What financial advantages does bioaccess® provide for clinical research?
bioaccess® leverages R&D tax incentives that offer substantial financial benefits for clinical research, making it a preferred choice for Medtech and Biopharma firms.
What are the key regulatory requirements for first-in-human gene therapy trials in Latin America?
Key regulatory requirements include preclinical data, ethical approvals, and strict compliance with Good Clinical Practice (GCP) guidelines.
How can effective clinical study management services aid in regulatory compliance?
Effective clinical study management services can help by conducting feasibility assessments, selecting appropriate sites, performing compliance reviews, setting up studies, and managing projects efficiently.
What strategies can be implemented for effective patient recruitment in gene therapy trials?
Strategies include leveraging patient registries, collaborating with advocacy organizations, utilizing digital platforms for outreach, involving healthcare providers, and tailoring recruitment messages to address patient concerns and motivations.
Why is collaboration with healthcare providers important in patient recruitment?
Healthcare providers play a crucial role in educating patients about study opportunities, fostering trust, and encouraging referrals by providing clear and transparent information about the study’s scope and eligibility criteria.
List of Sources
- bioaccess: Accelerate First-in-Human Gene Therapy Trials in Latin America
- Latin America Cell and Gene Therapy Market to Grow at 24.40% CAGR till 2034 (https://towardshealthcare.com/insights/latin-america-cell-and-gene-therapy-market-sizing)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC11361337)
- Latin America’s Landscape For Medtech Clinical Trials (https://clinicalleader.com/doc/latin-america-s-landscape-for-medtech-clinical-trials-0001)
- Should You Conduct Your Medical Device Clinical Trial In Latin America? – Journal for Clinical Studies (https://journalforclinicalstudies.com/conduct-medical-device-clinical-trial-latin-america)
- Understand Regulatory Requirements for First-in-Human Trials
- New regulations for clinical research in Brazil | Licks Attorneys (https://lickslegal.com/post/new-regulations-for-clinical-research-in-brazil)
- Master ANVISA Guidelines for Medtech Complianc… | bioaccess® (https://bioaccessla.com/blog/master-anvisa-guidelines-for-medtech-compliance-a-step-by-step-approach)
- bioaccessla.com (https://bioaccessla.com/blog/how-to-conduct-first-in-human-studies-cro-in-brazil-a-step-by-step-guide)
- What to Know About Brazil’s New Clinical Research Law (https://psi-cro.com/brazil-new-clinical-research-law)
- Implement Effective Patient Recruitment Strategies
- 2025 Trends In Patient Recruitment: From Disruption To Precision (https://clinicalleader.com/doc/trends-in-patient-recruitment-from-disruption-to-precision-0001)
- onestudyteam.com (https://onestudyteam.com/blog/clinical-trial-metrics-to-evaluate-patient-recruitment-campaigns)
- Enrollment in Clinical Trials: Statistics and Patient Recruitment Strategies | Power (https://withpower.com/guides/enrollment-in-clinical-trials-statistics-and-patient-recruitment-strategies)
- A Primer on the Importance of Recruitment and Retention in Clinical Trials – ACRP (https://acrpnet.org/2023/04/18/a-primer-on-the-importance-of-recruitment-and-retention-in-clinical-trials)
- Utilization of Real-World Data to Enhance Recruitment and Retention of Clinical Research Participants – ACRP (https://acrpnet.org/2019/08/13/utilization-of-real-world-data-to-enhance-recruitment-and-retention-of-clinical-research-participants)
- Design Robust Clinical Trial Protocols
- What is a Clinical Trial Protocol? A Guide to Its Design | IntuitionLabs (https://intuitionlabs.ai/articles/clinical-trial-protocol-guide)
- Clinical Trial Biostatistics | PPD (https://ppd.com/what-is-a-cro/clinical-trial-biostatistics)
- Comprehensive Guide to Clinical Trial Protocol Design & Development | Clinical Leader (https://clinicalleader.com/topic/clinical-trial-protocol-design-development)
- What are your views on Gene Therapy? – DelveInsight Business Research (https://delveinsight.com/blog/what-are-your-views-on-gene-therapy)
- Collaborate with Experienced Clinical Research Organizations
- appliedclinicaltrialsonline.com (https://appliedclinicaltrialsonline.com/view/insights-capturing-collaborative-value)
- Wide inconsistency observed among clinical trials’ startup time: Tufts report (https://fiercebiotech.com/cro/wide-inconsistency-observed-among-clinical-trials-startup-time-tufts-report)
- Contract Research Organization [CRO] Services Market, 2034 (https://fortunebusinessinsights.com/industry-reports/contract-research-organization-cro-services-market-100864)
- Building Strategic Sponsor-CRO Partnerships: The International Element Changing Clinical Trials (https://pharmasalmanac.com/articles/building-strategic-sponsor-cro-partnerships)
- Ensure Effective Data Management and Analysis
- infocentric.com.au (https://infocentric.com.au/2022/04/28/data-management-quotes)
- 20 Data Science Quotes by Industry Experts (https://coresignal.com/blog/data-science-quotes)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC12574785)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC6170092)
- Prioritize Ethical Considerations in Trial Design
- As genome-editing trials become more common, informed consent is changing (https://genome.gov/news/news-release/As-genome-editing-trials-become-more-common-informed-consent-is-changing)
- bioaccessla.com (https://bioaccessla.com/blog/10-key-insights-for-first-in-human-clinical-trials-success)
- Participants’ understanding of informed consent in clinical trials: A systematic review and updated meta-analysis (https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0295784)
- Clinical Trial Recruitment Statistics: Market Data Report 2026 (https://gitnux.org/clinical-trial-recruitment-statistics)
- Bioethics Quotes – 12 quotes on Bioethics Science Quotes – Dictionary of Science Quotations and Scientist Quotes (https://todayinsci.com/QuotationsCategories/B_Cat/Bioethics-Quotations.htm)
- Implement Post-Trial Follow-Up and Monitoring
- Long-Term Follow-Up of Patients Receiving Cell and Gene Therapy Products – Laszlo Irinyi, Barbara Mintzes, Julia Warning, Laura Collie, Amanda Rush, Cameron J. Turtle, Jennifer A. Byrne, 2026 (https://journals.sagepub.com/doi/10.1177/10430342251403439)
- (PDF) Participant perspectives on a phase I/II ocular gene therapy trial (NCT02077361) (https://researchgate.net/publication/334247062_Participant_perspectives_on_a_phase_III_ocular_gene_therapy_trial_NCT02077361)
- Post-trial monitoring of a randomised controlled trial of intensive glycaemic control in type 2 diabetes extended from 10 years to 24 years (UKPDS 91) (https://thelancet.com/journals/lancet/article/PIIS0140-6736(24)00537-3/fulltext)
- Long-term Follow-up of Trial Participants by Linkage to Routinely Collected Data (https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2719129)
- Gene Therapy Trials: Ensuring Engagement & Compliance (https://premier-research.com/perspectives/long-term-follow-up-in-gene-therapy-trials-ensuring-patient-engagement-regulatory-compliance)
- Invest in Continuous Education and Training for Research Teams
- What are your views on Gene Therapy? – DelveInsight Business Research (https://delveinsight.com/blog/what-are-your-views-on-gene-therapy)
- Elevate Training to Overcome Site Burden and Reduce Protocol Deviations by 35-50% | WCG (https://wcgclinical.com/insights/elevate-training-to-overcome-site-burden-and-reduce-protocol-deviations-by-35-50)
- The Value of Continuing Education and Professional Development for Clinical Trial Managers (https://briogrp.com/the-value-of-continuing-education-and-professional-development-for-clinical-trial-managers)
- Quotes on Genes (https://medium.com/@mnemko/quotes-on-genes-64f9f457d1f)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC7813291)
- Develop Effective Communication Strategies for Research Teams
- Improving Communication in Clinical Research – SOCRA Blog (https://socra.org/blog/improving-communication-in-clinical-research)
- 35 Quotes about Communication to Inspire Collaboration (https://vibe.us/blog/35-quotes-about-communication?srsltid=AfmBOooQLQDGUOC05WEdeh_hNiZcnxPPzP8x3jg1-fbqr4klhFUsiSzn)
- 36 Workplace Communication Quotes to Inspire Your Team (https://trinet.com/insights/workplace-communication-quotes)
- Communication Strategies (https://cdc.gov/health-literacy/php/research-summaries/communication-strategies.html)

Leave a Reply