Introduction
Navigating the medical device approval landscape in Peru presents significant challenges, particularly due to the complexities of the DIGEMID regulatory framework. This guide provides a clear, step-by-step approach to mastering the approval process, equipping aspiring manufacturers and distributors with the essential tools to meet necessary requirements.
As regulations evolve and timelines vary, how can you effectively streamline your application to avoid common pitfalls and ensure timely market entry? Understanding these dynamics is crucial for success in the Medtech sector.
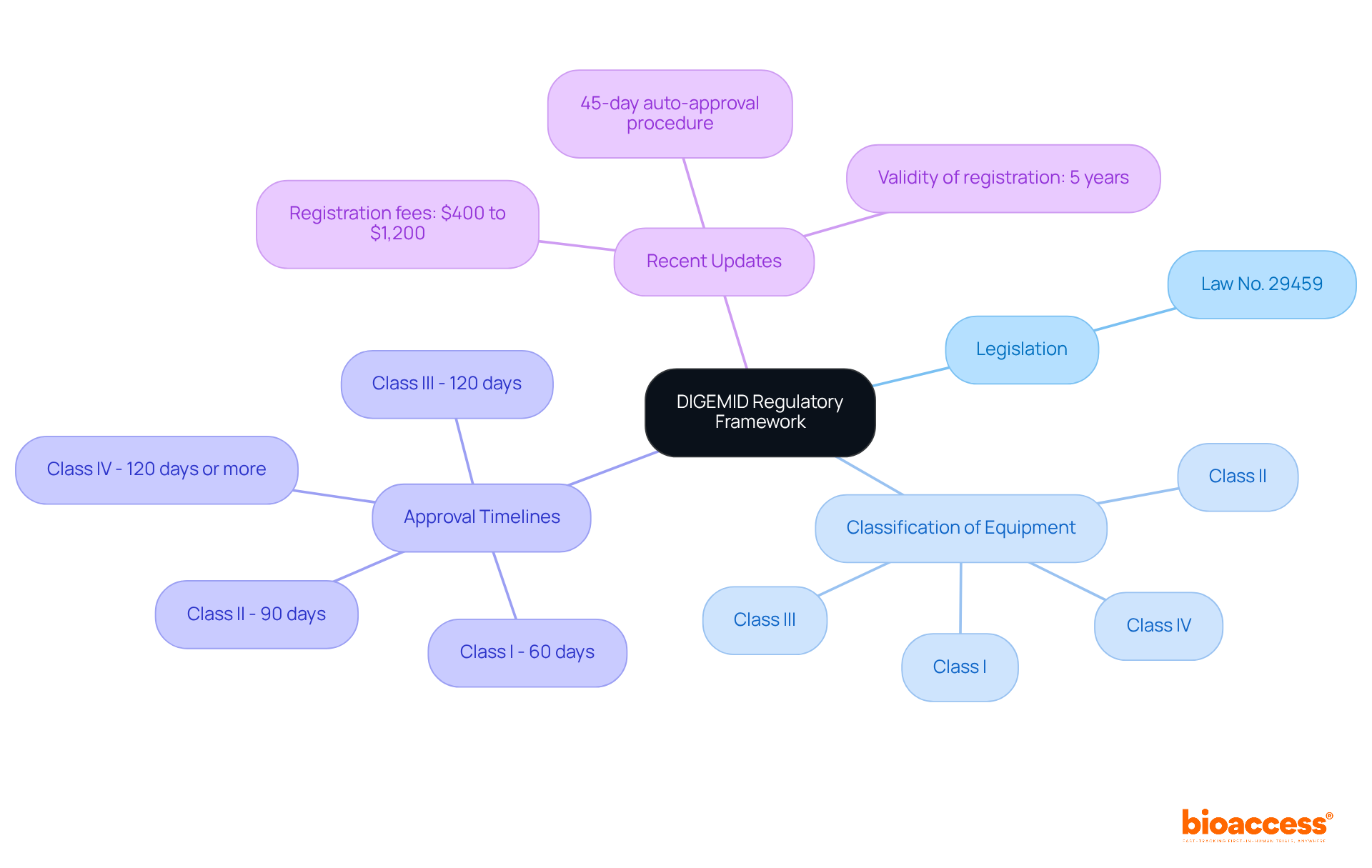
Understand the DIGEMID Regulatory Framework
Navigating the requires a solid understanding of the and its . This agency oversees the enrollment, monitoring, and quality assurance of medical equipment in the country. Here are the key aspects you need to know:
- Legislation: The cornerstone of is . This law outlines the requirements for registration and market access, ensuring that products meet essential safety and efficacy standards.
- : Medical instruments are classified into four categories (I, II, III, and IV) based on their risk level. Understanding this classification is crucial, as it determines the required for each item. For example, Class I products generally need less documentation than Class IV products, which demand thorough technical and safety evaluations.
- : The evaluation process can vary significantly depending on the item’s category. Class II and III devices typically undergo evaluations that can last between 6 to 12 months, while Class IV devices may take even longer due to their complexity. Being aware of these timelines is vital for effective submission planning.
- : It’s important to stay updated on any recent regulatory changes, such as the introduction of a 45-day auto-approval procedure for certain applications, which can expedite the approval timeline under specific conditions. Additionally, , and the validity of registration is five years-key factors for understanding the financial and long-term implications of the approval process.
By understanding these foundational elements, you will be better prepared to navigate the complexities of the . Collaborating with professionals like Ana Criado, Director of Regulatory Affairs at bioaccess, who possesses extensive experience in regulatory navigation and consulting for medical devices, can pave a smoother path to compliance and market entry.
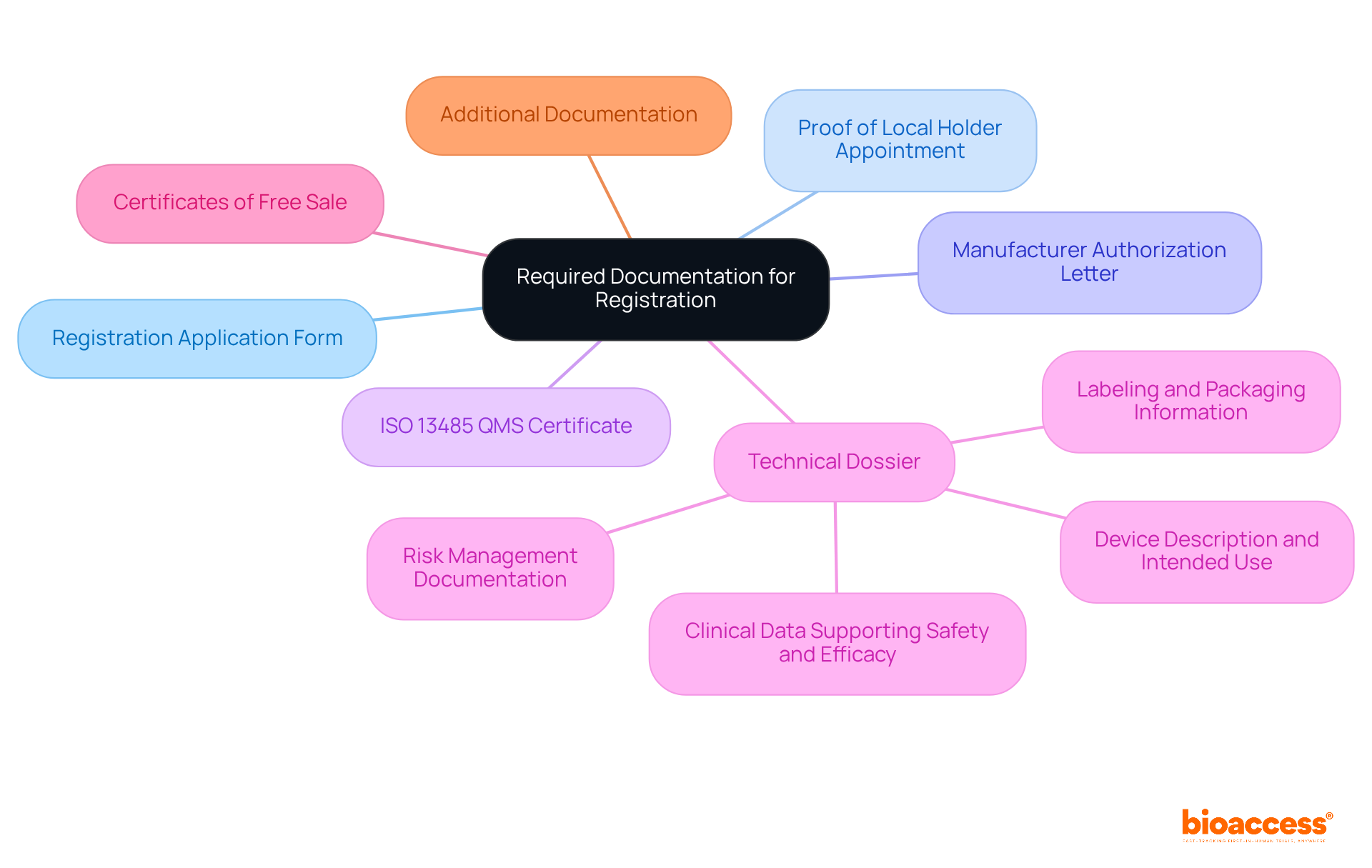
Gather Required Documentation for Registration
Before sending your application file to the authority, it’s crucial to gather all required paperwork to guarantee a seamless approval experience. The following checklist outlines the necessary documents:
- Registration Application Form: Complete the official application form supplied by the relevant authority.
- Proof of Local Holder Appointment: Designate a local representative or holder responsible for overseeing the enrollment process in Peru. This appointment is critical, as the local holder must have a in the country to facilitate communication with DIGEMID.
- : Obtain a letter from the manufacturer authorizing the local holder to act on their behalf, ensuring .
- : Ensure that your quality management system complies with , a prerequisite for all . Adherence to these standards is essential, as it indicates the safety and effectiveness of the equipment.
- : Prepare a comprehensive that includes:
- Device description and intended use
- supporting safety and efficacy
- Risk management documentation
- Labeling and packaging information
- : Provide certificates from nations where the product is already marketed, demonstrating and enhancing credibility.
- Additional Documentation: Depending on the device class, additional documents may be required, such as stability studies or post-market surveillance plans.
Having all these documents ready will streamline the and significantly reduce the likelihood of delays, ensuring a more efficient pathway to market entry in Peru.
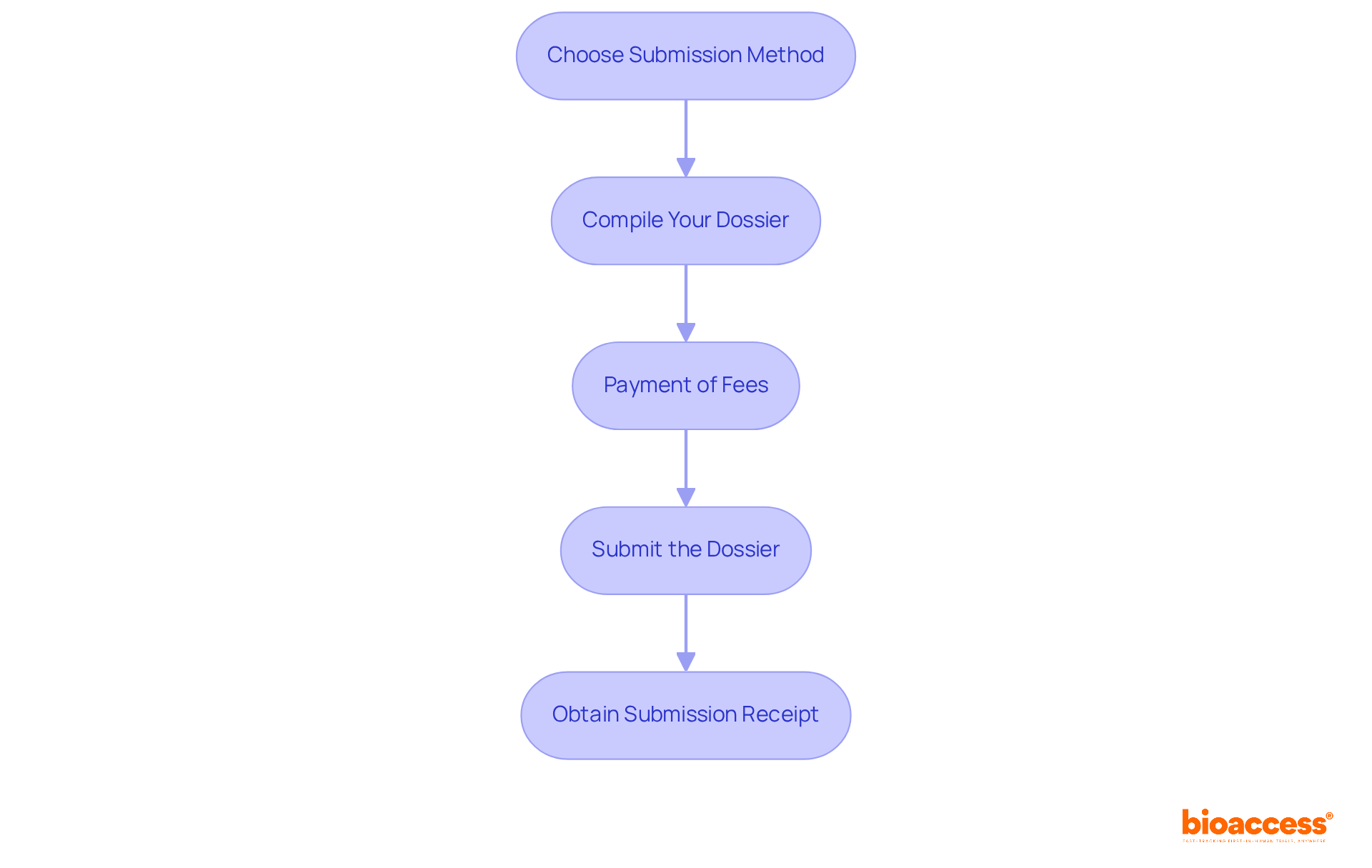
Submit Your Registration Dossier to DIGEMID
Once you have gathered all the required documentation, the next step is to to the . To ensure a , follow these steps:
- Choose Submission Method: Decide whether to submit your application online through the designated platform or in person at their offices. Stay updated on current , as they may change.
- Compile Your Dossier: Organize your documents according to the agency’s requirements. Ensure that all documents are complete, signed, and dated.
- : Prepare to pay the applicable , which are approximately 400 euros for Class I, 460 euros for Class II, 510 euros for Class III, and 580 euros for Class IV devices. Check the latest fee schedule on the ‘s website to ensure you include the correct amount.
- Submit the Dossier: If submitting electronically, follow the online instructions carefully. If submitting in person, ensure you have multiple copies of your dossier for submission.
- : After submission, request a receipt or confirmation of your application. This document is essential for tracking the status of your application.
By following these steps, you will ensure that your enrollment file is submitted accurately, setting the stage for the evaluation phase. Understanding as a Level 4 health authority by PAHO/WHO provides valuable insights into the , aiding in effectively navigating the submission procedure.
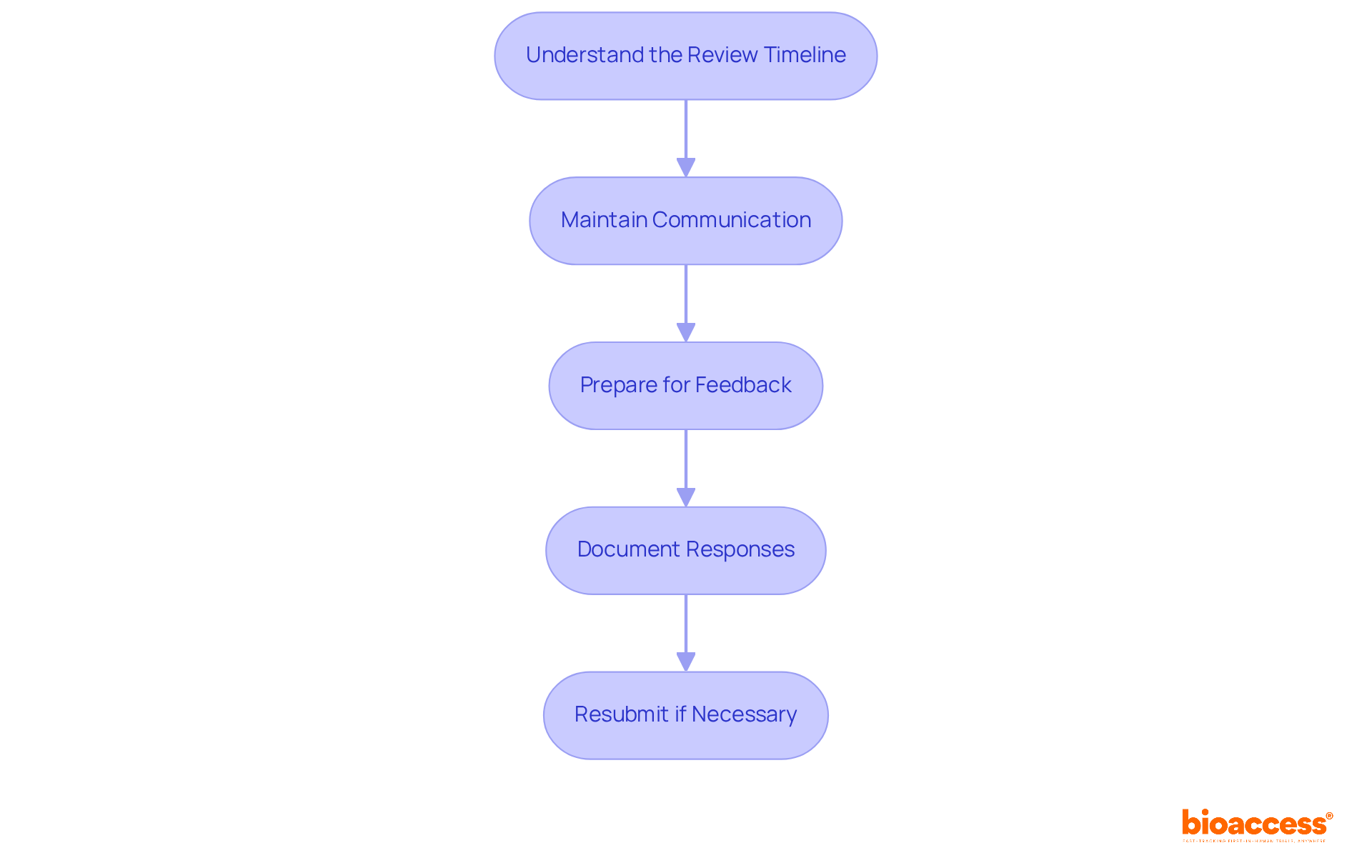
Navigate the Review Process and Respond to Feedback
After submitting your registration dossier, it enters the at DIGEMID. Understanding how to navigate this phase effectively is crucial for success:
- : The duration of the review varies by equipment category. Class I devices typically experience shorter timelines, often within a few weeks. In contrast, Class II and III devices may take 6 to 12 months, depending on their complexity and any additional information requests. Staying informed about these timelines is essential for managing expectations.
- : Keeping open lines of communication with the agency is vital. If you haven’t received updates within the expected timeframe, proactively follow up to inquire about your application’s status. This not only demonstrates your commitment but also helps build rapport with the agency.
- : During the review, DIGEMID may request additional information or clarification on specific aspects of your submission. Be ready to respond quickly and comprehensively to any questions, as timely communication can significantly influence the .
- : When addressing feedback, document your responses meticulously. Provide clear and concise explanations, and include any additional documentation requested. This organized approach aids in clarity and reflects professionalism.
- : If your application is not approved on the first attempt, carefully review the feedback, make the necessary adjustments, and resubmit your application as soon as possible. Understanding the reasons for rejection and addressing them effectively can .
By effectively navigating the and responding to feedback, you significantly increase your chances of achieving the for your .
Conclusion
Mastering the Peru DIGEMID device approval process is crucial for any medical device manufacturer looking to enter the Peruvian market. A solid understanding of the regulatory framework – including legislation, classification, and approval timelines – sets the stage for successful registration. By meticulously gathering the necessary documentation and following submission guidelines, stakeholders can confidently navigate the complexities of the DIGEMID process.
Key elements such as:
- Grasping the classification of medical devices
- Preparing a thorough registration dossier
- Effectively addressing feedback from the review process
are vital for ensuring compliance and speeding up market entry. Collaborating with seasoned professionals can significantly enhance the chances of a smooth approval journey, making it essential to leverage expert insights throughout the process.
Ultimately, staying updated on the latest regulations and adopting a proactive communication strategy with DIGEMID can greatly influence the success of device approvals. By adhering to this step-by-step guide, manufacturers can streamline their registration efforts and contribute to delivering safe and effective medical devices to the Peruvian healthcare system. Embracing these practices will pave a more efficient pathway to compliance and market success in Peru.
Frequently Asked Questions
What is the role of DIGEMID in Peru?
DIGEMID oversees the enrollment, monitoring, and quality assurance of medical equipment in Peru, ensuring that products meet safety and efficacy standards.
What legislation governs medical equipment regulation in Peru?
The cornerstone of medical equipment regulation in Peru is Law No. 29459, which outlines the requirements for registration and market access.
How are medical instruments classified in Peru?
Medical instruments are classified into four categories (I, II, III, and IV) based on their risk level, which determines the documentation and assessment procedures required for each item.
What are the approval timelines for medical devices in Peru?
Class II and III devices typically undergo evaluations lasting between 6 to 12 months, while Class IV devices may take longer due to their complexity.
What recent updates should applicants be aware of regarding the approval process?
Recent updates include the introduction of a 45-day auto-approval procedure for certain applications, which can expedite the approval timeline under specific conditions.
What are the registration fees and validity period for medical devices in Peru?
Registration fees range from $400 to $1,200, and the validity of registration is five years.
How can professionals assist in navigating the DIGEMID approval process?
Collaborating with experienced professionals, such as regulatory affairs consultants, can help streamline the compliance and market entry process for medical devices.
List of Sources
- Understand the DIGEMID Regulatory Framework
- DIGEMID’S Approval Process: A Guide for Manufacturers and Exporters (https://conicalpharmaceuticals.com/news-events/digemids-approval-process)
- Year in review: life sciences law in Peru (https://lexology.com/library/detail.aspx?g=61e8f32a-500b-4930-bfd3-900cf0fb40e7)
- Medical device registration in Peru – a brief guide | MDRC (https://mdrc-consulting.com/peru-medical-device-registration-guide-en)
- Medical Device Registration in Peru – DIGEMID Requirements (https://thema-med.com/en/medical-device-registration-in-peru)
- Gather Required Documentation for Registration
- Medical Device Registration in Peru – DIGEMID Requirements (https://thema-med.com/en/medical-device-registration-in-peru)
- Medical device registration in Peru – a brief guide | MDRC (https://mdrc-consulting.com/peru-medical-device-registration-guide-en)
- Medical Device Registration in Peru (https://emergobyul.com/services/medical-device-registration-peru)
- Our Guide For How to Register Medical Devices In Peru (https://regdesk.co/blog/guide-how-to-register-medical-devices-peru)
- Submit Your Registration Dossier to DIGEMID
- Medical Device Registration in Peru – DIGEMID Requirements (https://thema-med.com/en/medical-device-registration-in-peru)
- Peru’s Growing Medical Device Market In Latin America (https://meddeviceonline.com/doc/peru-s-growing-medical-device-market-in-latin-america-0001)
- Medical device registration in Peru – a brief guide | MDRC (https://mdrc-consulting.com/peru-medical-device-registration-guide-en)
- Navigate the Review Process and Respond to Feedback
- Peru DIGEMID (https://medenvoyglobal.com/in-country-representation/peru-digemid)
- Medical Device Registration in Peru – DIGEMID Requirements (https://thema-med.com/en/medical-device-registration-in-peru)
- Tips and Best Practices for Regulatory Reviews (https://mastercontrol.com/gxp-lifeline/can-i-get-a-translation-please-tips-to-ensuring-productive-communications-with-regulatory-authorities-part-two)
- Medical device regulatory environment: Peru, Chile, Ecuador, Costa Rica, Panama, Colombia, Paraguay, El Salvador, Uruguay, Bolivia | MDRC (https://mdrc-consulting.com/latam-medical-device-regulatory-environment)
- 6 Smart Regulatory Communication Strategies for Small Biotechs | PPD (https://ppd.com/blog/winning-regulatory-trust-six-smart-strategies-for-emerging-biotechs)

Leave a Reply