Introduction
Navigating the complexities of clinical research in Peru demands a solid grasp of the General Directorate of Medicines, Supplies, and Drugs (DIGEMID) and its regulatory framework. Researchers can significantly enhance their outcomes by mastering the DIGEMID online portal, a crucial tool for submitting applications and managing compliance. Yet, with the intricacies of regulatory guidelines and the potential for common pitfalls, how can researchers ensure their submissions are both timely and compliant? This article provides a comprehensive tutorial designed to empower researchers to effectively utilize the DIGEMID portal, streamline their submission processes, and boost their chances of success in securing clinical trial approvals.
Understand the Regulatory Landscape of DIGEMID in Peru
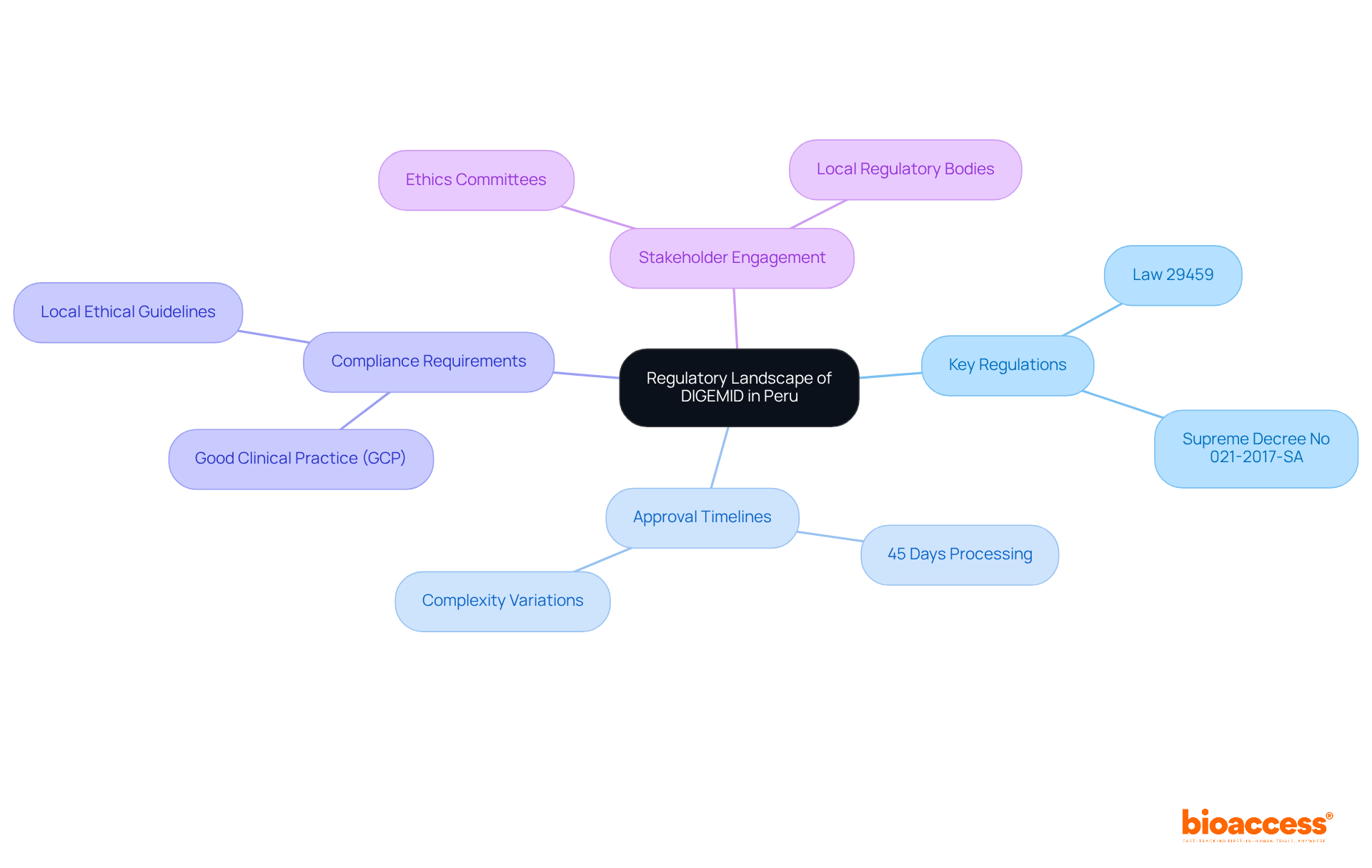
The General Directorate of Medicines, Supplies, and Drugs in Peru plays a pivotal role in overseeing research studies, ensuring the safety and effectiveness of medical products. Understanding the regulatory landscape is essential for researchers, as it involves grasping governing in Peru, including:
- Key Regulations: Law 29459 establishes the framework for and , outlining essential that must be met.
- : DIGEMID typically processes applications within 45 days. However, this timeline can vary based on the complexity of the submission, with a total of 26 research trials documented in Peru from 1999 to 2024.
- : Adhering to (GCP) and local ethical guidelines is vital for maintaining .
- : Engaging with key stakeholders, including ethics committees and local regulatory bodies, is crucial for effectively navigating the approval process.
By understanding these elements, researchers can enhance their preparedness for the submission process and better anticipate potential challenges in obtaining in Peru.
Navigate the DIGEMID Online Portal: Step-by-Step Access Guide
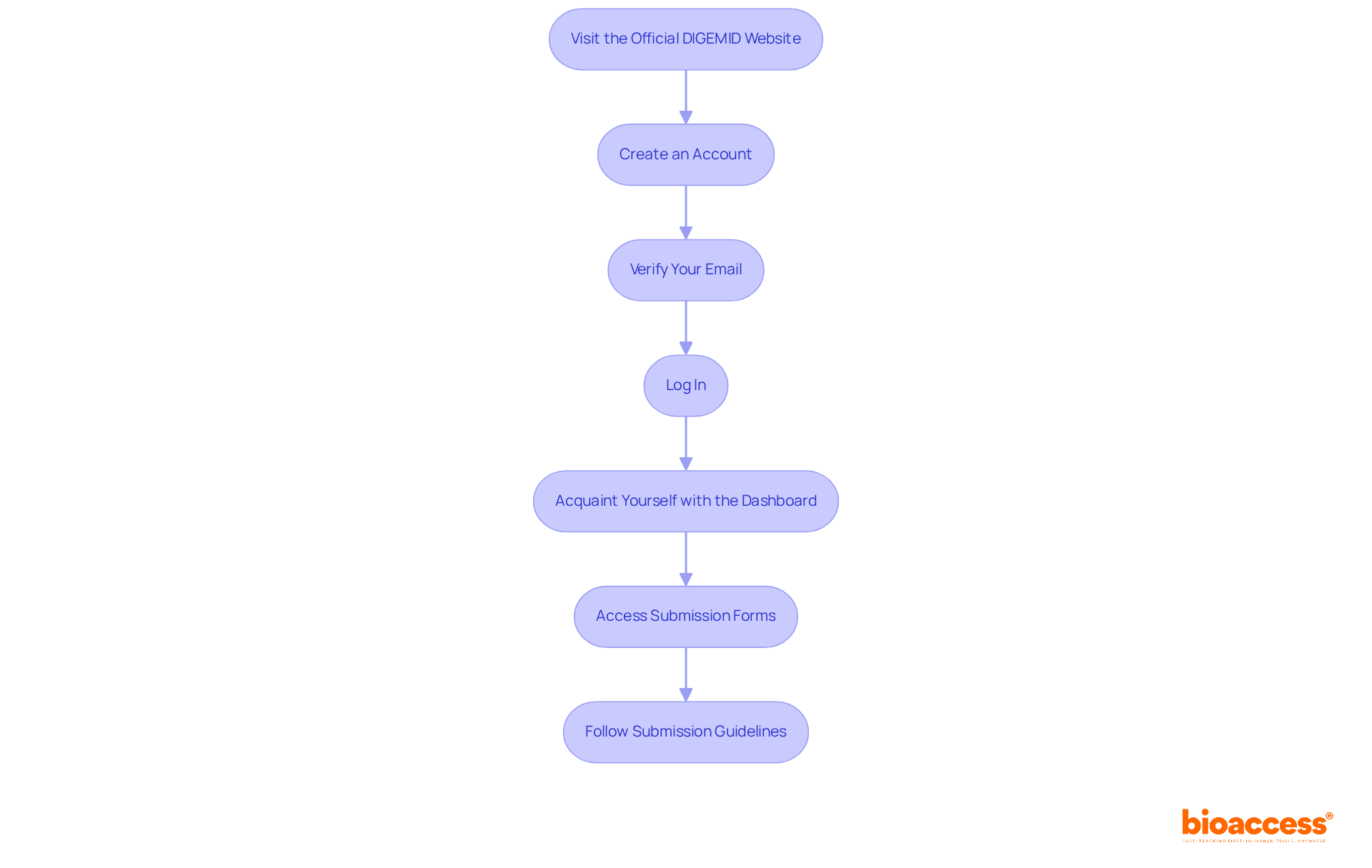
Accessing the DIGEMID online portal is a straightforward process when you follow these essential steps:
- Visit the : Navigate to DIGEMID’s official portal.
- Create an Account: Click on the ‘Register’ option to initiate account creation. Fill in the necessary details, including your name, email, and professional credentials.
- Verify Your Email: After registration, check your email for a . Click on it to activate your account.
- Log In: Return to the portal and log in using your credentials.
- Acquaint Yourself with the Dashboard: After logging in, take time to explore the dashboard to comprehend its layout and features, such as entry tracking and document uploads.
- : Navigate to the ‘Submissions’ section to locate the forms needed for your .
- Follow : Adhere to the specific guidelines provided for each form, ensuring compliance with document formats and required attachments.
By following the , researchers can efficiently navigate the portal and prepare their entries effectively. Successful user experiences suggest that knowledge of the greatly enhances the registration and application process, making it a valuable resource for .
Prepare and Submit Your Clinical Research Dossier: Compliance and Best Practices
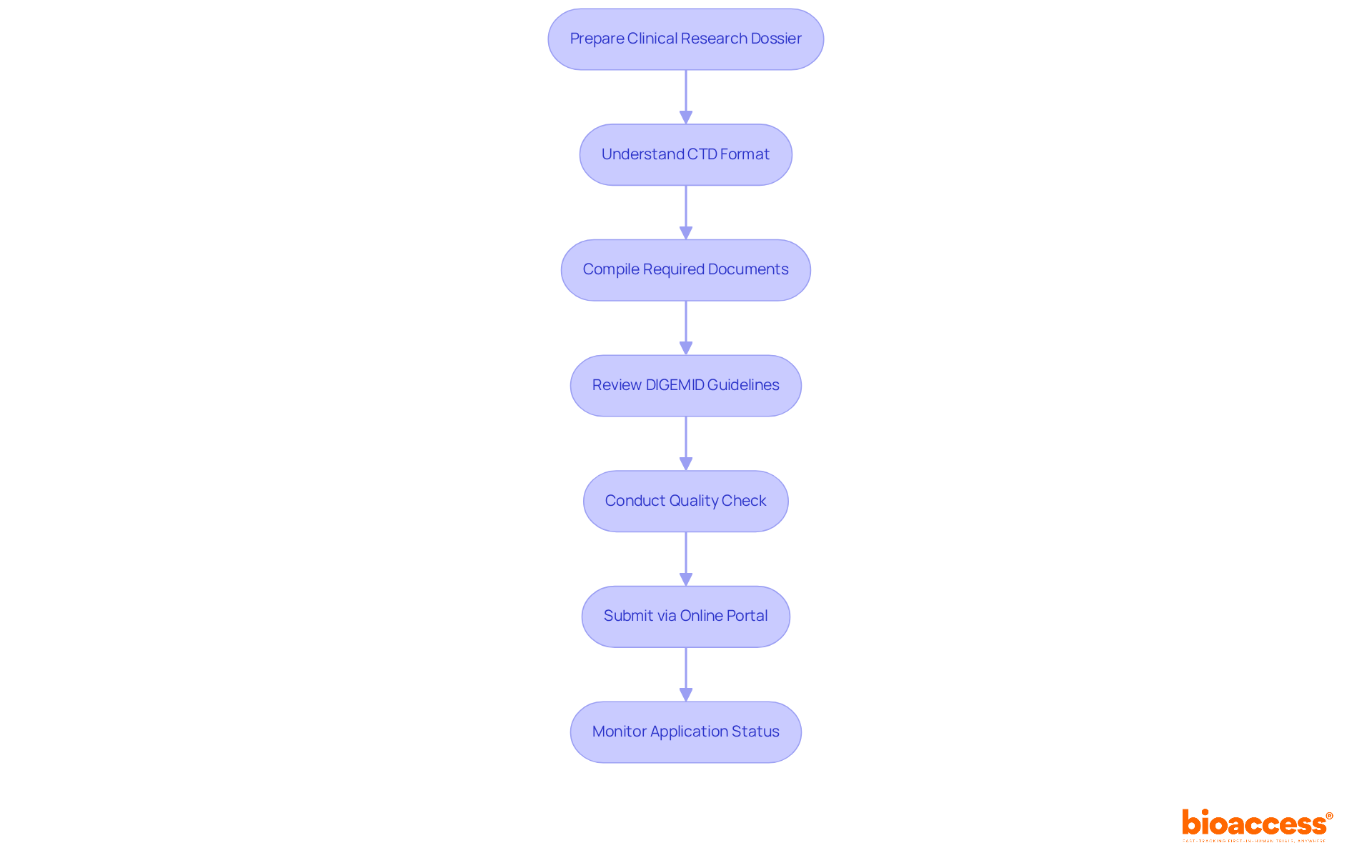
Preparing a for presentation to the regulatory body is a critical task that demands meticulous attention to detail and strict adherence to established guidelines. Understanding the is essential for success. Familiarize yourself with the , which is organized into five modules:
- Administrative Information
- Quality
- Nonclinical Study Reports
- Clinical Study Reports
- Labeling
This structure not only ensures but also facilitates efficient review by the relevant authority.
Next, compile all required documents. Gather essential items such as the study protocol, informed consent forms, investigator’s brochure, and safety reports. Ensuring these documents are complete and accurate is vital for a smooth submission process. Have you considered how missing information could delay your application?
Compliance with guidelines is non-negotiable. Review thoroughly to confirm that all papers meet the required standards. Pay close attention to formatting, language, and completeness, as non-compliance can lead to significant delays or outright rejections. Did you know that a substantial percentage of submissions are rejected due to common errors?
Conducting a quality check is the next step. Before sending, carry out a thorough assessment of the dossier to identify any inconsistencies or missing information. Engaging a colleague for a second review can provide additional insights and help catch errors that may have been overlooked.
When it comes to submission, make use of the . Ensure that all documents are uploaded in the correct format and that you receive confirmation of your upload. This step is critical for maintaining a record of your application. After submission, actively monitor the status of your application through the portal. Be prepared to respond promptly to any requests for additional information from DIGEMID, as timely communication can significantly impact the approval timeline.
By following the and these best practices, researchers can enhance the quality of their submissions and improve their chances of prompt approval. Notably, statistics indicate that many entries contain common errors, such as incorrect formatting and incomplete documentation. Therefore, a thorough understanding of the CTD format and diligent preparation are essential for success in the regulatory landscape of Peru. Moreover, leveraging Bioaccess’s extensive -including feasibility studies, site selection, compliance assessments, trial setup, import permits, project management, and reporting-can further simplify the submission process and ensure conformity to regulatory requirements.
Manage Post-Submission Expectations: Review Process and Compliance Follow-Up
After submitting your to DIGEMID, it’s essential to manage expectations regarding the effectively:
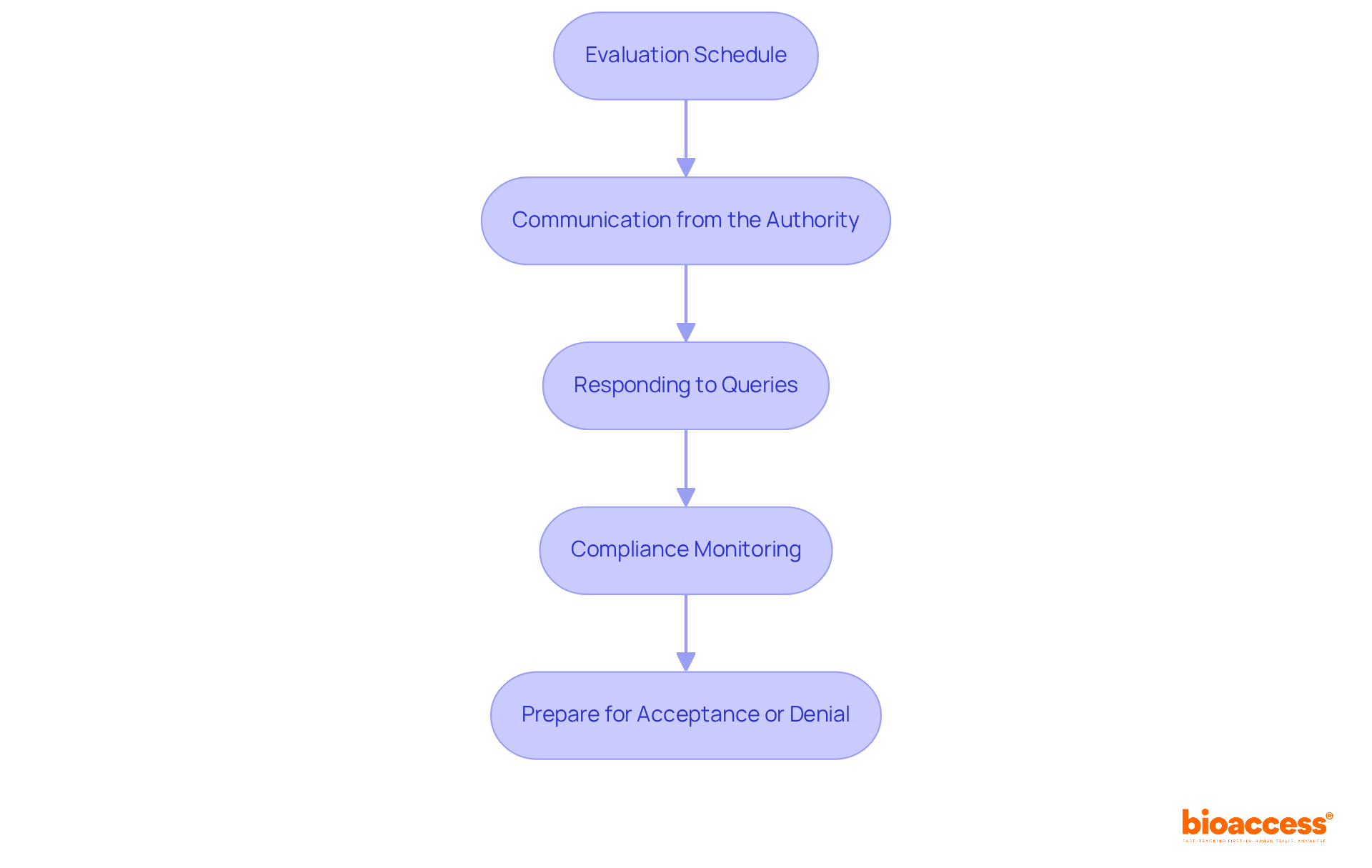
- Evaluation Schedule: The health authority typically . However, this period may fluctuate depending on the dossier’s complexity and the current volume of applications.
- Communication from the Authority: Expect to receive communications from the authority concerning your submission. This may involve , which are vital for a .
- Responding to Queries: If the agency requests further information, respond quickly and thoroughly to avoid delays. Ensure your responses are clear, well-documented, and directly address the queries raised.
- Compliance Monitoring: Throughout the assessment period, to all . Keep of any interactions with the relevant authority and ensure that all study activities conform to the approved protocols.
- Prepare for : Be aware that the assessment outcome can result in either . If approved, be ready to implement the study according to the approved protocol. In the case of rejection, carefully examine the feedback from DIGEMID to identify and rectify any issues before resubmission.
By effectively managing post-submission expectations, researchers can navigate the confidently while ensuring compliance throughout.
Conclusion
Mastering the Peru DIGEMID online portal is essential for researchers navigating the complexities of clinical trials in the country. Understanding the regulatory framework and following the outlined steps can significantly enhance the chances of successful submissions and compliance with local regulations.
Key insights emphasize the importance of:
- Comprehending the regulatory landscape
- Engaging with stakeholders
- Adhering to best practices when preparing and submitting clinical research dossiers
Moreover, managing post-submission expectations and promptly responding to queries from DIGEMID are vital components of the approval process.
Leveraging the resources available through the DIGEMID online portal not only streamlines the application process but also ensures that researchers remain compliant with the latest regulations. Embracing these practices empowers researchers to contribute effectively to the advancement of medical research in Peru, ultimately benefiting public health and fostering innovation in the field.
Frequently Asked Questions
What is the role of DIGEMID in Peru?
The General Directorate of Medicines, Supplies, and Drugs (DIGEMID) in Peru oversees research studies and ensures the safety and effectiveness of medical products.
Why is it important for researchers to understand the regulatory landscape in Peru?
Understanding the regulatory landscape is essential for researchers as it involves grasping key laws, guidelines, and processes governing clinical trials, which can enhance their preparedness for the submission process and help anticipate potential challenges.
What is the key regulation governing medical device registration and clinical trials in Peru?
Law 29459 establishes the framework for medical device registration and clinical trials in Peru, outlining essential compliance requirements that must be met.
What are the typical approval timelines for clinical trial applications submitted to DIGEMID?
DIGEMID typically processes applications within 45 days, although this timeline can vary based on the complexity of the submission.
How many research trials have been documented in Peru from 1999 to 2024?
A total of 26 research trials have been documented in Peru from 1999 to 2024.
What compliance requirements must researchers adhere to in Peru?
Researchers must adhere to Good Clinical Practice (GCP) and local ethical guidelines to maintain research integrity and ensure participant safety.
Who are the key stakeholders that researchers should engage with during the approval process?
Researchers should engage with key stakeholders, including ethics committees and local regulatory bodies, to effectively navigate the approval process.
List of Sources
- Understand the Regulatory Landscape of DIGEMID in Peru
- DIGEMID’s 45-day auto-approvals trigger safety warning (https://gabionline.net/policies-legislation/digemid-s-45-day-auto-approvals-trigger-safety-warning)
- Clinical trials by start date Peru 1999-2024| Statista (https://statista.com/statistics/1559899/peru-number-new-clinical-trial?srsltid=AfmBOopbc5Is-B86AjT1fFfHvWiC6yudtlhbkR7Cq8hRWMoSRyxlJsqj)
- Clinical trials in Peru (https://ibanet.org/clinical-trials-peru)
- Clinical trials by phase Peru 2025| Statista (https://statista.com/statistics/1559915/peru-clinical-trials-phase?srsltid=AfmBOopsiFc2ov_UKepez99_N44nsV6ESPRNi7tVuBMaX-huubh4fR-X)
- Navigate the DIGEMID Online Portal: Step-by-Step Access Guide
- TOP 25 REGULATORS QUOTES (of 88) | A-Z Quotes (https://azquotes.com/quotes/topics/regulators.html)
- (PDF) Counterfeit medicines in Peru: A retrospective review (1997-2014) (https://researchgate.net/publication/299647412_Counterfeit_medicines_in_Peru_A_retrospective_review_1997-2014)
- Using government data to understand the use and availability of medicines for hypertension and diabetes: lessons from Peru (https://tandfonline.com/doi/full/10.1186/s40545-022-00481-5)
- 4 Quotes that Underscore the Importance of Compliance (https://compliancebridge.com/4-quote-that-underscore-importance-of)
- Prepare and Submit Your Clinical Research Dossier: Compliance and Best Practices
- Dossier Format and CTD Requirements for DIGEMID Submissions – medical affairs compliance – PharmaRegulatory.in – India’s Regulatory Knowledge Hub (https://pharmaregulatory.in/dossier-format-and-ctd-requirements-for-digemid-submissions-medical-affairs-compliance)
- Common Technical Document (CTD) Structure & Module Overview (https://pubrica.com/services/medical-writing/regulatory-writing/ctd-structure-overview-modules)
- Statistical errors in manuscripts submitted to Biochemia Medica journal – Biochemia Medica (https://biochemia-medica.com/en/journal/19/3/10.11613/BM.2009.028)
- Manage Post-Submission Expectations: Review Process and Compliance Follow-Up
- Drug Development Timelines: How Health Canada’s 30-Day Regulatory Review Process Could Provide an Advantage | Altasciences (https://altasciences.com/resource-center/blog/drug-development-timelines-how-health-canadas-30-day-regulatory-review-process)
- Service Standards for Drug Submission Evaluations (Pharmaceuticals and Biologic Products) under the Food and Drug Regulations – Health Canada – Canada.ca (https://canada.ca/en/health-canada/corporate/about-health-canada/legislation-guidelines/acts-regulations/service-standards-high-volume-regulatory-authorizations/2018-2019/service-standards-drug-submission-evaluations-pharmaceuticals-biologic-products-under-food-drug-regulations.html)
- 4 Quotes that Underscore the Importance of Compliance (https://compliancebridge.com/4-quote-that-underscore-importance-of)

Leave a Reply