Introduction
In the intricate landscape of medical device regulation, COFEPRIS serves as a pivotal authority in Mexico, ensuring the safety and efficacy of health products. As the Federal Commission for Protection from Sanitary Risks, COFEPRIS governs the approval process and adapts to the evolving needs of the industry, streamlining regulations to foster innovation while maintaining stringent quality standards.
With the updates set for 2025, manufacturers are now navigating a more transparent and efficient regulatory environment, which underscores the critical importance of compliance for successful market entry. This article delves into the structure and functions of COFEPRIS, the classification of medical devices, and the essential steps for navigating the registration process. It highlights the role of clinical trials and the significance of adherence to ethical practices.
Understanding these elements is vital for stakeholders aiming to thrive in Mexico’s dynamic medical device market.
Explore COFEPRIS: Mexico’s Regulatory Authority for Medical Devices
The Federal Commission for Protection from Sanitary Risks (COFEPRIS), Mexico’s primary regulatory body for healthcare products, operates under the Ministry of Health. Its mandate includes overseeing the safety, efficacy, and quality of a broad spectrum of medical products, which encompasses compliance with . Governed by the General Health Law and various specific regulations, COFEPRIS plays a crucial role in the approval and ongoing oversight of health products.
As we move into 2025, the organization continues to evolve, refining its processes to enhance efficiency and transparency for stakeholders. Recent updates to its guidelines demonstrate a commitment to simplifying the complexities previously associated with . For example, manufacturers are now required to provide a when seeking changes to expiry dates, ensuring that products maintain their quality throughout their lifecycle. This requirement underscores the significance of rigorous in upholding product integrity, as the critical nature of these guidelines cannot be overstated; adherence to is vital for manufacturers aiming to successfully navigate the Mexican market. Non-compliance can result in substantial legal and operational challenges. A notable case study illustrating the impact of compliance is the implementation of NOM-241-SSA1-2021, which delineates . This regulation mandates that manufacturers follow , ensuring they adhere to stringent standards throughout the design, development, and distribution processes to maintain consistent quality in . Complying with these standards not only mitigates risks but also enhances the credibility of manufacturers in the eyes of regulators and consumers.
Expert opinions emphasize the importance of . As Pedro Omar Sánchez Neri noted, recent policy changes have significantly simplified prior standards, improving coherence and transparency for stakeholders. This evolution in regulatory practices facilitates smoother market entry for innovative medical devices and reinforces the necessity of maintaining high-quality standards in the industry. Furthermore, bioaccess® offers comprehensive , including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting, which are essential for adeptly navigating regulatory requirements. For instance, bioaccess® has effectively assisted clients in adapting to recent regulatory updates, ensuring that their submissions meet the new requirements efficiently. Collaboration with Pure Global enables a streamlined registration process that eases access to multiple nations, further simplifying the pathway for producers looking to expand their reach.
Overall, understanding the framework, roles, and oversight system is crucial for producers and stakeholders aiming to introduce healthcare instruments in Mexico. By staying informed about the latest updates and ensuring compliance, companies can effectively mitigate risks and seize the opportunities presented by this dynamic market.
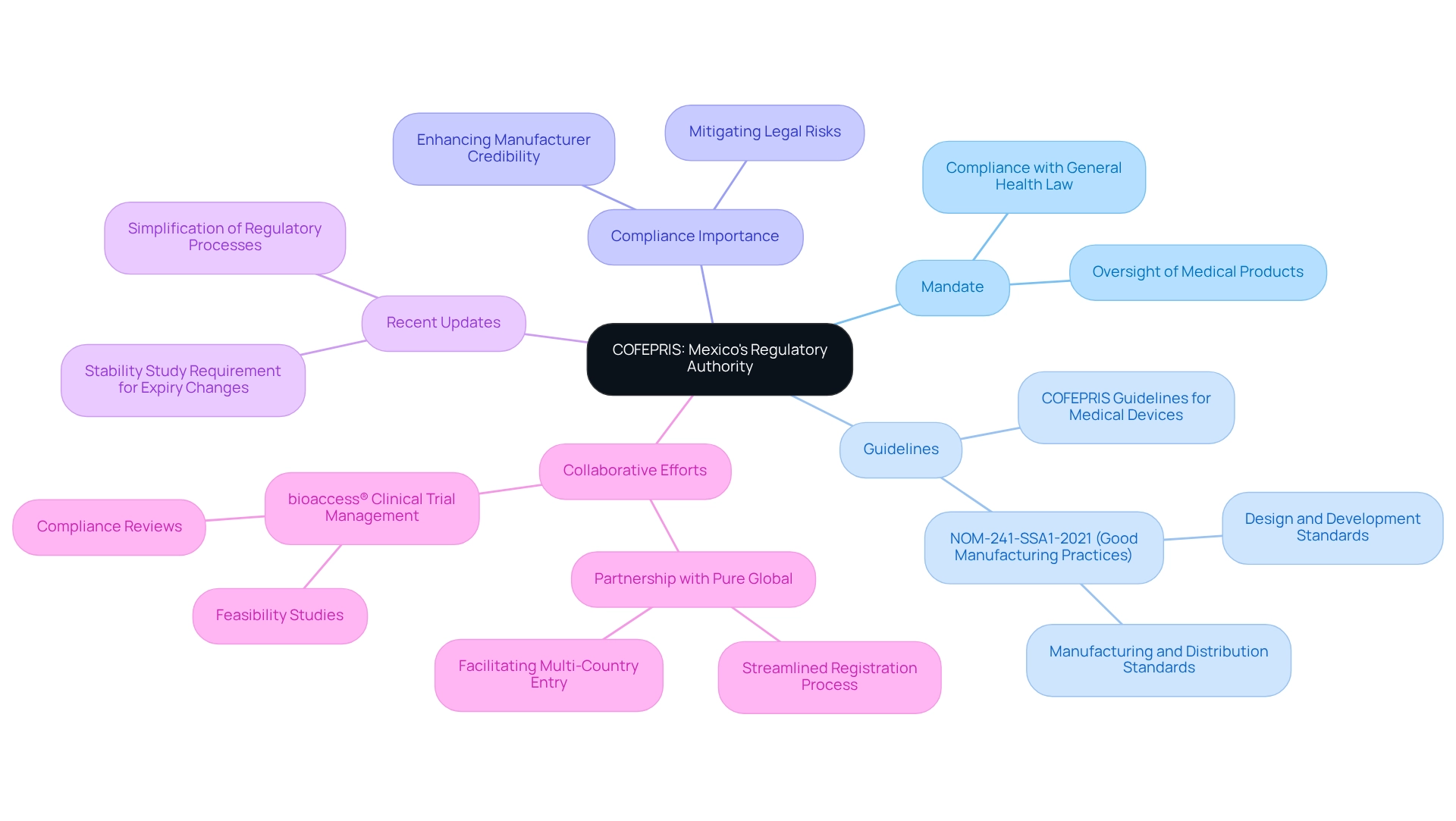
Understand Medical Device Classification Under COFEPRIS Regulations
According to the , medical instruments are categorized into three primary groups based on risk:
- Class I (low risk)
- )
- I (high risk)
typically face minimal regulatory oversight, whereas , such as implants, undergo extensive scrutiny. This classification is crucial, as it dictates the necessary documentation, clinical information, and testing procedures for each type of apparatus according to . Manufacturers must compile a detailed dossier that aligns with the assigned classification to streamline the approval process in accordance with .
Notably, the that do not require registration, providing a strategic advantage for manufacturers aiming to expedite market entry. As emphasized by the Mexican Federal Commission for Protection against Sanitary Risk (COFEPRIS), ‘[insert quote here].’
In 2025, , given that recent updates have refined the risk classification criteria, significantly impacting approval timelines. For example, the best-case scenario for registration may take as little as six months, while deficiencies in documentation can extend the process to 26 months. Understanding these classifications and associated statistics is essential for effectively navigating the compliance landscape in line with .
Furthermore, insights from the case study on strategic market entry in Mexico underscore the importance of comprehensive planning and adherence to local regulations for successful market access. Additionally, the emphasis on sustainability and social responsibility within the Medtech sector necessitates that manufacturers align their practices with evolving compliance expectations.
With bioaccess® leading the way in facilitating across Latin America—including expertise in Early-Feasibility Studies, , Pilot Studies, Pivotal Studies, and Post-Market Follow-Up Studies—companies can navigate these complexities more effectively. , recognized as a Level 4 health authority by PAHO/WHO, further highlights the significance of regulatory compliance in the region.
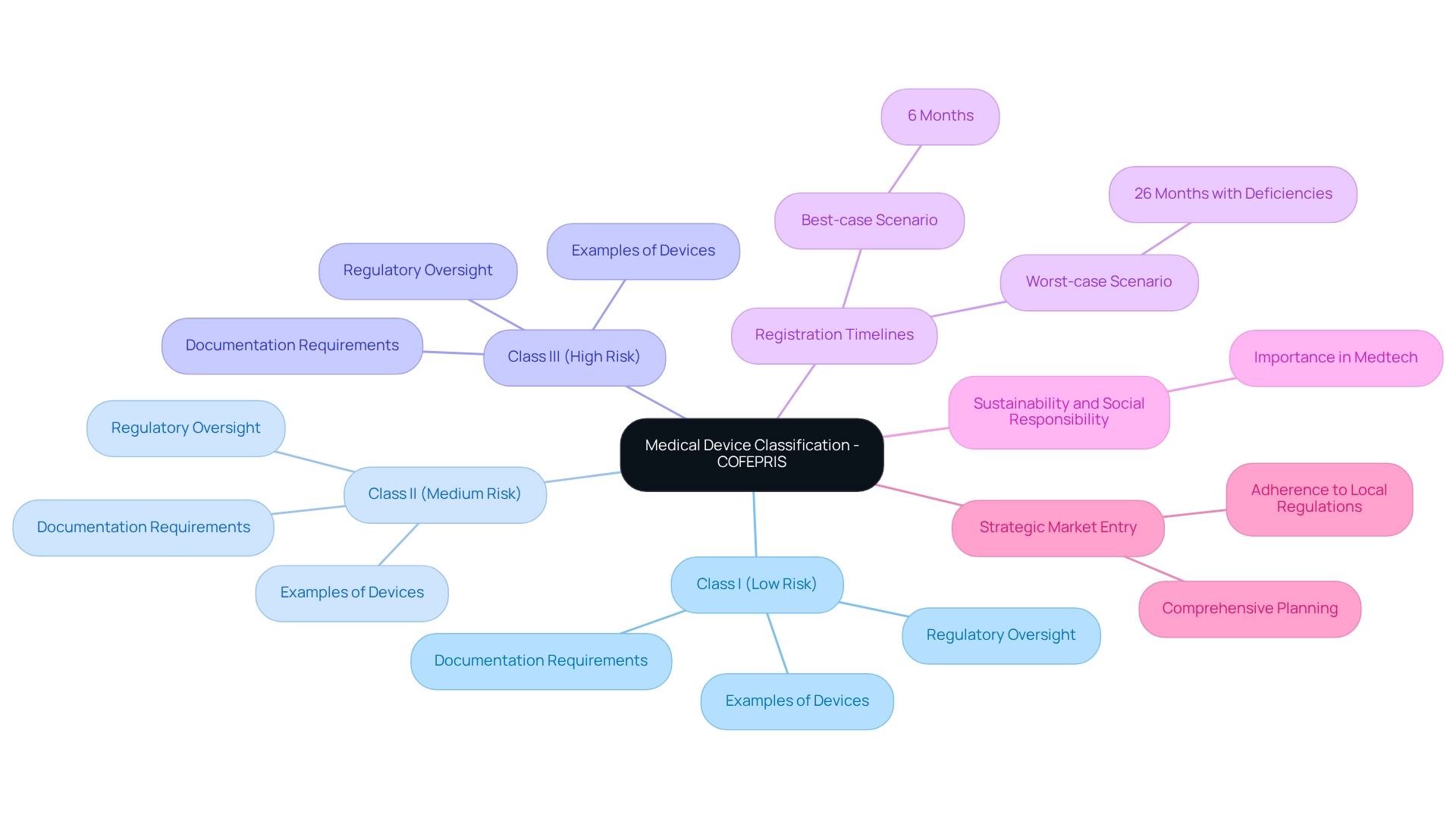
Navigate the COFEPRIS Registration Process for Medical Devices
Navigating the registration process for medical products involves several essential steps:
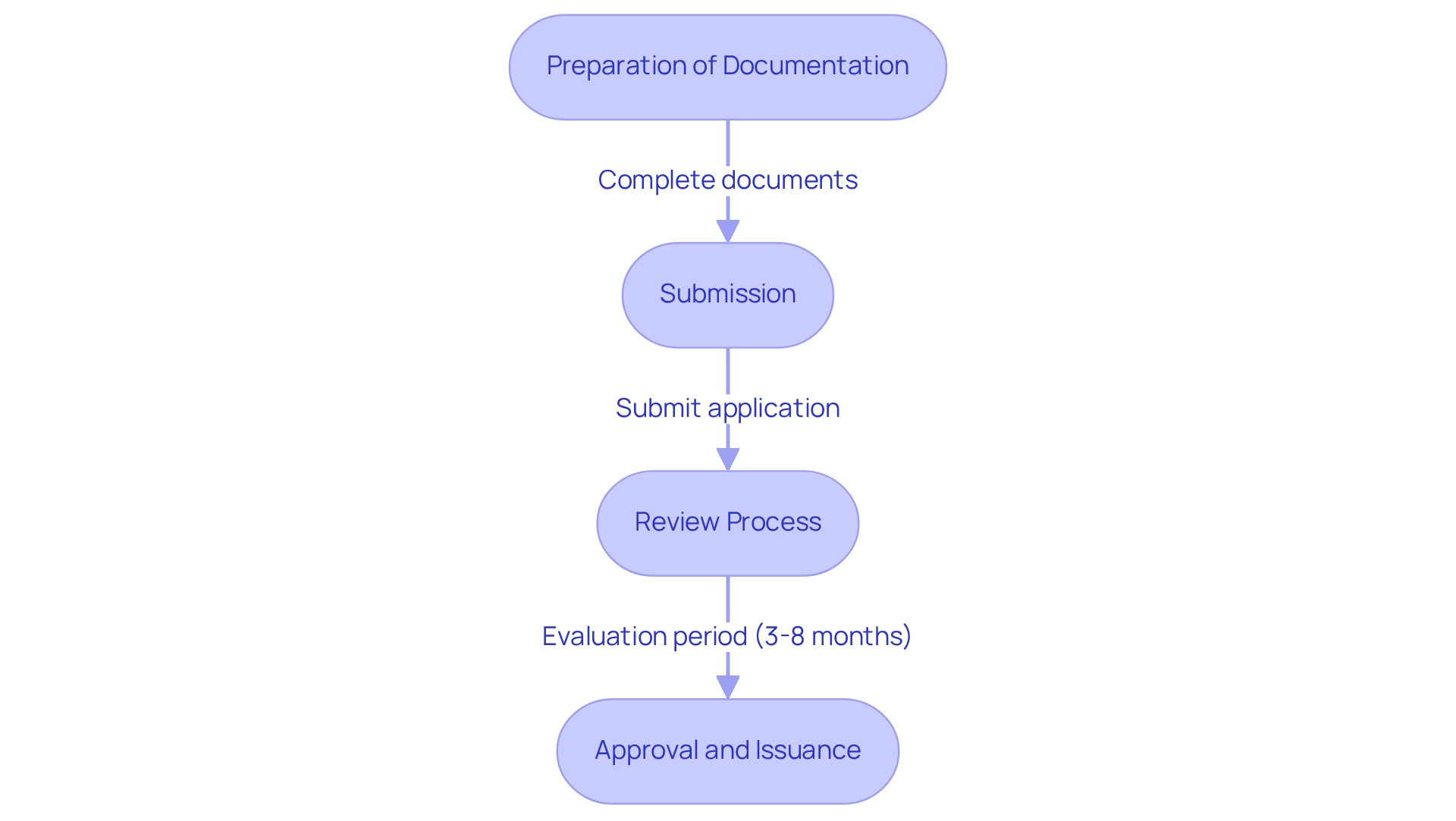
- Preparation of Documentation: Compile all necessary documents, including technical specifications, , and . This foundational step is vital for a smooth application process. Leveraging bioaccess®’s expertise in , including feasibility studies and site selection, can streamline this phase, ensuring compliance with local requirements.
- Submission: Submit the through the relevant online platform, ensuring that all documents are complete and accurate to avoid delays. Bioaccess® can assist in reviewing and providing feedback on study documents to comply with country requirements, enhancing the likelihood of a successful submission.
- Review Process: will perform a comprehensive evaluation of the application, which usually requires between 3 to 8 months, based on the categorization of the product. Companies must be proactive in managing this timeline to ensure timely market access. Engaging with experienced partners like bioaccess® can provide insights into navigating this timeline effectively.
- Approval and Issuance: Following a successful evaluation, the will grant a , allowing the product to be sold in Mexico. Grasping this procedure is essential for adhering to the and for promoting the swift progress of healthcare products in the market. Companies should also be aware that , necessitating renewal documentation to be submitted 150 days prior to expiration to maintain compliance and market access. Proactive management of renewals is essential for maintaining market access.
Review Clinical Trials and Testing Requirements for COFEPRIS Registration
According to , are mandated to validate the safety and efficacy of Class II and III healthcare instruments. The key requirements include:
- Study Design: Trials must adhere to international standards, particularly , ensuring ethical and scientific quality.
- Ethics Approval: Prior to initiating trials, obtaining approval from an is essential to safeguard participant welfare.
- Data Submission: A comprehensive registration dossier must be submitted, encompassing detailed methodology, results, and statistical analyses from .
- : Following approval, manufacturers are obligated to conduct to maintain compliance.
Understanding the is crucial for manufacturers to effectively navigate the registration process and ensure their medical devices meet the necessary legal standards. Moreover, the anticipated benefits of research in local communities must be reasonably assured before proceeding, reinforcing the ethical considerations inherent in . This ensures that the research not only complies with standards but also benefits the communities involved.
Recent developments, such as the launch of the Digital Platform for Research and (Digipris) by COFEPRIS in February 2023, facilitate the online submission and management of , streamlining the approval process. Furthermore, it is important to note that there is currently no information available regarding waiver requirements for consent, which is a significant aspect of the regulatory landscape. The head of the health institution is tasked with establishing strict supervision for research involving greater than minimal risk, particularly to protect vulnerable populations, such as minors. This supervision is critical in ensuring that ethical standards are upheld throughout the research process.
In this context, bioaccess™ plays a pivotal role as , collaborating with Caribbean Health Group to position Barranquilla as a premier destination for . This partnership, supported by Colombia’s Minister of Health, aims to enhance the , ensuring that ethical standards and community benefits are prioritized. A case study on amendments to research protocols underscores the importance of timely submissions to maintain ethical standards and participant safety throughout the research lifecycle, highlighting that delays can compromise both ethics and participant welfare.
Conclusion
The regulatory landscape for medical devices in Mexico is undergoing significant transformation, with COFEPRIS at the forefront of these changes. Understanding COFEPRIS’s structure and functions, as well as the classification of medical devices, empowers stakeholders to navigate the registration process effectively. Compliance with COFEPRIS regulations transcends mere legal obligation; it is essential for ensuring the safety, efficacy, and quality of medical products in the market.
The classification of medical devices into risk categories directly influences the approval process, underscoring the necessity for thorough documentation and adherence to established standards. Given that the timeline for registration can vary dramatically based on compliance, manufacturers must prioritize preparation and collaborate with experienced partners to facilitate smoother navigation through the regulatory requirements.
Moreover, the emphasis on clinical trials and ethical practices highlights a commitment to patient safety and product integrity. Recent updates to COFEPRIS regulations reflect an ongoing effort to enhance transparency and streamline processes, ultimately benefiting both manufacturers and consumers.
In conclusion, staying informed about COFEPRIS’s evolving regulatory framework is crucial for stakeholders aiming to succeed in Mexico’s dynamic medical device market. By embracing compliance, understanding risk classifications, and prioritizing ethical standards, manufacturers can not only mitigate risks but also seize the opportunities that this promising market offers. This proactive approach will ensure a sustainable and successful future in the healthcare landscape.
Frequently Asked Questions
What is COFEPRIS and what is its primary role?
COFEPRIS, the Federal Commission for Protection from Sanitary Risks, is Mexico’s primary regulatory body for healthcare products operating under the Ministry of Health. Its main role is to oversee the safety, efficacy, and quality of various medical products, ensuring compliance with its guidelines for medical devices.
What regulations govern COFEPRIS?
COFEPRIS operates under the General Health Law and various specific regulations that guide its processes and ensure the approval and ongoing oversight of health products.
How is COFEPRIS evolving as we approach 2025?
As we move into 2025, COFEPRIS is refining its processes to enhance efficiency and transparency for stakeholders, including simplifying the complexities of regulatory compliance through recent updates to its guidelines.
What is the significance of the stability or aging study report in COFEPRIS guidelines?
Manufacturers are now required to provide a stability or aging study report when seeking changes to expiry dates, which ensures that medical products maintain their quality throughout their lifecycle and underscores the importance of rigorous compliance with COFEPRIS guidelines.
What are the consequences of non-compliance with COFEPRIS guidelines?
Non-compliance with COFEPRIS guidelines can lead to substantial legal and operational challenges for manufacturers, impacting their ability to successfully navigate the Mexican market.
What is NOM-241-SSA1-2021 and its relevance to COFEPRIS?
NOM-241-SSA1-2021 outlines Good Manufacturing Practices (GMP) for healthcare products and mandates that manufacturers adhere to COFEPRIS guidelines, ensuring consistent quality in medical device production.
How do recent policy changes affect stakeholders?
Recent policy changes have simplified prior standards, improving coherence and transparency, which facilitates smoother market entry for innovative medical devices and reinforces the necessity of high-quality standards.
What services does bioaccess® provide in relation to COFEPRIS?
Bioaccess® offers comprehensive clinical trial management services, including compliance reviews, trial setup, and project management, which are essential for navigating regulatory requirements and adapting to recent updates.
Why is it important for producers to understand COFEPRIS’s framework?
Understanding COFEPRIS’s framework, roles, and oversight system is crucial for producers and stakeholders aiming to introduce healthcare instruments in Mexico, as it helps mitigate risks and seize opportunities in the market.
List of Sources
- Explore COFEPRIS: Mexico’s Regulatory Authority for Medical Devices
- regdesk.co (https://regdesk.co/cofepris-guidance-on-changes-to-sanitary-registry-introduction)
- COFEPRIS Mexico Medical Device Regulations | Pure Global (https://pureglobal.com/markets/mexico/cofepris-medical-device-regulations)
- Mexico’s COFEPRIS Publishes the Final Version of the GMP Standard (https://emergobyul.com/news/mexicos-cofepris-publishes-final-version-gmp-standard)
- Understand Medical Device Classification Under COFEPRIS Regulations
- Navigating the regulatory landscape of fast-growing markets | Mexico (https://congenius.ch/regulatory-landscape-mexico)
- regdesk.co (https://regdesk.co/resource-library/mexico)
- Navigate the COFEPRIS Registration Process for Medical Devices
- slideshare.net (https://slideshare.net/slideshow/cofepris-mexico-medical-device-registration/272076933)
- COFEPRIS medical device and IVD registration and approval in Mexico (https://emergobyul.com/services/cofepris-medical-device-and-ivd-registration-and-approval-mexico)
- COFEPRIS Mexico Medical Device Regulations | Pure Global (https://pureglobal.com/markets/mexico/cofepris-medical-device-regulations)
- Review Clinical Trials and Testing Requirements for COFEPRIS Registration
- clinregs.niaid.nih.gov (https://clinregs.niaid.nih.gov/country/canada/mexico)
- Clinical Research Regulation For Mexico | ClinRegs (https://clinregs.niaid.nih.gov/country/mexico)
- Mexico Clinical Trials Market Size & Trend Analysis 2035 (https://expertmarketresearch.com/reports/mexico-clinical-trials-market)

Leave a Reply