Introduction
Virtual clinical trials are revolutionizing the landscape of medical research. They provide unprecedented access to diverse patient populations and significantly enhance the efficiency of study processes. Research directors stand to gain a multitude of advantages from this innovative approach, including:
- Improved recruitment rates
- Reduced costs
- The ability to adapt study designs based on real-time feedback
However, as the shift towards virtual trials accelerates, research leaders must navigate the challenges and ethical considerations that accompany this transformation. How can they effectively address these issues while maximizing the potential benefits of this emerging paradigm?
bioaccess: Accelerating Virtual Clinical Trials with Global Agility
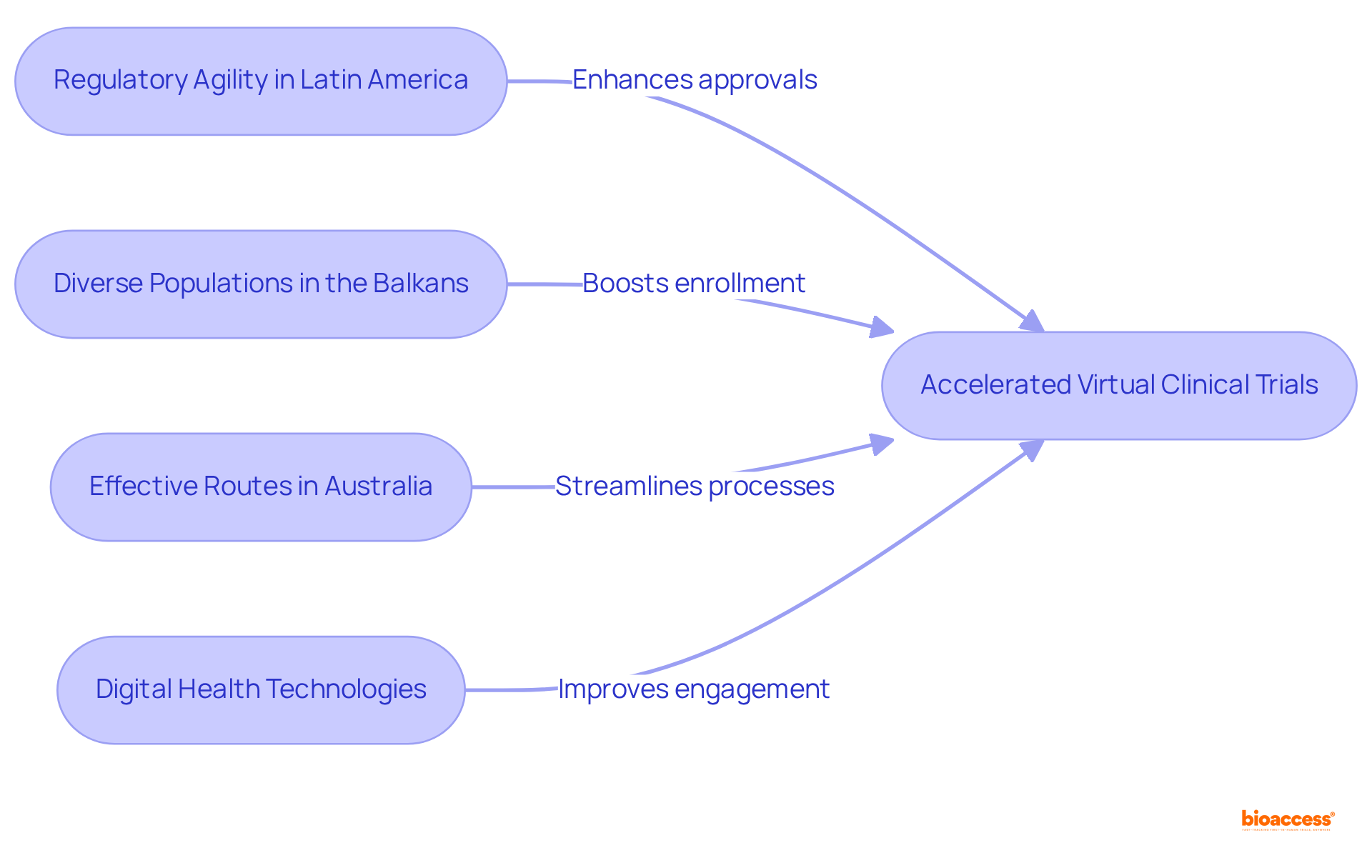
bioaccess® leverages the regulatory agility of Latin America, the diverse populations of the Balkans, and the effective routes in Australia to enhance . This strategic amalgamation facilitates within an impressive timeframe of 4-6 weeks and . Such agility positions bioaccess® as an indispensable partner for research directors seeking to , ensuring timely access to . Recent advancements in , including the , further underscore the significance of this approach, as the industry increasingly embraces innovative methods to and elevate user engagement.
Enhanced Patient Recruitment: The Key Advantage of Virtual Clinical Trials
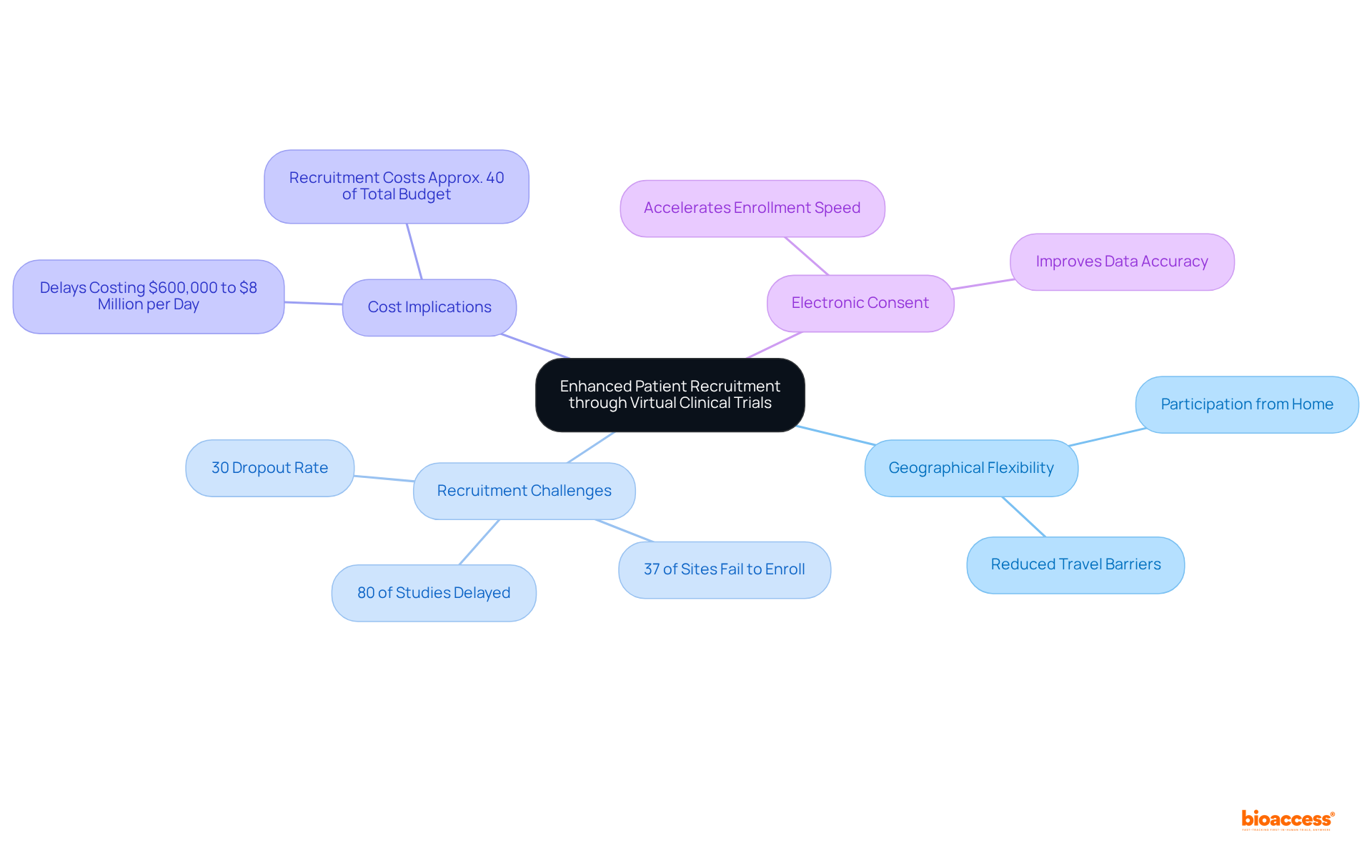
significantly by eliminating , allowing individuals to participate from the comfort of their homes. This flexibility not only boosts enrollment rates but also aligns seamlessly with the schedules and lifestyles of potential participants.
Research indicates that approximately 80% of studies are delayed or terminated due to , underscoring the urgent need for . Digital platforms are crucial in , ensuring a more representative sample. Notably, 37% of research sites fail to enroll sufficient participants, which highlights the importance of these platforms in overcoming recruitment hurdles.
Furthermore, delays can cost sponsors between $600,000 and $8 million for each day a study postpones a product’s development and launch, reinforcing the necessity for . The implementation of has been shown to , streamlining the recruitment process.
Consequently, not only enhance accessibility but also foster greater inclusivity in medical research, ultimately leading to more successful outcomes.
Cost-Effectiveness: How Virtual Clinical Trials Reduce Research Expenses
notably by minimizing the need for physical locations, travel costs, and associated overheads. By leveraging technology for and data collection, organizations can significantly , leading to .
For instance, can , particularly regarding expenses tied to participant travel and the personnel required for on-site observation. The typical budget reductions for can range from $4,300 to $600,000, depending on the study’s characteristics.
The is particularly appealing for . Furthermore, with 50% of individuals more likely to participate in studies that offer home care alternatives, , thereby enhancing the financial viability of these innovative approaches.
 — the larger the slice, the greater the savings from that category. Each slice of the pie shows how much each cost factor contributes to the total savings in virtual clinical trials — the larger the slice, the greater the savings from that category.](https://images.tely.ai/telyai/vsfswxcc-each-slice-of-the-pie-shows-how-much-each-cost-factor-contributes-to-the-total-savings-in-virtual-clinical-trials-the-larger-the-slice-the-greater-the-savings-from-that-category.webp)
Flexibility in Study Design: Adapting to Patient Needs with Virtual Trials
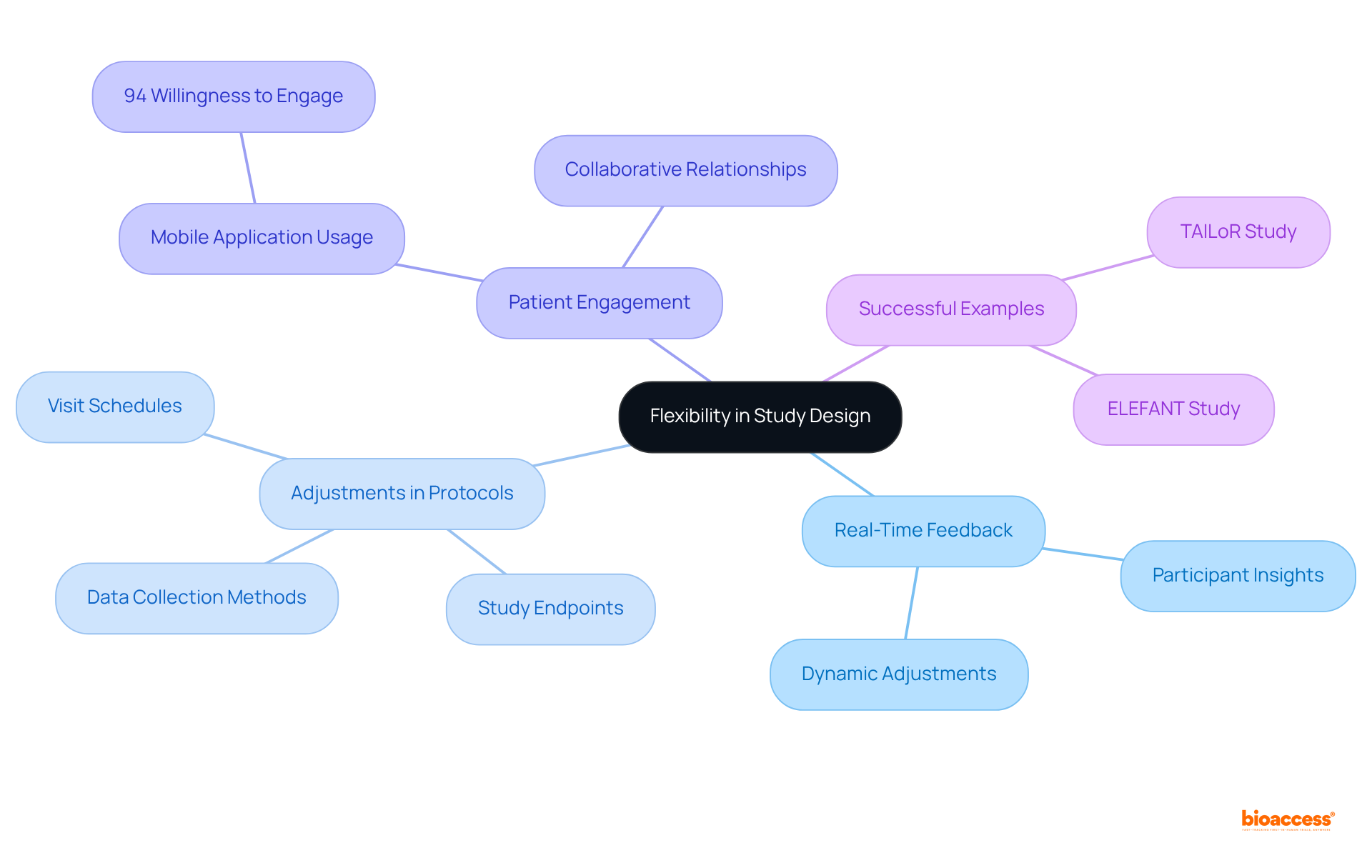
present exceptional , enabling researchers to modify protocols based on from participants. This flexibility encompasses , data collection methods, and even study endpoints, all tailored to meet and needs. Such responsiveness not only boosts but also significantly enhances retention rates.
Notably, a Medidata survey revealed that 94% of respondents expressed a willingness to engage with . As we approach 2025, the ability to dynamically modify is becoming increasingly vital, particularly in virtual environments where patient-focused strategies are essential.
Successful examples of , such as the ELEFANT and TAILoR studies, illustrate how integrating patient insights can yield more effective and efficient research outcomes, ultimately fostering a more between researchers and participants.
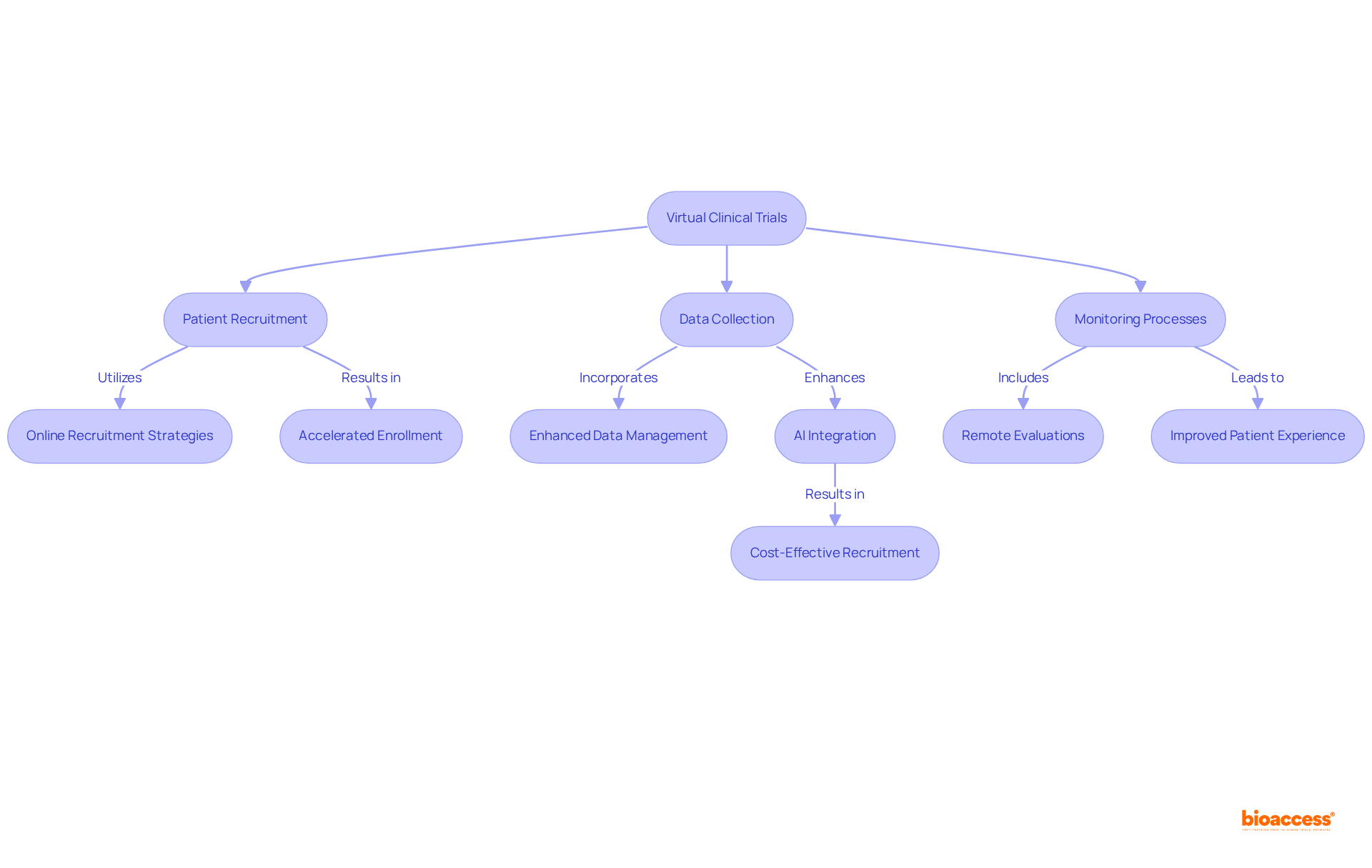
Technology Integration: Streamlining Data Collection in Virtual Trials
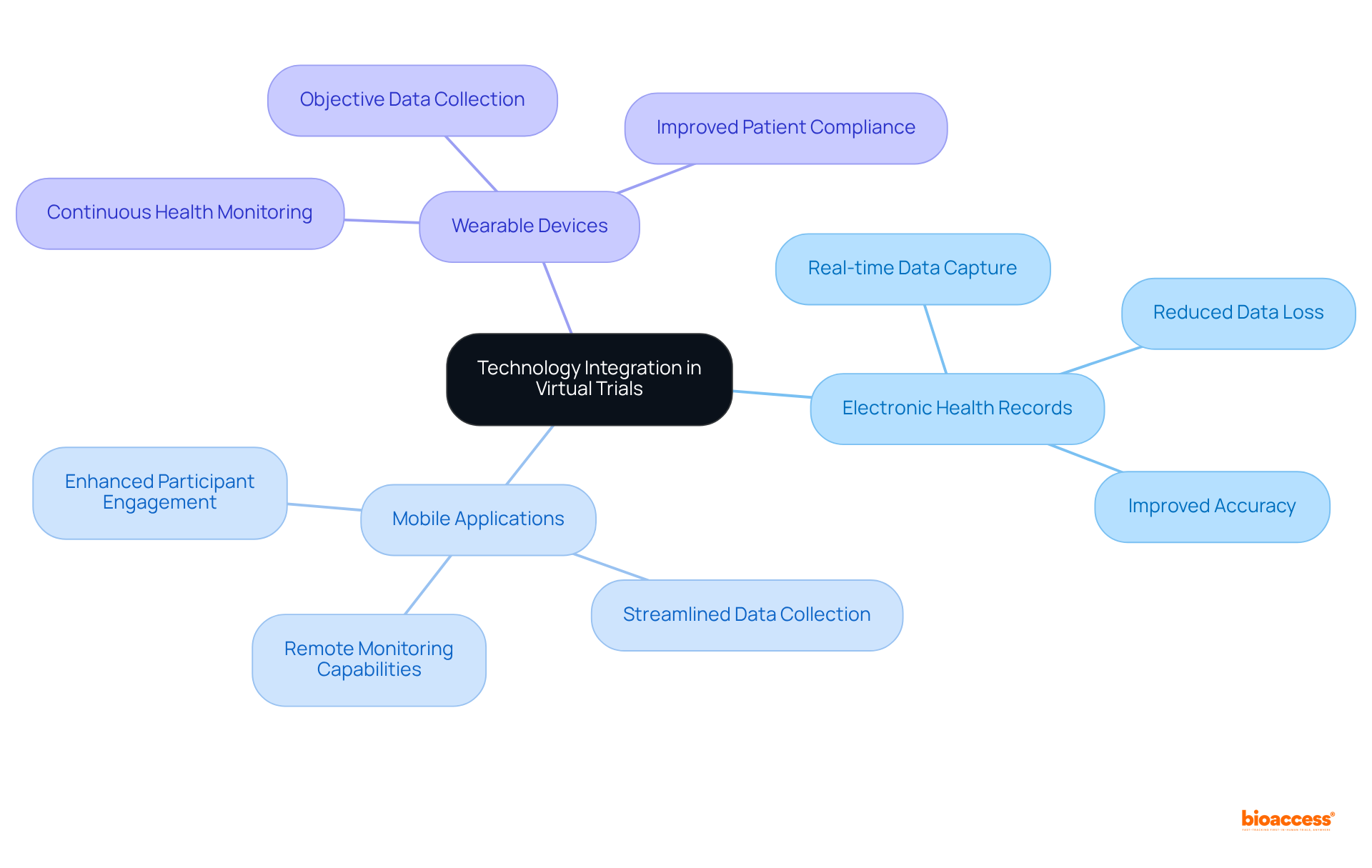
The integration of technology in significantly enhances through , , and . These innovative tools facilitate and monitoring, thereby reducing the risk of and ensuring greater accuracy. Such advancements in technology not only enable more but also play a crucial role in improving .
Accelerated Timelines: Speeding Up Research with Virtual Clinical Trials
significantly enhance research timelines by , , and monitoring processes. By employing remote evaluations and advanced digital tools for data management, these studies can progress at an accelerated pace, facilitating quicker analysis and reporting of results.
For instance, online recruitment strategies yield 4.17 times more individuals per day compared to traditional methods, with the median cost per enrollee notably lower at $72 versus $199 for offline recruitment. This efficiency proves vital for expediting the introduction of to the market.
Furthermore, in 2022/23, there were 952,789 individuals involved in in England, an increase of over 220,000 compared to pre-pandemic figures, underscoring the growing importance of in enhancing participation. The integration of AI and machine learning can , by an average of six months per asset.
Additionally, setbacks in medical studies can cost firms over $35,000 daily for each study, highlighting the of inefficiencies in traditional approaches. As continue to evolve, they are poised to transform the landscape of medical research, ensuring that reach patients more swiftly.
However, it is essential to acknowledge that participation in carries potential risks, which must be thoroughly assessed. As Samruddhi Yardi noted, ‘ are essential for progressing medical understanding and enhancing healthcare by evaluating the safety and effectiveness of new treatments before they are widely available.
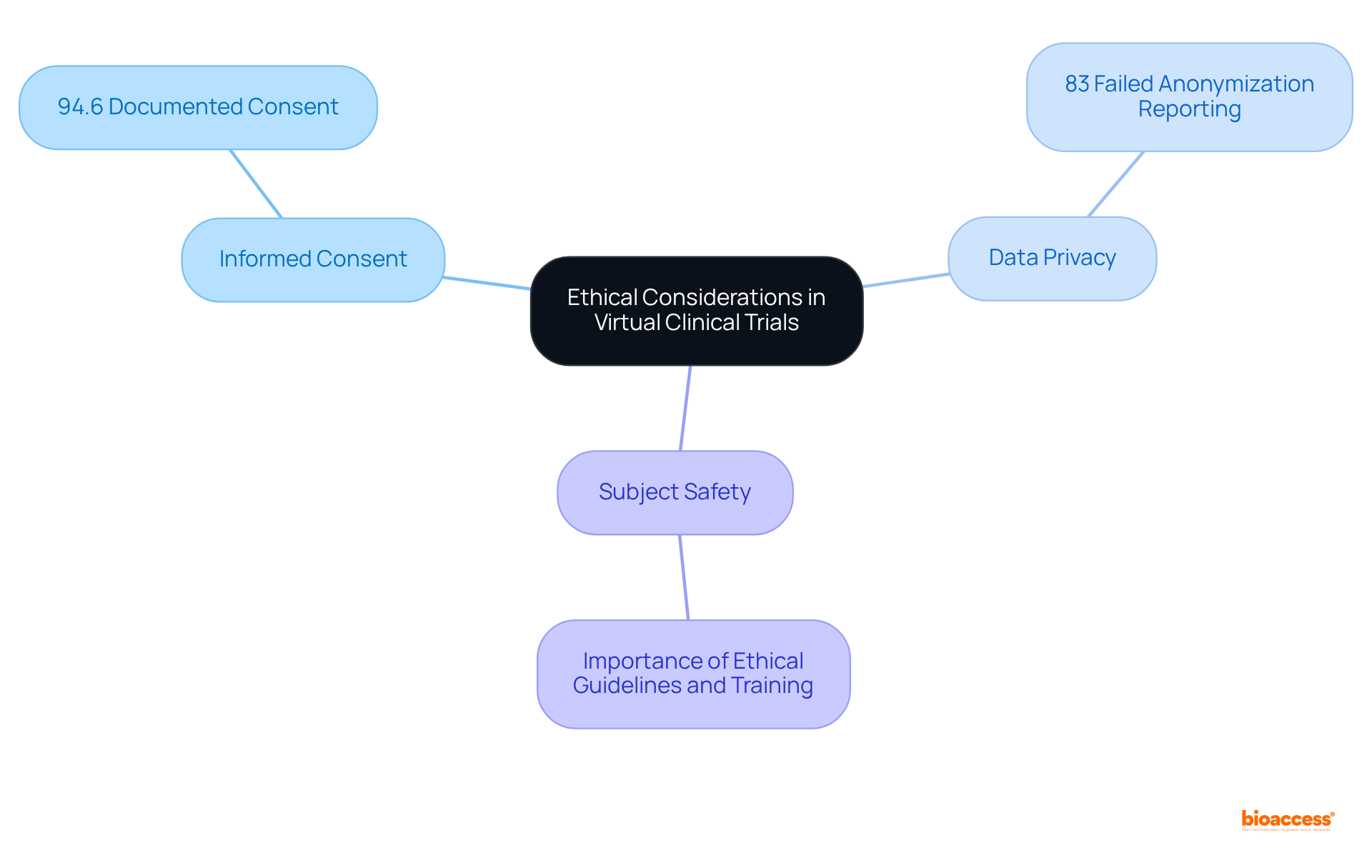
Ethical Considerations: Ensuring Compliance in Virtual Clinical Trials
Carrying out virtual clinical trials necessitates a stringent approach to , particularly regarding , , and . Research directors must ensure that all interactions during virtual clinical trials adhere to , thereby guaranteeing that individuals are fully informed about the study’s nature and their rights.
For instance, as of 2023, was documented in 94.6% of research studies, highlighting the . Furthermore, ; studies indicate that 83% of studies failed to adequately report on the anonymization of data, potentially undermining trust among subjects.
To ensure compliance, directors should and utilize , which have gained popularity for their ability to streamline the consent process while ensuring user comprehension.
By prioritizing these , research leaders can foster trust and integrity in the research process, ultimately enhancing participation and study success.
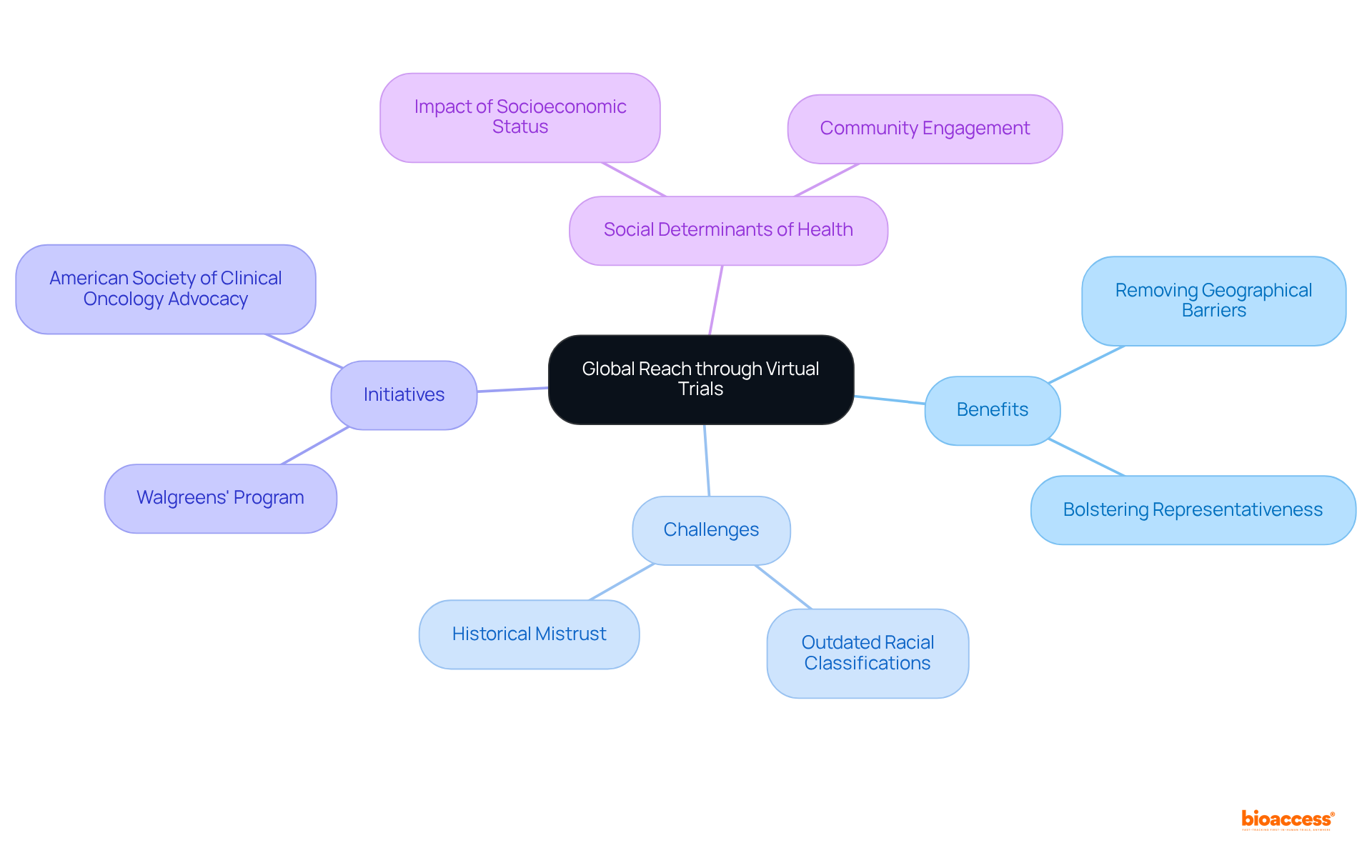
Global Reach: Accessing Diverse Patient Populations through Virtual Trials
significantly enhance researchers’ access to by removing geographical barriers. This inclusivity not only facilitates the from varied backgrounds but also bolsters the representativeness of study outcomes.
However, challenges such as can hinder effective recruitment and representation. By utilizing digital platforms, researchers can engage underrepresented groups effectively, ensuring that findings are relevant and applicable to a broader audience.
For instance, initiatives like Walgreens’ research program aim to improve racial and ethnic diversity by incorporating both in-person and virtual participation methods. Additionally, the American Society of Clinical Oncology advocates for , underscoring the necessity for diverse representation.
It is also essential to consider (SDOH), which significantly influence participation in studies, especially among marginalized communities. As we approach 2025, addressing —including the legacy of distrust stemming from events such as the Tuskegee Syphilis Experiment—will be crucial for ensuring that medical research reflects the demographics of the wider community.
Engaging with experts like Arthur L. Caplan, who stresses the , can provide valuable insights into overcoming these challenges. Ultimately, the focus on through will be vital for promoting fairness in medical research.
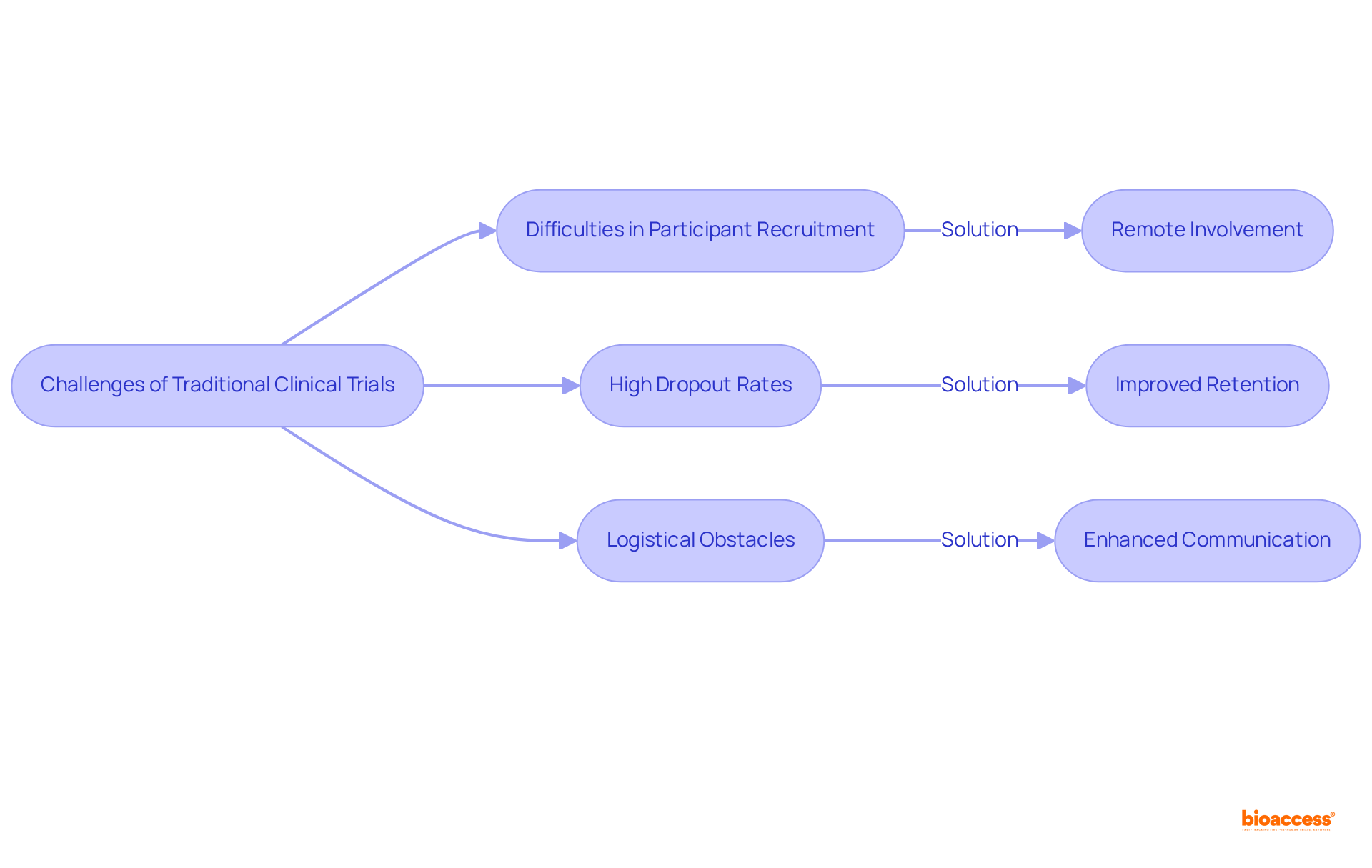
Overcoming Challenges: How Virtual Trials Address Traditional Clinical Trial Limitations
effectively address various constraints related to conventional studies, such as difficulties in , , and logistical obstacles. For instance, only 31% of 114 studies in the UK achieved enrollment targets, underscoring the challenges in patient recruitment.
By facilitating remote involvement, these significantly reduce the challenges associated with travel and time commitments, leading to . Research indicates that 90% of follow-up participants expressed a willingness to engage again if the study were extended, with virtual experiments achieving .
Furthermore, the integration of , enhances communication and enables real-time tracking of participant involvement in , thereby boosting the overall efficiency of the study.
This innovative approach effectively addresses frequent , such as lengthy onboarding processes and high attrition rates, with than traditional methods, ultimately resulting in more robust and .
Future Trends: Innovations Shaping the Landscape of Virtual Clinical Trials
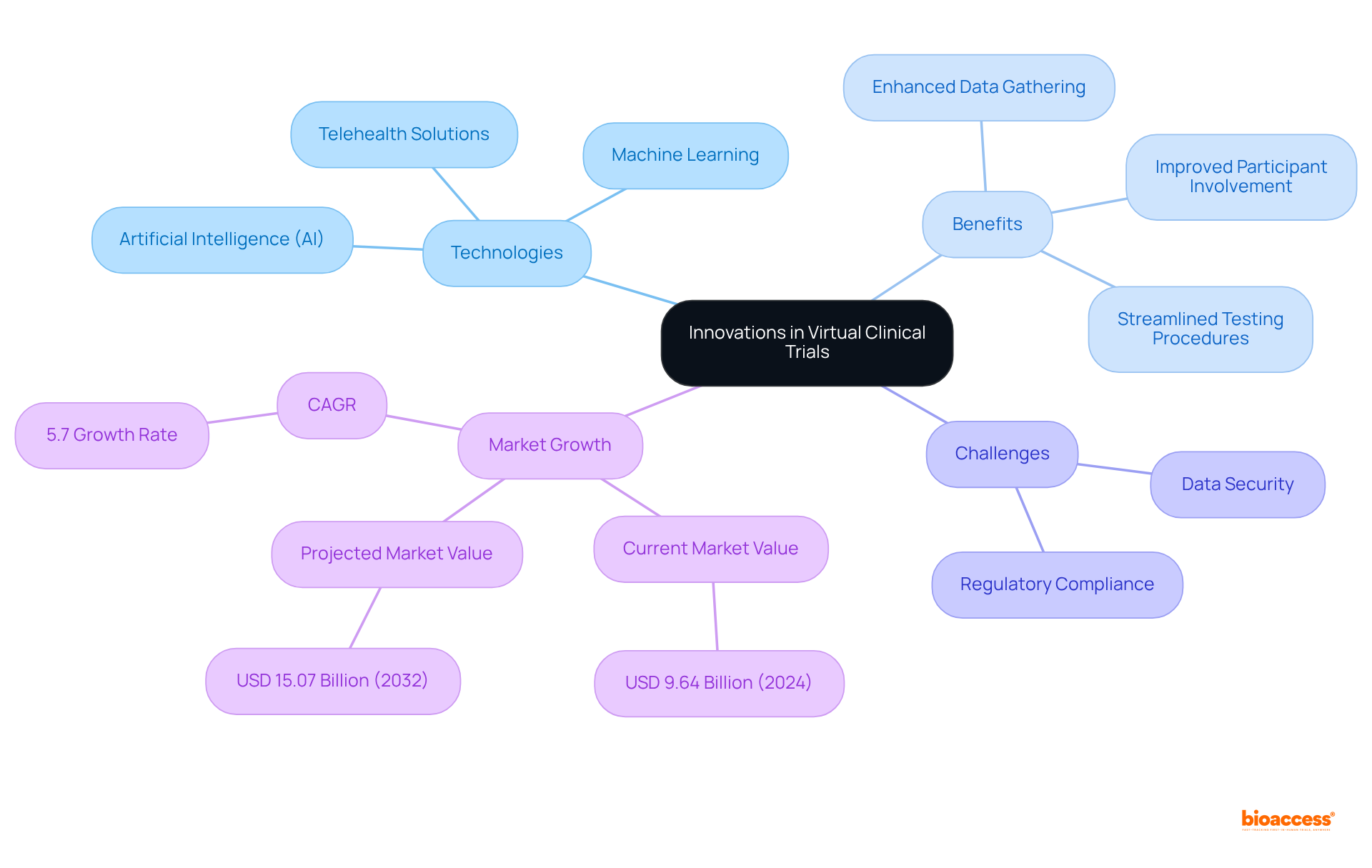
The future of virtual is advancing rapidly, driven by innovations in technology such as , machine learning, and telehealth solutions. These advancements are enhancing , , and streamlining . For example, AI and machine learning are progressively employed to examine extensive datasets, allowing for more accurate selection of individuals and real-time tracking of results. Furthermore, telehealth solutions enable remote consultations and evaluations, allowing participants to take part from the comfort of their homes, thus enhancing .
Significantly, fewer than 5% of adult cancer patients enroll in research studies, emphasizing the essential function of remote involvement in tackling recruitment issues. Statistics suggest that the virtual medical trials market was valued at USD 9.64 billion in 2024 and is expected to expand at a compound annual growth rate (CAGR) of 5.7%, reaching USD 15.07 billion by 2032. This growth is driven by the increasing demand for and the integration of advanced technologies in . However, challenges such as and regulatory compliance remain significant concerns that must be addressed.
As research directors navigate this evolving landscape, embracing these innovations while being mindful of potential obstacles will be crucial for maximizing the effectiveness and efficiency of clinical studies. Ultimately, this will lead to , making collaboration essential for overcoming the challenges faced in clinical research.
Conclusion
Virtual clinical trials are revolutionizing the medical research landscape, presenting research directors with a multitude of benefits that enhance efficiency, inclusivity, and adaptability. By harnessing technology and innovative methodologies, these trials not only bolster patient recruitment and retention but also markedly reduce costs and streamline processes. This transition towards virtual studies signifies a critical juncture in clinical research, fostering a more responsive and patient-centered approach.
The key advantages emphasized throughout the article include:
- Improved recruitment capabilities, which dismantle geographical barriers and promote greater diversity among participant demographics.
- The cost-effectiveness of virtual trials, enabling organizations to allocate resources more judiciously while achieving elevated retention rates.
- Flexibility in study design and the integration of advanced technologies allowing for real-time modifications based on participant feedback, ultimately enhancing research outcomes.
As the future of clinical trials progresses, embracing innovations and tackling the challenges associated with virtual studies will be imperative. Research directors are urged to prioritize ethical considerations, ensure compliance, and leverage the potential of emerging technologies to optimize their research impact. By doing so, they can contribute to a more equitable and efficient medical research landscape, paving the way for expedited and more reliable outcomes that benefit both researchers and patients alike.
Frequently Asked Questions
What is bioaccess and how does it enhance virtual clinical trials?
bioaccess® leverages the regulatory agility of Latin America, diverse populations of the Balkans, and effective routes in Australia to enhance virtual clinical trials. This combination facilitates ethical approvals within 4-6 weeks and boosts enrollment rates by 50%.
What are the advantages of virtual clinical trials in patient recruitment?
Virtual clinical trials enhance patient enrollment by eliminating geographical barriers, allowing individuals to participate from home. This flexibility aligns with participants’ schedules and lifestyles, leading to higher enrollment rates and greater inclusivity in medical research.
What challenges do traditional studies face regarding patient recruitment?
Approximately 80% of studies are delayed or terminated due to recruitment challenges, with 37% of research sites failing to enroll sufficient participants. Delays can cost sponsors between $600,000 and $8 million for each day a study is postponed.
How does electronic consent (e-consent) impact the recruitment process?
The implementation of electronic consent (e-consent) accelerates enrollment speed, streamlining the recruitment process and addressing challenges in participant recruitment.
What are the cost benefits of virtual clinical trials?
Virtual clinical trials reduce research expenses by minimizing the need for physical locations, travel costs, and associated overheads. They can decrease study costs by approximately 25-30%, leading to budget reductions ranging from $4,300 to $600,000.
Why are virtual clinical trials appealing to startups and smaller research organizations?
The cost-effectiveness of virtual clinical trials is particularly appealing to startups and smaller organizations as they significantly lower staffing and facility costs, enhancing financial viability. Additionally, 50% of individuals are more likely to participate in studies that offer home care alternatives, leading to retention rates of 95%.
List of Sources
- bioaccess: Accelerating Virtual Clinical Trials with Global Agility
- antidote.me (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- grandviewresearch.com (https://grandviewresearch.com/industry-analysis/virtual-clinical-trials-market)
- 10 Trends and Statistics for Clinical Trials in 2023 (https://xtalks.com/10-trends-and-statistics-for-clinical-trials-in-2023-3377)
- grandviewresearch.com (https://grandviewresearch.com/industry-analysis/us-virtual-clinical-trials-market-report)
- snsinsider.com (https://snsinsider.com/reports/virtual-clinical-trials-market-2493)
- Enhanced Patient Recruitment: The Key Advantage of Virtual Clinical Trials
- antidote.me (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- clinicaltrialsarena.com (https://clinicaltrialsarena.com/comment/virtual-clinical-trials-healthcare-trends)
- Improving Participant Recruitment in Clinical Trials: Comparative Analysis of Innovative Digital Platforms (https://jmir.org/2024/1/e60504)
- biospace.com (https://biospace.com/virtual-clinical-trials-market-size-to-reach-us-21-5-bn-by-2030)
- From AI to telemedicine: Transforming clinical trial recruitment in 2024 (https://labiotech.eu/expert-advice/clinical-trial-recruitment)
- Cost-Effectiveness: How Virtual Clinical Trials Reduce Research Expenses
- How to Cut Costs in Clinical Trials – Anju (https://anjusoftware.com/insights/eclinical/edc/cut-costs-clinical-trials)
- Decentralized clinical trials in the trial innovation network: Value, strategies, and lessons learned – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10465321)
- finance.yahoo.com (https://finance.yahoo.com/news/virtual-clinical-trials-market-analysis-144900382.html)
- Registry-based trials: a potential model for cost savings? – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC7682727)
- 83bar.com (https://83bar.com/benefits-of-virtual-clinical-trials-vs-traditional-clinical-trials)
- Flexibility in Study Design: Adapting to Patient Needs with Virtual Trials
- princetonmedicalinstitute.com (https://princetonmedicalinstitute.com/2023/12/15/exploring-adaptive-clinical-trials-flexibility-in-study-design)
- jmir.org (https://jmir.org/2023/1/e44171)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC7677786)
- becarispublishing.com (https://becarispublishing.com/doi/10.57264/cer-2024-0011)
- Decentralized Clinical Trials – Key Trends and Statistics | Medidata Solutions (https://medidata.com/en/decentralized-clinical-trials-key-trends-and-statistics)
- Technology Integration: Streamlining Data Collection in Virtual Trials
- lifebit.ai (https://lifebit.ai/blog/clinical-trial-technology-trends)
- straitsresearch.com (https://straitsresearch.com/report/virtual-clinical-trials-market)
- Clinical Trials Statistics and Facts (2026) (https://media.market.us/clinical-trials-statistics)
- 7 Key Findings from the 2022 Clinical Trial Operations Technology Survey (https://florencehc.com/blog-post/7-key-findings-from-the-2022-clinical-trial-operations-technology-survey)
- ahajournals.org (https://ahajournals.org/doi/10.1161/CIRCULATIONAHA.119.040798)
- Accelerated Timelines: Speeding Up Research with Virtual Clinical Trials
- 83bar.com (https://83bar.com/benefits-of-virtual-clinical-trials-vs-traditional-clinical-trials)
- drug-dev.com (https://drug-dev.com/clinical-analytics-solutions-reducing-clinical-cost-budget-variations-with-state-of-the-art-data-lifecycle-management-solutions)
- Clinical Trials Statistics and Facts (2026) (https://media.market.us/clinical-trials-statistics)
- mckinsey.com (https://mckinsey.com/industries/life-sciences/our-insights/unlocking-peak-operational-performance-in-clinical-development-with-artificial-intelligence)
- Online Patient Recruitment in Clinical Trials: Systematic Review and Meta-Analysis – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC7673977)
- Ethical Considerations: Ensuring Compliance in Virtual Clinical Trials
- nature.com (https://nature.com/articles/s41598-024-64139-9)
- Considerations For Improving Patient Recruitment Into Clinical Trials (https://clinicalleader.com/doc/considerations-for-improving-patient-0001)
- jpeds.com (https://jpeds.com/article/S0022-3476(20)30990-2/abstract)
- Clinical Trials Statistics and Facts (2026) (https://media.market.us/clinical-trials-statistics)
- Structural under-reporting of informed consent, data handling and sharing, ethical approval, and application of Open Science principles as proxies for study quality conduct in COVID-19 research: a systematic scoping review (https://gh.bmj.com/content/8/5/e012007)
- Global Reach: Accessing Diverse Patient Populations through Virtual Trials
- clinicalleader.com (https://clinicalleader.com/doc/patient-diversity-in-clinical-trials-how-to-meet-the-need-0001)
- statnews.com (https://statnews.com/2022/06/23/needed-clearer-explanation-importance-diversity-in-clinical-trials)
- The Importance of Diversity in Clinical Trials and the Impact of FDA Guidance – Clinical Trial Listing Database & Insights | CenterWatch (https://wcgclinical.com/insights/the-importance-of-diversity-in-clinical-trials-and-the-impact-of-fda-guidance)
- antidote.me (https://antidote.me/blog/why-racial-diversity-in-clinical-trials-is-so-important)
- Overcoming Challenges: How Virtual Trials Address Traditional Clinical Trial Limitations
- mdpi.com (https://mdpi.com/2227-9032/13/14/1708)
- lindushealth.com (https://lindushealth.com/blog/strategies-for-improving-patient-retention-in-clinical-trials)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC9789500)
- journals.plos.org (https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0235500)
- Unique Considerations for Patient Retention in Decentralized Clinical Trials – ACRP (https://acrpnet.org/2023/02/22/unique-considerations-for-patient-retention-in-decentralized-clinical-trials)
- Future Trends: Innovations Shaping the Landscape of Virtual Clinical Trials
- grandviewresearch.com (https://grandviewresearch.com/industry-analysis/virtual-clinical-trials-market)
- bioaccessla.com (https://bioaccessla.com/blog/7-key-trends-shaping-the-future-of-virtual-clinical-trials-for-medical-devices)
- straitsresearch.com (https://straitsresearch.com/report/virtual-clinical-trials-market)
- futuredatastats.com (https://futuredatastats.com/virtual-clinical-trials-market?srsltid=AfmBOoqtjTa5jGvi4NoNU0br-7CkMstBYLUS4l-Ujnr0DjHaHFHWwiFX)
- snsinsider.com (https://snsinsider.com/reports/virtual-clinical-trials-market-2493)

Leave a Reply