Introduction
Brazil’s evolving clinical trial landscape offers MedTech firms a pivotal chance to conduct first-in-human studies.
With recent reforms such as Law No. 14.874/2024, the approval process has been significantly streamlined, enabling faster timelines and adherence to international standards like ICH-GCP.
Many sponsors find themselves overwhelmed by Brazil’s intricate regulatory landscape.
What if sponsors tapped into local expertise and formed strategic partnerships to boost their trial outcomes and take full advantage of Brazil’s speed and cost benefits?
Understand Brazil’s Regulatory Framework for Clinical Trials
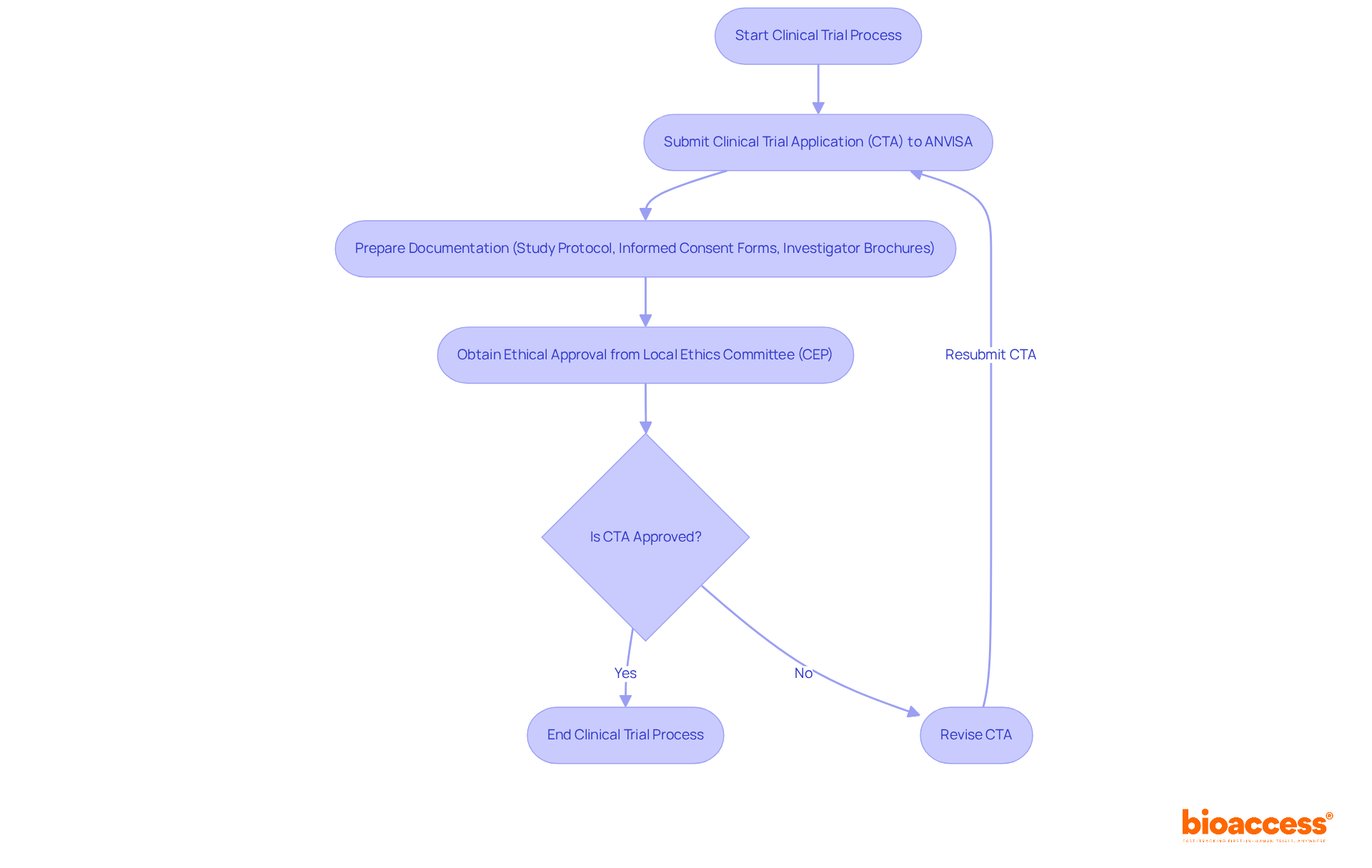
Understanding Brazil’s regulatory framework is crucial for MedTech firms aiming to conduct a medtech clinical trial in Brazil efficiently. Brazil’s regulatory structure for research involving human subjects is primarily overseen by ANVISA (Agência Nacional de Vigilância Sanitária), which manages the authorization and oversight of studies in this area. Recent reforms, particularly Law No. 14.874/2024, have significantly streamlined the approval process, reducing the average timeline from 265 days to just 90 days. This law mandates adherence to ICH-GCP (International Council for Harmonisation – Good Clinical Practice) standards. This ensures that all studies uphold high ethical and scientific quality.
To commence a clinical study, sponsors must submit a Clinical Trial Application (CTA) to ANVISA, which necessitates comprehensive documentation, including:
- Study protocol
- Informed consent forms
- Investigator brochures
Additionally, ethical approval from a local ethics committee (CEP) is necessary prior to submission to ANVISA. Understanding these requirements and timelines is vital for MedTech firms eager to speed up their first-in-human studies in the region.
Furthermore, sponsors should be aware of the specific requirements for various product types, such as medical devices and pharmaceuticals, as these can differ significantly. Navigating Brazil’s regulatory landscape can be daunting for MedTech firms, particularly when conducting a medtech clinical trial in Brazil due to varying requirements for different product types. Collaborating with local specialists or dedicated CROs knowledgeable about the Brazilian regulatory environment, such as bioaccess®, can improve compliance and efficiency in the research process. Currently, Brazil boasts 4,995 research studies recruiting and a total of 18,099 studies listed. This robust environment makes it an attractive option for early-stage investigations. By partnering with experts like bioaccess®, firms can navigate these complexities and expedite their research efforts.
Plan Logistics and Documentation for Efficient Trial Execution
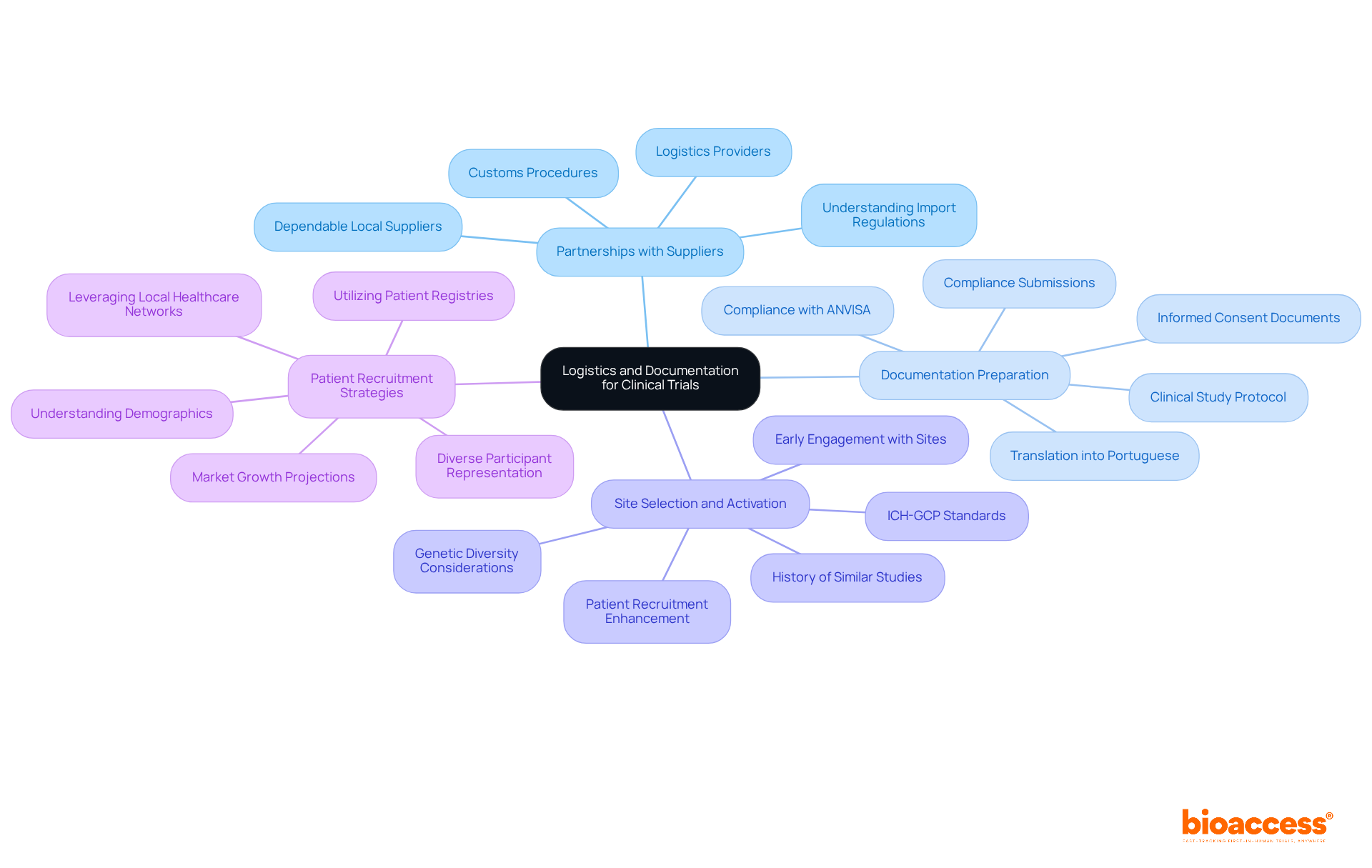
Navigating the complexities of early-stage studies in Latin America, especially for first-in-human research, is essential for conducting a successful medtech clinical trial in Brazil. A well-structured logistics strategy should encompass the following key elements:
- Forge partnerships with dependable local suppliers and logistics providers to ensure the prompt delivery of materials for research studies. Understanding Brazil’s complex import regulations and customs procedures is essential for any medtech clinical trial in Brazil. Delays in these areas can significantly impact study timelines. With bioaccess®, the approval process for studies can be expedited to as little as 4-8 weeks for ethics approvals, making efficient supply chain management even more essential.
- Documentation Preparation: All trial-related documents must be meticulously prepared and translated into Portuguese, as mandated by ANVISA. This includes the clinical study protocol, informed consent documents, and compliance submissions. Incomplete or improperly formatted documents can lead to significant delays in approval, jeopardizing the entire study. It’s crucial to comply with ANVISA’s requirements for a smooth approval process in medtech clinical trial in Brazil, and bioaccess® can help navigate these regulatory pathways effectively.
- Site Selection and Activation: Choose research sites that adhere to ICH-GCP standards and have a demonstrated history in conducting similar studies. Engaging with sites early in the planning process can facilitate quicker activation and enhance patient recruitment efforts. The region’s unique genetic diversity and high prevalence of chronic diseases provide significant opportunities for targeted studies in the context of a medtech clinical trial in Brazil, supported by bioaccess®’s established networks that enable 50% faster enrollment with pre-qualified patient populations.
- Patient Recruitment Strategies: Develop targeted recruitment strategies that leverage local healthcare networks and patient registries. A thorough understanding of the demographics and health profiles of the Brazilian population can improve recruitment efforts for the medtech clinical trial in Brazil and ensure diverse participant representation. The market for medical studies in the region is anticipated to expand at a CAGR of 3.9% from 2025 to 2032, underscoring the rising need for efficient strategies in patient recruitment. Bioaccess® has effectively utilized LATAM trial data for FDA IDE and PMA submissions, showcasing the importance of GCP-compliant information in speeding up patient access and enrollment.
By prioritizing these logistical elements, sponsors can not only enhance their study outcomes but also position themselves for success in a competitive landscape.
Establish Strategic Partnerships to Enhance Trial Success
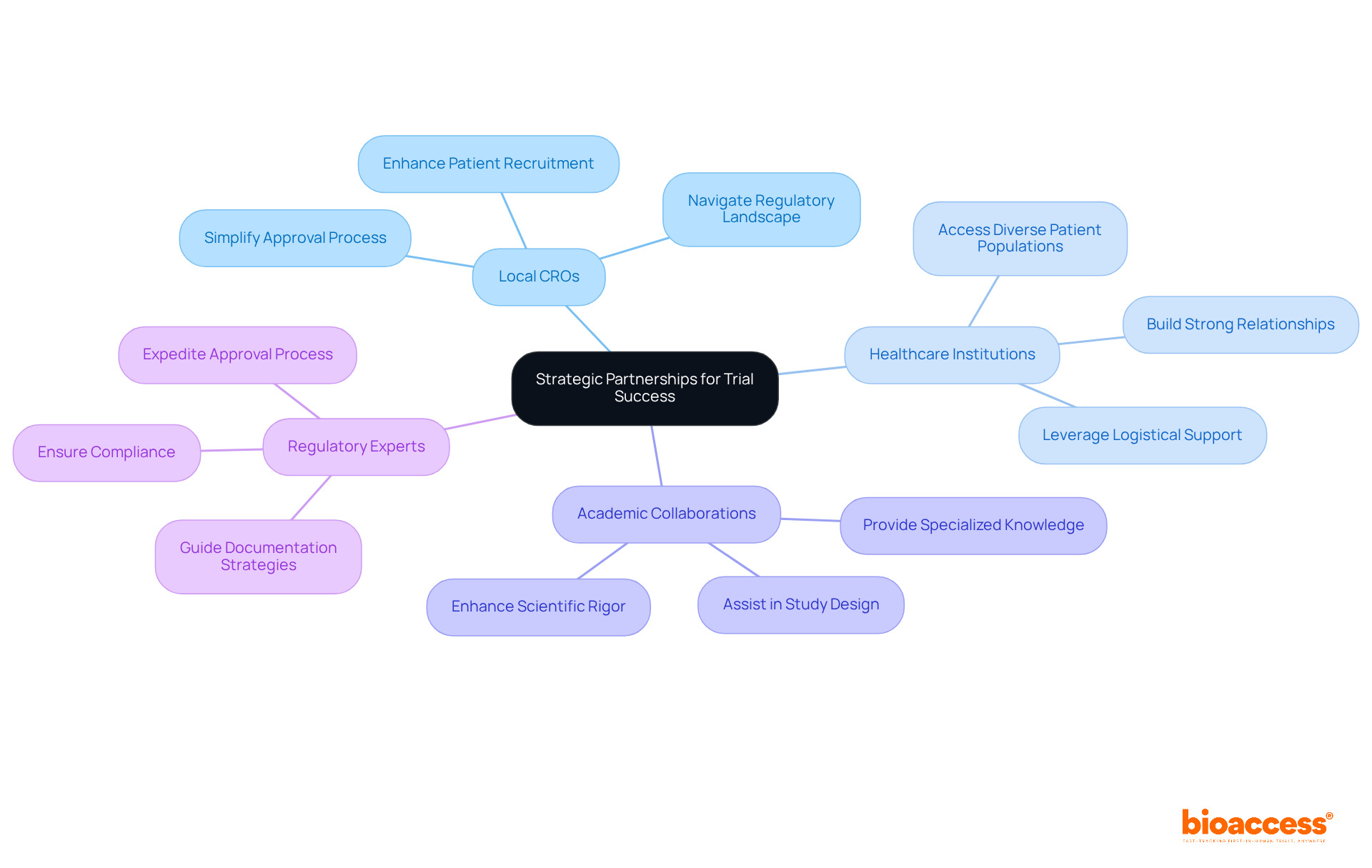
Navigating the complexities of medtech clinical trial Brazil can be daunting, especially for first-in-human studies. Collaborating with local Contract Research Organizations (CROs), healthcare institutions, and compliance consultants provides essential insights and resources. Here are best practices for establishing effective partnerships:
- Engage Local CROs: Collaborating with a local CRO that focuses on first-in-human studies can simplify the approval submission process and improve patient recruitment initiatives. Local CROs have a deep understanding of ANVISA’s requirements and can effectively navigate the regulatory landscape, ensuring compliance with ICH-GCP standards.
- Collaborate with Healthcare Institutions: By building strong relationships with hospitals and research centers, you can tap into diverse patient populations, enhancing recruitment strategies. These institutions often have established networks and logistical support that can significantly enhance the efficiency of execution in studies.
- Utilize Academic Collaborations: Partnering with universities and research institutions can enhance the scientific rigor of studies. Academic collaborators assist in study design, data evaluation, and grant access to specialized knowledge, which is essential for the success of medical research.
- Utilize Local Regulatory Experts: Engaging regulatory consultants familiar with Brazilian laws can ensure compliance and expedite the approval process. These experts offer guidance on best practices for documentation and submission strategies, helping to navigate the complexities of local regulations.
By cultivating these strategic alliances, sponsors can enhance operational capacities, reduce risks, and ultimately boost the success rates of their medtech clinical trial Brazil and other studies in Latin America. This collaborative approach not only accelerates healthcare development but also positions Brazil as a leader in innovative research within the MedTech and Biopharma sectors.
Implement Proactive Risk Management and Adaptation Strategies
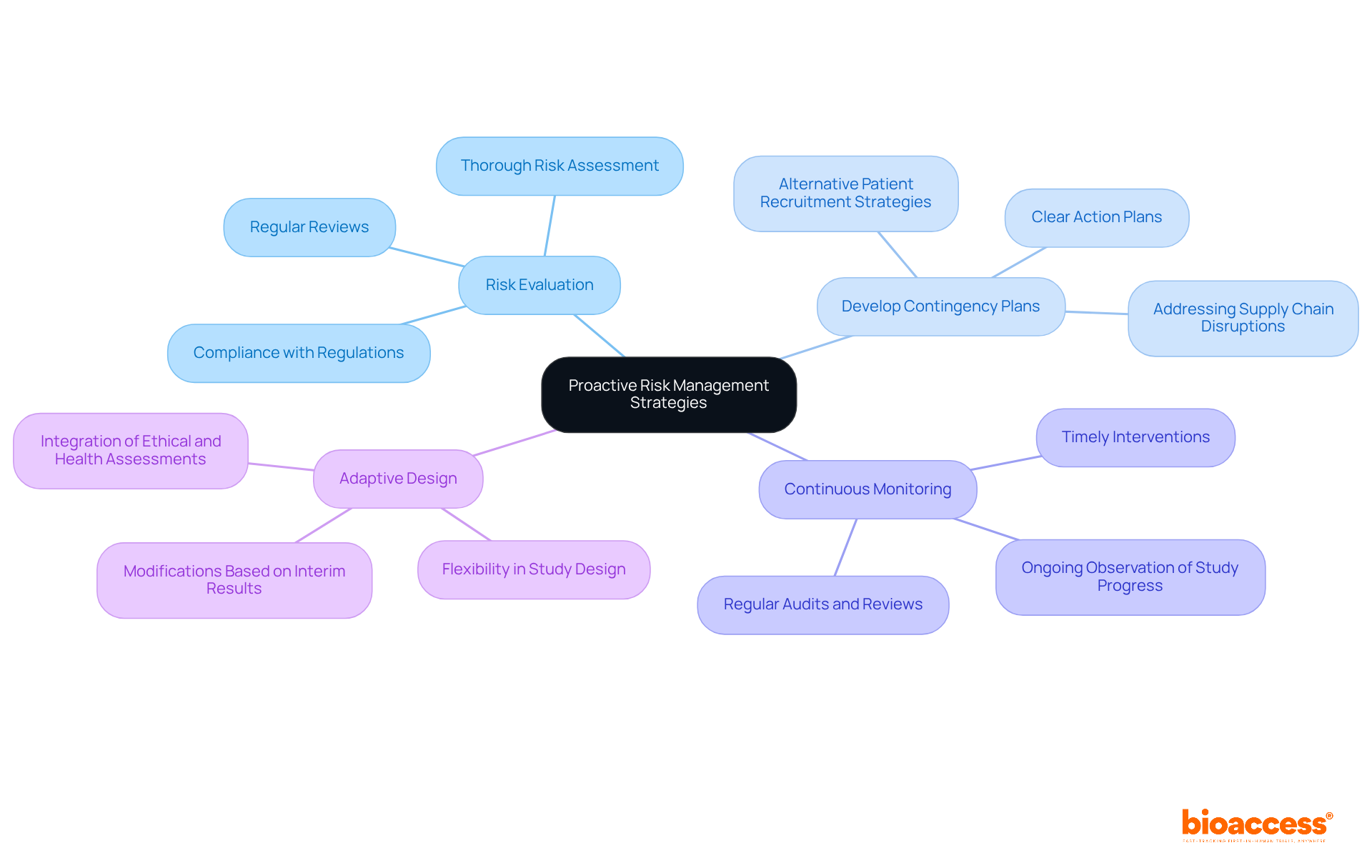
In Brazil’s evolving clinical research landscape, proactive risk management is not just beneficial; it’s essential for success. By identifying potential risks early, sponsors can develop robust mitigation strategies that enhance study integrity and compliance. Here are key practices for effective risk management:
- Risk Evaluation: Perform a thorough risk evaluation at the study’s start to pinpoint potential obstacles related to compliance with regulations, patient recruitment, and logistical concerns. Regular reviews of this assessment are essential. They help adapt to changes in the environment and ensure continuous monitoring of potential risks.
- Develop Contingency Plans: Establish contingency plans for identified risks, including alternative strategies for patient recruitment and addressing supply chain disruptions. Having a clear plan in place can reduce delays and keep the study on track, especially in a market where approval timelines can be significantly shortened.
- Continuous Monitoring: Implement a system for ongoing observation of study progress and adherence to regulatory requirements. Regular audits and reviews can help identify issues early, allowing for timely interventions. This is vital in the region, where Research Ethics Committees (CEPs) now have a maximum period of 30 days to issue an opinion, enhancing the need for proactive oversight.
- Adaptive Design: Consider utilizing adaptive designs that allow for modifications based on interim results. This flexibility can enhance the study’s efficiency and effectiveness, particularly in the region’s dynamic regulatory environment, where integrating ethical and health assessments can streamline processes.
Ultimately, embracing these strategies empowers sponsors to navigate the medtech clinical trial Brazil landscape with confidence and agility. This approach not only safeguards participant welfare but also positions sponsors to leverage Brazil’s strategic advantages in speed and cost efficiency for early-stage clinical trials.
Conclusion
While Brazil offers a promising landscape for MedTech clinical trials, many firms find themselves grappling with the complexities of its regulatory environment. Understanding the regulatory framework and the streamlined approval processes from recent reforms is essential for success. Companies can significantly reduce timelines and enhance the quality of their studies by navigating this robust regulatory environment, overseen by ANVISA. This sets the stage for effective first-in-human trials, making Brazil an attractive destination for innovative research.
Key strategies for success include:
- Meticulous planning of logistics and documentation
- Establishing strategic partnerships
- Implementing proactive risk management
Working closely with local experts, like Contract Research Organizations (CROs), not only helps meet ICH-GCP standards but also streamlines patient recruitment. Additionally, recognizing the unique characteristics of Brazil’s diverse population can enhance recruitment strategies and improve study outcomes. By focusing on these best practices, MedTech firms can navigate the complexities of clinical trials in Brazil and position themselves favorably in the competitive landscape.
Ultimately, embracing these insights not only fosters operational efficiency but also reinforces Brazil’s role as a strategic hub for early-stage clinical trials. To thrive in this dynamic market, firms must act decisively, leveraging local insights and regulatory advantages to drive their innovations forward. By taking proactive steps today, firms can capitalize on Brazil’s advantages in speed, cost efficiency, and patient access, paving the way for successful clinical trials and ultimately advancing healthcare solutions.
Frequently Asked Questions
What is the primary regulatory authority overseeing clinical trials in Brazil?
The primary regulatory authority overseeing clinical trials in Brazil is ANVISA (Agência Nacional de Vigilância Sanitária).
What recent reforms have impacted the clinical trial approval process in Brazil?
Recent reforms, particularly Law No. 14.874/2024, have streamlined the approval process, reducing the average timeline from 265 days to just 90 days.
What standards must clinical trials in Brazil adhere to?
Clinical trials in Brazil must adhere to ICH-GCP (International Council for Harmonisation – Good Clinical Practice) standards to ensure high ethical and scientific quality.
What documentation is required for a Clinical Trial Application (CTA) to ANVISA?
The documentation required for a Clinical Trial Application (CTA) includes a study protocol, informed consent forms, and investigator brochures.
Is ethical approval necessary before submitting a CTA to ANVISA?
Yes, ethical approval from a local ethics committee (CEP) is necessary prior to the submission of a Clinical Trial Application to ANVISA.
Are there different requirements for various product types in clinical trials?
Yes, there are specific requirements for various product types, such as medical devices and pharmaceuticals, which can differ significantly.
How can MedTech firms improve compliance and efficiency in the Brazilian regulatory landscape?
MedTech firms can improve compliance and efficiency by collaborating with local specialists or dedicated CROs knowledgeable about the Brazilian regulatory environment, such as bioaccess®.
What is the current status of clinical research studies in Brazil?
Currently, Brazil boasts 4,995 research studies recruiting and a total of 18,099 studies listed, making it an attractive option for early-stage investigations.
Why is Brazil considered a strategic advantage for early-stage clinical trials?
Brazil is considered a strategic advantage for early-stage clinical trials due to its speed in approval processes, cost efficiency, and robust patient recruitment capabilities.
List of Sources
- Understand Brazil’s Regulatory Framework for Clinical Trials
- REBEC (https://ensaiosclinicos.gov.br/news/547)
- Brazil Just Changed The Rules For Clinical Trial Approvals — What Sponsors Need To Know | bioaccess® (https://bioaccessla.com/blog/brazil-anvisa-parallel-review-clinical-trial-approvals-2026)
- Brazil Cuts Clinical Trial Approval Timeline to 90 Days | bioaccess®: Fast-Tracking First-in-Human Trials, Anywhere posted on the topic | LinkedIn (https://linkedin.com/posts/bioaccess_clinicaltrials-brazil-anvisa-activity-7447976698958106627-98MW)
- Brazil Clinical Trials Market Size, Share | Analysis Report [2032] (https://fortunebusinessinsights.com/brazil-clinical-trials-market-112310)
- Plan Logistics and Documentation for Efficient Trial Execution
- Brazil Clinical Trials Market Size, Share | Analysis Report [2032] (https://fortunebusinessinsights.com/brazil-clinical-trials-market-112310)
- Brazil Clinical Trials Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/clinical-trials-market/brazil)
- Unlocking Brazil’s Clinical Trial Opportunity: A Strategic Roadmap for Healthcare Leaders (https://lek.com/insights/life-sciences-pharma/unlocking-brazils-clinical-trial-opportunity-strategic-roadmap)
- Clinical trials initiated in Brazil 2024| Statista (https://statista.com/statistics/1067453/brazil-number-clinical-trials-initiated?srsltid=AfmBOoqsuWcvCDHgnKZZUoESuwbtvzOOwY8LS_UkZzs5Z2pfHI9PAic4)
- Establish Strategic Partnerships to Enhance Trial Success
- Brazil Clinical Trials Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/clinical-trials-market/brazil)
- Unlocking Brazil’s Clinical Trial Opportunity: A Strategic Roadmap for Healthcare Leaders (https://lek.com/insights/life-sciences-pharma/unlocking-brazils-clinical-trial-opportunity-strategic-roadmap)
- Brazil accounts for 1.7% share of global clinical trial activity in 2021 (https://clinicaltrialsarena.com/marketdata/brazil-accounts-for-1-7-share-of-global-clinical-trial-activity-in-2021)
- Implement Proactive Risk Management and Adaptation Strategies
- New regulations for clinical research in Brazil | Licks Attorneys (https://lickslegal.com/post/new-regulations-for-clinical-research-in-brazil)
- Current scenario and future perspectives of clinical research in Brazil: a national survey – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10898894)
- From Risk to Readiness: Clinical Development Trends Shaping 2026 (https://contractpharma.com/exclusives/from-risk-to-readiness-clinical-development-trends-shaping-2026)
- 50 Risk Management Quotes: Wisdom for Smart Decision-making | ITD World (https://itdworld.com/blog/leadership/risk-management-quotes)

Leave a Reply