Introduction
In the rapidly evolving landscape of clinical research, head-to-head comparisons have emerged as a vital tool for determining the effectiveness and safety of various interventions. These structured studies not only guide clinical decisions but also influence regulatory approvals and market access strategies, particularly in the context of advanced therapies for complex diseases like cancer.
However, as the complexity of these comparisons increases, how can researchers ensure they are effectively designed and executed to yield meaningful results? This article delves into the intricacies of mastering head-to-head comparisons in clinical trials, offering a step-by-step guide to navigate the challenges and maximize the impact of these essential studies.
Define Head-to-Head Comparisons in Clinical Trials
Structured studies utilize to assess two or more interventions, therapies, or products in order to determine their . are crucial for evaluating the effectiveness of new therapies against established standards of care, thereby guiding clinical decisions, regulatory approvals, and market access strategies. In 2025, the significance of these experiments is underscored by the rapid advancements in cancer medicine, which have yielded a multitude of options for patients. As the care landscape evolves, the development of effective becomes increasingly complex, necessitating innovative approaches such as adaptive designs and biomarker-based patient selection.
Real-world examples illustrate the effectiveness of direct comparisons. For instance, the implementation of has proven vital in enhancing by identifying the most effective therapies available. These studies not only provide conclusive data on but also by informing well-considered treatment choices.
Statistics reveal that medical studies typically require the participation of thousands of patients and demand tens of thousands of hours of research effort. However, the challenges of late-stage Phase III studies, which often necessitate large participant numbers, can be alleviated by designing smaller yet powerful experiments. Such studies can accelerate enrollment and information gathering, ultimately leading to faster advancements in medical research. By maximizing the information obtained from each study, healthcare providers can ensure the best possible care options for their patients through . Moreover, with bioaccess®’s expertise in managing (EFS), (FIH), and pivotal studies, coupled with their expedited patient enrollment strategies that achieve 50% quicker recruitment and $25K savings through FDA-ready data, the regulatory implications of these studies are reinforced, underscoring their importance in the evolving landscape of .
Establish Clear Objectives for Your Comparisons
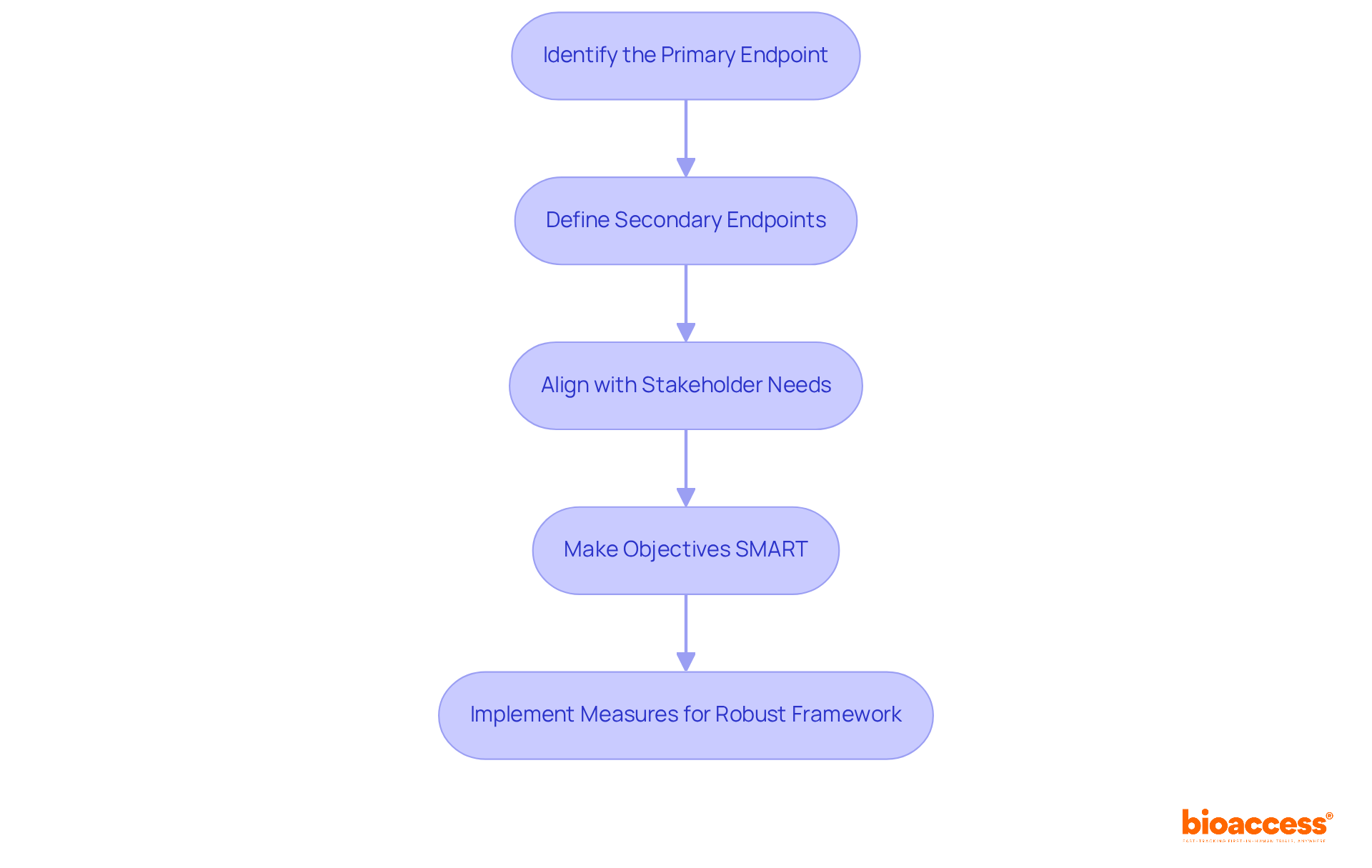
To establish clear objectives for your , it is essential to follow a structured approach:
- Identify the : Determine the main outcome you wish to measure, such as efficacy, safety, or quality of life. This clarity is crucial for guiding the research focus.
- Define : Outline additional outcomes that will provide further insights into the interventions being compared. These can include metrics like patient-reported outcomes or biomarker assessments.
- Align with Stakeholder Needs: Ensure that your objectives meet the expectations of , sponsors, and the medical community. This alignment is vital for gaining support and facilitating the approval process.
- : Ensure that your objectives are . For instance, a particular goal might be to demonstrate a 20% within six months of care. This not only clarifies the goals but also enhances the ability to make when evaluating results effectively.
By implementing these measures, you can establish a robust framework for your that fosters significant comparisons and impactful results.
Select Appropriate Study Designs for Effective Comparisons
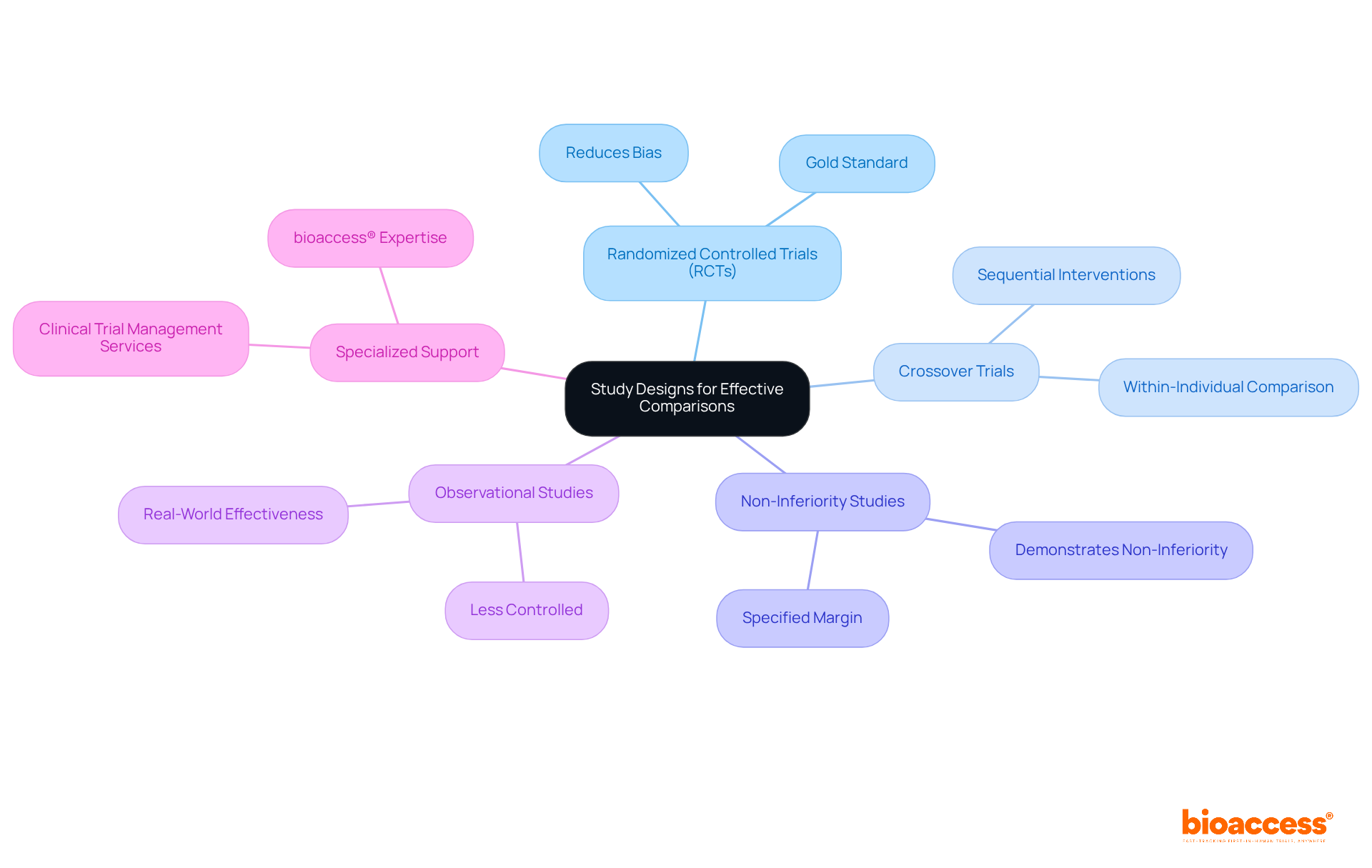
When selecting a study design for , it is essential to consider the following options:
- (RCTs): Recognized as the gold standard for clinical studies, RCTs effectively reduce bias by randomly allocating participants to various intervention groups.
- : In these trials, participants receive both interventions sequentially, allowing for direct comparison within the same individual.
- Non-Inferiority Studies: These studies are designed to demonstrate that a new treatment is not worse than an existing treatment by a specified margin.
- Observational Studies: Although less controlled, observational studies can provide valuable insights into real-world effectiveness and safety when RCTs are not feasible.
In the context of conducting these trials in , leveraging comprehensive , such as those offered by bioaccess®, can significantly enhance during the process. Their expertise in ensures that your study design aligns with your objectives, the nature of the interventions, and the target population. It is crucial to choose a design that best fits your research goals while considering the specialized support available through bioaccess®.
Implement Robust Data Collection and Analysis Techniques
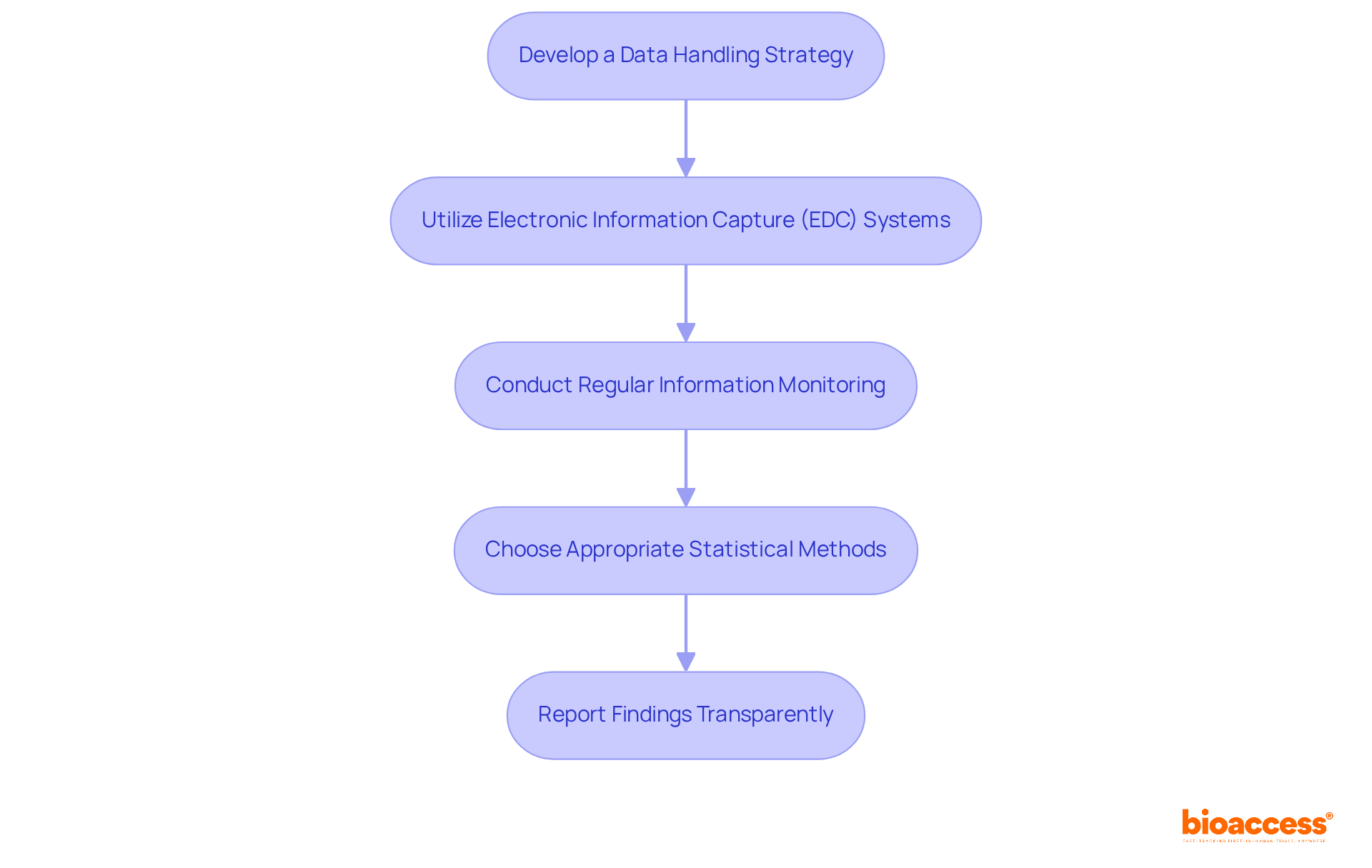
To implement robust , adhere to the following steps:
- Develop a : Formulate a comprehensive plan that details how information will be collected, stored, and analyzed. This strategy must ensure compliance with , outlining team members’ roles and responsibilities, information handling procedures, and compliance measures.
- Utilize : Leverage to streamline information collection processes. These systems facilitate real-time data input, significantly reducing errors associated with traditional paper-based methods. By 2025, the integration of advanced EDC systems is expected to enhance data quality and operational efficiency in .
- Conduct : Implement systematic monitoring processes to guarantee accuracy and completeness throughout the trial. Routine audits and inspections can identify inconsistencies early, allowing for prompt adjustments and safeguarding information integrity.
- Choose Appropriate : Select . Employing the right methods is essential for effective data analysis and can profoundly affect the validity of your findings.
- : Ensure that results are reported in accordance with established guidelines, such as CONSORT, to uphold transparency and credibility. Clear reporting not only enhances the trustworthiness of the research but also aids in better understanding and application of the findings in clinical practice.
Conclusion
Mastering head-to-head comparisons in clinical trials is essential for evaluating the efficacy and safety of new therapies against established standards. These structured studies not only inform clinical decisions but also play a pivotal role in regulatory approvals and market access strategies, especially in the rapidly evolving landscape of cancer medicine. As the complexity of these comparisons increases, innovative methodologies and clear objectives become paramount for successful outcomes.
Throughout this article, we have discussed key components of effective head-to-head comparisons. Establishing clear objectives, selecting appropriate study designs, and implementing robust data collection and analysis techniques are critical steps in ensuring that these studies yield meaningful results. Real-world examples highlight the positive impact of these comparisons on patient-centered care and economic efficiency in healthcare, emphasizing their necessity in clinical research.
Ultimately, the significance of head-to-head comparisons cannot be overstated. As the medical field continues to advance, embracing these methodologies will be crucial for delivering optimal care to patients. Stakeholders are encouraged to prioritize these comparisons in their research endeavors, thereby contributing to a more informed and effective healthcare landscape.
Frequently Asked Questions
What are head-to-head comparisons in clinical trials?
Head-to-head comparisons in clinical trials are structured studies that assess two or more interventions, therapies, or products to determine their relative effectiveness and safety profiles.
Why are head-to-head comparisons important?
They are crucial for evaluating the effectiveness of new therapies against established standards of care, guiding clinical decisions, regulatory approvals, and market access strategies.
How has the significance of head-to-head comparisons changed in recent years?
The significance has increased due to rapid advancements in cancer medicine, which have resulted in many treatment options for patients, making effective comparisons more complex.
What innovative approaches are being used in head-to-head comparisons?
Innovative approaches include adaptive designs and biomarker-based patient selection to enhance the effectiveness of these comparisons.
How do real-world examples demonstrate the effectiveness of head-to-head comparisons?
Randomized head-to-head comparisons have been vital in enhancing patient-centered care by identifying the most effective therapies, providing conclusive data on treatment effectiveness, and improving economic efficiency in healthcare.
What are some challenges associated with medical studies?
Medical studies typically require thousands of patient participants and significant research effort, particularly in late-stage Phase III studies that often necessitate large participant numbers.
How can the challenges of Phase III studies be addressed?
By designing smaller yet powerful experiments, which can accelerate enrollment and information gathering, leading to faster advancements in medical research.
What benefits do head-to-head comparisons provide to healthcare providers?
They maximize the information obtained from each study, ensuring that healthcare providers can offer the best possible care options for their patients.
How does bioaccess® contribute to the management of clinical studies?
Bioaccess® specializes in managing Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), and pivotal studies, with expedited patient enrollment strategies that achieve 50% quicker recruitment and $25K savings through FDA-ready data.
What are the regulatory implications of head-to-head comparisons?
The regulatory implications are reinforced by the importance of these studies in the evolving landscape of medical research, particularly in Latin America.
List of Sources
- Define Head-to-Head Comparisons in Clinical Trials
- gene.com (https://gene.com/stories/two-heads-are-better-than-one)
- pubmed.ncbi.nlm.nih.gov (https://pubmed.ncbi.nlm.nih.gov/22152420)
- pubmed.ncbi.nlm.nih.gov (https://pubmed.ncbi.nlm.nih.gov/22205018)
- Establish Clear Objectives for Your Comparisons
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC4917792)
- Set—and achieve—SMART-er goals [2026] (https://asana.com/resources/smart-goals)
- physio-pedia.com (https://physio-pedia.com/SMART_Goals)
- scilife.io (https://scilife.io/blog/smart-quality-goals-pharma)
- Implement Robust Data Collection and Analysis Techniques
- bioaccessla.com (https://bioaccessla.com/blog/10-key-insights-to-enhance-your-phase-research-in-clinical-trials)
- Exploring Data Quality Management within Clinical Trials – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC5801732)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC3326906)
- journals.sagepub.com (https://journals.sagepub.com/doi/full/10.1089/hum.2022.150)
- Data Management in Clinical Research: Best Practices | Datavant (https://datavant.com/clinical-research/clinical-data-management)

Leave a Reply