Introduction
Navigating the complexities of clinical trial management in Brazil can be daunting for sponsors seeking to introduce innovative therapies. With a regulatory framework governed by ANVISA that emphasizes ethical considerations and streamlined approval processes, understanding best practices in this arena is crucial for success.
How can sponsors effectively leverage Brazil’s evolving regulations and robust patient recruitment strategies to enhance their trial outcomes and ensure compliance?
Let’s explore the essential practices that can help sponsors turn the complexities of clinical trials into a strategic advantage in Latin America.
Understand Brazil’s Regulatory Framework for Clinical Trials
Navigating Brazil’s regulatory landscape for clinical trial management can be a complex endeavor, yet understanding its nuances is essential for success. Brazil’s regulatory system for research involving human participants is primarily overseen by ANVISA, which manages the authorization and supervision of studies. Key regulations include:
- Resolution 466/2012: This regulation outlines ethical considerations and mandates ethical committee approval before initiating trials. It emphasizes the need for informed consent and the protection of participants’ rights, ensuring that ethical standards are upheld throughout the research process.
- Law No. 14.874/2024: This recent legislation aims to simplify the authorization process, significantly cutting timelines from an average of 265 days to a firm limit of 90 business days for applications related to research involving human subjects. This modification enhances Brazil’s appeal as a destination for clinical trial management and global research studies, especially for early-stage evaluations.
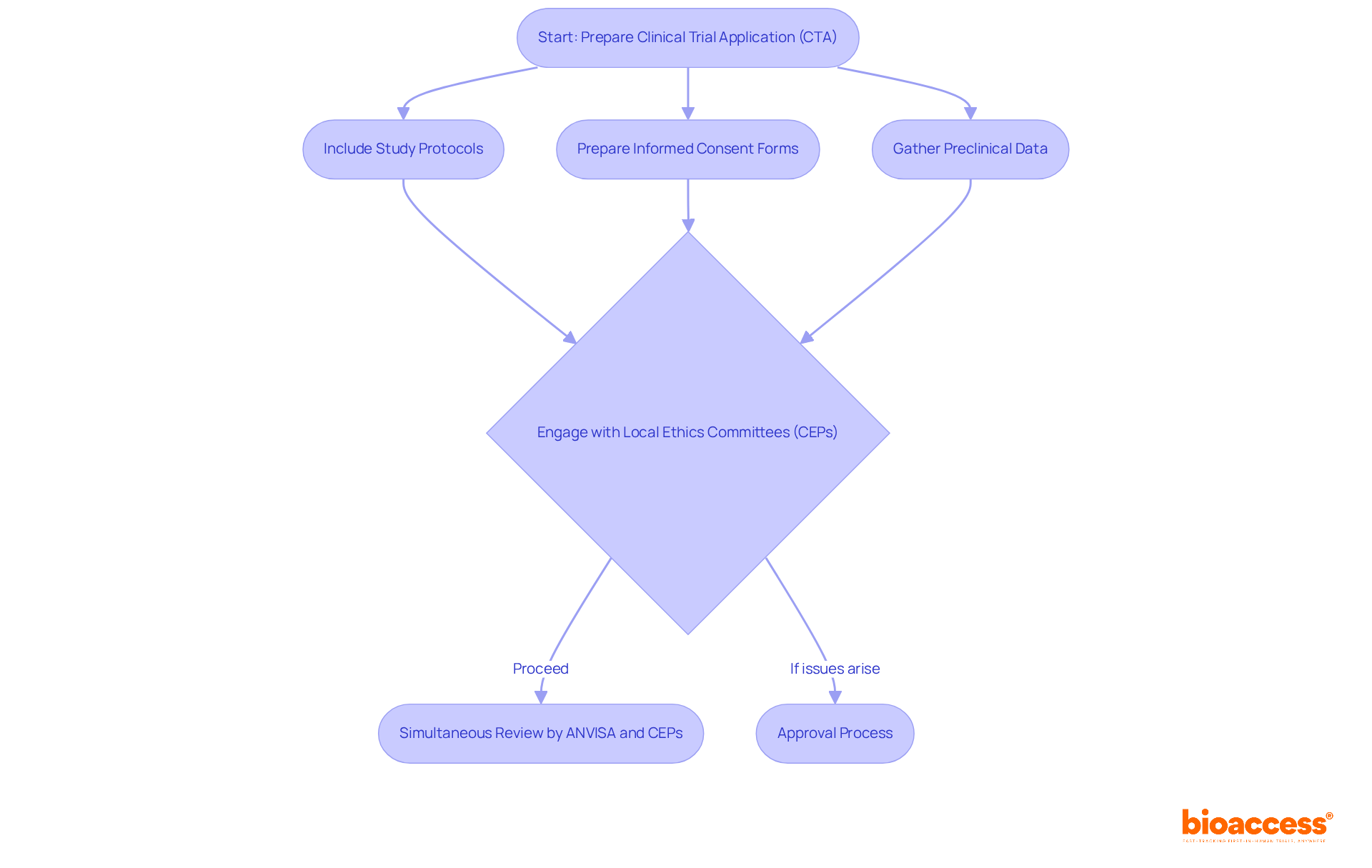
To successfully navigate this framework, sponsors must prepare a Clinical Trial Application (CTA) that includes detailed study protocols, informed consent forms, and evidence of preclinical data, which is essential for clinical trial management in Brazil. Grasping these requirements is crucial for guaranteeing timely endorsements and adherence to local regulations.
Sponsors should actively engage with local ethics committees (CEPs) and the National Commission for Ethics in Research (CONEP) to ensure a smooth approval process. Early involvement with these entities can enable smoother approvals and boost the credibility of the study. The new framework allows for simultaneous review by ANVISA and ethics committees, further expediting the process and reducing the time to market for innovative therapies. By proactively engaging with regulatory bodies and ethics committees, sponsors can significantly enhance their chances of timely approvals and successful study outcomes.
Optimize Site Selection and Patient Recruitment Strategies
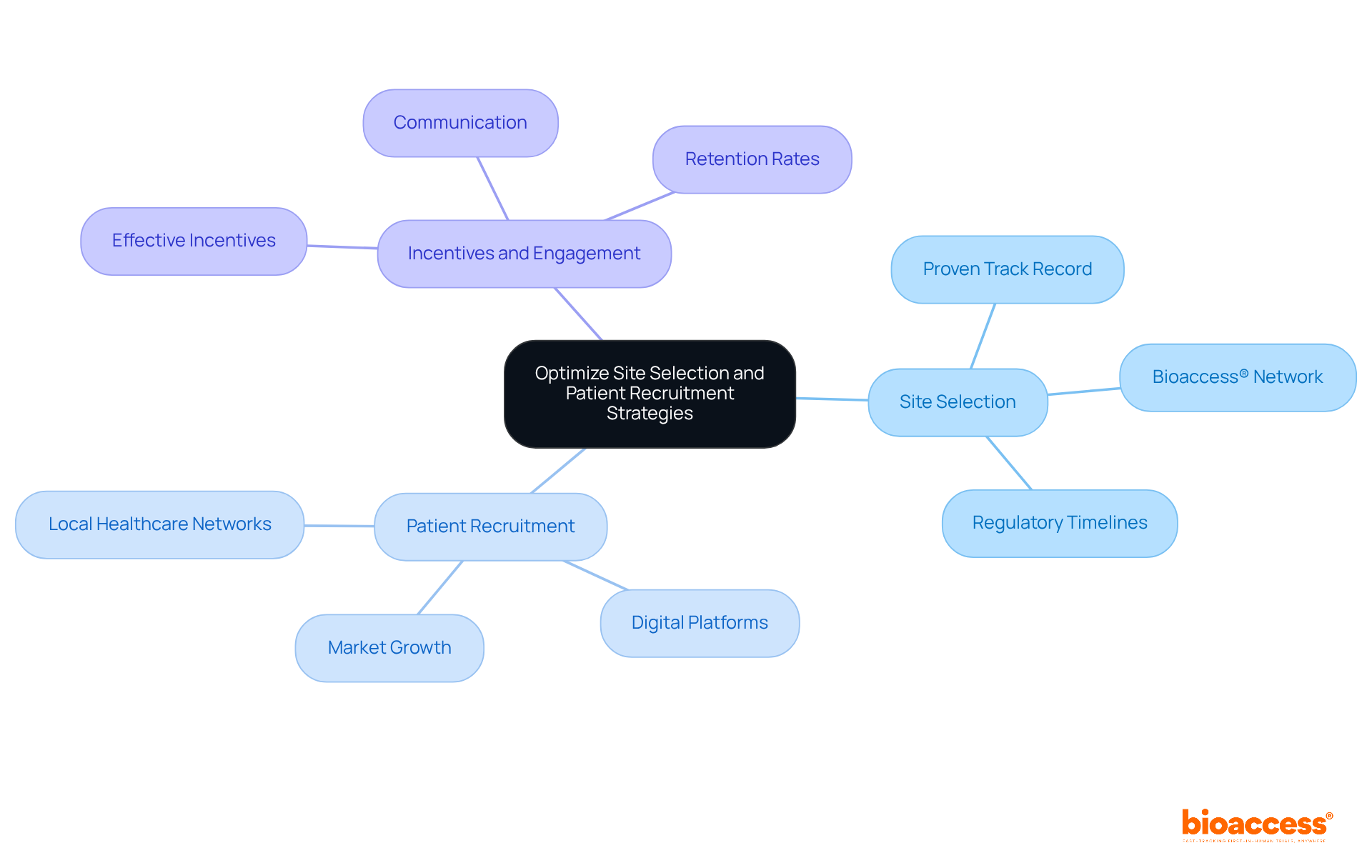
Selecting the right research sites is not just important; it’s a decisive factor in the success of clinical trial management in Brazil. Here are best practices:
- Site Selection: Opt for sites with a proven track record in clinical research and direct access to the target patient population. Utilize bioaccess®’s extensive network of over 50 pre-qualified research sites to ensure adherence to ICH-GCP standards and operational efficiency. This network enables quicker site activation, essential for adhering to regulatory timelines established by ANVISA, which regulates study approvals in the region. For instance, previous evaluations conducted through bioaccess® have demonstrated a 40% reduction in site activation time, allowing sponsors to initiate studies more swiftly.
- Patient Recruitment: How can local healthcare networks and community outreach programs help you connect with potential participants? Engaging these networks and utilizing digital platforms and social media can broaden outreach, particularly among underrepresented populations. The market for clinical trial management Brazil in patient recruitment services is anticipated to expand significantly, reaching USD 144.4 million by 2033, emphasizing the growing significance of effective recruitment strategies. Successful case studies, such as those from MedTech startups that partnered with bioaccess®, illustrate how targeted recruitment strategies can lead to a 30% increase in enrollment rates.
- Incentives and Engagement: Implementing effective incentives and maintaining consistent communication are essential for enhancing participant retention rates. This can involve educational materials regarding the study and its benefits, which are crucial for building trust and commitment among participants. Testimonials from previous clients highlight that clear communication and participant engagement greatly enhance retention and satisfaction, with one client observing a retention rate of over 85% in their recent study.
By refining these strategies, sponsors can not only enhance their study outcomes but also elevate Brazil’s reputation in clinical trial management Brazil.
Incorporate Early Feasibility Studies to Enhance Trial Outcomes
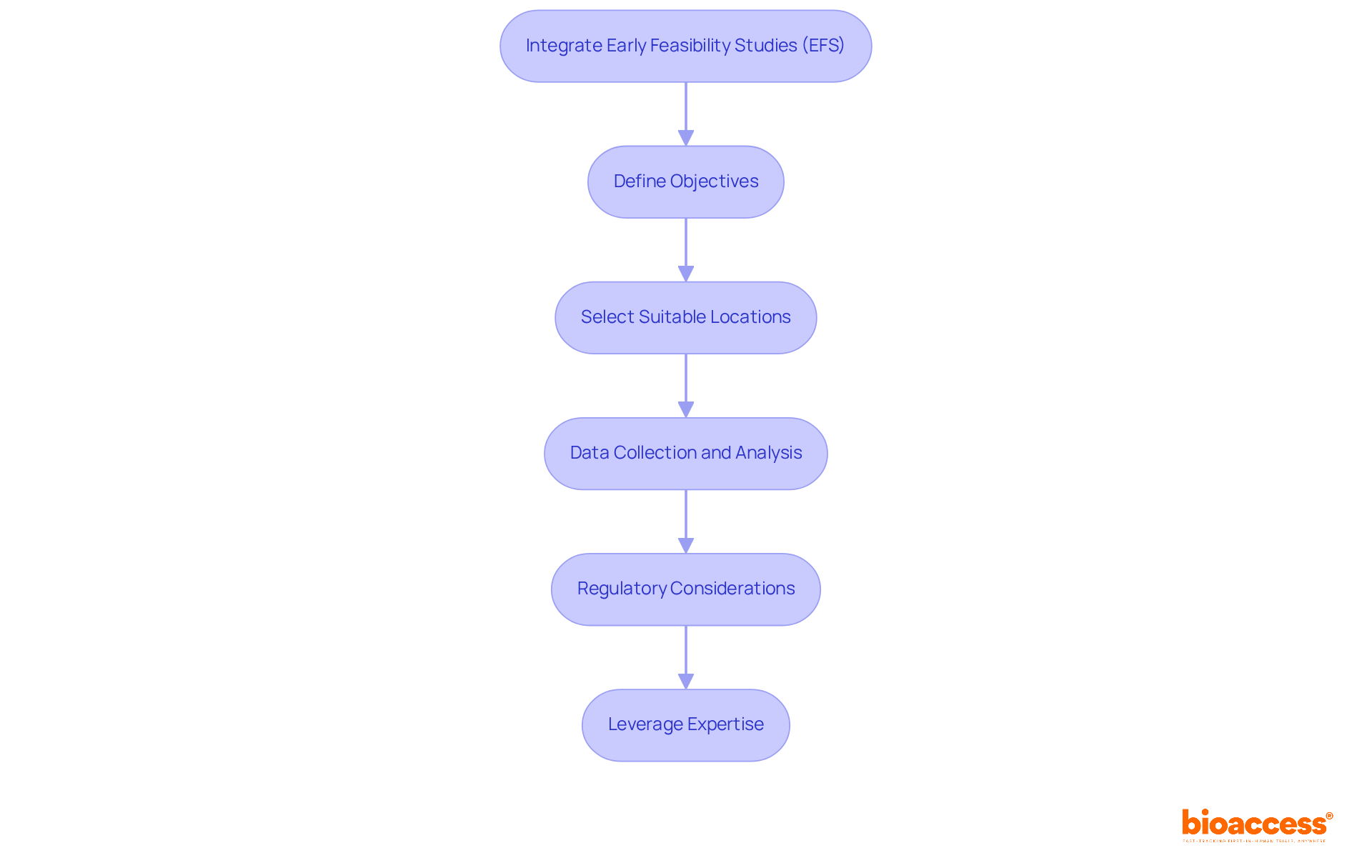
Integrating Early Feasibility Studies (EFS) into your research strategy is essential for navigating Brazil’s complex regulatory environment and is a critical component of clinical trial management Brazil to improve study outcomes. Here’s how to effectively implement EFS:
- Define Objectives: Establish clear objectives for the EFS, focusing on critical questions related to the safety, efficacy, and usability of the medical device or treatment. This clarity will guide the study design and data collection processes.
- Select Suitable Locations: Choose research sites experienced in conducting EFS and with access to the target patient population. This selection is crucial for improving data quality and participant engagement, ultimately leading to more reliable outcomes.
- Data Collection and Analysis: Implement robust data collection methodologies to capture insights on patient responses and device performance. Examining this data will guide subsequent phases of the study and enable necessary protocol adjustments.
- Regulatory Considerations: Ensure adherence to ANVISA regulations by submitting the EFS as part of the overall application for the study. EFS can be conducted before all preclinical testing has been completed, allowing for flexibility in the development process. This proactive approach can facilitate faster approvals, typically within 30 to 90 days, and enhance the credibility of the study. As pointed out by Paola Antonini, EFS are effective tools for understanding and predicting investigator and study site performance, which is crucial given that more than 90% of research studies fail due to factors like limited patient recruitment.
Many studies struggle to recruit enough patients, leading to high failure rates. By integrating EFS, sponsors can significantly improve their chances of success in clinical trial management Brazil, as these studies not only mitigate risks but also refine study designs. By utilizing the benefits of the local regulatory framework and patient recruitment capabilities, sponsors can expedite their development timelines effectively. Moreover, leveraging bioaccess®’s expertise in navigating the complexities of FIH studies can further improve the efficiency and success of your research strategy. Embracing EFS not only mitigates risks but also positions your research for greater success in the competitive landscape of clinical trials.
Ensure Compliance with ICH-GCP Standards and Local Regulations
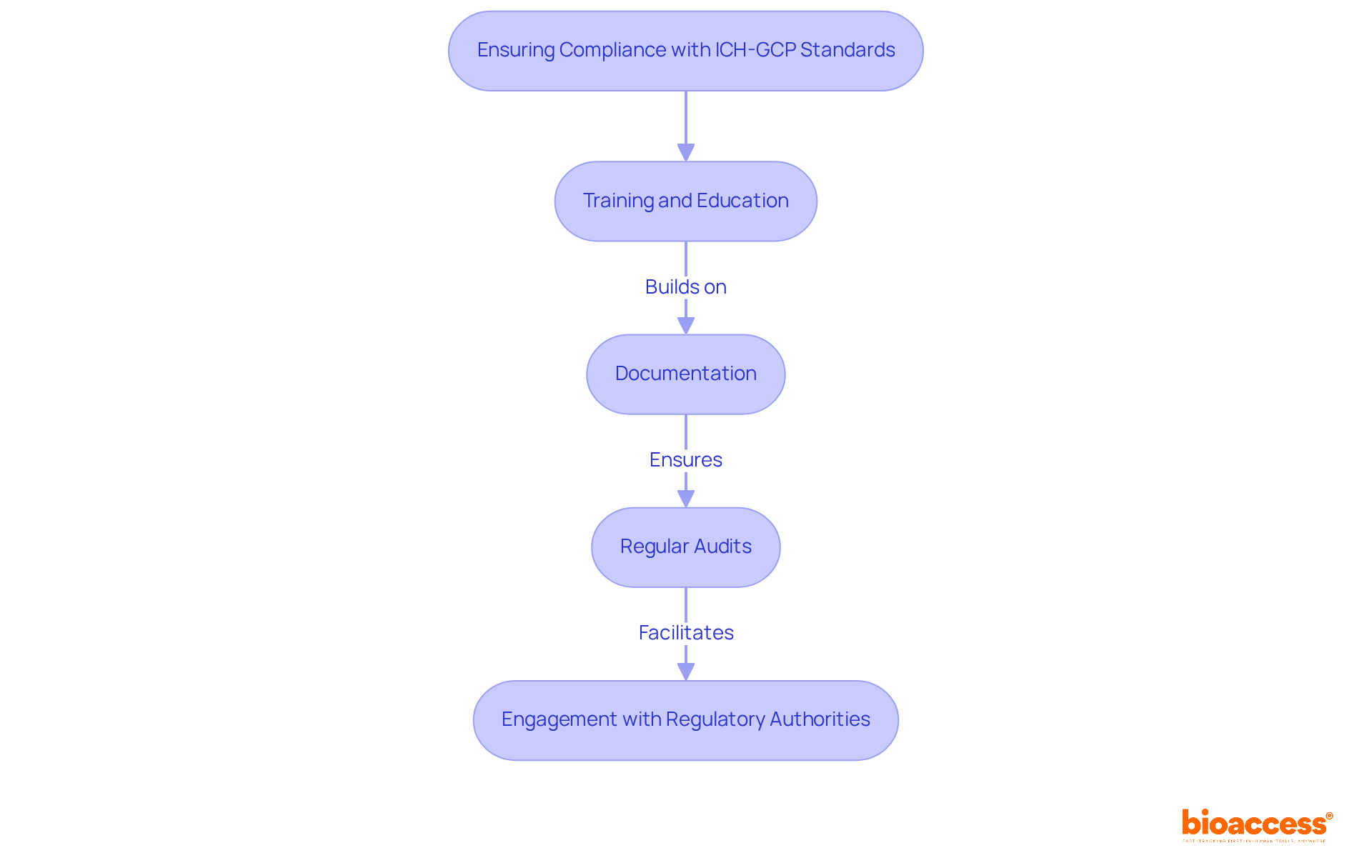
In Brazil, adherence to ICH-GCP standards and local regulations is not just a requirement; it’s a cornerstone for the success of First-in-Human investigations. Here are key practices to ensure adherence:
- Training and Education: Implement comprehensive training programs for all trial staff on ICH-GCP guidelines and local regulations, emphasizing ethical considerations and responsibilities in clinical research. Regular refresher training should be conducted every two years to maintain high standards of compliance.
- Documentation: Maintain meticulous records of all trial-related activities, including informed consent, protocol amendments, and adverse event reporting. Essential sponsor-specific documents must be retained according to applicable regulatory requirements, ensuring that this documentation is critical for regulatory inspections and audits.
- Regular Audits: Without regular audits, compliance issues may go unnoticed, jeopardizing the integrity of the study. Conduct internal audits to assess compliance with ICH-GCP and local regulations. This proactive approach can identify potential issues before they escalate, ensuring that study processes are proportionate to risks and data significance, as outlined in the latest ICH E6(R3) guidelines.
- Engagement with Regulatory Authorities: Foster open communication with ANVISA and local ethics committees to stay informed about regulatory changes and expectations. This engagement can facilitate smoother authorizations, enhance study credibility, and ensure that studies are conducted in accordance with ethical principles and regulatory requirements. With bioaccess®’s expertise, sponsors can navigate the approval process more efficiently, benefiting from 4-8 week ethics approvals and FDA/EMA-ready trial data.
Prioritizing compliance ensures the integrity of clinical trials. It protects participant safety and enhances the likelihood of successful regulatory submissions, ultimately accelerating the path to market for innovative therapies. Ultimately, a steadfast commitment to compliance can be the difference between a trial’s success and failure in the competitive landscape of clinical research.
Conclusion
Navigating the complexities of clinical trial management in Brazil is not just a challenge; it’s an opportunity for those who are prepared. To master clinical trial management in Brazil, one must:
- Command a thorough understanding of the regulatory framework
- Execute strategic site selection
- Implement effective patient recruitment
- Uphold rigorous compliance standards
Harnessing the distinctive features of Brazil’s clinical trial landscape can significantly enhance the chances of success in conducting innovative research.
Key insights from this article emphasize the necessity of:
- Engaging with ANVISA and local ethics committees early in the process to effectively navigate the regulatory landscape
- Streamlining site selection by utilizing pre-qualified research sites to drastically reduce activation times
- Employing targeted patient recruitment strategies that are essential for achieving enrollment goals
Furthermore, incorporating Early Feasibility Studies not only mitigates risks but also refines trial designs, leading to more reliable outcomes.
As clinical trials in Brazil evolve, embracing these best practices will elevate study success rates and enhance Brazil’s standing as a premier location for clinical research. By prioritizing compliance and leveraging local expertise, sponsors can accelerate their development timelines and bring innovative therapies to market more efficiently. Ultimately, the proactive engagement with Brazil’s regulatory landscape will not only enhance trial outcomes but also set a benchmark for future innovations in global healthcare.
Frequently Asked Questions
What is the primary regulatory authority overseeing clinical trials in Brazil?
The primary regulatory authority overseeing clinical trials in Brazil is ANVISA, which manages the authorization and supervision of studies involving human participants.
What are the key regulations that govern clinical trials in Brazil?
Key regulations include Resolution 466/2012, which outlines ethical considerations and mandates ethical committee approval, and Law No. 14.874/2024, which simplifies the authorization process and reduces application timelines.
What does Resolution 466/2012 require for clinical trials?
Resolution 466/2012 requires ethical committee approval before initiating trials, emphasizes informed consent, and ensures the protection of participants’ rights throughout the research process.
How does Law No. 14.874/2024 impact the authorization process for clinical trials in Brazil?
Law No. 14.874/2024 aims to simplify the authorization process by reducing application timelines from an average of 265 days to a firm limit of 90 business days for research involving human subjects.
What is a Clinical Trial Application (CTA) and what should it include?
A Clinical Trial Application (CTA) is a submission prepared by sponsors that includes detailed study protocols, informed consent forms, and evidence of preclinical data, which are essential for clinical trial management in Brazil.
How can sponsors enhance their chances of timely approvals for clinical trials in Brazil?
Sponsors can enhance their chances of timely approvals by actively engaging with local ethics committees (CEPs) and the National Commission for Ethics in Research (CONEP) early in the process, as well as by leveraging the new framework that allows for simultaneous review by ANVISA and ethics committees.
What benefits does the new regulatory framework offer for clinical trials in Brazil?
The new regulatory framework offers benefits such as expedited approvals, reduced time to market for innovative therapies, and enhanced credibility of the study through early engagement with regulatory bodies and ethics committees.
List of Sources
- Understand Brazil’s Regulatory Framework for Clinical Trials
- Brazil Just Changed The Rules For Clinical Trial Approvals — What Sponsors Need To Know | bioaccess® (https://bioaccessla.com/blog/brazil-anvisa-parallel-review-clinical-trial-approvals-2026)
- How Brazil’s New Law Is Redefining The Country’s Clinical Research (https://clinicalleader.com/doc/how-brazil-s-new-law-is-redefining-the-country-s-clinical-research-0001)
- Brazil Clinical Trials Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/clinical-trials-market/brazil)
- Brazil Clinical Trials Market Size, Share | Analysis Report [2032] (https://fortunebusinessinsights.com/brazil-clinical-trials-market-112310)
- Brazil Cuts Clinical Trial Approval Timeline to 90 Days | bioaccess®: Fast-Tracking First-in-Human Trials, Anywhere posted on the topic | LinkedIn (https://linkedin.com/posts/bioaccess_clinicaltrials-brazil-anvisa-activity-7447976698958106627-98MW)
- Optimize Site Selection and Patient Recruitment Strategies
- How Many Vials for Brazil? Efficiency+ Brings Statistical Innovation to Trial Operations (https://asabiopreport.substack.com/p/how-many-vials-for-brazil-efficiency)
- Brazil Clinical Trial Patient Recruitment Services Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/clinical-trial-patient-recruitment-services-market/brazil)
- Brazil Clinical Trials Market Size & Future Growth 2035 (https://expertmarketresearch.com/reports/brazil-clinical-trials-market)
- Best Practices For First In Human Trials In Brazil: Strategies For Success | bioaccess® (https://bioaccessla.com/blog/best-practices-for-first-in-human-trials-in-brazil-strategies-for-success)
- Incorporate Early Feasibility Studies to Enhance Trial Outcomes
- Early feasibility studies on devices: “doing it sooner” to avoid trial failure | Meditrial (https://meditrial.net/2022/09/early-feasibility-studies-on-devices-doing-it-sooner-to-avoid-trial-failure)
- Early Feasibility Studies: Top 6 Considerations | MED Institute (https://medinstitute.com/blog/early-feasibility-studies-top-6-considerations)
- Early Feasibility Studies (EFS) Program (https://fda.gov/medical-devices/investigational-device-exemption-ide/early-feasibility-studies-efs-program)
- Clinical Trial Success Rates by Therapeutic Area 2026-27 Data Analysis (https://ccrps.org/clinical-research-blog/clinical-trial-success-rates-by-therapeutic-area-2026-27-data-analysis)
- Ensure Compliance with ICH-GCP Standards and Local Regulations
- 7 Key Responsibilities for ICH‑GCP Compliance (https://cognivia.com/ich-gcp-compliance-responsibilities)
- The Importance of ICH GCP (https://ccrps.org/clinical-research-blog/the-importance-of-ich-gcp)
- Discover the 11 Principles of ICH GCP E6(R3) | Medicover MICS (https://medicover-mics.com/the-11-principles-of-ich-good-clinical-practice)
- What Is ICH GCP, And How Does It Impact Clinical Planning? (https://clinicalleader.com/doc/what-is-ich-gcp-and-how-does-it-impact-clinical-planning-0001)

Leave a Reply