Introduction
Navigating the complexities of clinical trial applications (CTAs) for biologics is no small feat, especially as regulatory landscapes evolve and grow more intricate. Submitting a CTA to ALIMS isn’t just a formality; it’s a pivotal step that can significantly influence the success of a clinical study. This guide outlines essential steps and best practices to ensure a smooth submission process, while also addressing common pitfalls that can derail even the most meticulously prepared applications. What strategies can enhance the likelihood of approval and streamline the path to clinical research success?
Understanding the Medtech landscape is crucial, as it presents unique challenges that require innovative solutions. Bioaccess plays a vital role in navigating these challenges, offering insights and support that can make a difference in the approval process. By collaborating effectively and leveraging expertise, stakeholders can significantly improve their chances of success in clinical research.
In summary, the importance of collaboration cannot be overstated. As you move forward, consider the next steps that will position your application for success.
Understand Clinical Trial Applications for Biologics
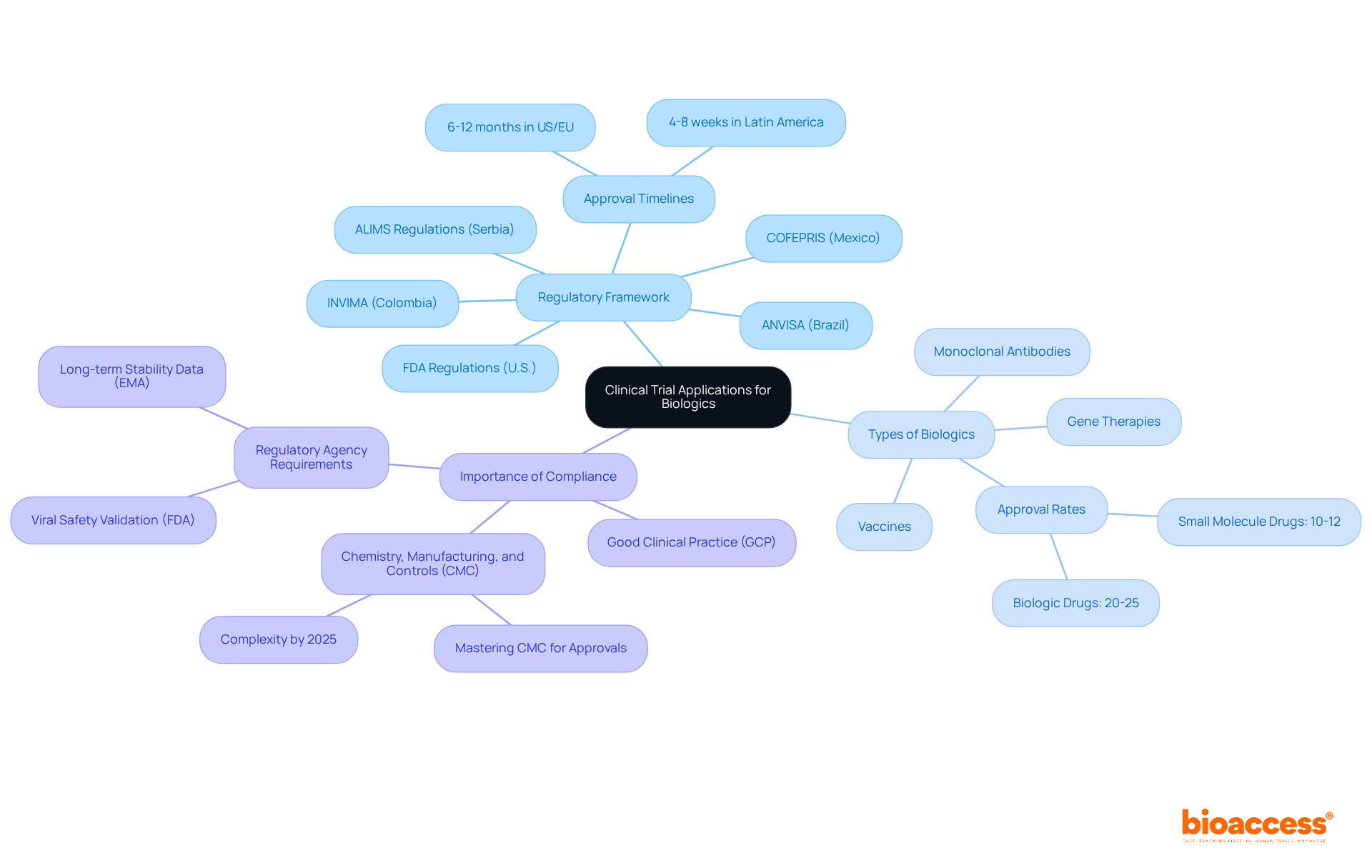
The represents formal requests submitted to regulatory authorities, seeking permission to conduct clinical studies. Understanding the purpose and components of the is crucial for ensuring compliance and achieving successful approval. Key elements include:
- : Familiarize yourself with the specific regulations governing biologics in your jurisdiction, such as the FDA in the U.S. or ALIMS in Serbia. In Latin America, bioaccess® collaborates with various regulatory agencies, including INVIMA (Colombia), ANVISA (Brazil), and COFEPRIS (Mexico), achieving -significantly faster than the 6-12 months typically required in the US/EU.
- Types of Biologics: Recognize the different categories of biologics, including monoclonal antibodies, vaccines, and gene therapies, as each may have unique requirements. For instance, biologic drugs boast an , which is notably higher than the 10%-12% approval rate for small molecule drugs.
- : Highlight the necessity of adhering to to ensure the integrity of the study and the safety of participants. Mastering Chemistry, Manufacturing, and Controls (CMC) for biologics is essential for achieving approvals and maintaining compliance throughout product lifecycles.
Stay informed about ALIMS regulations for biologics clinical studies, particularly concerning , as these guidelines evolve to reflect global harmonization efforts. By 2025, CMC requirements for biologics are expected to become increasingly complex, necessitating comprehensive data on product characterization and quality assurance.
By grasping these foundational concepts and understanding how bioaccess® can expedite your , you will be better prepared to navigate the complexities of the application process. This preparation ultimately enhances the likelihood of successful study outcomes.
Gather Required Documentation for CTA Submission
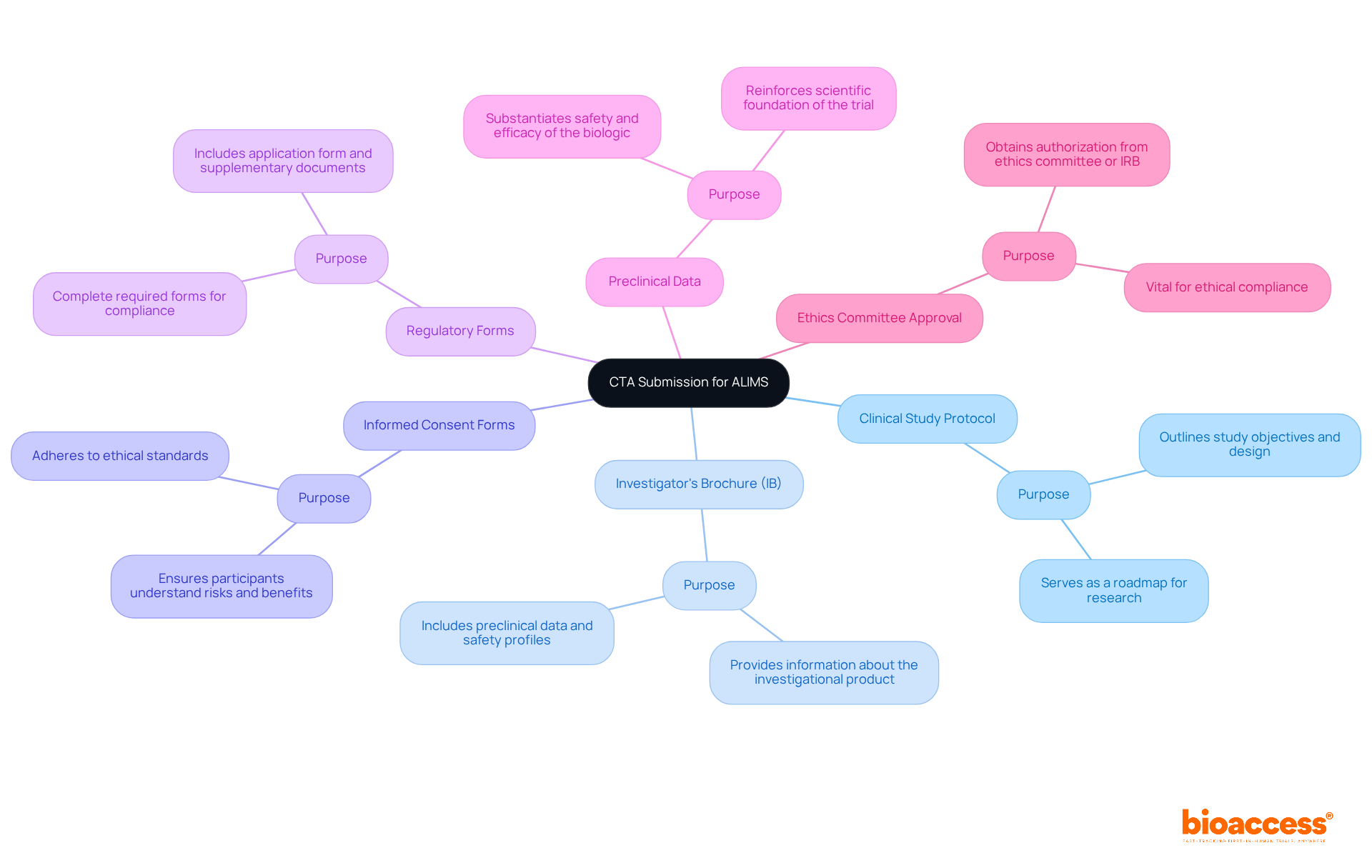
Assembling a comprehensive collection of materials is essential to ensure a to ALIMS for biologics. This structured guide will help you navigate the process effectively:
- : This essential document outlines the study’s objectives, design, methodology, and statistical considerations, serving as a roadmap for the research.
- : A critical resource that provides comprehensive information about the investigational product, including and safety profiles, which are vital for informed decision-making.
- : These forms must be clear and adhere to ethical standards, ensuring participants fully understand the study’s risks and benefits before enrollment.
- : Complete all required mandated by ALIMS for CTA submission to ALIMS for biologics, including the application form and any supplementary documents necessary for compliance.
- : Incorporate results from preclinical studies that substantiate the safety and efficacy of the biologic, reinforcing the trial’s scientific foundation.
- : Obtain authorization from an ethics committee or institutional review board (IRB) before presenting, as this is vital for ethical compliance.
By diligently assembling these documents, you significantly enhance the chances of a seamless submission process. Statistics indicate that common documentation errors can lead to delays, underscoring the importance of thorough preparation. Successful for biologics exemplify clarity and adherence to regulatory standards, further emphasizing the necessity of a well-structured CTA submission to ALIMS for biologics. As noted by clinical research experts, a robust is vital for guiding the trial and ensuring compliance with regulatory expectations.
Submit the Clinical Trial Application to ALIMS
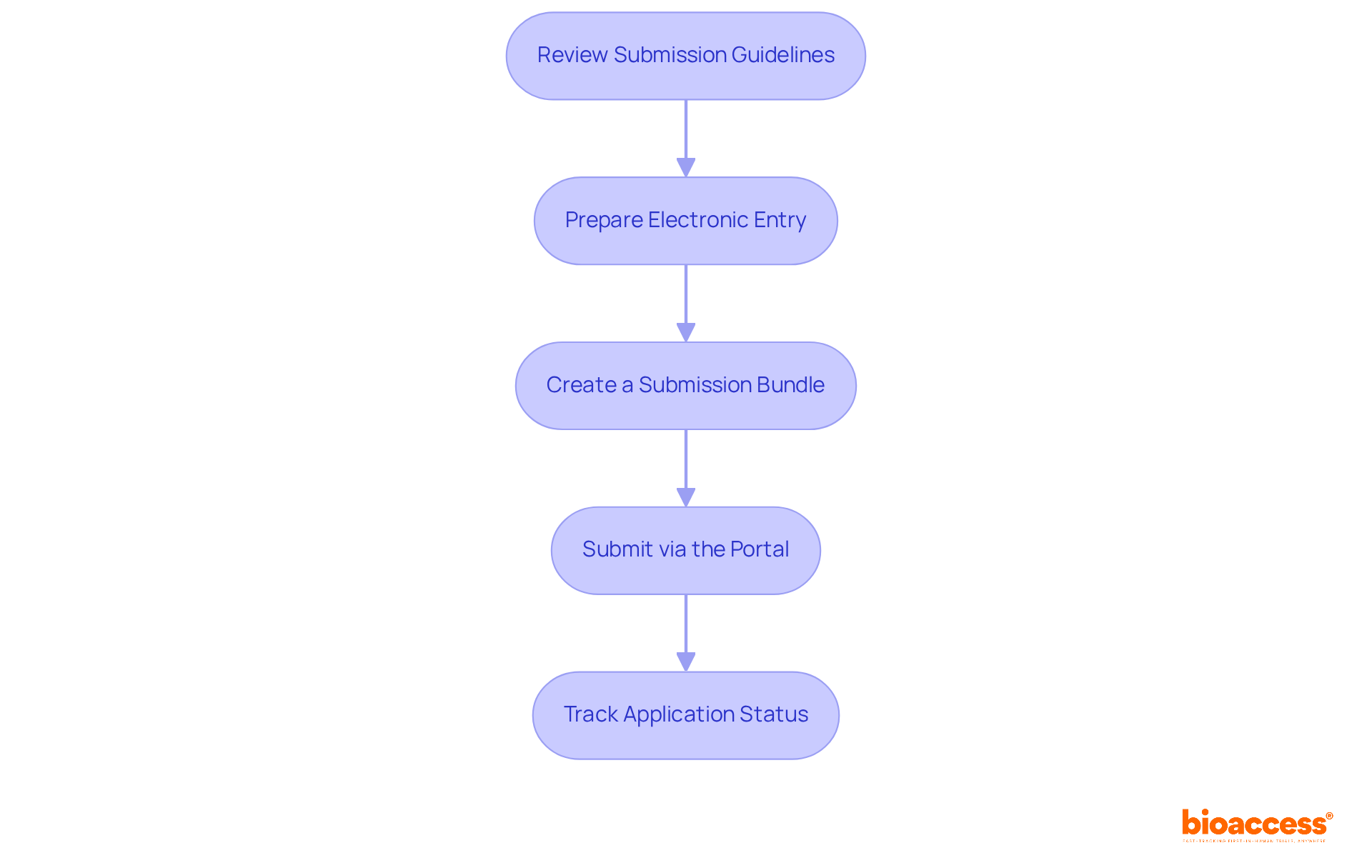
To successfully submit your to ALIMS, follow these :
- Review : Thoroughly examine the specific to ensure full compliance with all requirements. Non-compliance may result in a Not Satisfactory Notice (NSN) for significant deficiencies during the review process.
- : Most entries must be made electronically. Arrange all files in line with specifications, usually in eCTD format, to enable a seamless review process.
- : Compile all necessary documents into a cohesive collection. This should include a cover letter that succinctly summarizes the application and highlights key points.
- Submit via the Portal: Access the portal for uploading your package. Confirm that you receive an acknowledgment of your application to ensure it is officially logged.
- : After sending, actively monitor the status of your application through the ALIMS portal. Be prepared to promptly address any queries or requests for additional information from the review team. Note that all CTAs are subject to a 30-day default review period from the date of receipt of the completed application.
By following these steps, you can improve the efficiency and accuracy of your CTA submission to ALIMS for biologics, ultimately facilitating a smoother approval process. Previous analyses indicated an industry benchmark for , underscoring the importance of following these guidelines.
Troubleshoot Common Issues in CTA Submission
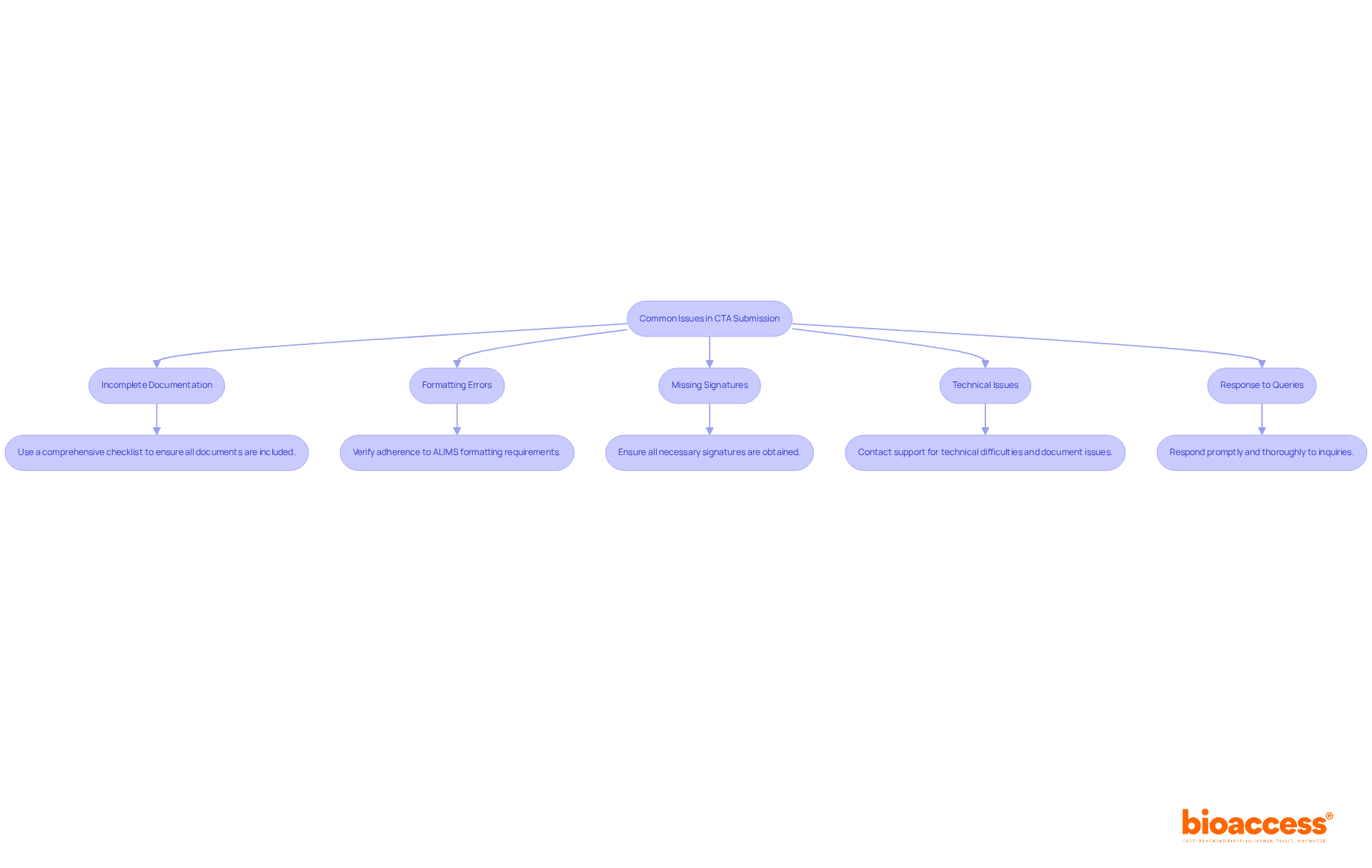
Despite careful preparation, challenges can arise during the CTA filing process. Understanding these is crucial for . Here are some strategies to troubleshoot effectively:
- : Confirm that all necessary documents are included in your entry. Utilize a to ensure thoroughness before sending, as incomplete entries can lead to significant delays. As Sonal Gadekar points out, ” can help life sciences companies refine their application strategies and reduce the risk of setbacks.”
- : Rigorously verify that all files adhere to ALIMS formatting requirements. Incorrect formats frequently cause submission delays, so double-checking can save valuable time.
- Missing Signatures: Ensure that all necessary signatures are obtained on forms and documents. The absence of required signatures can lead to outright rejection of your application, making a final review essential.
- : If you encounter technical difficulties, promptly reach out to support for assistance. Document any issues you experience for future reference, as this can help in resolving similar problems later.
- Response to Queries: Respond promptly and thoroughly to any inquiries regarding your application. Delays in your responses can prolong the review process, potentially impacting your project timeline. Keep in mind that the average response time for queries from ALIMS is crucial, as .
By proactively addressing these and emphasizing the importance of thorough documentation, you can significantly enhance the likelihood of a successful CTA submission to ALIMS for biologics.
Conclusion
Navigating the complexities of Clinical Trial Applications (CTAs) for biologics is essential for advancing innovative therapies. A comprehensive understanding of the submission process to ALIMS is crucial. Successful submissions hinge on grasping the regulatory framework, gathering the appropriate documentation, and meticulously following established guidelines. By mastering these elements, stakeholders can significantly enhance their chances of obtaining timely approvals for their clinical studies.
Key insights discussed throughout this article highlight the importance of compliance with Good Clinical Practice (GCP) guidelines and the necessity of assembling a complete set of required documents. Recognizing common pitfalls, such as incomplete documentation and formatting errors, is crucial for minimizing delays and maximizing the likelihood of a successful application. Additionally, employing a structured approach to address potential issues can streamline the submission experience.
Ultimately, the successful submission of CTAs to ALIMS for biologics not only facilitates the advancement of innovative therapies but also contributes to the broader landscape of medical research. By adhering to best practices and continuously refining submission strategies, organizations can play a pivotal role in bringing vital biologic treatments to market. Emphasizing thorough preparation and proactive problem-solving will enhance individual project outcomes and foster a more efficient regulatory environment for future clinical trials.
Frequently Asked Questions
What is a CTA submission for biologics?
A CTA submission for biologics is a formal request submitted to regulatory authorities, seeking permission to conduct clinical studies involving biologic products.
Why is understanding the CTA submission important?
Understanding the CTA submission is crucial for ensuring compliance with regulations and achieving successful approval for clinical studies.
What regulatory frameworks should be considered for biologics?
It is important to familiarize yourself with the specific regulations governing biologics in your jurisdiction, such as the FDA in the U.S. or ALIMS in Serbia, as well as other regulatory agencies in Latin America like INVIMA, ANVISA, and COFEPRIS.
What are the different types of biologics?
Different categories of biologics include monoclonal antibodies, vaccines, and gene therapies, each of which may have unique regulatory requirements.
What is the approval rate for biologic drugs compared to small molecule drugs?
Biologic drugs have an approval rate of approximately 20%-25%, which is higher than the 10%-12% approval rate for small molecule drugs.
Why is compliance with Good Clinical Practice (GCP) guidelines important?
Compliance with GCP guidelines is essential to ensure the integrity of the study and the safety of participants involved in clinical trials.
What is the significance of Chemistry, Manufacturing, and Controls (CMC) for biologics?
Mastering CMC for biologics is essential for achieving regulatory approvals and maintaining compliance throughout the product lifecycle.
How are ALIMS regulations for biologics expected to evolve?
By 2025, CMC requirements for biologics are expected to become more complex, necessitating comprehensive data on product characterization and quality assurance.
How can bioaccess® assist with the CTA submission process?
Bioaccess® can expedite clinical studies and market entry in Latin America, helping to navigate the complexities of the CTA submission to ALIMS for biologics.
List of Sources
- Understand Clinical Trial Applications for Biologics
- CMC for Biologics Explained: Ultimate Guide to Quality, Compliance, and Regulatory Lessons – PharmaRegulatory.in – India’s Regulatory Knowledge Hub (https://pharmaregulatory.in/cmc-for-biologics-explained-ultimate-guide-to-quality-compliance-and-regulatory-lessons)
- Clinical Trial Success Rates: How Many Drugs Make It to Market? (Latest Approval Stats) (https://patentpc.com/blog/clinical-trial-success-rates-how-many-drugs-make-it-to-market-latest-approval-stats)
- New FDA Guidance: Further Enhancing Clinical Trial Participation – EmVenio (https://emvenio.com/news-and-resources/new-fda-guidance-clinical-trial-participation)
- FDA policy sets new clinical trial requirements for drug approvals (https://europeanpharmaceuticalreview.com/news/271500/fda-policy-new-clinical-trial-requirements-us-drug-approvals)
- Navigating Regulatory Challenges in Biologics: A Case Study in Overcoming Clinical Holds (https://veristat.com/blog/navigating-regulatory-challenges-in-biologics-a-case-study-in-overcoming-clinical-holds)
- Gather Required Documentation for CTA Submission
- FDA policy sets new clinical trial requirements for drug approvals (https://europeanpharmaceuticalreview.com/news/271500/fda-policy-new-clinical-trial-requirements-us-drug-approvals)
- agingresearch.org (https://agingresearch.org/blog/new-york-times-article-on-clinical-trials-features-quote-from-the-alliance-for-aging-research)
- Clinical Trials Quotes (2 quotes) (https://goodreads.com/quotes/tag/clinical-trials)
- sofpromed.com (https://sofpromed.com/what-are-the-documents-required-for-clinical-trial-applications-to-regulatory-authorities-in-europe)
- Submit the Clinical Trial Application to ALIMS
- Benchmarking R&D success rates of leading pharmaceutical companies: an empirical analysis of FDA approvals (2006–2022) (https://sciencedirect.com/science/article/pii/S1359644625000042)
- Guidance Document For Clinical Trial Sponsors: Clinical Trial Applications – Canada.ca (https://canada.ca/en/health-canada/services/drugs-health-products/drug-products/applications-submissions/guidance-documents/clinical-trials/clinical-trial-sponsors-applications.html)
- antidote.me (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- Troubleshoot Common Issues in CTA Submission
- 94% of compliance officers say: No documentation? It’s not done (https://ama-assn.org/practice-management/physician-health/94-compliance-officers-say-no-documentation-it-s-not-done)
- Importance of Documentation – Expert and Influential Leader Quotes : Erase Your Risk with Proper Documentation (https://forensicnotes.com/importance-of-documentation-expert-and-influential-leader-quotes)
- acdis.org (https://acdis.org/articles/note-associate-editorial-director-quotes-note)
- antidote.me (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- How to Avoid Common Causes of Regulatory Submission Rejections (https://freyrsolutions.com/blog/common-causes-of-regulatory-submission-rejections-and-how-to-avoid-them)

Leave a Reply