Introduction
Navigating the approval process for medical devices in India presents a complex challenge, significantly shaped by the stringent regulations imposed by the India Food and Drug Administration (FDA). Stakeholders aiming to penetrate this dynamic market must grasp the crucial role of the Central Drugs Standard Control Organization (CDSCO) in upholding rigorous safety and efficacy standards. Yet, the journey to securing approval is riddled with obstacles, including extensive documentation requirements and potential delays in review timelines.
How can companies streamline their submissions and effectively address these hurdles to ensure timely market access? Understanding these challenges is essential for any organization looking to thrive in the Medtech landscape. By leveraging insights into the regulatory environment and adopting strategic approaches, stakeholders can enhance their chances of success.
Understand the Role of the India Food and Drug Administration
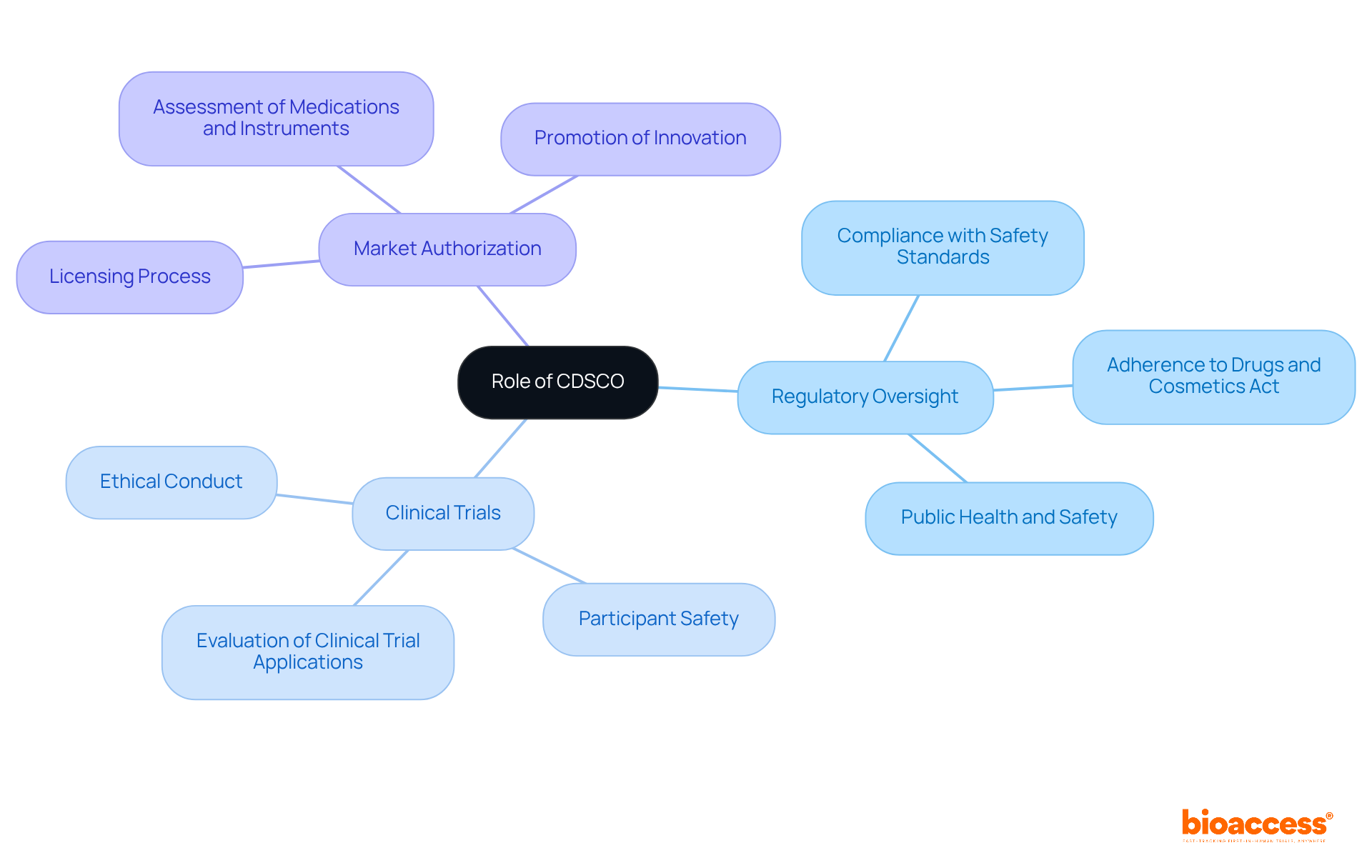
The Central Drugs Standard Control Organization (CDSCO), which operates under the , plays a pivotal role in overseeing the endorsement of medications and medical instruments. This ensures compliance with rigorous safety, effectiveness, and quality criteria before they enter the market. Understanding the is essential for stakeholders aiming to navigate the approval process effectively.
Regulatory Oversight: The CDSCO supervises the , ensuring adherence to the . The oversight of the is crucial for maintaining public health and safety. By prioritizing compliance, the organization fosters a trustworthy environment for both consumers and industry players.
Clinical Trials: The organization evaluates , emphasizing ethical conduct and participant safety. This scrutiny is vital for fostering trust in the . Stakeholders must recognize that a thorough understanding of these regulations can significantly enhance their chances of successful submissions.
Market Authorization: After a comprehensive assessment, the regulatory body issues licenses for medications and instruments, a critical step for companies looking to enter the Indian market. Successful certifications of medical instruments highlight the commitment of the to promoting innovation while ensuring regulatory adherence.
Industry leaders stress that a comprehensive understanding of the CDSCO’s functions can greatly improve the chances of successful submissions, ultimately leading to quicker market access and better patient outcomes. With bioaccess® providing extensive -including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting-stakeholders can leverage expert support to navigate the efficiently.
The ” (GSR-104 E) implemented by the regulatory body streamlines the method for conducting bioavailability (BA) and bioequivalence (BE) studies, reflecting the organization’s commitment to modernizing regulatory practices. Statistics on FDA authorization timelines in India indicate that timely submissions can lead to quicker market access, underscoring the importance of understanding the CDSCO’s operational framework.
Gather Required Documentation for Medical Device Approval
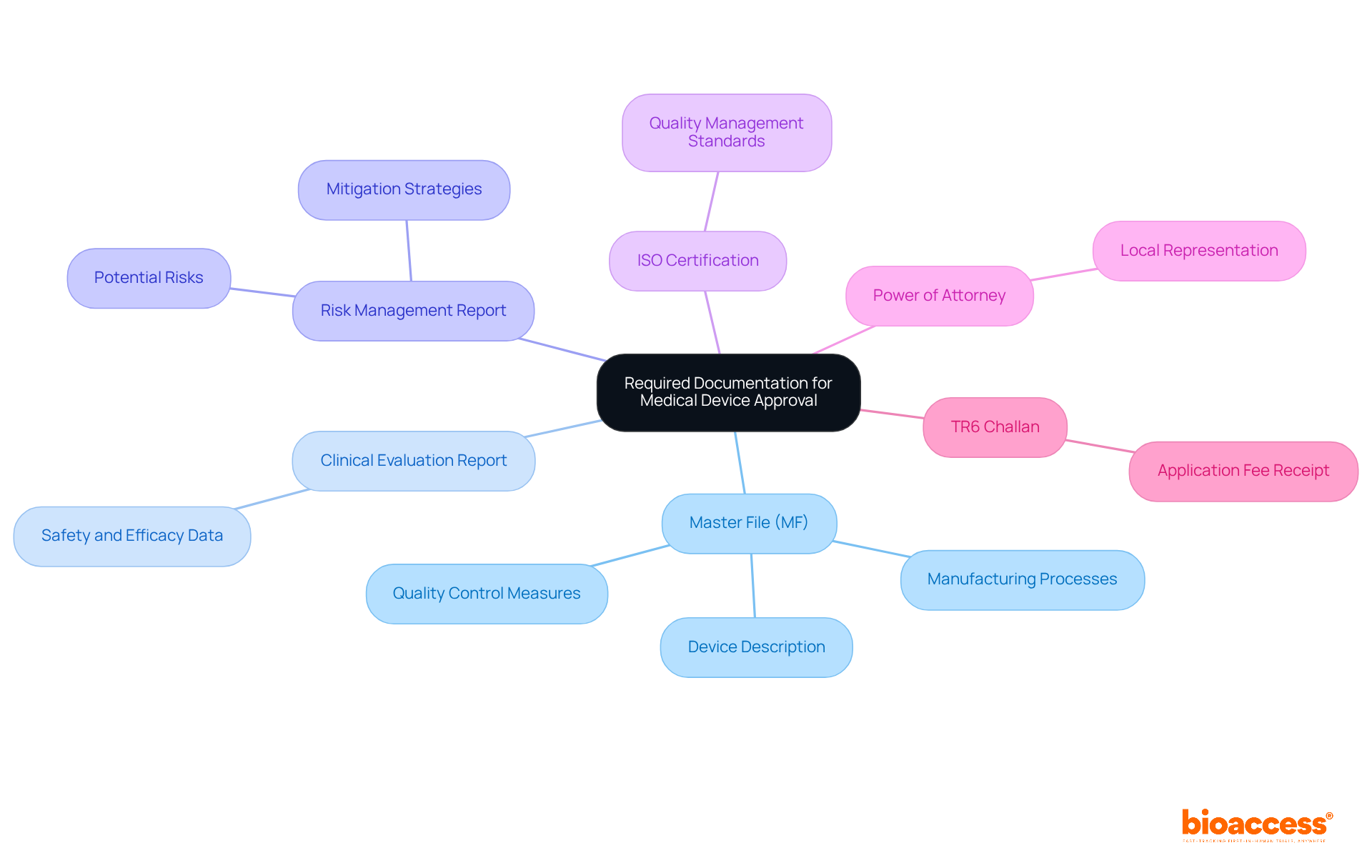
To effectively navigate the in India, it’s crucial to gather comprehensive documentation, with a particular focus on the . This file is essential as it contains detailed information about the apparatus, including its design, manufacturing processes, and quality control measures. Experts emphasize that a well-prepared DMF can significantly enhance the chances of prompt approval, as nearly 30% of in India face delays due to documentation issues.
In addition to the DMF, the following documents are necessary:
- : This report demonstrates the safety and efficacy of the device based on , which is vital for .
- : This documentation outlines potential risks associated with the equipment and the strategies for their mitigation, ensuring compliance with safety standards.
- : Proof of compliance with international quality management standards is often required for , reinforcing the manufacturer’s commitment to quality.
- Power of Attorney: If relevant, this document permits a local representative to act on behalf of the manufacturer during the approval procedure, facilitating smoother communication with regulatory authorities.
- TR6 Challan: A fee receipt confirming payment of the application fee, which is a prerequisite for processing the submission.
Having these documents prepared in advance will streamline the submission process and help avoid unnecessary delays, ultimately enhancing the chances of successful submissions to the India FDA in 2026.
Submit Your Application to the India FDA
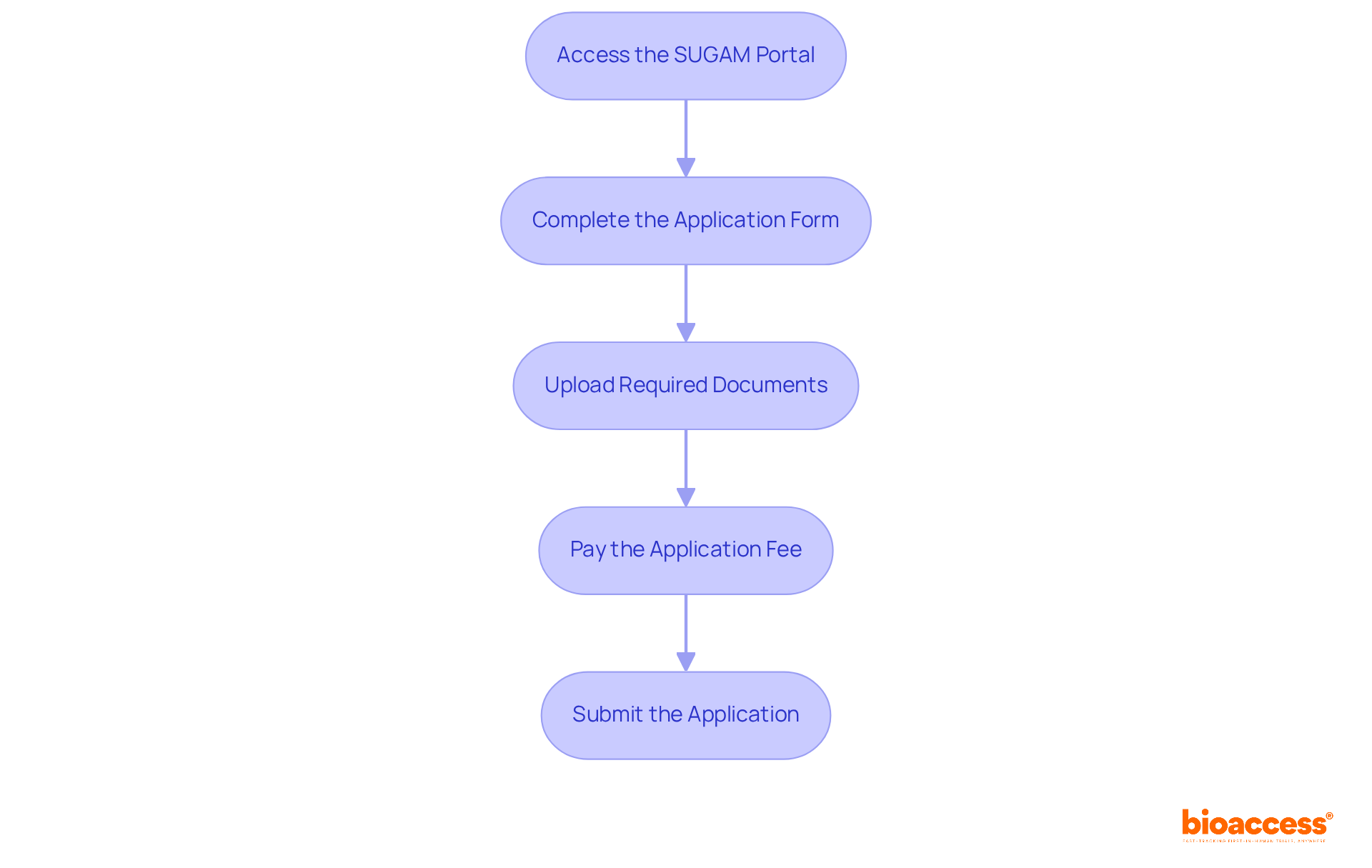
To successfully submit your application to the India FDA, follow these essential steps:
- Access the : Start by visiting the online submission portal, SUGAM. If you don’t have an account, create one to proceed.
- Complete the Application Form: Fill out the MD-14 application form for medical equipment accurately, ensuring all product details are correct. As the Central Drugs Standard Control Organization states, ‘ means obtaining to manufacture, import, or sell an item in India.’
- Upload Required Documents: Attach all necessary documentation, including the , , and any additional supporting materials.
- Pay the : Submit the TR6 Challan as proof of payment. The varies by classification, with Class A items typically priced around ₹5,000, while Classes B and C range from ₹10,000 to ₹50,000. Understanding these fees is crucial, as “.”
- Submit the Application: Before finalizing your submission, thoroughly review all information for accuracy and completeness. Once confirmed, . Utilizing the , enhancing transparency and efficiency in .
Upon submission, you will receive an acknowledgment receipt, which is vital for tracking your application’s status. Regulatory advisors emphasize that using the streamlines the process, improving transparency and efficiency in .
Navigate Challenges in the Approval Process
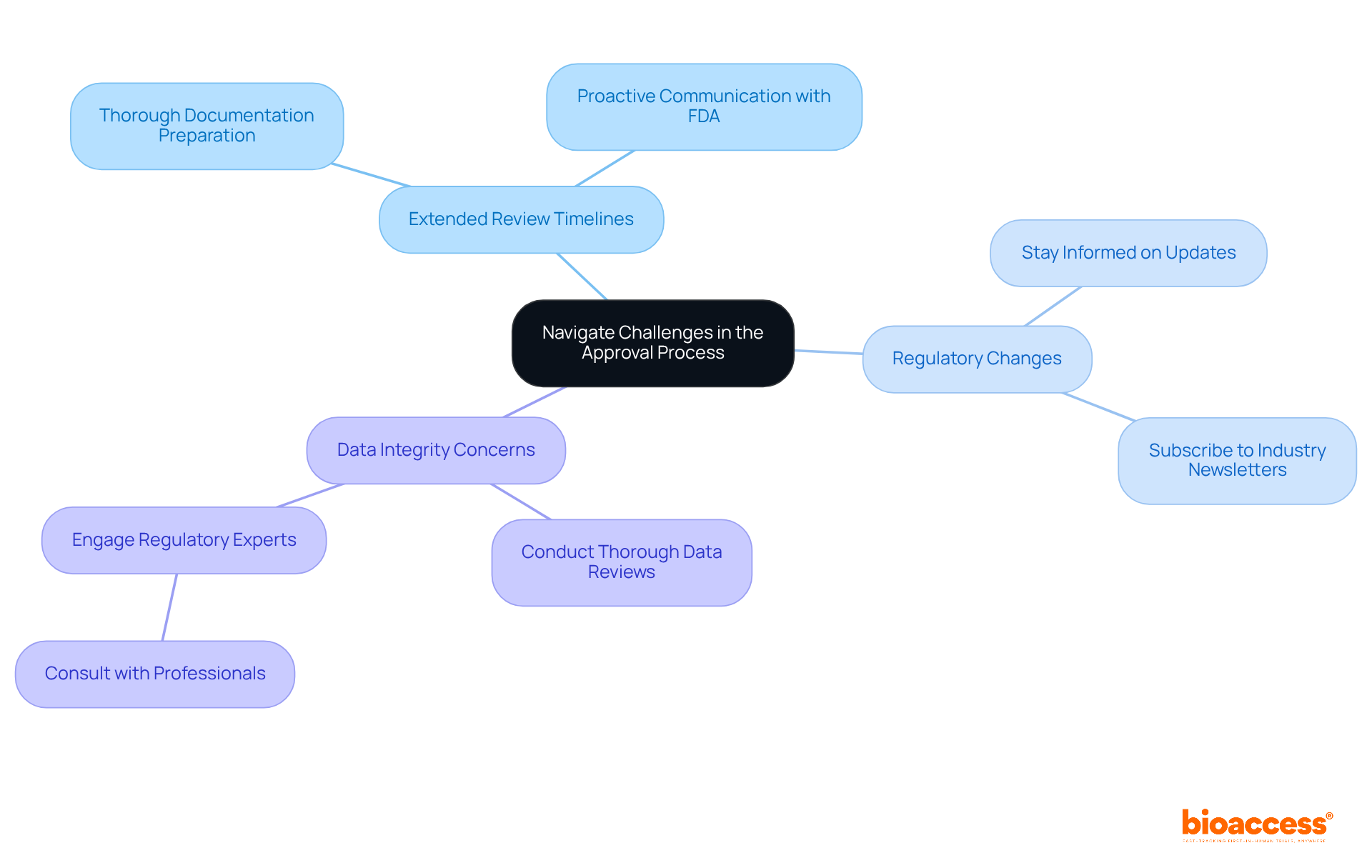
The approval procedure for medical devices in India poses several challenges that can significantly impact timelines. Understanding these obstacles is crucial for anyone involved in . Here are key challenges and strategies to navigate them effectively:
- : The India FDA often experiences delays in reviewing applications, with average timelines extending beyond initial expectations. Recent reports indicate that review times can vary widely, with some applications facing delays of several months due to incomplete submissions or regulatory complexities. To alleviate these delays, ensure that all documentation is thoroughly prepared and precise before submission. This proactive approach can avert requests for additional information that prolong the review duration.
- : The regulatory landscape is dynamic, with frequent updates that can affect application requirements. Staying informed about these changes is essential. Regularly check the (CDSCO) website and subscribe to industry newsletters to receive timely updates on regulatory shifts that may impact your application.
Effective communication with the India Food and Drug Administration is crucial. If issues arise during the review, do not hesitate to reach out for clarification or support. Engaging with the FDA early can help resolve potential issues and simplify the authorization pathway.
- : The integrity of submitted data is paramount. Inaccuracies can lead to significant delays or outright rejections. Conduct thorough reviews of all data and documentation prior to submission to ensure compliance with FDA standards.
Additionally, leveraging the expertise of professionals like Ana Criado, Director of at bioaccess, can provide valuable insights into navigating these challenges. With her extensive experience in and biomedical engineering, she can assist applicants in . Moreover, bioaccess offers comprehensive , including feasibility studies, site selection, , trial organization, import permits, project oversight, and reporting. These services can greatly enhance the efficiency and success of the authorization cycle.
By anticipating these challenges and preparing accordingly, applicants can significantly improve their chances of successfully navigating the .
Conclusion
Understanding the complexities of the India Food and Drug Administration (FDA) approval process is crucial for stakeholders looking to successfully bring medical devices to market. The Central Drugs Standard Control Organization (CDSCO) plays a pivotal role in ensuring that products meet stringent safety and efficacy standards. By mastering this approval process, stakeholders can significantly enhance their chances of timely market access and improved patient outcomes.
Key insights reveal the critical importance of thorough documentation, including the Device Master File and Clinical Evaluation Report, which are essential for successful submissions. Additionally, navigating the challenges posed by extended review timelines and regulatory changes necessitates proactive preparation and effective communication with the FDA. Engaging expert services, such as those provided by bioaccess, can further streamline the process and help mitigate potential obstacles.
Ultimately, adopting a well-informed approach to the India FDA approval process not only facilitates compliance but also fosters innovation within the medical device sector. Stakeholders are encouraged to remain vigilant, continuously update their knowledge of regulatory requirements, and utilize available resources to effectively navigate this complex landscape. By embracing these strategies, they will not only increase the likelihood of successful applications but also contribute to the advancement of healthcare solutions in India.
Frequently Asked Questions
What is the Central Drugs Standard Control Organization (CDSCO)?
The CDSCO operates under the India Food and Drug Administration and is responsible for overseeing the endorsement of medications and medical instruments, ensuring they meet safety, effectiveness, and quality criteria before entering the market.
What is the role of the CDSCO in regulatory oversight?
The CDSCO supervises the approval process for new drugs and medical devices, ensuring compliance with the Drugs and Cosmetics Act, which is crucial for maintaining public health and safety.
How does the CDSCO evaluate clinical trials?
The organization evaluates clinical trial applications with an emphasis on ethical conduct and participant safety, which is vital for fostering trust in the clinical research process.
What is the process for market authorization by the CDSCO?
After a comprehensive assessment, the CDSCO issues licenses for medications and medical instruments, which is essential for companies seeking to enter the Indian market.
How can understanding the CDSCO’s functions benefit stakeholders?
A comprehensive understanding of the CDSCO’s functions can significantly improve the chances of successful submissions, leading to quicker market access and better patient outcomes.
What services does bioaccess® provide to assist stakeholders?
Bioaccess® offers extensive clinical trial management services, including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting.
What are the ‘New Drugs and Clinical Trials Rules-2018’?
These rules implemented by the CDSCO streamline the process for conducting bioavailability (BA) and bioequivalence (BE) studies, reflecting the organization’s commitment to modernizing regulatory practices.
Why is it important to understand the CDSCO’s operational framework?
Understanding the CDSCO’s operational framework is important because timely submissions can lead to quicker market access, as indicated by statistics on FDA authorization timelines in India.
List of Sources
- Understand the Role of the India Food and Drug Administration
- US FDA asks Indian pharma leaders to make quality, their top priority (https://m.economictimes.com/industry/healthcare/biotech/pharmaceuticals/us-fda-asks-indian-pharma-leaders-to-make-quality-their-top-priority/articleshow/34679309.cms)
- Introduction (https://cdsco.gov.in/opencms/opencms/en/About-us/Introduction)
- (PDF) An analysis of drug approvals in India over past 5 years (https://researchgate.net/publication/385179765_An_analysis_of_drug_approvals_in_India_over_past_5_years)
- INDIA’S GLOBAL LEADERSHIP IN US-FDA APPROVALS (https://pharmaknowledgecentre.com/indias-global-leadership-in-us-fda-approvals)
- India’s Unique Opportunity and Responsibility as Pharmacy to the World (https://fda.gov/news-events/fda-voices/indias-unique-opportunity-and-important-responsibility-pharmacy-world)
- Gather Required Documentation for Medical Device Approval
- 6 Important Quotes from Medtech Leaders (https://mddionline.com/business/6-important-quotes-from-medtech-leaders)
- 7 quotes from 2023 to guide you on the medtech market (https://tiinatyni.com/blogi/7-quotes-from-2023-to-guide-you-on-the-medtech-market)
- Regulatory Tips & Pointers from a Former FDA Reviewer (https://greenlight.guru/blog/regulatory-tips-pointers-from-a-former-fda-reviewer)
- Indian Regulatory Dossier preparation – Things to remember (https://morulaa.com/things-to-remember-while-preparing-your-regulatory-dossier)
- MFDS 2024 Medical Device Report: Key Statistics | Martin King posted on the topic | LinkedIn (https://linkedin.com/posts/martink2_mfds-the-2024-medical-device-approval-report-activity-7341797005997883395-G9ym)
- Submit Your Application to the India FDA
- CDSCO Medical Device Registration (https://pharmadocx.com/cdsco-medical-device-registration)
- Medical Device Registration in India: Fees, Docs & Process (https://registerkaro.in/medical-device-registration)
- Clinical research entities asked to register by April 1 (https://m.economictimes.com/industry/healthcare/biotech/pharmaceuticals/clinical-research-entities-asked-to-register-by-april-1/articleshow/118741465.cms)
- CDSCO Registration Fees & License Fees for Medical Devices in India 2025 (https://omcmedical.com/blog/cdsco-and-license-fees)
- CDSCO Medical Devices Approval Cost in India (https://artixio.com/post/cdsco-medical-device-approval-cost)
- Navigate Challenges in the Approval Process
- Working With An Unpredictable US FDA: Navigating Year Two Of A Changing Agency (https://insights.citeline.com/pink-sheet/pink-sheet-perspectives/working-with-an-unpredictable-us-fda-navigating-year-two-of-a-changing-agency-QGWLIIUHNNB3FCBNNPLK55G2QU)
- Medical Device FDA Approval Process [+Timeline] (https://greenlight.guru/blog/fda-medical-device-approval-process)
- Avoiding FDA Roadblocks: Key Insights from Review of CRLs on OpenFDA (https://lumanity.com/perspectives/avoiding-fda-roadblocks-key-insights-from-review-of-crls-on-openfda)
- Factors Influencing Clearance Time for Medical Devices (https://mddionline.com/medical-device-regulations/factors-influencing-fda-clearance-time-for-medical-devices-evolution-of-a-critical-regulatory-pathway)
- FDA Staffing Constraints Extend Planning and Review Timelines | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/fda-staffing-constraints-extend-planning-review-timelines)

Leave a Reply