Introduction
In the rapidly evolving landscape of medical technology, achieving ISO 13485 certification stands as a pivotal milestone for manufacturers seeking to ensure quality and compliance. This certification not only enhances operational efficiency but also strategically positions companies in a market where regulatory standards are continuously tightening.
However, with a multitude of audit firms available, how can Medtech startups and established companies effectively navigate the maze of options to identify the right partner for their certification journey?
This article presents a curated list of the top ISO 13485 audit firms in Mexico, each offering unique strengths and services that can significantly influence a company’s path to compliance and market readiness.
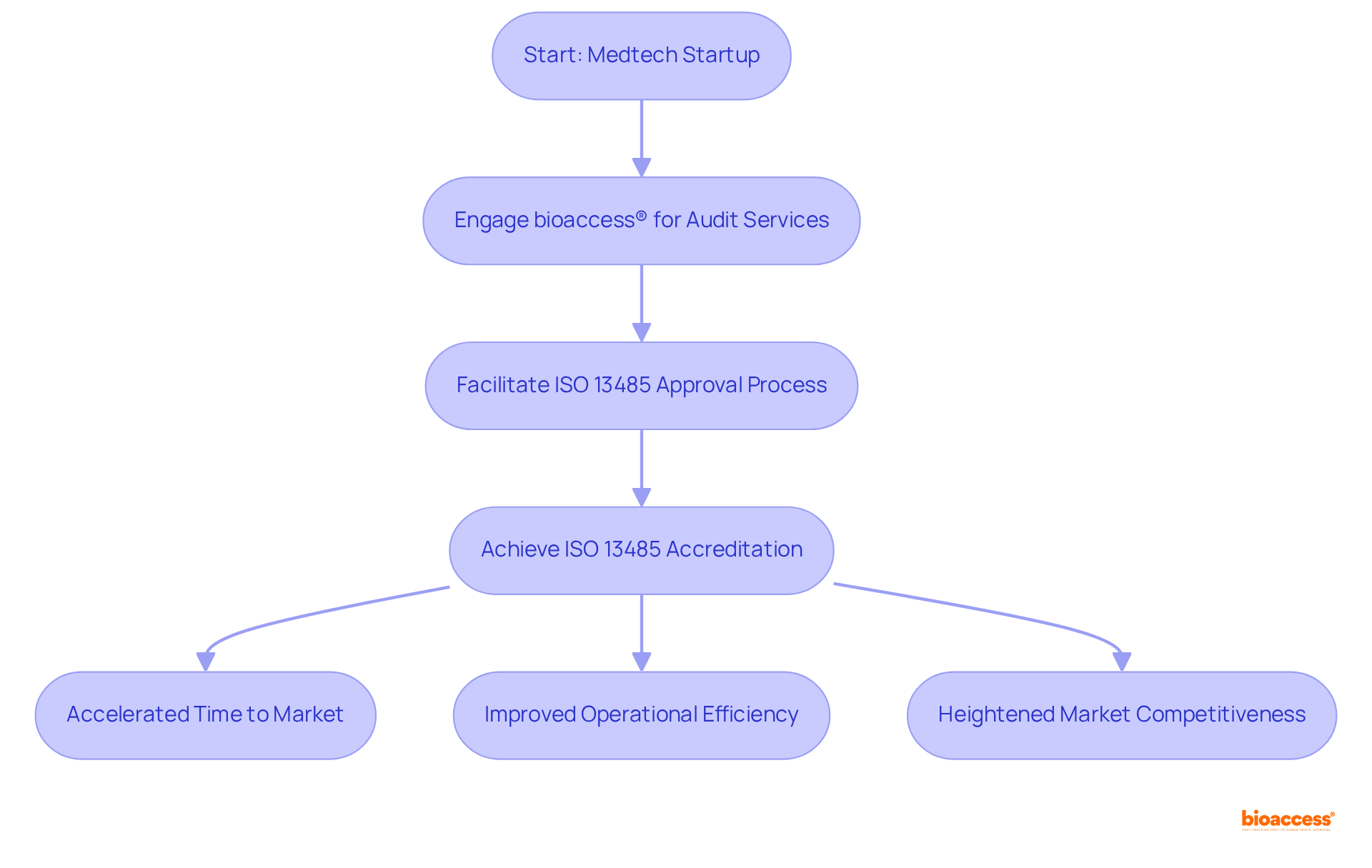
bioaccess: Accelerated ISO 13485 Audit Services for Medtech Startups
bioaccess® specializes in expedited tailored for and is recognized in the , leveraging its profound understanding of , the Balkans, and Australia.
In 2025, the average duration to achieve is significantly reduced, with bioaccess® facilitating this process to ensure startups can secure approval within weeks rather than months. This swift approach not only accelerates but also substantially enhances standards, establishing bioaccess® as a vital ally for innovative medical device firms.
Successful cases of demonstrate the tangible benefits of this qualification, as outlined in the , including improved operational efficiency and heightened market competitiveness.
Industry leaders consistently highlight the essential role of in nurturing a , underscoring its significance within the Medtech sector.
Intertek: Global Leader in ISO 13485 Certification and Auditing
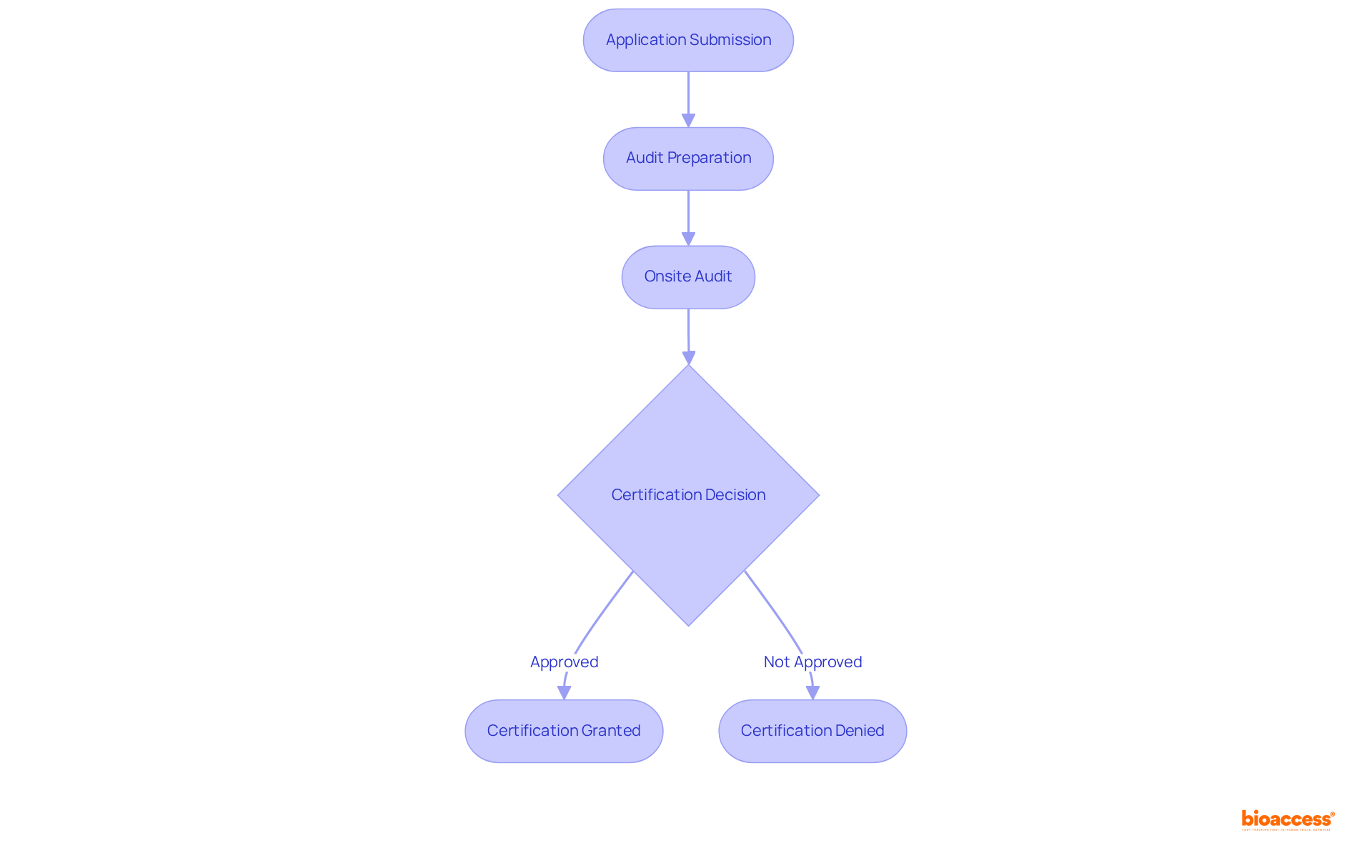
Intertek is recognized on the audit firms Mexico list as a global leader in , offering essential support to as they navigate the complexities of .
With a vast network of specialists, Intertek ensures that clients receive tailored assistance, including:
This comprehensive approach not only aids producers in meeting but also fosters a culture of .
In 2025, Intertek issued a substantial number of approvals, exemplifying its commitment to within the Medtech sector.
As the industry evolves, Intertek’s role in facilitating and driving innovation becomes increasingly vital, positioning manufacturers for success in a competitive landscape.
DNV: Specialized ISO 13485 Certification for Medical Devices
DNV provides specialized services for , tailored specifically for the . With experienced auditors who possess a profound understanding of the unique challenges manufacturers encounter, DNV offers that enhance the approval process. Recognized as an auditing entity by the , DNV bolsters its credibility in the approval process.
In 2025, DNV’s auditing services were acknowledged for their effectiveness in navigating the complexities of regulations, with the average duration for . This efficiency is critical, especially as the global is projected to grow from approximately $25 billion in 2023 to $40 billion by 2032, underscoring the increasing significance of regulatory standards in the industry.
Furthermore, DNV’s engagement in the , streamlining and enhancing assurance and safety for medical devices. empowers clients to confidently demonstrate adherence to international standards, thereby enhancing their market readiness and fostering trust among stakeholders.
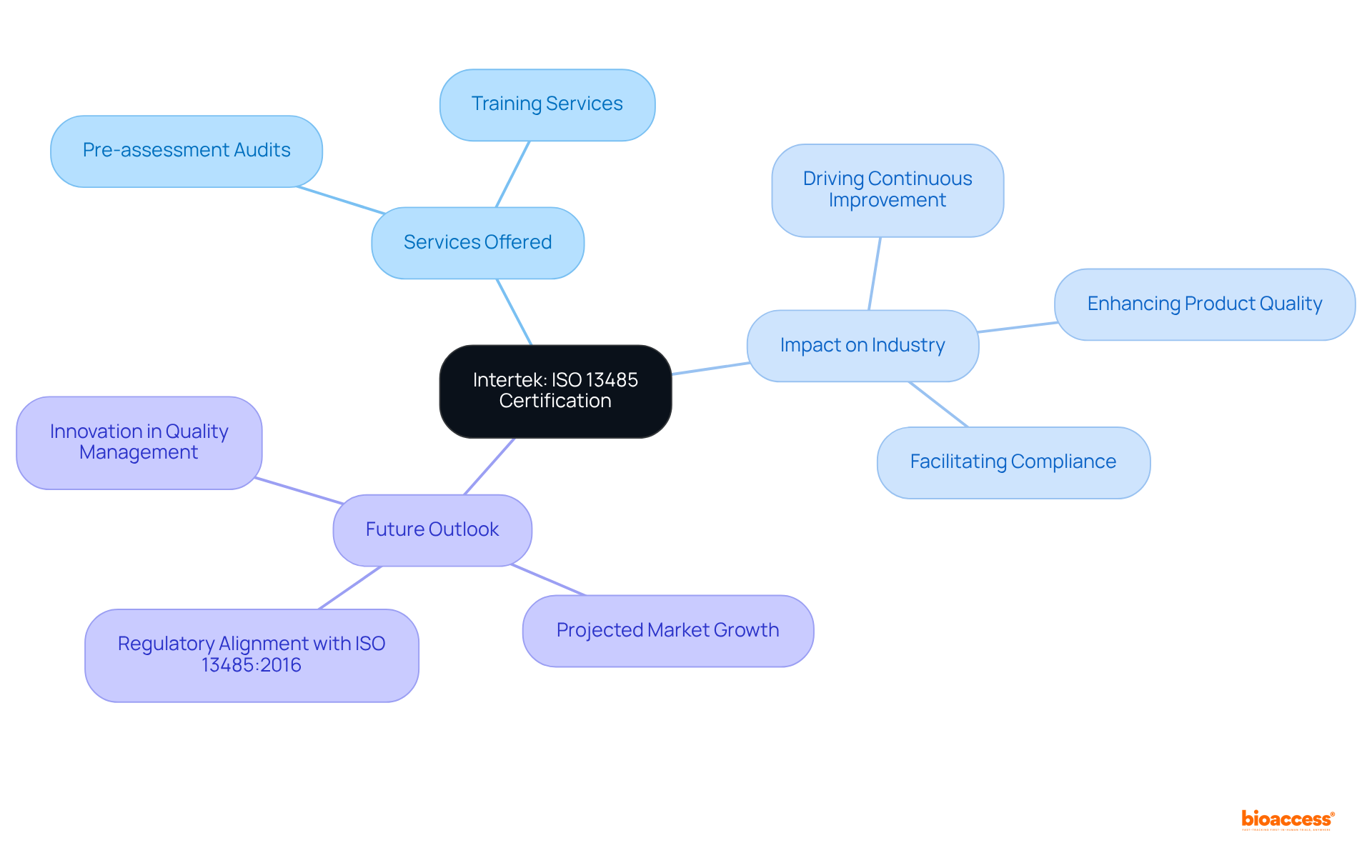
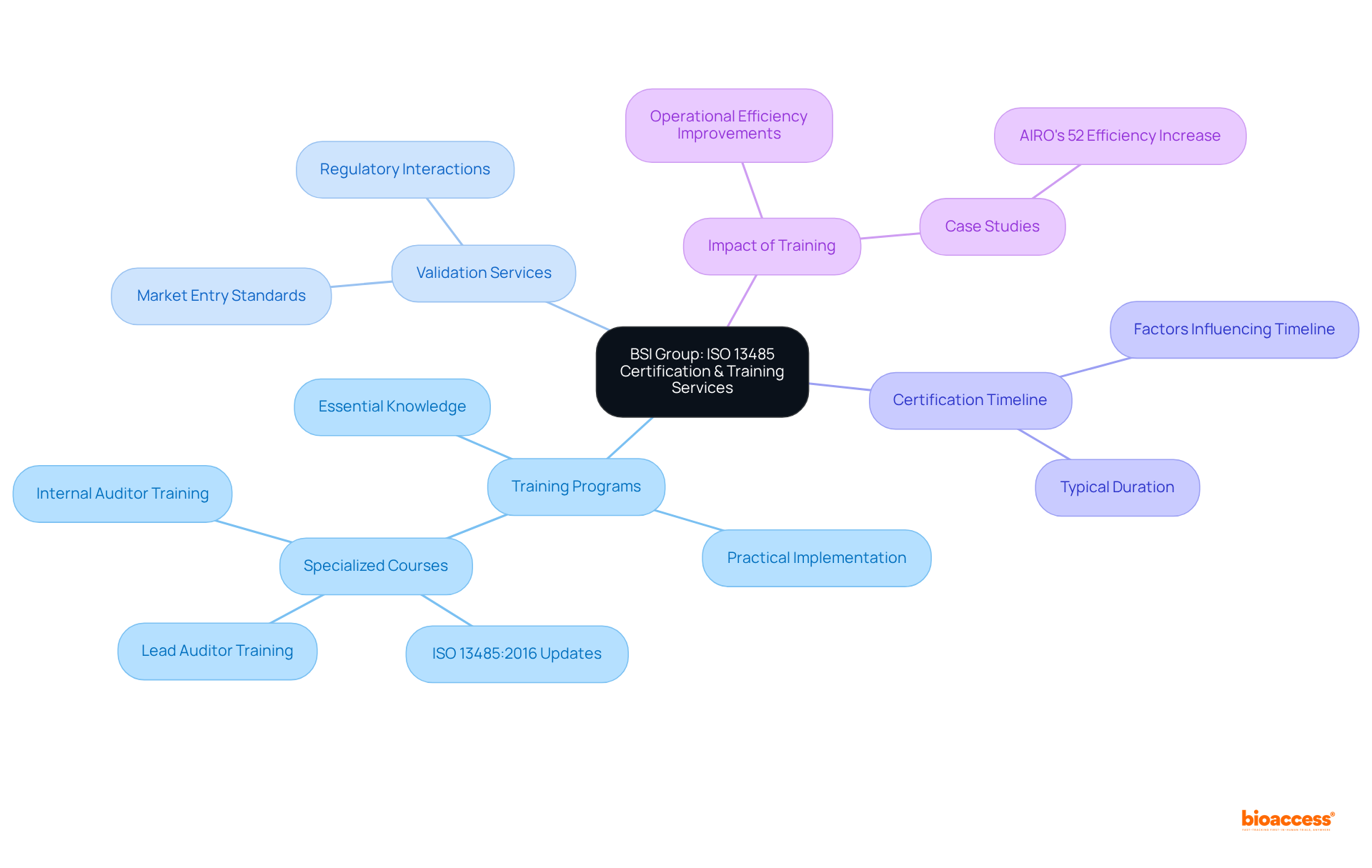
BSI Group: Comprehensive ISO 13485 Certification and Training Services
BSI Group provides a comprehensive range of , as listed among the . Their training programs equip organizations with the essential knowledge required to establish and maintain effective (QMS). By emphasizing practical implementation, these programs support both startups and established companies in navigating the complexities of compliance, ensuring that their performance systems are robust and appropriately scaled to meet operational demands.
The validation services offered by BSI are crucial for companies aiming to fulfill the stringent standards necessary for market entry. This not only bolsters their credibility within the industry but also streamlines interactions with regulatory bodies. Typically, organizations can expect to within a few months, according to the , depending on their level of preparedness and the complexity of their management systems.
Recent training initiatives by BSI have included specialized courses that focus on the , particularly its implications for medical device manufacturers. These programs underscore the importance of ensuring patient safety and compliance with regulations, both of which are vital for the successful commercialization of innovative products. Case studies illustrate how organizations participating in BSI’s training have significantly improved their adherence rates and operational efficiencies, highlighting the tangible benefits of investing in management training. For instance, AIRO achieved a 52% increase in bone ablation efficiency, demonstrating the impact of . Moreover, with over 15 years of experience in , bioaccess® recognizes the necessity of a suitably scaled quality management system, as emphasized by Medtech consultant David Amor, who also stresses the .
TÜV SÜD: Rigorous ISO 13485 Auditing and Certification
TÜV SÜD is recognized for its comprehensive , which are crucial for and feature in the list to help them meet stringent regulatory standards. Their experienced auditors conduct thorough assessments, ensuring adherence to all pertinent regulations and industry best practices. This meticulous approach not only enhances but also fosters .
With an average audit duration of approximately [insert specific duration] that meets industry benchmarks, TÜV SÜD facilitates a , promoting smoother market access for innovative . Their commitment to quality and safety is exemplified by successful instances, such as [insert specific examples], showcasing their vital role in aiding Medtech companies in achieving and enhancing their market visibility.
Furthermore, TÜV SÜD’s recent recognition as a highlights their expertise and authority in the sector.
Maven Professional Services: Tailored ISO 13485 Certification Solutions
Maven Professional Services stands at the forefront of customized . Their team of experts collaborates closely with clients to thoroughly understand their unique requirements, crafting that ensure compliance. This bespoke approach not only streamlines the but also enhances the overall , positioning clients for success in a competitive landscape.
PJR: Customer-Focused ISO 13485 Certification Services
PJR (Perry Johnson Registrars) distinguishes itself through its customer-focused , meticulously tailored to meet the specific needs of medical device manufacturers. Their dedicated team prioritizes client support, providing throughout the qualification process. This unwavering commitment to excellence and not only simplifies the regulatory journey but also empowers manufacturers to secure efficiently.
With a typical qualification timeframe that aligns with industry standards, PJR assures clients of their ability to navigate the complexities of compliance with confidence. The advantages of PJR’s support extend beyond mere certification; they cultivate a collaborative environment that enhances understanding and implementation of management systems, ultimately leading to improved product quality and regulatory readiness.
Case studies exemplify how PJR’s customized approach has effectively facilitated for various Medtech firms, emphasizing the importance of , which are crucial according to the ISO 13485 audit firms Mexico list for achieving and maintaining high industry standards.
Importantly, PJR’s efficiency is on par with the , which occur within , highlighting the critical importance of speed in the .
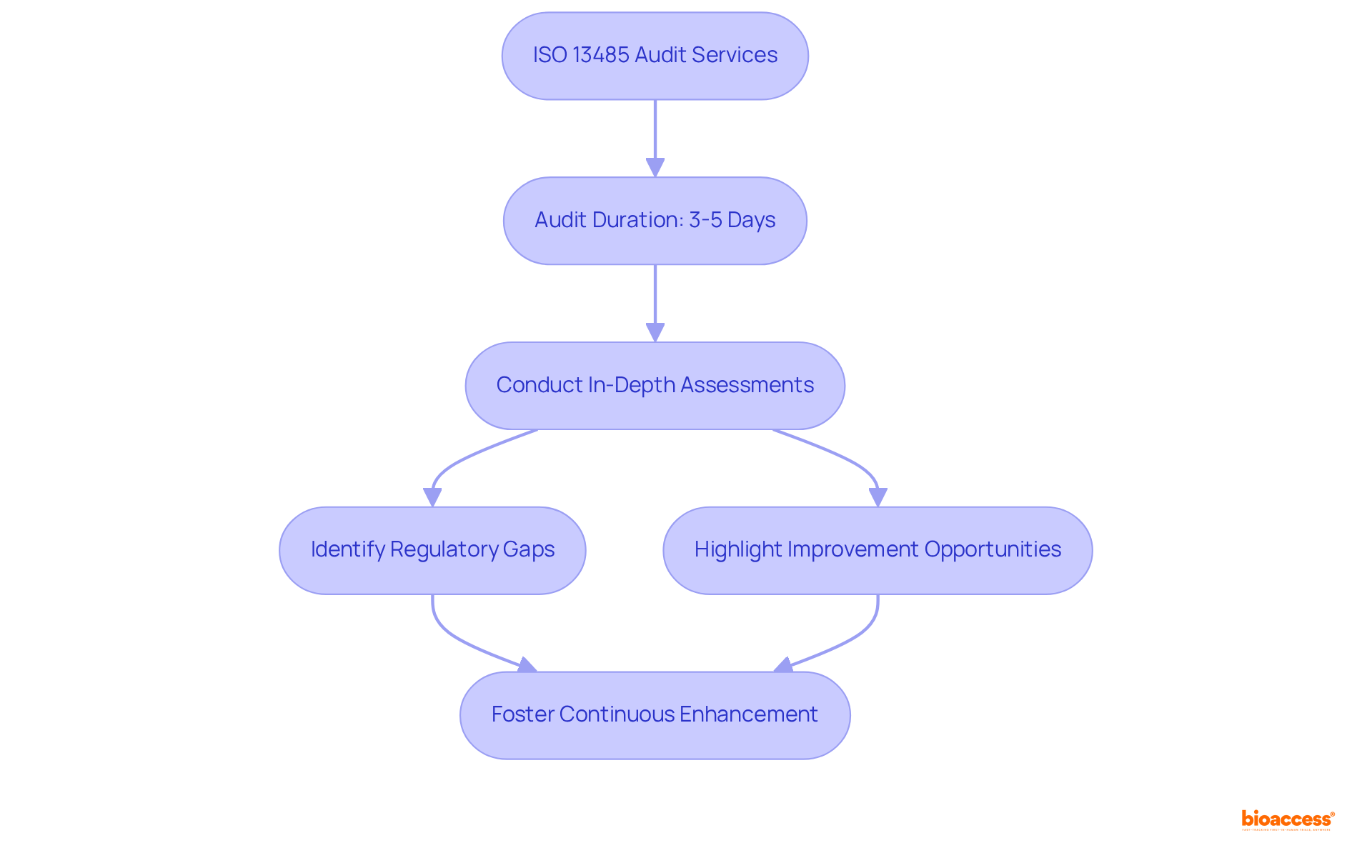
Pro QC International: Detailed ISO 13485 Audit Services
Pro QC International offers a comprehensive list of , providing for to ensure . Their seasoned auditors perform that not only identify but also highlight opportunities for improvement, fostering a culture of continuous enhancement.
With an average audit duration of approximately 3 to 5 days, consistent with industry best practices, Pro QC underscores the necessity of to uphold within the Medtech sector. This commitment to excellence is reflected in the successful compliance stories of numerous manufacturers, illustrating how .
As noted in the 2025 Medical Device Industry Report, companies are increasingly struggling to meet management system requirements, making . Moreover, the report on the impact of internal silos indicates that dismantling these barriers through thorough evaluations can markedly improve quality outcomes.
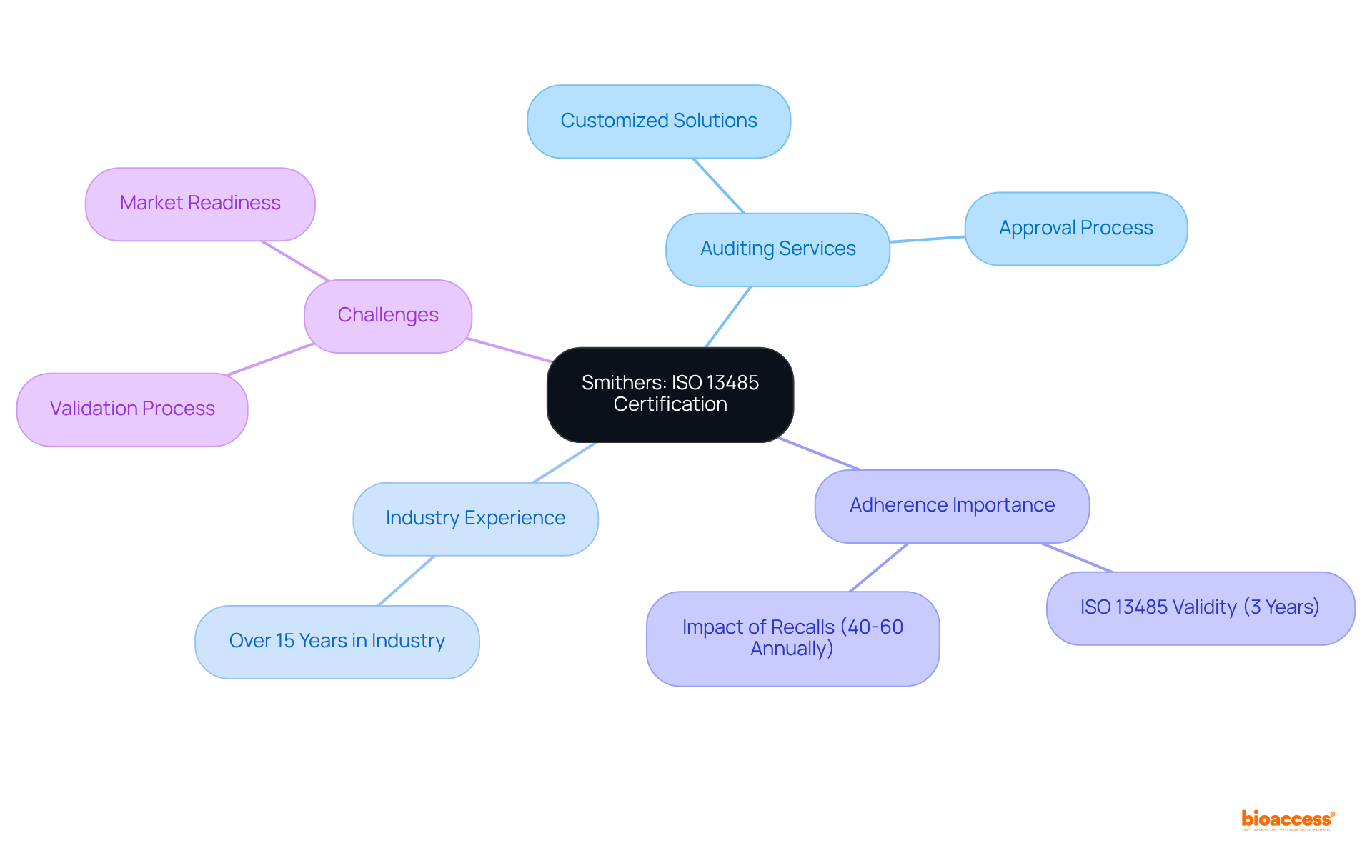
Smithers: ISO 13485 Certification and Auditing for Medical Devices
Smithers specializes in providing audit firms Mexico list and auditing services tailored specifically for . With over , their expert team is adept at navigating the unique challenges faced by this sector. They provide customized solutions that facilitate adherence, allowing clients to manage the with confidence. Notably, is valid for three years, requiring manufacturers to uphold rigorous management systems to ensure compliance.
Furthermore, with approximately 40-60 over the past five years, the critical importance of adherence and in the cannot be overstated. Smithers addresses key , guiding clients in overcoming obstacles and optimizing their path to market readiness.
DAC Audit Services: Expert ISO 13485 Compliance Audits
DAC Audit Services excels in providing expert specifically tailored for , which can be found in the . With a team of seasoned auditors, they conduct to guarantee that organizations adhere to all . This unwavering commitment to quality and compliance not only facilitates efficient certification but also empowers clients in their mission to introduce to the market.
Conclusion
The landscape of ISO 13485 audit firms in Mexico is crucial for ensuring that medical device manufacturers adhere to the stringent regulatory standards essential for market success. By highlighting ten key firms, this article emphasizes the necessity of specialized auditing services tailored to the unique challenges of the Medtech sector. These firms not only facilitate compliance but also enhance operational efficiencies, ultimately contributing to improved product quality and safety.
Key insights reveal that companies such as:
- bioaccess®
- Intertek
- DNV
- BSI Group
provide comprehensive services that streamline the certification process, reduce time to market, and foster a culture of continuous improvement. The focus on tailored solutions, rigorous auditing processes, and expert guidance underscores the critical nature of ISO 13485 compliance in upholding high industry standards and enhancing competitiveness.
As the Medtech industry continues to evolve, the importance of engaging with proficient ISO 13485 audit firms cannot be overstated. Manufacturers are urged to view compliance not merely as a regulatory obligation but as a strategic advantage that can accelerate their innovations to market swiftly and safely. Investing in quality management systems and expert auditing services is essential for sustaining trust and ensuring the safety of medical devices that impact lives.
Frequently Asked Questions
What is bioaccess and what services does it provide?
bioaccess® specializes in expedited ISO 13485 audit services tailored for Medtech startups, leveraging its understanding of regulatory frameworks across Latin America, the Balkans, and Australia.
How does bioaccess facilitate the ISO 13485 approval process for startups?
In 2025, bioaccess® significantly reduces the duration to achieve ISO 13485 approval in Latin America, enabling startups to secure approval within weeks rather than months.
What are the benefits of ISO 13485 accreditation for Medtech companies?
Successful ISO 13485 accreditation leads to improved operational efficiency and heightened market competitiveness for Medtech companies.
Why is ISO 13485 compliance important in the Medtech sector?
Industry leaders emphasize that ISO 13485 compliance nurtures a culture of quality and safety, making it essential within the Medtech sector.
Who is Intertek and what role do they play in ISO 13485 certification?
Intertek is a global leader in ISO 13485 accreditation and auditing services, providing essential support to medical device manufacturers in navigating compliance complexities.
What services does Intertek offer to clients?
Intertek offers pre-assessment audits and training services to help producers meet local and global regulatory standards and foster continuous improvement.
What was Intertek’s achievement in 2025 regarding ISO 13485 approvals?
In 2025, Intertek issued a substantial number of ISO 13485 approvals, showcasing its commitment to enhancing product quality and safety in the Medtech sector.
What specialized services does DNV provide for ISO 13485 certification?
DNV offers specialized ISO 13485 audit services tailored for the medical device industry, providing valuable insights to enhance the approval process.
How long does the ISO 13485 certification process typically take with DNV?
The average duration for ISO 13485 certification with DNV typically spans from three to six months.
What initiatives does DNV engage in to enhance regulatory procedures?
DNV’s involvement in the TCP III initiative facilitates the sharing of audit reports, streamlining regulatory procedures and enhancing assurance and safety for medical devices.
How does DNV support clients in demonstrating adherence to international standards?
DNV’s commitment to excellence and safety empowers clients to confidently demonstrate adherence to international standards, enhancing their market readiness and fostering trust among stakeholders.
List of Sources
- bioaccess: Accelerated ISO 13485 Audit Services for Medtech Startups
- akitra.com (https://akitra.com/five-key-steps-for-achieving-iso-certification)
- cognidox.com (https://cognidox.com/blog/medical-device-technical-file-requirements-what-you-need-to-know)
- fastercapital.com (https://fastercapital.com/content/ISO-13485–Enhancing-Medical-Device-Quality-with-ISO-Certification.html)
- 6 Important Quotes from Medtech Leaders (https://mddionline.com/business/6-important-quotes-from-medtech-leaders)
- scispot.com (https://scispot.com/blog/your-lab-guide-to-iso-13485)
- Intertek: Global Leader in ISO 13485 Certification and Auditing
- ISO Certification Market Size and Share | Statistics – 2034 (https://factmr.com/report/iso-certification-market)
- assuranceinaction.intertek.com (https://assuranceinaction.intertek.com/post/102imcr/kiss-keep-it-sweet-and-successful)
- verifiedmarketreports.com (https://verifiedmarketreports.com/product/iso-certification-market)
- intertek.com (https://intertek.com/blog/2024/11-26-understanding-21-cfr-820)
- dataintelo.com (https://dataintelo.com/report/iso-certification-service-market)
- DNV: Specialized ISO 13485 Certification for Medical Devices
- dnv.ae (https://dnv.ae/news/2024)
- dataintelo.com (https://dataintelo.com/report/global-management-systems-certification-market)
- linkedin.com (https://linkedin.com/posts/héctor-salomón-galindo-alvarado_dnv-has-been-recognized-as-auditing-organization-activity-7220065003813437441-xIZn)
- dnv.com (https://dnv.com/news/2024/scpa-tcp-iii)
- BSI Group: Comprehensive ISO 13485 Certification and Training Services
- arthrolase.com (https://arthrolase.com/news)
- scribd.com (https://scribd.com/document/564157183/Bsi-Iso13485-Product-Guide-Uk-En)
- greenlight.guru (https://greenlight.guru/blog/why-medical-device-startups-need-to-implement-a-right-sized-qms-with-david-amor)
- bsigroup.com (https://bsigroup.com/en-US/products-and-services/standards/iso-13485-quality-management-system)
- bsigroup.com (https://bsigroup.com/en-US/training-courses/iso-13485-quality-management-for-medical-devices-training-courses)
- TÜV SÜD: Rigorous ISO 13485 Auditing and Certification
- tidepool.org (https://tidepool.org/blog/tidepools-open-source-qms-gets-iso-13485-certified)
- tuvsud.com (https://tuvsud.com/en-us/industries/healthcare-and-medical-devices/medical-devices-and-ivd/medical-device-market-approval-and-certification/medical-device-regulation)
- linkedin.com (https://linkedin.com/posts/tuvsud-medicaldevices_medicaldevices-mdsap-webinar-activity-7232355891117780993-4hoA)
- iioa.global (https://iioa.global/tuv-sud-publishes-its-2024-annual-report)
- meddeviceonline.com (https://meddeviceonline.com/doc/key-qms-considerations-for-your-medical-device-startup-0001)
- Maven Professional Services: Tailored ISO 13485 Certification Solutions
- sushvin.com (https://sushvin.com/medical-devices-compliance-updates-newsletter-June-2025.html)
- linkedin.com (https://linkedin.com/pulse/medical-device-news-update-january-2025-naveen-agarwal-ph-d–lumbe)
- clutch.co (https://clutch.co/profile/forward-security)
- llcbuddy.com (https://llcbuddy.com/data/quality-management-systems-statistics)
- PJR: Customer-Focused ISO 13485 Certification Services
- 25 quotes on customer experience from CX professionals (https://callminer.com/blog/25-quotes-on-customer-experience-from-cx-professionals)
- greenlight.guru (https://greenlight.guru/blog/quality-challenges)
- salesforce.com (https://salesforce.com/ca/hub/service/famous-customer-service-quotes)
- 101 Of The Best Customer Experience Quotes (https://forbes.com/sites/blakemorgan/2019/04/03/101-of-the-best-customer-experience-quotes)
- qualtrics.com (https://qualtrics.com/blog/customer-service-quotes-to-inspire-your-team)
- Pro QC International: Detailed ISO 13485 Audit Services
- 5 Important Things for Life Sciences Companies to Consider | MasterControl (https://mastercontrol.com/gxp-lifeline/quality-inspiration)
- greenlight.guru (https://greenlight.guru/blog/quality-challenges)
- Smithers: ISO 13485 Certification and Auditing for Medical Devices
- linkedin.com (https://linkedin.com/posts/sacramento-valley-manufacturing-alliance_a-closer-look-at-iso-13485-for-medical-device-activity-7222376823207215104-B6vQ)
- modusadvanced.com (https://modusadvanced.com/resources/blog/quality-certifications-for-manufacturing-as9100-vs.-iso-9001)
- The Top 5 Challenges in Medical Device Regulatory Compliance and How to Overcome Them – Nerac (https://nerac.com/the-top-5-challenges-in-medical-device-regulatory-compliance-and-how-to-overcome-them)
- marketresearch.com (https://marketresearch.com/Business-Research-Company-v4006/Medical-Device-Safety-Testing-Global-41463921)
- smithers.com (https://smithers.com/resources)
- DAC Audit Services: Expert ISO 13485 Compliance Audits
- greenlight.guru (https://greenlight.guru/blog/quality-challenges)
- 130+ Compliance Statistics & Trends to Know for 2026 (https://secureframe.com/blog/compliance-statistics)
- simplerqms.com (https://simplerqms.com/iso-13485-audit)
- azquotes.com (https://azquotes.com/quotes/topics/auditors.html)
- dacaudit.com (https://dacaudit.com/iso-13485-medical-device-manufacturing)

Leave a Reply