Introduction
Navigating the complex landscape of regulatory submissions for alpha-emitter trials is no small feat. Yet, it is essential for the success of clinical research in this innovative field. Organizations that master the intricacies of regulatory frameworks and submission strategies stand to gain a competitive edge, ensuring compliance and accelerating approval timelines.
However, the challenge remains: how can companies effectively engage with regulatory authorities? Adapting practices to meet evolving standards while maintaining high-quality submissions is crucial. This is where understanding the Medtech landscape becomes vital. By addressing key challenges, organizations can position themselves for success in a rapidly changing environment.
Understand Regulatory Frameworks for Alpha-Emitter Trials
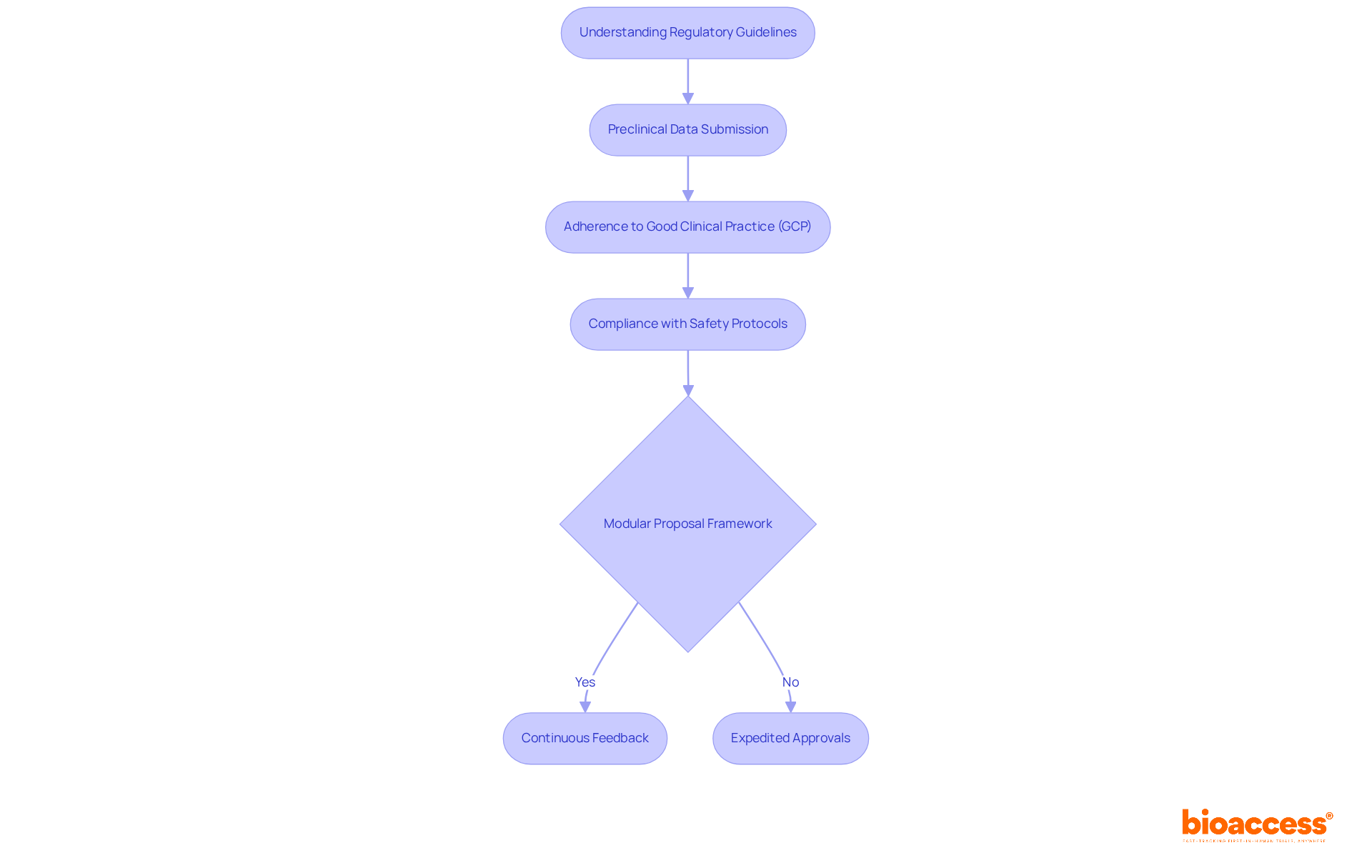
Navigating the alpha-emitter trials is crucial for clinical research success. Understanding the set by regulatory authorities like the FDA and EMA is essential. is a key requirement, demonstrating both safety and efficacy. Moreover, adherence to ensures that trials are conducted ethically and uphold the highest quality standards. is vital to mitigate associated risks.
can significantly streamline the review process. This framework allows for data submission in sections, facilitating continuous feedback from oversight bodies as each section is evaluated. Such an approach enhances communication and enables sponsors to address potential issues early in the submission process, ultimately leading to expedited approvals. further underscore the importance of these practices, reinforcing the need for robust oversight strategies in this evolving field.
Bioaccess stands ready to support your needs. Our extensive services include:
- Feasibility studies
- Site selection
- Setup
- Import permits
- Project management
- Comprehensive reporting on study status and adverse events
We ensure a thorough and compliant process, paving the way for successful outcomes in your research endeavors.
Implement Effective Submission Strategies for Alpha-Emitter Trials
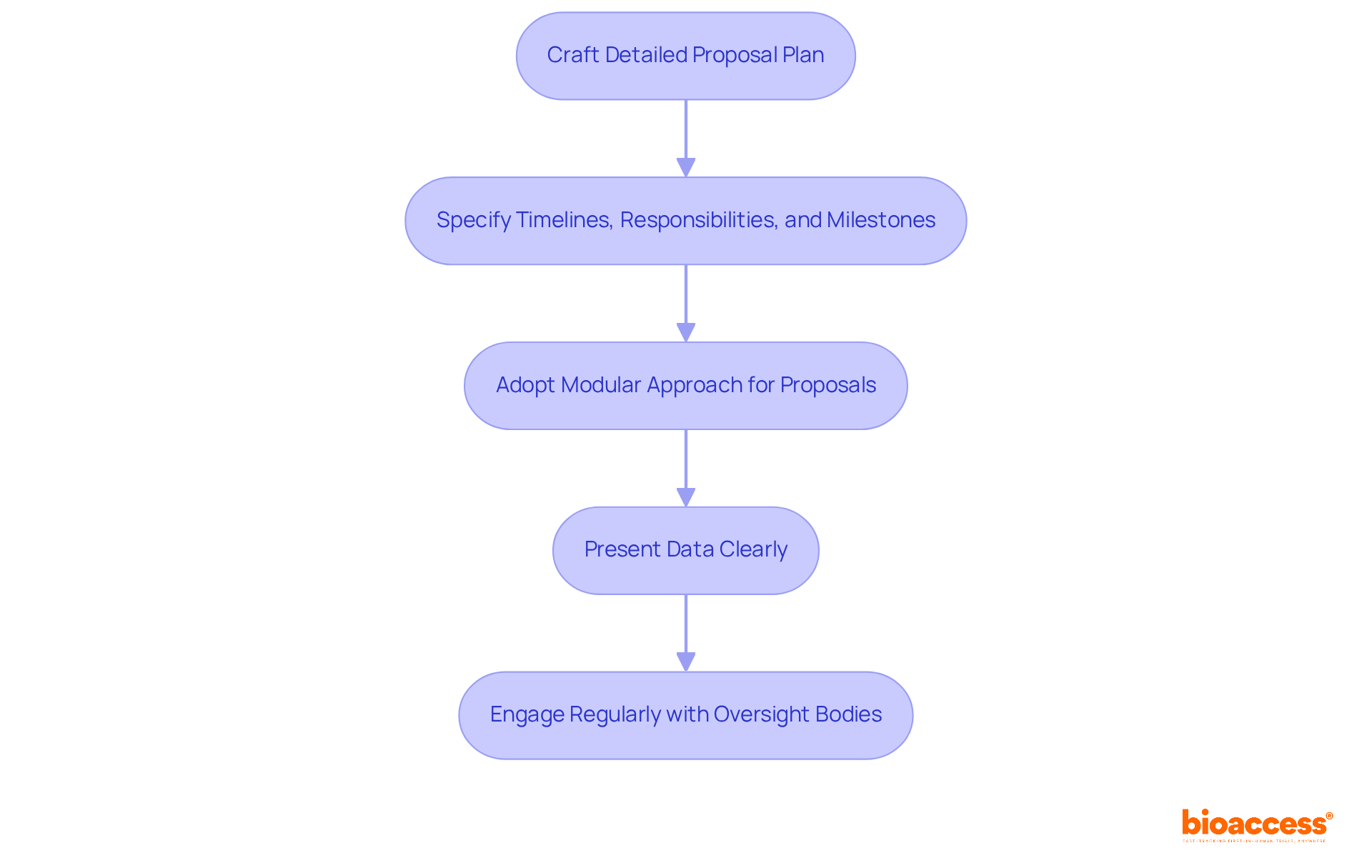
To enhance the likelihood of successful approvals, organizations must adopt a . This involves crafting a detailed proposal plan that specifies . A modular approach to proposals is especially beneficial, enabling that can efficiently tackle as they emerge. Furthermore, will streamline the review process.
Timely and regular interaction with oversight bodies is crucial. This engagement offers perspectives that can enhance application strategies and proactively address possible challenges. By adopting these practices, organizations can significantly enhance their efficiency in proposals and . The importance of cannot be overstated.
Foster Collaboration with Regulatory Authorities and Stakeholders
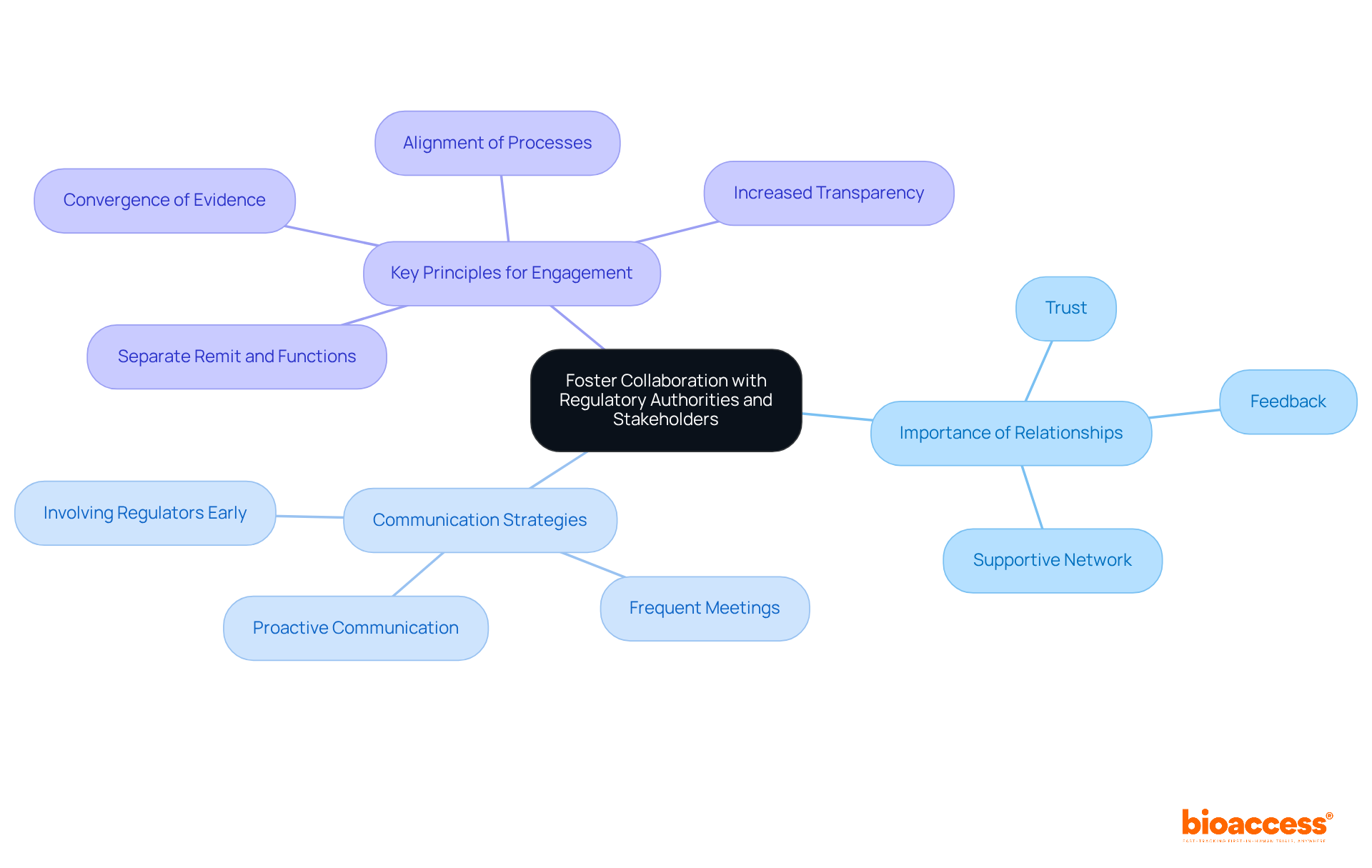
Building strong connections with oversight bodies and stakeholders, such as INVIMA, is crucial for providing . Organizations must prioritize to foster trust and facilitate smoother interactions. Frequent meetings with oversight representatives provide valuable feedback and insights that guide submission strategies, ultimately improving the chances of approval. Additionally, connecting with and industry associations is essential, as these links help entities stay informed about and best practices. By nurturing these connections, organizations can establish a supportive network that significantly enhances their capacity to manage the complex compliance environment efficiently.
Successful in alpha-emitter trials underscores the importance of proactive communication. Companies that maintain consistent communication with oversight agencies, including INVIMA, report faster resolution of compliance matters and a better understanding of evidence requirements. Katherine Ruiz, a specialist in compliance matters for medical devices and in vitro diagnostics in Colombia, emphasizes that proactive and transparent communication is vital for establishing . Expert insights suggest that involving regulators early in the planning phase, rather than presenting finalized solutions, can lead to more favorable outcomes. This approach not only but also aligns the objectives of all parties involved, ultimately driving faster patient access to innovative therapies.
As the regulatory environment continues to evolve, particularly in 2026, the emphasis on effective communication with regulatory bodies like INVIMA will be paramount. Organizations that prioritize building strong relationships with these stakeholders will be better positioned to navigate challenges and capitalize on opportunities in the . Furthermore, adopting four key principles for future interactions – separate remit and functions, convergence of evidence, alignment of processes, and increased transparency – can provide a structured approach for organizations to enhance their .
Adapt and Optimize Submission Practices Based on Feedback
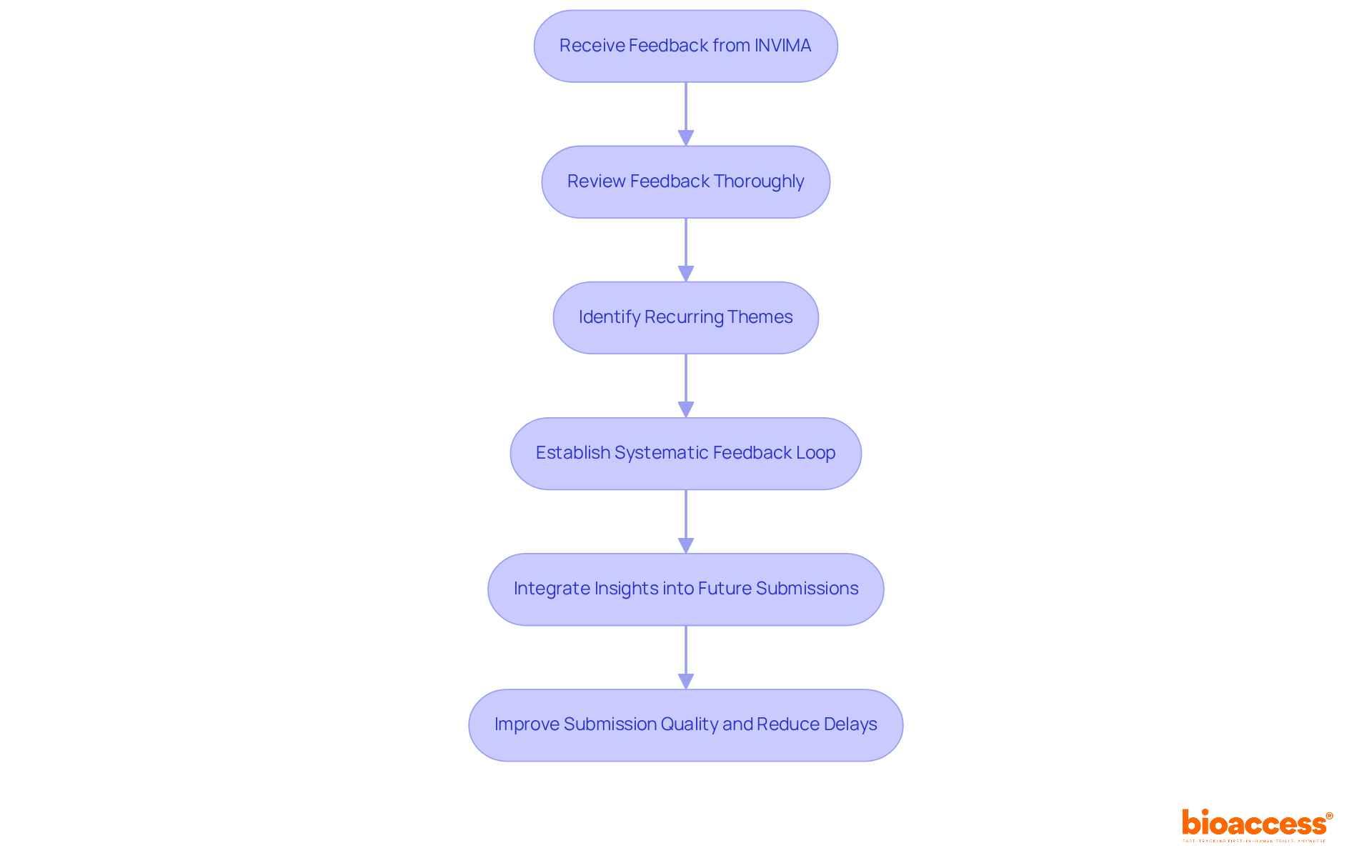
Organizations must view , such as INVIMA (Colombia National Food and Drug Surveillance Institute), as a crucial opportunity for growth, particularly in terms of . INVIMA is instrumental in inspecting and supervising the marketing and manufacturing of health products, including , and is recognized as a Level 4 health authority by the Pan American Health Organization/World Health Organization. After each submission, a thorough review of the feedback is essential to pinpoint recurring themes and areas for improvement specific to .
Establishing a enables organizations to consistently refine their practices for proposals, ensuring that insights gained are effectively integrated into future submissions. This proactive strategy not only elevates the quality of entries but also signals to oversight bodies a strong . Regular training sessions focused on and updates in compliance requirements can significantly bolster an entity’s presentation capabilities.
Statistics reveal that submissions benefiting from oversight feedback see a notable , with 73% ultimately receiving approval after assessment. Moreover, organizations that actively engage in refining their application processes based on feedback can reduce median delays, which can extend up to 435 days for requests requiring resubmissions. By fostering a culture of and responsiveness to compliance insights, organizations can enhance their chances of and expedite the approval process.
Katherine Ruiz, an expert in Regulatory Affairs for and In Vitro Diagnostics in Colombia, underscores the necessity of understanding local regulatory frameworks to navigate these challenges effectively.
Conclusion
Navigating the complexities of regulatory submission support for alpha-emitter trials is crucial for the success of clinical research efforts. A solid grasp of the regulatory frameworks established by authorities like the FDA and EMA, combined with a commitment to ethical practices and safety protocols, forms the bedrock of effective submissions. By embracing a modular approach to proposals and fostering open communication with regulatory bodies, organizations can significantly boost their chances of timely approvals and successful trial outcomes.
Key insights from the article underscore the importance of:
- Systematic submission strategies
- Proactive stakeholder engagement
- Integration of feedback from regulatory authorities
Organizations that prioritize these elements are better positioned to navigate the evolving regulatory landscape, ultimately leading to improved compliance and faster access to innovative therapies. The emphasis on collaboration with oversight bodies, such as INVIMA, further highlights the necessity of building strong relationships to facilitate smoother interactions and enhance submission quality.
In conclusion, the path to successful regulatory submission for alpha-emitter trials is paved with:
- Strategic planning
- Continuous improvement
- Commitment to collaboration
By adopting these best practices, organizations can not only optimize their submission processes but also contribute to the advancement of medical science and the timely delivery of groundbreaking therapies to patients in need. Actively engaging with regulatory authorities and staying informed about evolving guidelines will be essential for future success in this dynamic field.
Frequently Asked Questions
Why is understanding regulatory frameworks important for alpha-emitter trials?
Understanding regulatory frameworks is crucial for the success of clinical research in alpha-emitter trials as it ensures compliance with guidelines set by regulatory authorities like the FDA and EMA.
What are the key requirements for alpha-emitter trials?
Key requirements include comprehensive preclinical data demonstrating safety and efficacy, adherence to Good Clinical Practice (GCP), and compliance with safety protocols for handling radioactive materials.
How does the FDA’s modular proposal framework benefit the submission process?
The FDA’s modular proposal framework allows for data submission in sections, facilitating continuous feedback from oversight bodies, enhancing communication, and enabling sponsors to address potential issues early, which can lead to expedited approvals.
What recent guidelines have been issued by the EMA regarding alpha-emitter trials?
Recent EMA guidelines for regulatory submission support alpha-emitter trials in 2026 emphasize the importance of robust oversight strategies in this evolving field.
What services does Bioaccess offer to support clinical study management?
Bioaccess offers services including feasibility studies, site selection, setup, import permits, project management, and comprehensive reporting on study status and adverse events to ensure a thorough and compliant process.
List of Sources
- Understand Regulatory Frameworks for Alpha-Emitter Trials
- Regulatory Updates, January 2026- Caidya® (https://caidya.com/resources/global-regulatory-updates-on-clinical-trials-january-2026)
- FDA guidance on Oncology Therapeutic Radiopharmaceuticals (https://tracercro.com/resources/blogs/new-fda-guidance-on-oncology-therapeutic-radiopharmaceuticals)
- Makary says FDA will “end two-trial dogma” for approvals (https://pharmaphorum.com/news/makary-says-fda-will-end-two-trial-dogma-approvals)
- ema.europa.eu (https://ema.europa.eu/en/radiopharmaceuticals-scientific-guideline)
- EMA drafts concept paper for future radiopharmaceuticals guideline (https://raps.org/news-and-articles/news-articles/2024/10/ema-drafts-concept-paper-for-future-radiopharmaceu)
- Implement Effective Submission Strategies for Alpha-Emitter Trials
- Presenting Clinical Data for Regulatory Submission: A Stats Perspective (https://cytel.com/perspectives/presenting-clinical-data-for-regulatory-submission-a-stats-perspective)
- Factors Influencing Clearance Time for Medical Devices (https://mddionline.com/medical-device-regulations/factors-influencing-fda-clearance-time-for-medical-devices-evolution-of-a-critical-regulatory-pathway)
- intuitionlabs.ai (https://intuitionlabs.ai/articles/pharma-regulatory-submission-challenges)
- mckinsey.com (https://mckinsey.com/industries/life-sciences/our-insights/rewiring-pharmas-regulatory-submissions-with-ai-and-zero-based-design)
- Statistical Support for Regulatory Submissions | PharPoint (https://pharpoint.com/resources/statistical-support-regulatory-submissions-discussions)
- Foster Collaboration with Regulatory Authorities and Stakeholders
- Featured: Connecting Publishers with Subject Matter Experts (https://featured.com/questions/building-strong-regulatory-relationships)
- appliedclinicaltrialsonline.com (https://appliedclinicaltrialsonline.com/view/state-cro-and-sponsor-relationships)
- Regulatory, health technology assessment and company interactions: the current landscape and future ecosystem for drug development, review and reimbursement – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11574548)
- Robert J. Kueppers on Trust and Regulation (Trust Quotes #14) – Trusted Advisor Associates (https://trustedadvisor.com/trustmatters/robert-j-kueppers-on-trust-and-regulation-trust-quotes-14)
- Understanding the Influence and Impact of Stakeholder Engagement in Patient-centered Outcomes Research: a Qualitative Study – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC8993962)
- Adapt and Optimize Submission Practices Based on Feedback
- Presenting Clinical Data for Regulatory Submission: A Stats Perspective (https://cytel.com/perspectives/presenting-clinical-data-for-regulatory-submission-a-stats-perspective)
- Statistical Support for Regulatory Submissions | PharPoint (https://pharpoint.com/resources/statistical-support-regulatory-submissions-discussions)
- ideagen.com (https://ideagen.com/thought-leadership/blog/22-feedback-quotes-for-2022)
- 26 powerful quotes about feedback (https://netigate.net/articles/surveys/quotes-about-feedback)
- t-three.com (https://t-three.com/thinking-space/blog/7-inspiring-quotes-that-will-help-you-give-better-feedback)

Leave a Reply