Introduction
Preparation for regulatory inspections in the biologics sector is not just a formality; it’s a critical component that can determine the success or failure of a product’s approval. Organizations must navigate a complex landscape of documentation, quality management systems, and training to ensure compliance with stringent regulatory standards. This article explores the essential elements of an audit readiness package for biologics in Phase III, providing a step-by-step guide to assembling a comprehensive package that not only meets regulatory expectations but also enhances operational efficiency.
With the stakes so high, what common pitfalls could jeopardize this crucial preparation? How can organizations effectively avoid them?
Define Audit Readiness for Biologics
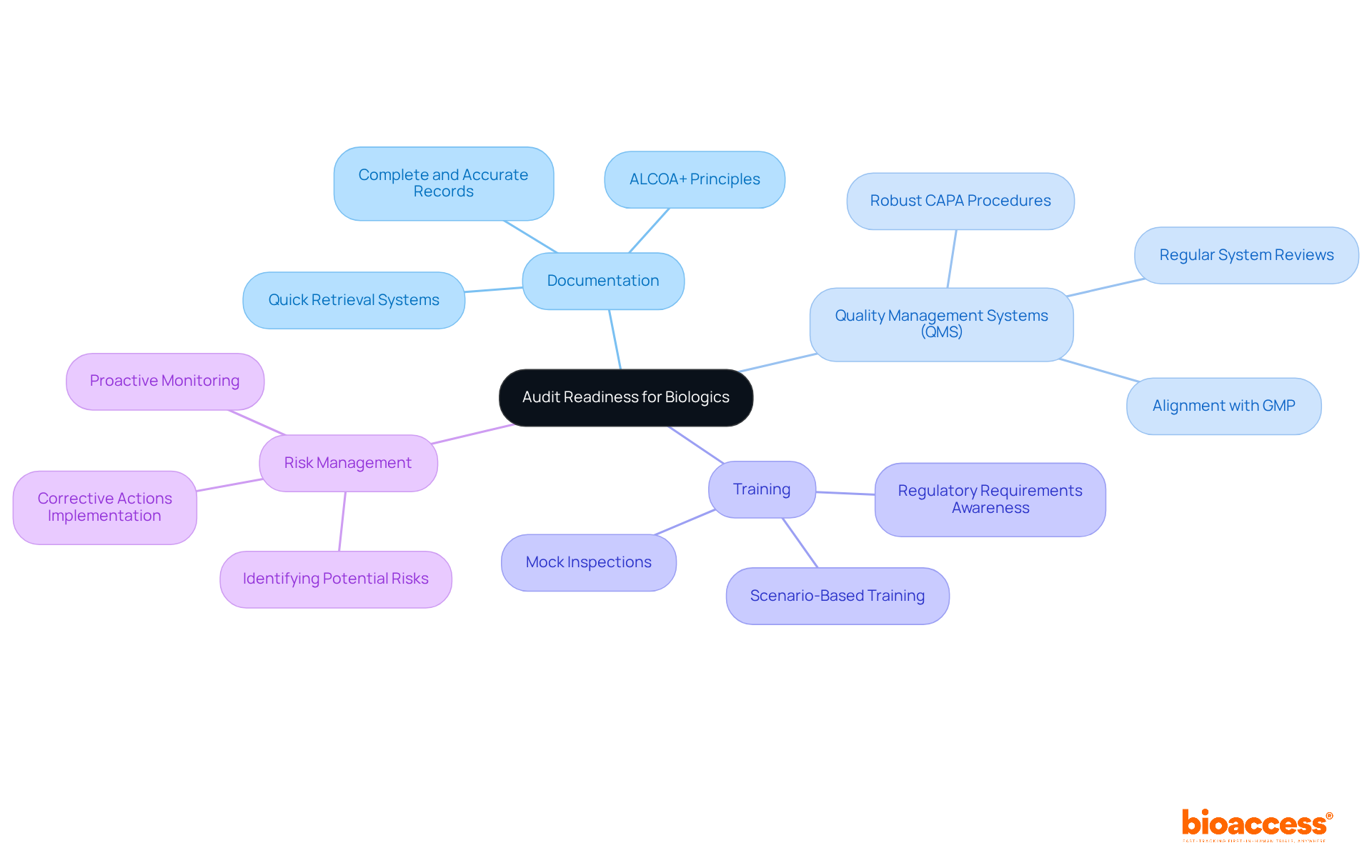
Preparation for biologics is essential for ensuring that organizations are fully equipped for concerning biological products. This preparation involves having all necessary documentation, processes, and systems in place to with regulatory standards. Key aspects of audit readiness include:
- Documentation: It’s crucial to ensure that all records related to the development, manufacturing, and testing of biologics are complete, accurate, and easily accessible. Experts like Ana Criado, Director of Regulatory Affairs and CEO of Mahu Pharma, highlight the significance of in fulfilling .
- : Implementing a robust QMS that aligns with and other is vital. Ana’s extensive experience as a regulatory consultant for international firms underscores the importance of a strong QMS in ensuring compliance with regulations.
- Training: All personnel involved in the production and oversight of biologics must be . This area is critical, and professionals like Katherine Ruiz, an expert in Regulatory Affairs for Medical Devices and In Vitro Diagnostics in Colombia, can offer valuable insights.
- : that could impact compliance or product quality is essential. By understanding these elements, organizations can improve their readiness for evaluations and ensure they meet the stringent requirements set by regulatory bodies.
Identify Key Components of the Audit Readiness Package
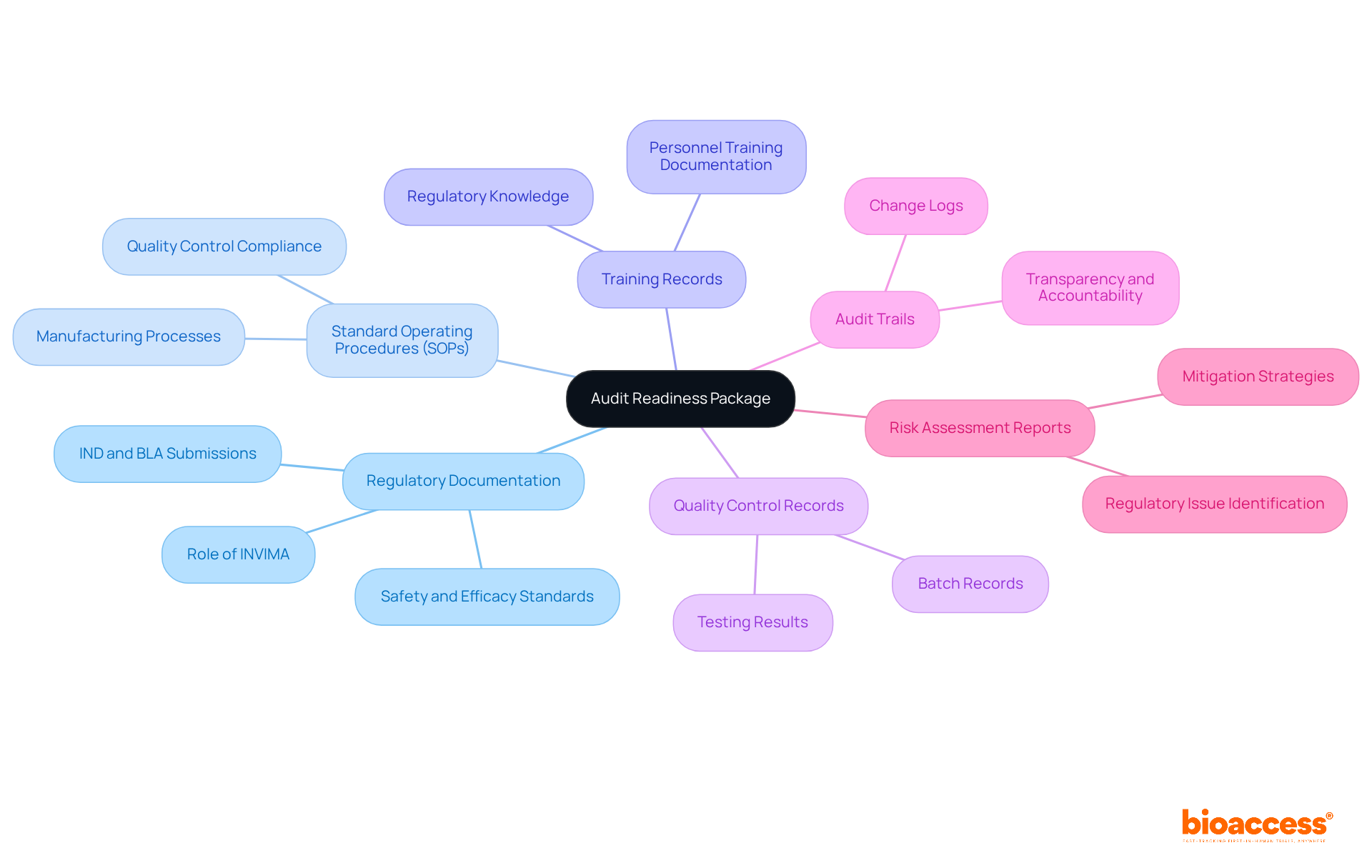
A successful is crucial for ensuring adherence and enabling effective evaluations. This package must include several essential elements:
- : Submissions to regulatory authorities, such as Investigational New Drug (IND) applications and Biologics License Applications (BLA), must be meticulously compiled. In Colombia, understanding the role of (Instituto Nacional de Vigilancia de Medicamentos y Alimentos) is vital, as it oversees the marketing and manufacturing of health products. This documentation serves as the foundation for demonstrating adherence to safety and efficacy standards, ensuring that products meet rigorous testing and documentation criteria before approval.
- : Detailed SOPs are indispensable, outlining the processes for manufacturing, testing, and quality control of biologics. These documents ensure that all operations comply with established protocols, thereby reducing risks during evaluations and aligning with ‘s regulatory expectations.
- : Comprehensive documentation of training sessions for personnel involved in the production and oversight of biologics is essential. These records must be thorough and traceable, confirming that staff are knowledgeable about regulatory requirements, which is crucial for maintaining operational integrity and meeting ‘s standards.
- : Organizations must maintain data evidencing the effectiveness of quality control measures. This includes batch records and testing results, which are critical for demonstrating product reliability and safety, as required by .
- : Comprehensive logs that track changes and updates to critical documents and processes are necessary for ensuring transparency and accountability. These examination trails provide a transparent record of adherence initiatives and assist the review process, conforming to ‘s oversight necessities.
- : Documentation of risk evaluations is essential for identifying and mitigating potential regulatory issues. These reports help entities proactively tackle vulnerabilities, thus improving overall compliance in line with ‘s guidelines.
By systematically gathering these components, organizations can develop an that not only meets regulatory expectations but also underscores their commitment to in the biologics sector, particularly in relation to ‘s supervision.
Compile the Audit Readiness Package Step-by-Step
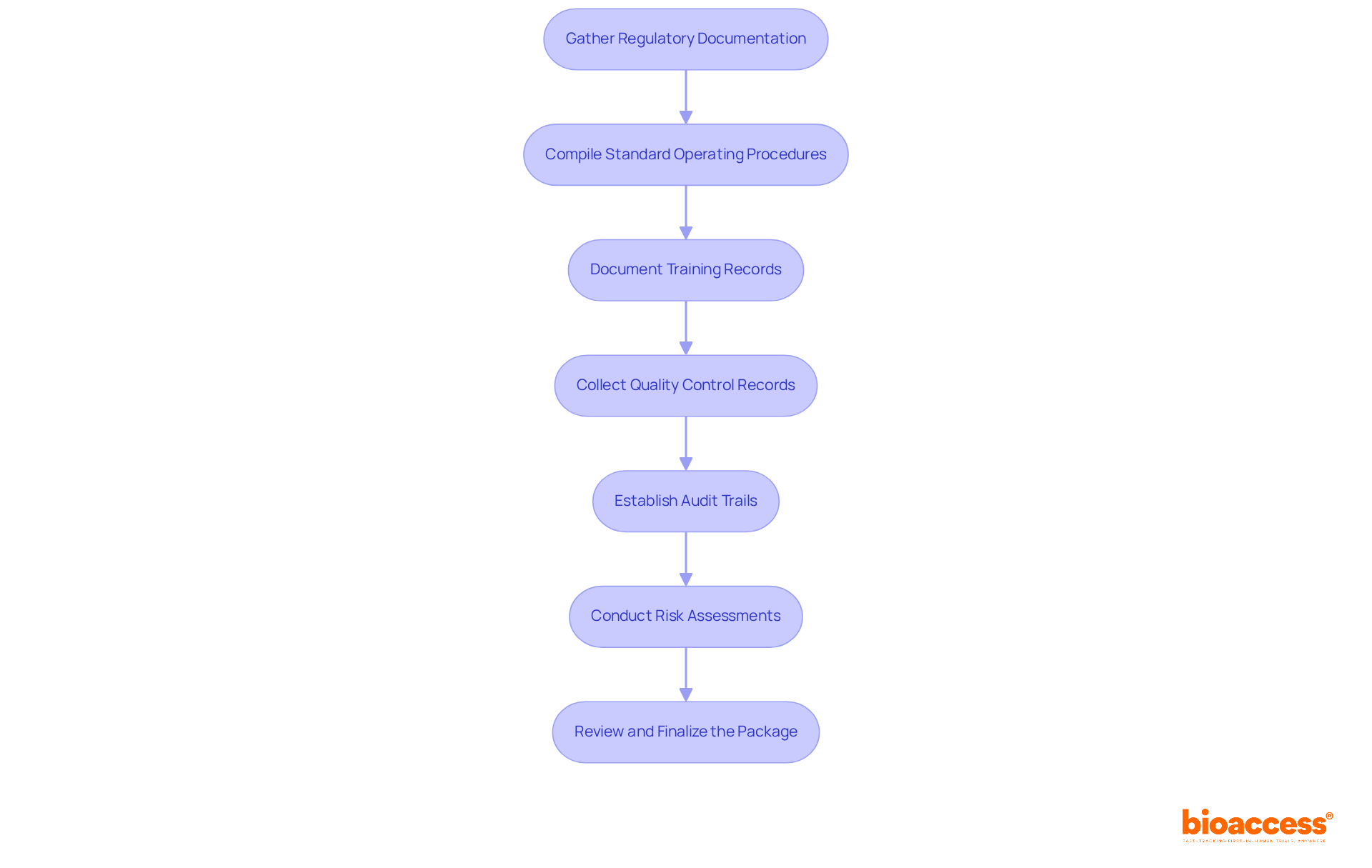
To effectively compile the for biologics, follow these structured steps:
- Gather : Assemble all pertinent submissions, including Investigational New Drug (IND) and Biologics License Application (BLA) documents. Arrange these in chronological order for easy access during the review. Did you know that approximately 30% of companies struggle with gathering these documents due to disorganization?
- Compile (SOPs): Review and consolidate all SOPs related to the production and testing of biologics. Ensure that these documents are current and accurately reflect operational practices. Experts emphasize that having up-to-date SOPs is crucial for demonstrating compliance during evaluations.
- : Establish a centralized repository for training records, confirming that all personnel involved in the review process have completed the necessary training programs. Organizations with are 40% more likely to succeed in inspections without significant issues.
- Collect Quality Control Records: Accumulate all , including batch records, testing results, and any deviations from established procedures, to showcase adherence and quality assurance. Thorough can significantly reduce the risk of non-compliance, as evidenced by successful case studies.
- Establish : Implement a robust system for tracking changes to critical documents, ensuring that all modifications are logged with timestamps and user identification for transparency. Maintaining clear enhances the credibility of your documentation.
- : Document any risk assessments conducted, detailing identified risks and corresponding mitigation strategies. Regular reviews of these assessments are essential for ongoing compliance. Industry experts recommend conducting these assessments at least quarterly to stay ahead of potential issues.
- : Perform a thorough final review of the compiled package to ensure completeness and accuracy. Make necessary adjustments to address any gaps before the evaluation. Integrating feedback from practice evaluations can provide valuable insights into areas needing enhancement.
By adhering to these steps, entities can develop an for that withstands regulatory scrutiny, ultimately enhancing their compliance stance and operational effectiveness.
Avoid Common Mistakes in Audit Readiness
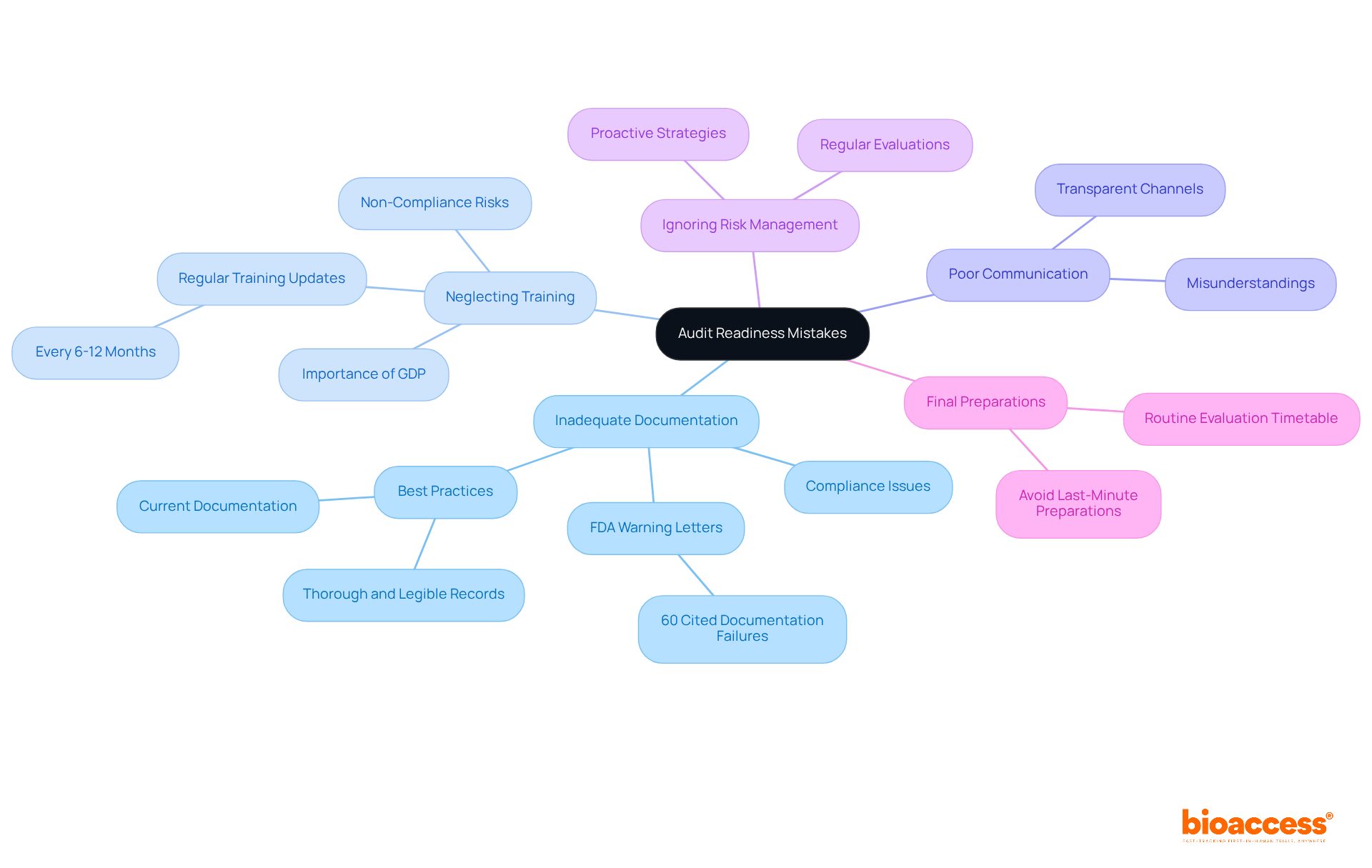
To enhance , organizations must avoid several common pitfalls:
- : Maintaining complete and accurate records is crucial. can lead to , as over 60% of FDA warning letters in recent years cited documentation failures. Ensure that all records are thorough, legible, and current to support review processes effectively. Remember, “If it isn’t documented, it didn’t happen.”
- Neglecting Training: can result in non-compliance during audits. Regularly updating ensures that all staff are well-versed in current regulations and (GDP). Training refreshers every 6-12 months can help uphold standards and readiness. Notably, 69% of regulatory professionals believe it is safer to err on the side of more documentation than less.
- Poor Communication: A lack of communication among team members can lead to misunderstandings and incomplete preparations. Creating transparent is vital to guarantee that everyone is aligned and aware of their duties in the evaluation process.
- Ignoring : Regular are essential to identify and mitigate potential regulatory issues. Organizations should implement a proactive strategy to address vulnerabilities before they escalate into significant problems.
- Final Preparations: Waiting until the last moment to prepare for a review can lead to oversights and mistakes. Establishing a routine evaluation preparation timetable ensures continuous compliance and enables comprehensive planning well before any assessment.
By addressing these frequent errors, entities can significantly boost their audit readiness package biologics phase iii and increase their likelihood of achieving positive assessment results. Case studies have shown that organizations emphasizing documentation and training experience a marked improvement in their evaluation outcomes.
Utilize Tools and Resources for Effective Preparation
To enhance , organizations must consider several essential tools and resources:
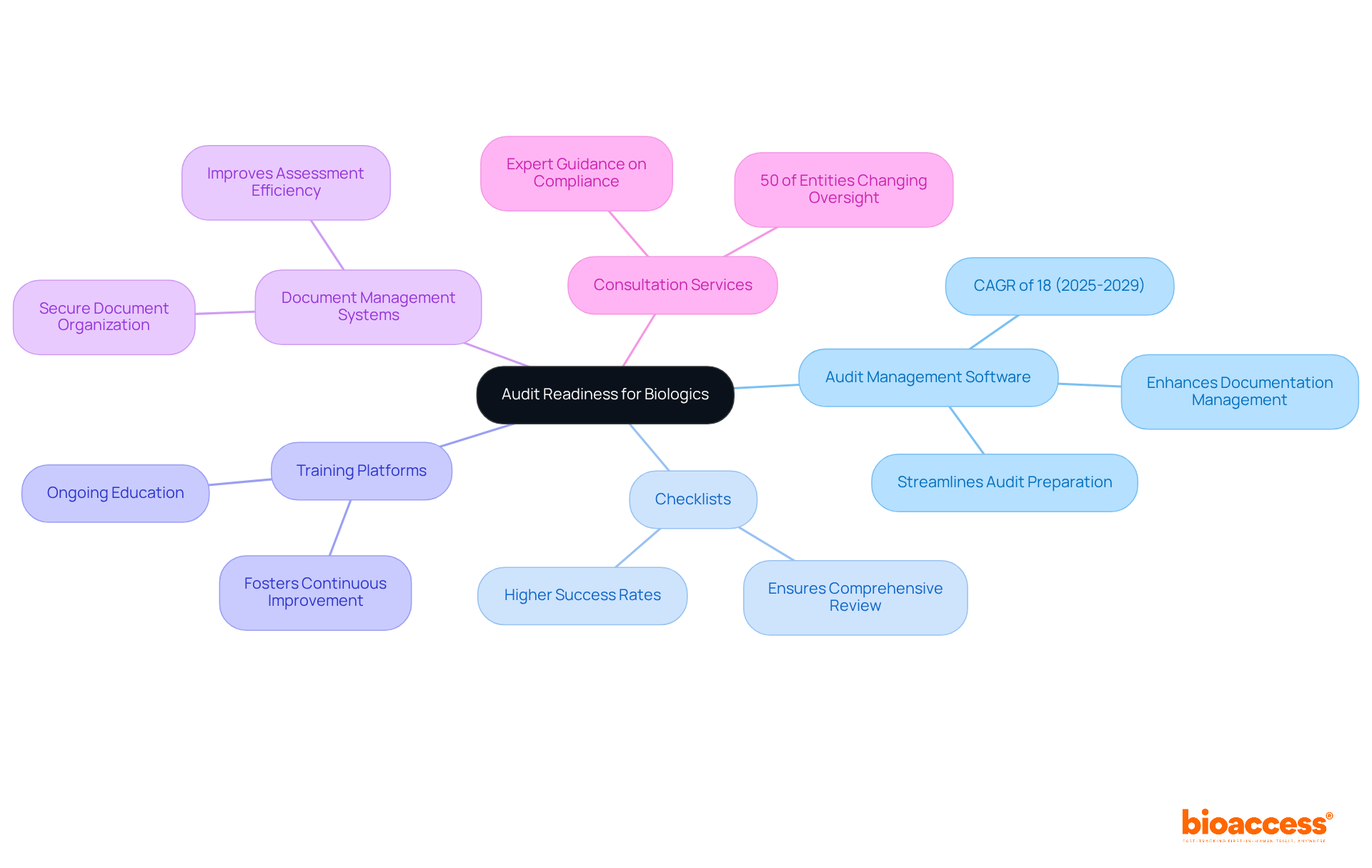
- : Implementing advanced can significantly streamline the audit preparation process. These solutions assist in enhanced documentation management and allow monitoring of , ultimately boosting efficiency and precision in preparation. Notably, the review software market is anticipated to expand at a CAGR of 18% from 2025 to 2029, underscoring the growing significance of embracing such tools.
- Checklists: Employing thorough is crucial. These checklists ensure that all essential elements are included in the review package, acting as a reliable guide throughout the preparation process. Research indicates that entities using structured checklists experience higher success rates in audit readiness.
- Training Platforms: Leveraging online training platforms for ongoing education is vital. These platforms equip staff with up-to-date knowledge on compliance and regulatory requirements, fostering a culture of continuous improvement and preparedness.
- : Utilizing robust allows organizations to securely organize and store critical documents. This guarantees easy access during evaluations, reducing the time spent seeking essential information and improving overall assessment efficiency.
- Consultation Services: Engaging with can provide invaluable insights. Their expertise can guide entities through complex regulatory landscapes, ensuring that all compliance aspects are thoroughly addressed. Significantly, 50% of entities are altering their third-party oversight due to shifts in U.S. enforcement, highlighting the necessity for effective tools in this evolving landscape.
By effectively utilizing these tools and resources, entities can significantly improve their , leading to a smoother and more efficient review process. Additionally, it is important to note that 32% of organizations incurred financial liabilities exceeding $1 million from audits, underscoring the financial implications of inadequate audit readiness.
Conclusion
Mastering the audit readiness package for biologics in Phase III is essential for organizations striving to meet regulatory standards and achieve successful evaluations. This thorough preparation involves not just meticulous documentation but also the establishment of robust quality management systems, comprehensive training, and effective risk management strategies. By concentrating on these core components, organizations can significantly bolster their readiness for regulatory inspections.
Key insights explored throughout the article include the necessity of assembling:
- Regulatory documentation
- Standard operating procedures
- Training records
- Quality control records
- Audit trails
Each of these elements is crucial in laying a solid foundation for compliance, especially concerning oversight by authorities like INVIMA. The article also pointed out common pitfalls to avoid, such as insufficient documentation and neglecting personnel training, which can jeopardize audit readiness.
The importance of a well-structured audit readiness package cannot be overstated. Organizations should leverage tools like:
- Audit management software
- Checklists
- Training platforms
to streamline their preparation process. By adopting best practices and proactively addressing potential challenges, entities can enhance their audit readiness, safeguard operational integrity, and improve their reputation within the biologics sector. Taking these steps is vital for navigating the complexities of regulatory compliance and achieving successful audit outcomes.
Frequently Asked Questions
What is audit readiness for biologics?
Audit readiness for biologics involves preparing organizations for regulatory inspections and evaluations by ensuring that all necessary documentation, processes, and systems are in place to demonstrate compliance with regulatory standards.
What are the key aspects of audit readiness?
Key aspects of audit readiness include thorough documentation, a robust Quality Management System (QMS), proper training of personnel, and effective risk management to identify and mitigate potential compliance issues.
Why is documentation important in audit readiness?
Documentation is crucial because it ensures that all records related to the development, manufacturing, and testing of biologics are complete, accurate, and easily accessible, fulfilling regulatory expectations.
What role does a Quality Management System (QMS) play in audit readiness?
A QMS is vital for aligning with Good Manufacturing Practices (GMP) and other regulatory requirements, ensuring compliance with regulations and maintaining product quality.
How important is training for personnel in the context of audit readiness?
Training is critical as all personnel involved in the production and oversight of biologics must be adequately trained and aware of regulatory requirements to maintain operational integrity.
What components should be included in an audit readiness package for biologics?
An audit readiness package should include regulatory documentation, Standard Operating Procedures (SOPs), training records, quality control records, audit trails, and risk assessment reports.
What is the significance of regulatory documentation in the audit readiness package?
Regulatory documentation, such as Investigational New Drug (IND) applications and Biologics License Applications (BLA), is essential for demonstrating adherence to safety and efficacy standards required by regulatory authorities.
What are Standard Operating Procedures (SOPs) and why are they important?
SOPs are detailed documents outlining the processes for manufacturing, testing, and quality control of biologics, ensuring compliance with established protocols and reducing risks during evaluations.
How do training records contribute to audit readiness?
Training records document the training sessions for personnel, confirming that staff are knowledgeable about regulatory requirements, which is crucial for maintaining compliance and operational integrity.
What is the purpose of maintaining quality control records?
Quality control records provide evidence of the effectiveness of quality control measures, including batch records and testing results, which are critical for demonstrating product reliability and safety.
Why are audit trails necessary in an audit readiness package?
Audit trails track changes and updates to critical documents and processes, ensuring transparency and accountability, which assists in the review process and conforms to regulatory oversight necessities.
What is the importance of risk assessment reports in audit readiness?
Risk assessment reports document evaluations of potential regulatory issues, helping organizations proactively address vulnerabilities and improve overall compliance with regulatory guidelines.
List of Sources
- Define Audit Readiness for Biologics
- “BioBoston Consulting: Leading Life Sciences and Biotechnology Strategy Experts” (https://biobostonconsulting.com/best-practices-for-validation-documentation-ensuring-compliance-and-audit-readiness)
- reedsmith.com (https://reedsmith.com/articles/fda-inspections-in-2025-heightened-rigor-data-driven-targeting-and-increased-surveillance)
- FDA Inspection Readiness in 2026: A Complete Guide & Checklist (https://thefdagroup.com/blog/how-to-prepare-for-an-fda-inspection)
- Audit Readiness: Meaning & Key Pharma Steps (https://masuuglobal.com/what-is-audit-readiness)
- Identify Key Components of the Audit Readiness Package
- Audit Readiness: Meaning & Key Pharma Steps (https://masuuglobal.com/what-is-audit-readiness)
- Regulatory Documentation: Importance & Examples (https://vaia.com/en-us/explanations/medicine/pharmacy/regulatory-documentation)
- Statistical Tools for Biologics Stability Testing (https://bioprocessintl.com/qa-qc/comprehensive-stability-assessment-of-biotechnological-products-degradation-mechanisms-statistical-tools-and-regulatory-compliance-part-1)
- Statistical and regulatory considerations in assessments of interchangeability of biological drug products – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC4046084)
- Compile the Audit Readiness Package Step-by-Step
- Audit Readiness Guide | 5 Essential Steps (https://cgmpconsulting.com/guide-to-audit-readiness)
- Statistical Tools for Biologics Stability Testing (https://bioprocessintl.com/qa-qc/comprehensive-stability-assessment-of-biotechnological-products-degradation-mechanisms-statistical-tools-and-regulatory-compliance-part-1)
- Audit Readiness Checklist: How Life Science Companies Can Prepare (https://wolfandco.com/resources/insights/audit-readiness-checklist-how-life-science-companies-can-prepare)
- Audit report quotes | AnalytiChem (https://biotrading.com/biotrading/quality/audit-report-quotes)
- Avoid Common Mistakes in Audit Readiness
- Top 5 Mistakes in Pharma Regulatory Audit Preparation USA (https://confiancapharmazon.com/pharma-regulatory-audit-preparation-usa-mistakes)
- 94% of compliance officers say: No documentation? It’s not done (https://ama-assn.org/practice-management/physician-health/94-compliance-officers-say-no-documentation-it-s-not-done)
- The Perils of Poor Documentation – Saiva AI (https://saiva.ai/the-perils-of-poor-documentation)
- The Impact of Poor Documentation on Regulatory Compliance – JAF Consulting, Inc. (https://jafconsulting.com/the-impact-of-poor-documentation-on-regulatory-compliance)
- Utilize Tools and Resources for Effective Preparation
- The Rising Cost of Software Compliance: 2025 Survey Highlights Growing Audit Frequency and Financial Impact on Businesses (https://businesswire.com/news/home/20250122806719/en/The-Rising-Cost-of-Software-Compliance-2025-Survey-Highlights-Growing-Audit-Frequency-and-Financial-Impact-on-Businesses)
- Audit Software Market Growth Analysis – Size and Forecast 2025-2029 | Technavio (https://technavio.com/report/audit-software-market-analysis)
- 110 security and compliance statistics for tech leaders to know in 2025 (https://vanta.com/resources/compliance-statistics)
- 130+ Compliance Statistics & Trends to Know for 2026 (https://secureframe.com/blog/compliance-statistics)
- Audit Software Market Size, Share & Analysis Report, 2025-2034 (https://gminsights.com/industry-analysis/audit-software-market)
