Introduction
Understanding the intricacies of clinical documentation is essential in today’s healthcare landscape, where precision and accuracy significantly impact patient care and organizational compliance. The Certified Clinical Documentation Specialist (CCDS) credential not only signifies a hallmark of expertise in this field but also opens doors to enhanced career opportunities and increased earning potential.
However, with evolving regulations and the complexities of medical record-keeping, aspiring professionals must navigate the path to certification effectively to fully leverage its benefits. How can they achieve this? The answer lies in a commitment to understanding the nuances of this vital discipline.
Define CCDS: Understanding Clinical Documentation Specialist Certification
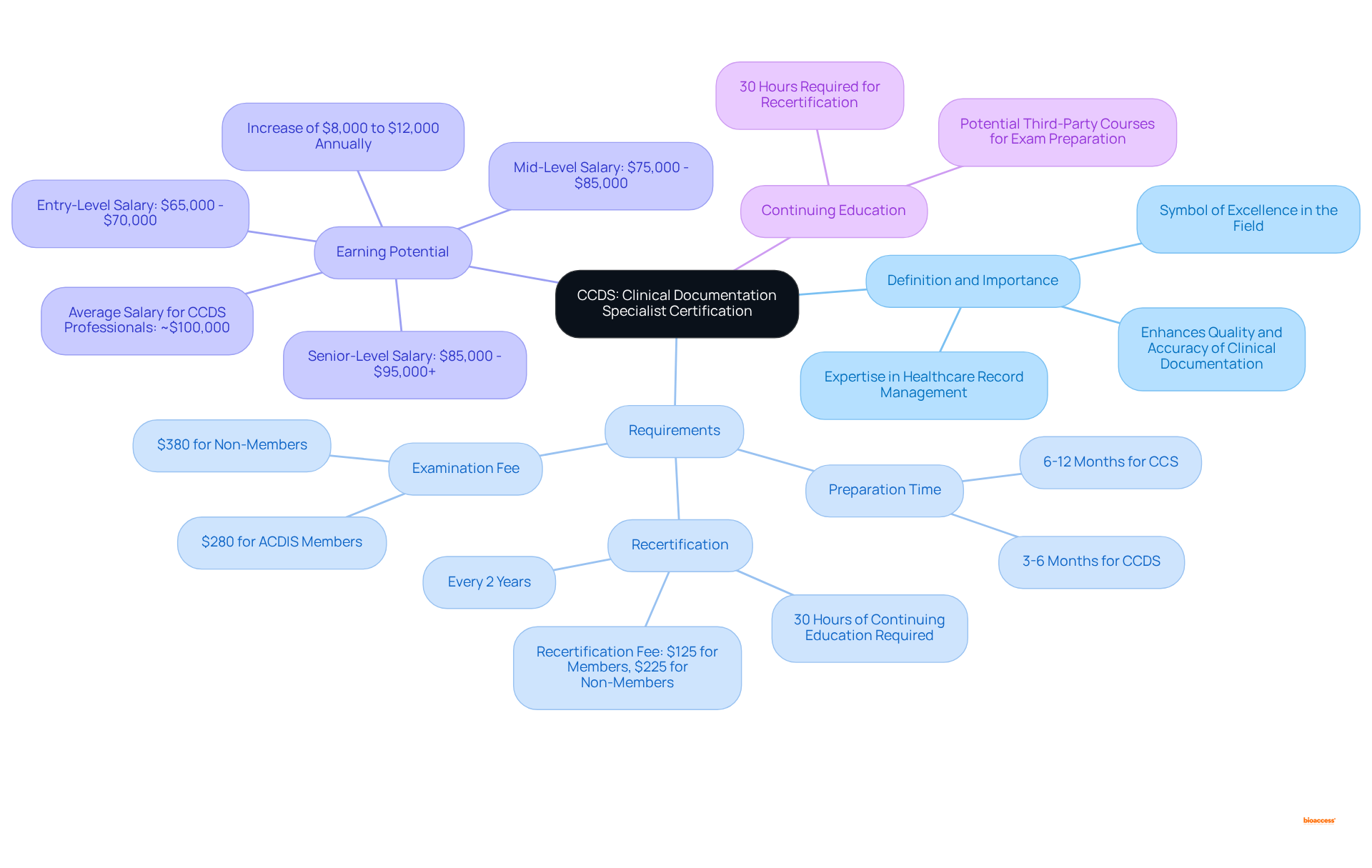
The serves as a professional accolade, signifying an individual’s expertise in , which relates to . This esteemed credential is awarded to healthcare professionals who demonstrate a comprehensive understanding of , , and regulatory requirements, reflecting the . Recognized as a symbol of excellence in the field, the indicates that the holder possesses the essential skills necessary to enhance the quality and accuracy of —an aspect crucial for patient care, billing, and compliance with healthcare regulations.
Qualified professionals often experience a significant increase in their earning potential, with this credential contributing between $8,000 and $12,000 to annual salaries. Typically, preparation for the credential spans 3-6 months, and maintaining the qualification necessitates , alongside the completion of 30 hours of continuing education. Furthermore, the examination fee is set at $280 for ACDIS members and $380 for non-members.
As the healthcare landscape evolves, the importance of maintaining high standards in medical records becomes increasingly clear. This qualification emerges as a valuable asset for individuals aspiring to excel in this domain.
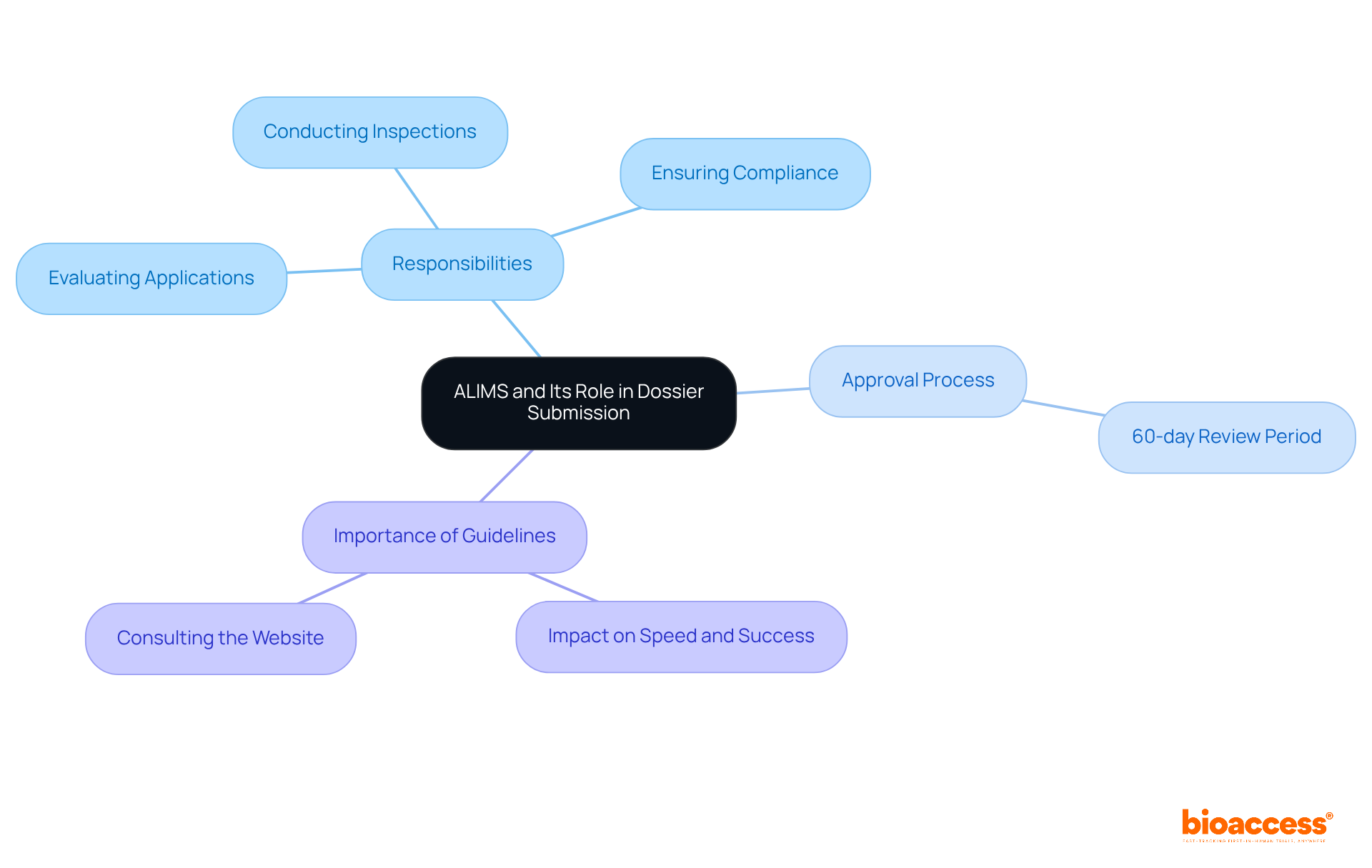
Explore the Importance of CCDS Certification in Clinical Research
This accreditation is critical in medical research, ensuring that documentation adheres to the highest standards of precision and thoroughness. Such meticulousness is essential for preserving the integrity of research trials, as it directly influences data quality and . The is crucial as play a pivotal role in accurately documenting all patient interactions and healthcare data, which is vital for the reliability of research outcomes. Statistics reveal that organizations employing professionals who understand experience significantly enhanced data integrity, leading to more dependable results. Moreover, the in certification bolsters the trustworthiness of research organizations, underscoring their commitment to excellence in record-keeping practices. By fostering a culture of , these organizations not only elevate their operational efficiency but also ensure adherence to regulatory standards, ultimately propelling the advancement of medical innovations.
Bioaccess supports this endeavor through , which encompass:
- Feasibility studies
- Site selection
- Trial setup
- Import permits
- Project management
- Reporting
Each of these services is instrumental in by guaranteeing that records are thorough and compliant with regulatory requirements, which reflects the . Furthermore, these services positively influence local economies by generating jobs and promoting international collaboration, thereby enhancing the overall healthcare landscape. However, common findings in source records, such as inadequate case histories and missing essential files, underscore the challenges in maintaining record integrity. Training and experience in effective record-keeping practices, grounded in , are crucial for research personnel to address these challenges. Additionally, the potential legal ramifications of insufficient records emphasize the necessity of proper qualifications in safeguarding the integrity of research studies.
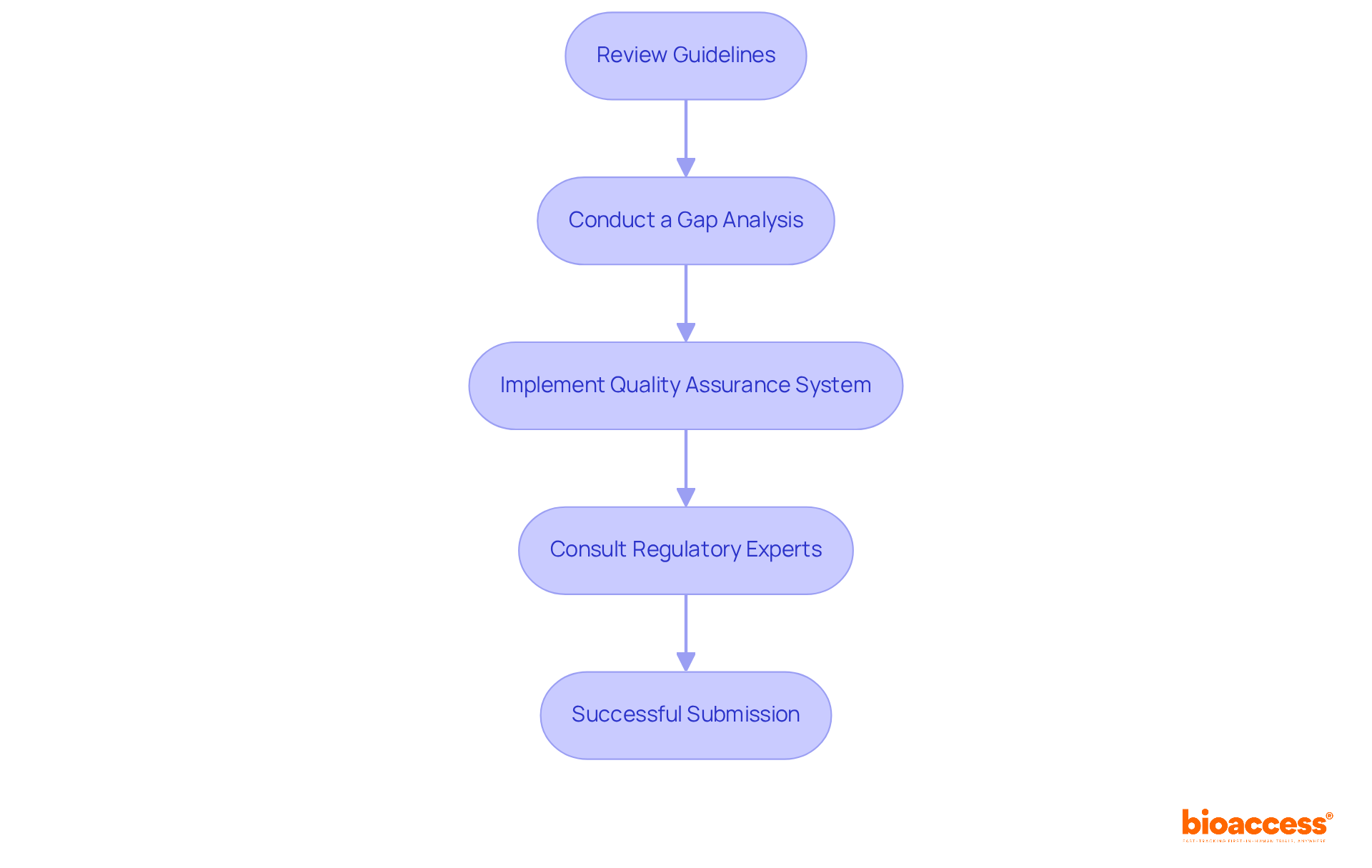
Outline the Steps to Achieve CCDS Certification
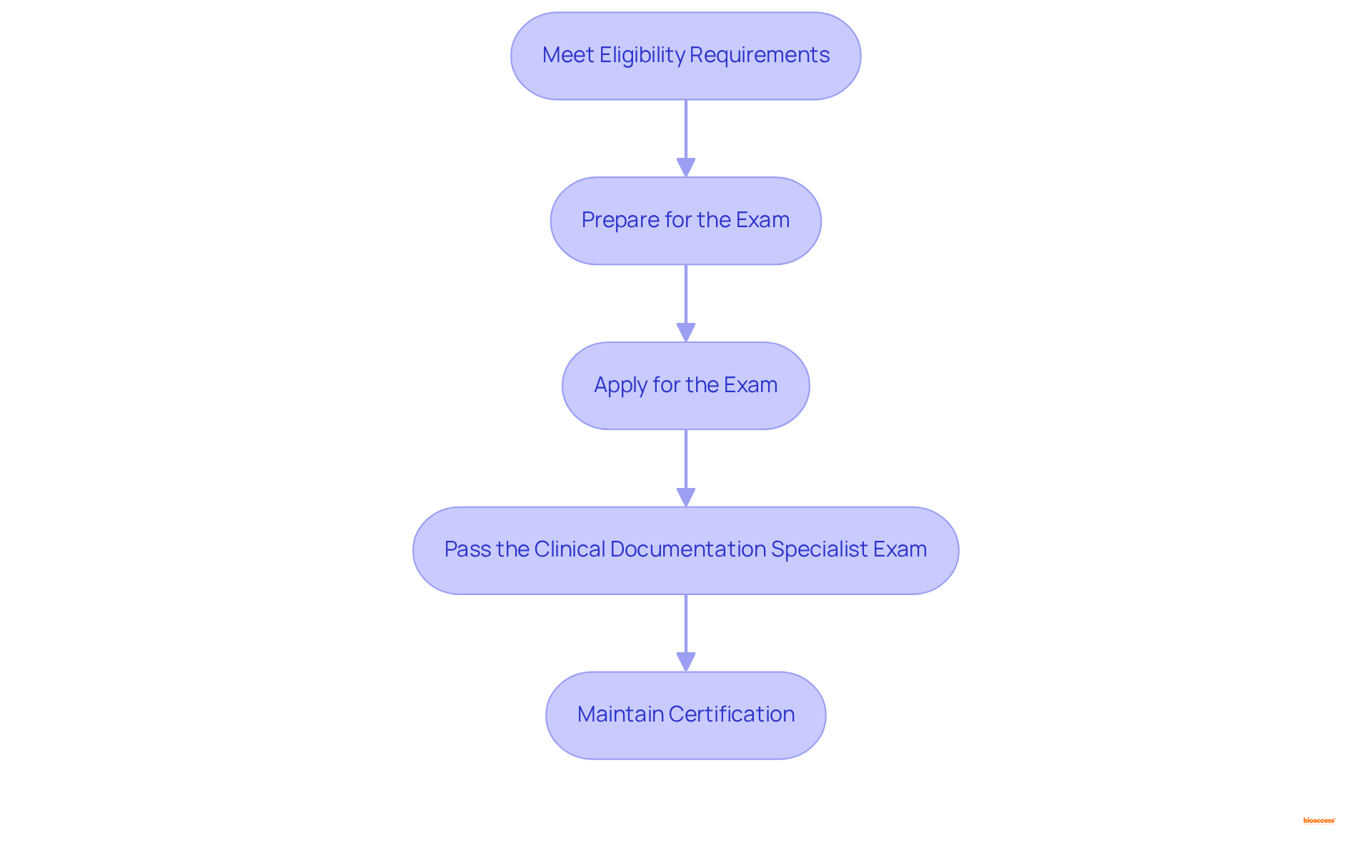
To attain certification, candidates must adhere to several crucial steps:
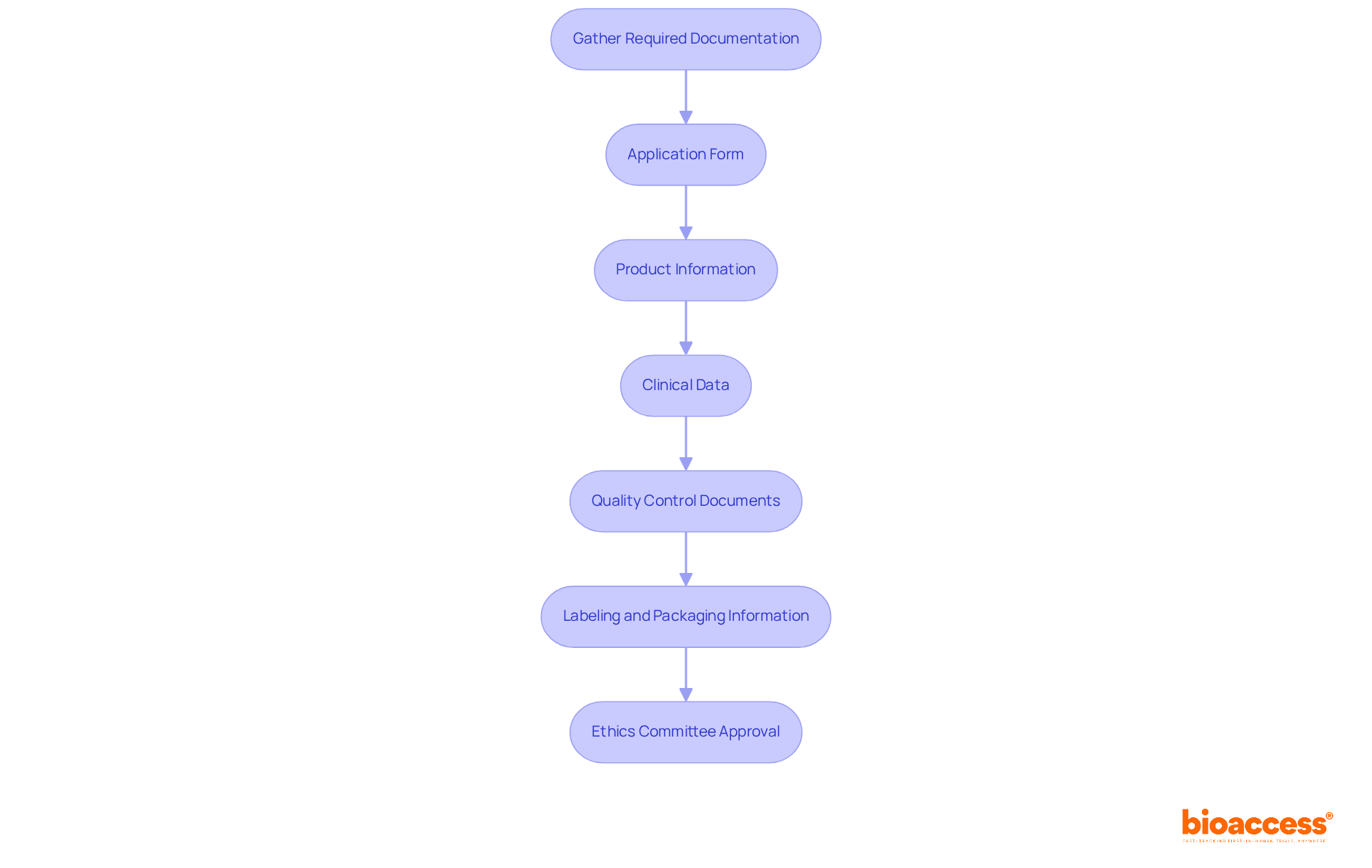
- Meet : Candidates typically require a background in healthcare, such as nursing or health information management, along with relevant work experience in . Specifically, a Bachelor’s degree necessitates a minimum of six years of full-time (CDM) experience, while an Associate’s degree requires at least seven years. For individuals without a degree, eight years of full-time CDM experience is mandatory.
- Prepare for the Exam: Preparation involves reviewing record-keeping standards, coding guidelines, and . Many candidates find success by enrolling in . Organized study materials for the certification exam preparation will soon be accessible.
- Apply for the Exam: Candidates must complete an online application through the ACDIS website and pay the required fees, which are $280 for ACDIS members and $380 for non-members, with an additional $100 fee for international candidates. The renewal fee for ACDIS members is $125, and $225 for non-members.
- Pass the : The exam consists of 120 questions that assess knowledge of and coding guidelines. Recent pass rates indicate a trend of approximately 79.20% for 2023, with previous years showing 75% for 2022 and 77% for 2021, reflecting the preparedness of candidates.
- Maintain Certification: After acquiring the credential, professionals must fulfill , which involve earning 30 continuing education units (CEUs) every two years, with a minimum of 20 CEUs from ACDIS. This ongoing education ensures that remain updated on evolving regulations and coding requirements. Furthermore, those holding the certification must recertify every two years to stay current in their field.
The Beta Exam for the relevant qualification is anticipated to commence in September 2025.
Examine the Benefits of CCDS Certification for Career Growth
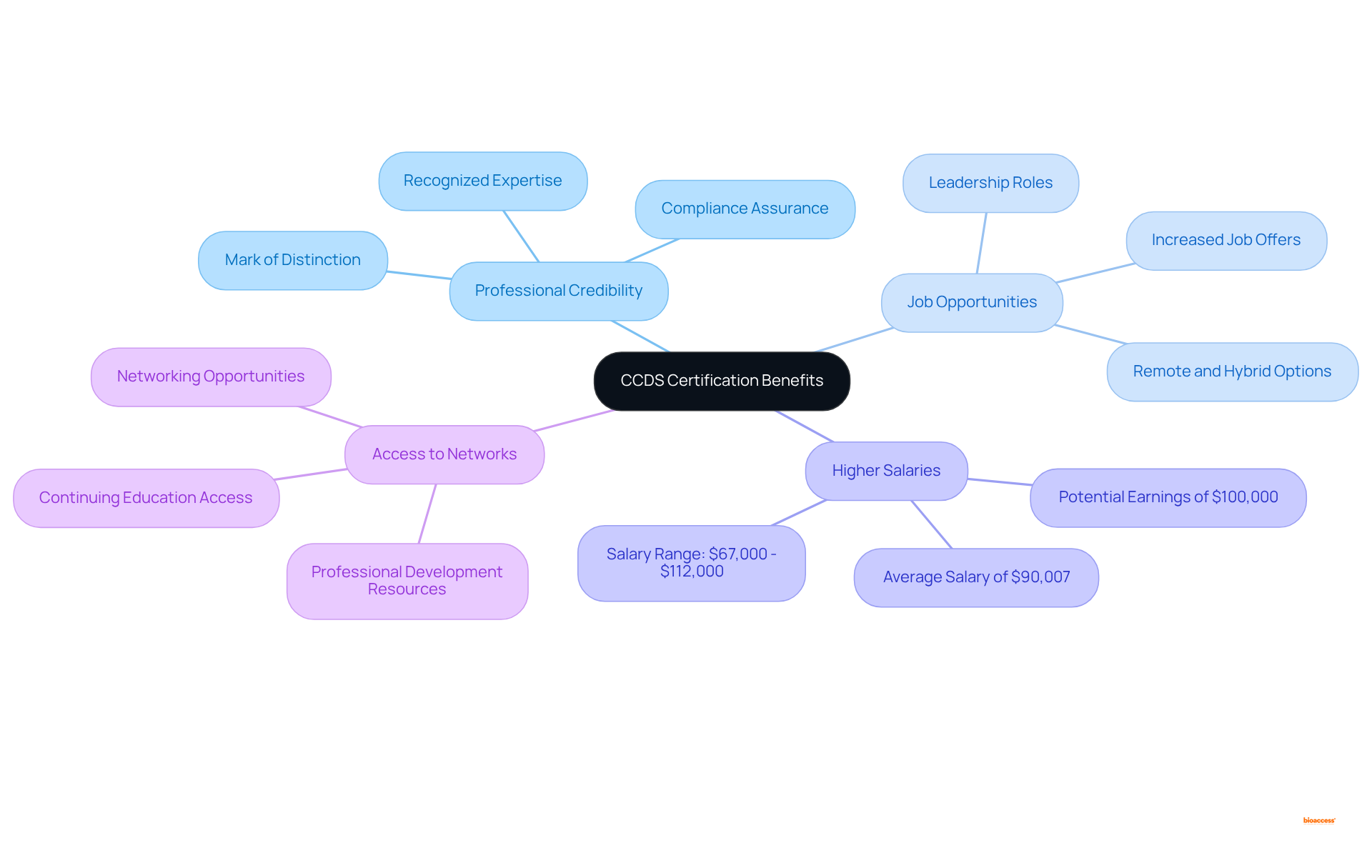
Achieving offers significant advantages for . This credential enhances , as employers recognize it as a . Consequently, this recognition can lead to increased job opportunities and the potential for advancement into .
Moreover, this accreditation is often associated with ; qualified professionals typically earn considerably more than their non-qualified counterparts. Additionally, the qualification provides that facilitate ongoing education and career development.
Discuss the Future of CCDS Certification in Clinical Documentation
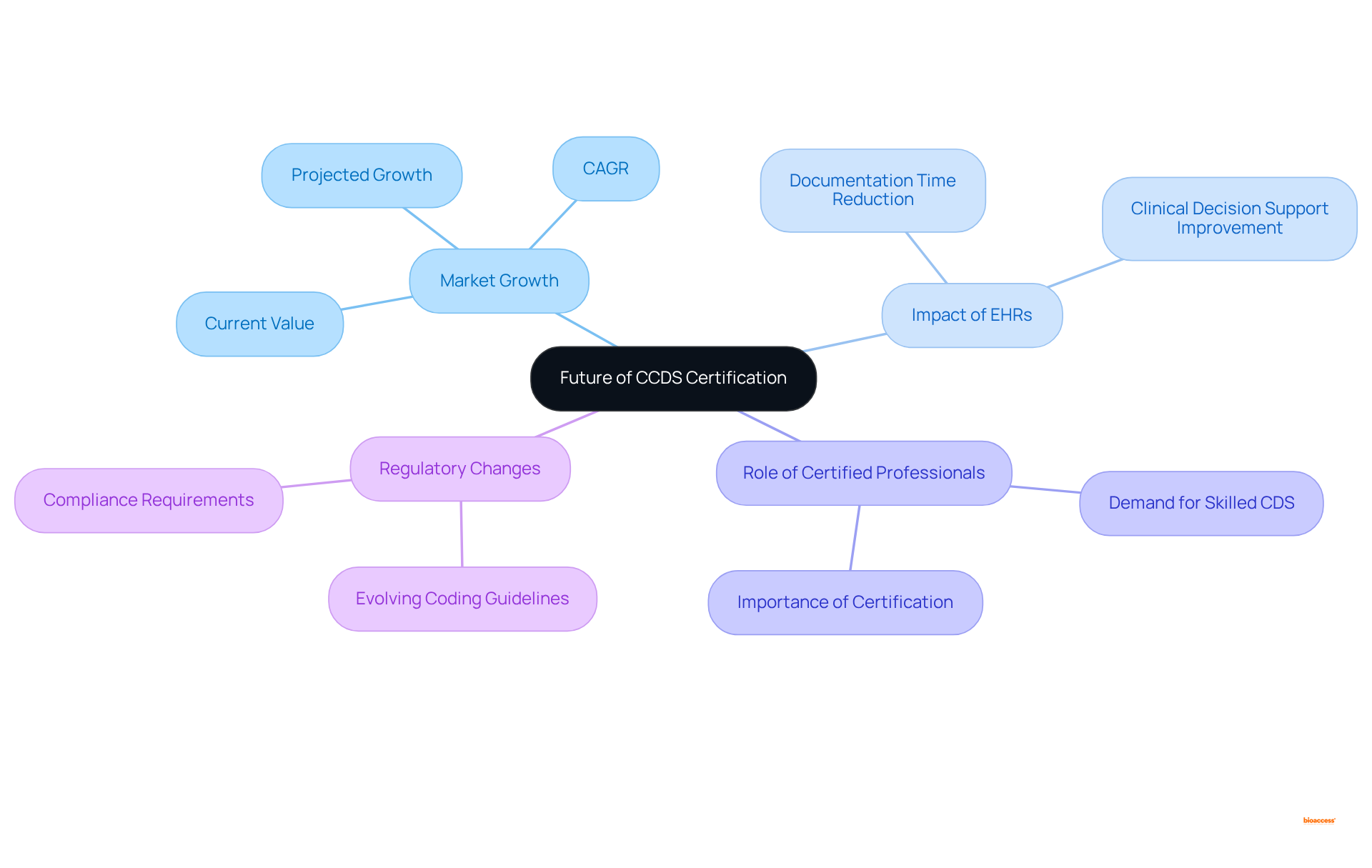
The prospects for this accreditation are set for significant growth as the healthcare industry increasingly prioritizes accurate medical records. The widespread adoption of electronic health records (EHRs)—with 78% of office-based physicians implementing these systems—has revolutionized documentation practices. This transformation has resulted in a reported 75% reduction in documentation time and an 85% improvement in . Such advancements not only enhance patient care but also escalate the demand for skilled . As regulatory frameworks and coding guidelines evolve, this credentialing will adapt to reflect these changes, ensuring that maintain the most up-to-date knowledge and skills. This ongoing evolution will solidify their role as essential contributors to , ultimately and optimizing reimbursement processes.
The is projected to expand from USD 4,205 million in 2024 to USD 7,499.52 million by 2032, at a compound annual growth rate (CAGR) of 7.5%. This anticipated growth underscores the increasing need for , highlighting the crucial role of qualified experts in navigating the complexities of modern healthcare. Furthermore, healthcare providers transitioning to value-based care models report a 50% enhancement in quality metrics, which emphasizes the critical importance of . Notably, 65% of CDI experts possess the credential, underscoring its significance within the industry. The remains promising, with a projected growth of 7% through 2029, further underscoring the necessity of .
Conclusion
Understanding the significance of the Certified Clinical Documentation Specialist (CCDS) certification is essential for healthcare professionals aiming to enhance their expertise in clinical documentation. This credential not only validates a professional’s skills in maintaining accurate healthcare records but also signifies their commitment to improving patient care, compliance, and operational efficiency within healthcare settings.
The CCDS certification process encompasses critical elements such as:
- Eligibility requirements
- Preparation strategies
- Ongoing education necessary to maintain this esteemed credential
The benefits of obtaining this certification extend beyond personal career growth; it plays a vital role in ensuring high standards in clinical documentation, thereby influencing the integrity of medical research and the overall quality of healthcare services.
As the healthcare landscape continues to evolve with advancements in technology and regulatory demands, the CCDS certification remains a crucial asset for professionals in the field. Embracing this certification not only enhances individual career prospects but also contributes to the broader mission of delivering quality healthcare. For those contemplating this path, pursuing CCDS certification can lead to rewarding opportunities and the chance to make a meaningful impact in the healthcare industry.
Frequently Asked Questions
What does CCDS stand for?
CCDS stands for Certified Clinical Documentation Specialist, which is a professional credential signifying expertise in healthcare record management.
What are the key responsibilities of a Certified Clinical Documentation Specialist?
A Certified Clinical Documentation Specialist is responsible for ensuring the quality and accuracy of clinical documentation, which is crucial for patient care, billing, and compliance with healthcare regulations.
How does obtaining CCDS certification impact earning potential?
Professionals with CCDS certification often experience a significant increase in their earning potential, with the credential contributing between $8,000 and $12,000 to annual salaries.
What is the typical preparation time for the CCDS certification exam?
Preparation for the CCDS certification exam typically spans 3 to 6 months.
What are the requirements to maintain CCDS certification?
To maintain CCDS certification, individuals must undergo recertification every two years and complete 30 hours of continuing education.
How much does it cost to take the CCDS certification exam?
The examination fee is $280 for ACDIS members and $380 for non-members.
Why is CCDS certification important in clinical research?
CCDS certification is critical in clinical research as it ensures that documentation adheres to high standards of precision and thoroughness, which is essential for data quality and regulatory compliance.
What role do Certified Clinical Documentation Specialists play in research?
Certified Clinical Documentation Specialists accurately document all patient interactions and healthcare data, which is vital for the reliability of research outcomes.
How does CCDS certification affect the integrity of research organizations?
Organizations employing CCDS-certified professionals experience enhanced data integrity, which bolsters the trustworthiness of research organizations and underscores their commitment to excellence in record-keeping practices.
What services does Bioaccess provide to support CCDS certification?
Bioaccess provides comprehensive clinical trial management services, including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting, all of which facilitate CCDS certification by ensuring thorough and compliant records.
What challenges exist in maintaining record integrity in clinical research?
Common challenges include inadequate case histories and missing essential files, which can undermine record integrity.
What principles are important for research personnel to address record-keeping challenges?
Training and experience in effective record-keeping practices grounded in ICH-GCP (International Council for Harmonisation – Good Clinical Practice) principles are crucial for research personnel.
What are the potential legal ramifications of insufficient records in research?
Insufficient records can lead to legal issues, emphasizing the necessity of proper qualifications to safeguard the integrity of research studies.
List of Sources
- Define CCDS: Understanding Clinical Documentation Specialist Certification
- intelycare.com (https://intelycare.com/career-advice/ccds-certification-overview-and-faq)
- sprypt.com (https://sprypt.com/blog/how-much-do-clinical-documentation-specialists-make)
- acdis.org (https://acdis.org/cdi-week/2024-cdi-week-industry-overview-survey)
- acdis.org (https://acdis.org/cdi-week/2023-cdi-week-industry-overview-survey)
- acdis.org (https://acdis.org/articles/note-associate-editorial-director-quotes-note)
- Explore the Importance of CCDS Certification in Clinical Research
- adiintellect.com (https://adiintellect.com/2024/07/05/accurate-documentation-in-clinical-trials)
- acdis.org (https://acdis.org/articles/note-associate-editorial-director-quotes-note)
- (PDF) Good documentation practice in clinical research (https://researchgate.net/publication/51469891_Good_documentation_practice_in_clinical_research)
- finenessinstitute.com (https://finenessinstitute.com/why-documentation-is-important-in-clinical-research)
- acdis.org (https://acdis.org/certification/ccds/about)
- Outline the Steps to Achieve CCDS Certification
- scdm.org (https://scdm.org/ccds-certification)
- vivian.com (https://vivian.com/certifications/ccds)
- ibhre.org (https://ibhre.org/about-ibhre/statistical-data)
- Examine the Benefits of CCDS Certification for Career Growth
- intelycare.com (https://intelycare.com/career-advice/ccds-certification-overview-and-faq)
- norwood.com (https://norwood.com/salaries-improving-for-cdi-specialist-according-to-acdis-annual-survey)
- payscale.com (https://payscale.com/research/US/Certification=Certified_Clinical_Documentation_Specialist_(CCDS)/Salary)
- sprypt.com (https://sprypt.com/blog/how-much-do-clinical-documentation-specialists-make)
- acdis.org (https://acdis.org/faq/what-are-benefits-ccds-certification)
- Discuss the Future of CCDS Certification in Clinical Documentation
- bls.gov (https://bls.gov/ooh/healthcare/medical-records-and-health-information-technicians.htm)
- linkedin.com (https://linkedin.com/pulse/state-cdi-coding-hiring-2025-actionable-market-insights-chris-madden-mf74c)
- Clinical Documentation Improvement Market Size, Share and Forecast 2032 (https://credenceresearch.com/report/clinical-documentation-improvement-market)
- sprypt.com (https://sprypt.com/blog/how-much-do-clinical-documentation-specialists-make)