Introduction
Diversity in clinical trials transcends being a mere regulatory checkbox; it stands as a critical element that significantly influences the validity and applicability of medical research outcomes. By incorporating a broader spectrum of participants from diverse racial, ethnic, and socioeconomic backgrounds, researchers can uncover essential differences in treatment responses, ultimately leading to more effective healthcare solutions. Yet, with less than 5% of eligible individuals participating in clinical studies, a pressing question emerges: how can the industry dismantle the barriers to inclusion and ensure that diverse populations are authentically represented in research? This article explores seven innovative strategies designed to enhance diversity in clinical trials, paving the way for more equitable health outcomes across all communities.
bioaccess®: Accelerating Diversity in Clinical Trials Through Global Agility
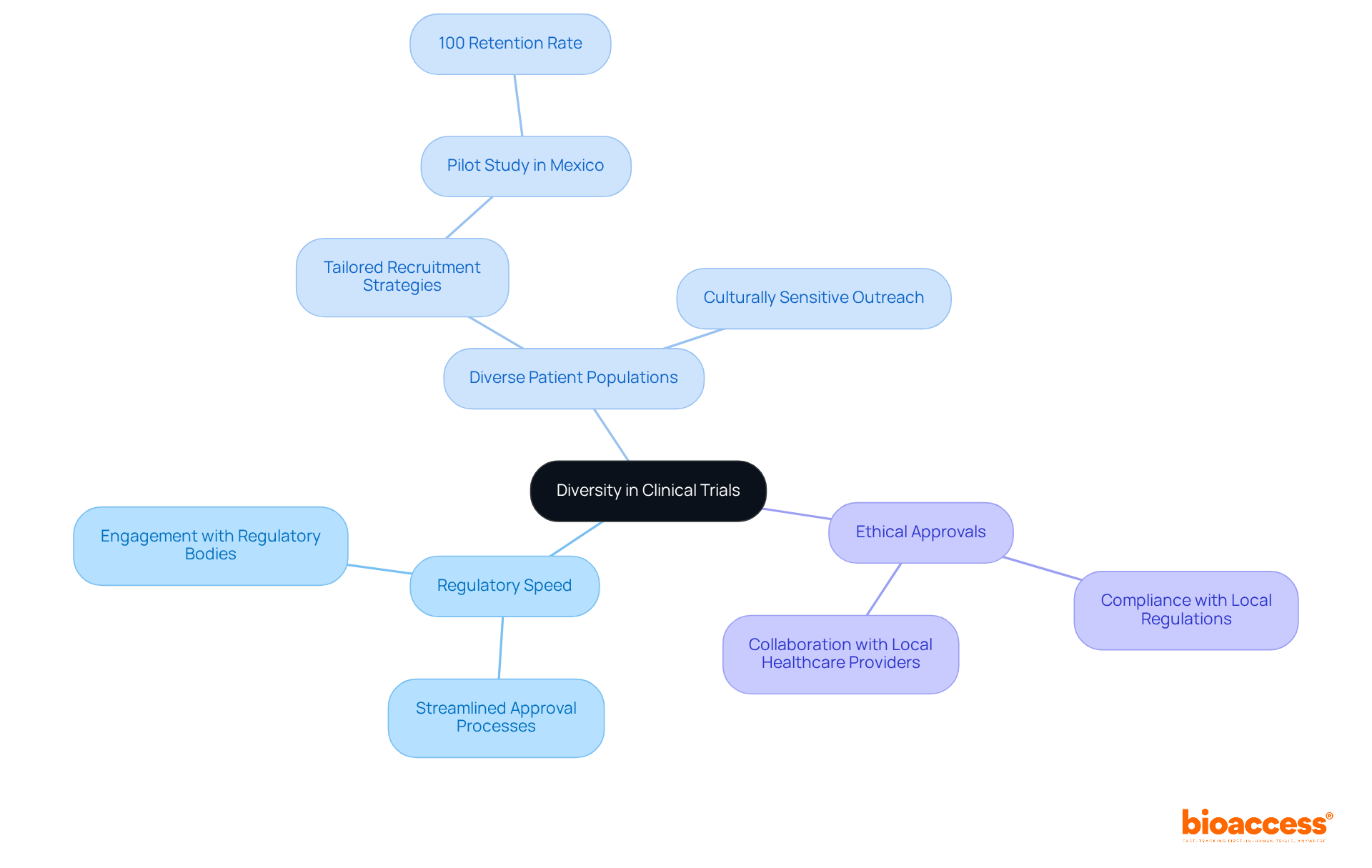
bioaccess® strategically leverages the , the of the Balkans, and Australia’s efficient pathways to expedite and enhance patient enrollment. This multifaceted strategy not only accelerates medical studies but also enhances diversity in , which is essential for producing comprehensive data that accurately reflects diverse patient reactions to therapies.
By judiciously selecting test sites, bioaccess® significantly improves the , addressing a critical gap in research. A compelling example is a pilot study on an electronic patient-reported outcome (ePRO) intervention for breast cancer patients in Mexico, which achieved an impressive 100% retention rate, showcasing the effectiveness of .
Furthermore, as the global research landscape evolves, the necessity for diversity in is underscored by the fact that fewer than 5% of eligible individuals worldwide participate in studies, with rural, Indigenous, and low-income communities often overlooked. Through and collaboration with local healthcare providers, bioaccess® enhances participant engagement and retention, ultimately contributing to the and improved health outcomes for all.
Diverse Representation: Ensuring Accurate Clinical Trial Outcomes
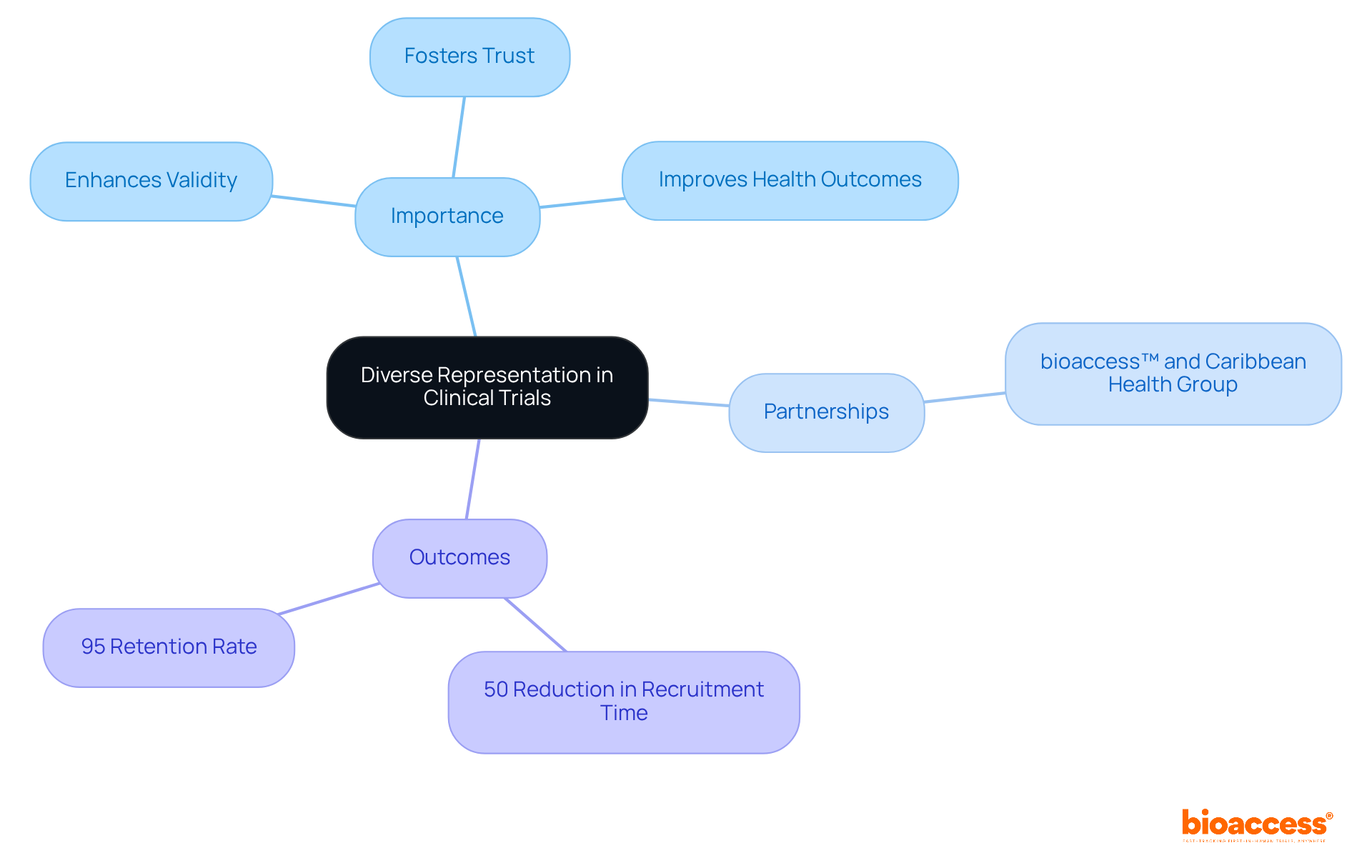
is essential for ensuring that the results are relevant to the wider population. By involving individuals from , studies can to uncover variations in drug effectiveness and safety among different groups. This inclusivity not only enhances the scientific validity of the research but also fosters among underrepresented groups, ultimately leading to and promoting .
The partnership between bioaccess™ and Caribbean Health Group aims to establish Barranquilla as a premier location for , supported by Colombia’s Minister of Health. This initiative and improves the overall efficiency of medical studies.
For instance, GlobalCare Clinical Trials’ collaboration with bioaccess™ has achieved over a 50% and an impressive 95% .
Barriers to Inclusion: Overcoming Challenges in Clinical Trial Diversity
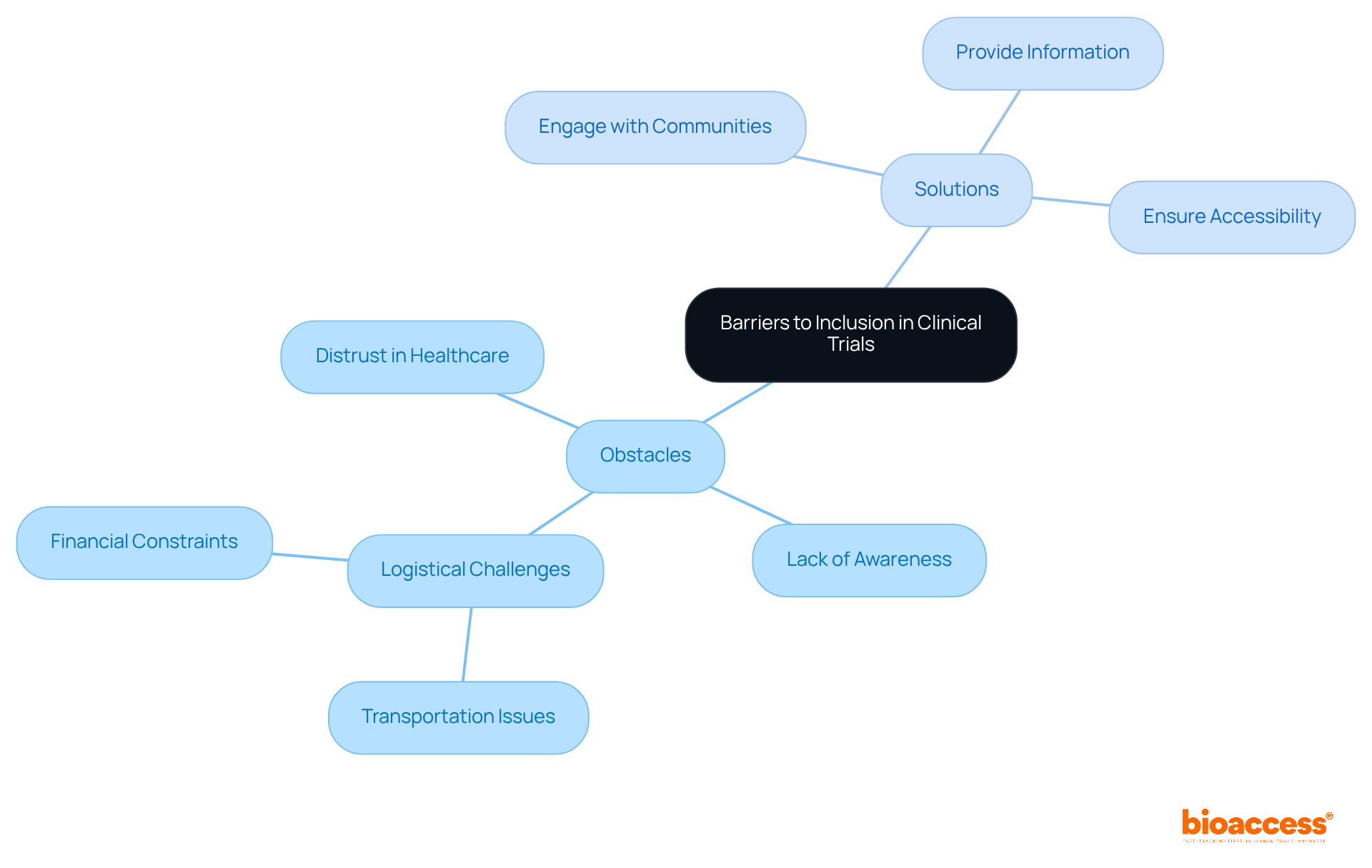
Obstacles to participation in frequently emerge from structural issues, including:
- Distrust in the healthcare system
- A lack of awareness regarding research opportunities
- and
To effectively address these challenges, it is crucial to:
- Engage directly with
- Provide comprehensive information about the benefits of participation
- Ensure that research sites are easily accessible
By tackling these barriers, we not only but also foster enduring relationships with diverse populations, thereby .
Leveraging Technology: Expanding Access to Diverse Populations in Trials
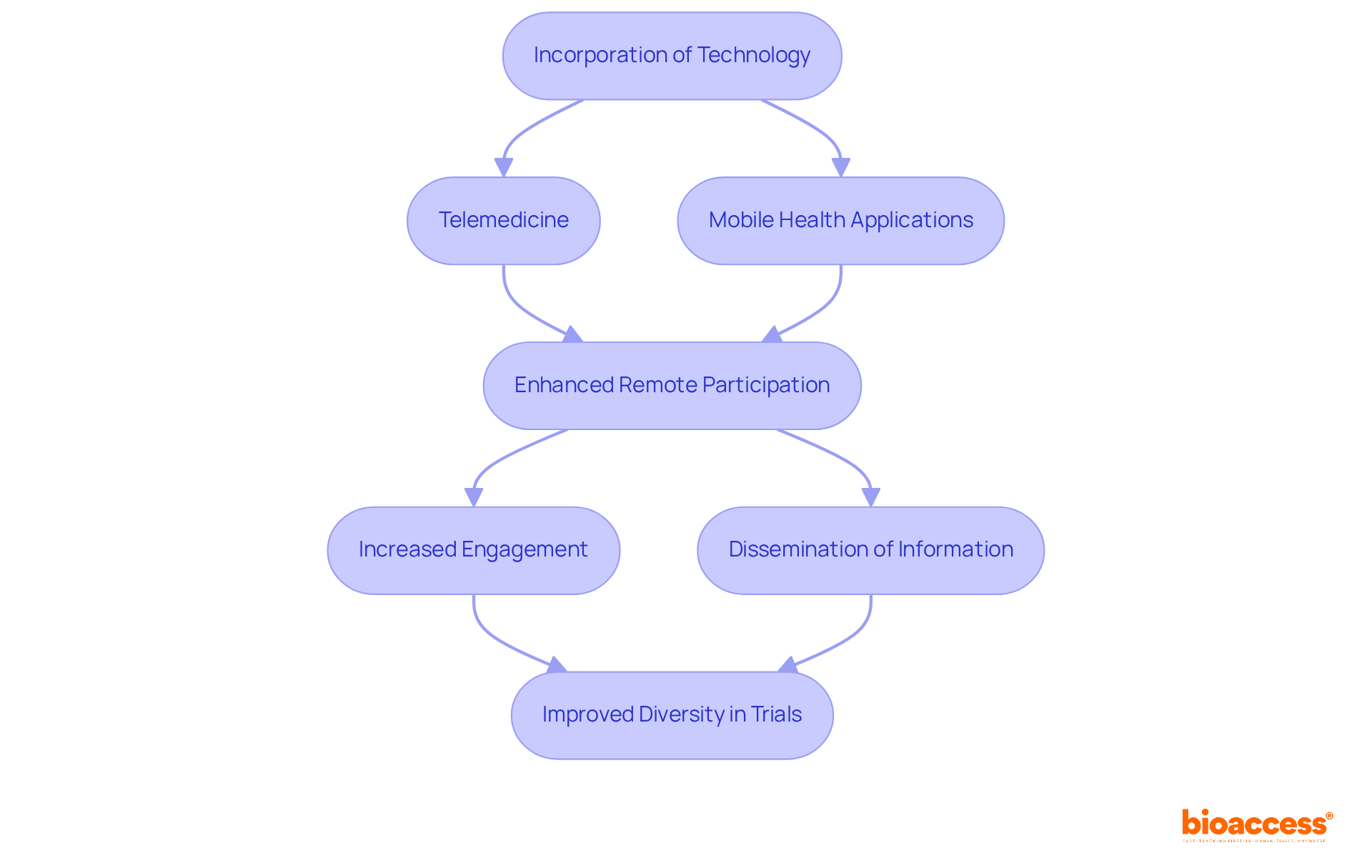
The incorporation of technology, particularly and , plays a crucial role in expanding access to research studies, which is essential for promoting across diverse groups. By facilitating remote participation, these tools minimize the necessity for in-person visits, thereby enhancing accessibility for individuals in .
For instance, has been effectively utilized in numerous medical studies targeting minority populations, allowing participants to engage from their homes—an especially beneficial option for individuals with mobility challenges or those living in remote areas. Furthermore, mobile wellness applications foster between participants and researchers, thereby increasing engagement and adherence to study protocols.
Digital platforms serve as vital resources for disseminating information about medical studies in multiple languages and formats, ensuring that potential participants are well-informed and motivated to participate. Recent advancements in these technologies have yielded promising outcomes, with studies indicating that can significantly improve from underserved demographics, ultimately leading to more generalizable findings and effectively addressing .
Additionally, , such as those offered by bioaccess, encompass essential components like study setup, import permits, and reporting procedures, all of which are critical for . These services ensure that assessments are designed and executed with a focus on , thereby enhancing participation from varied demographic groups.
As Gerald S. Bloomfield emphasizes, increasing awareness of medical research among underrepresented populations is vital, given that only 8% of individuals with cancer in the U.S. engage in research studies. This underscores the importance of leveraging technology and robust management services to in medical research.
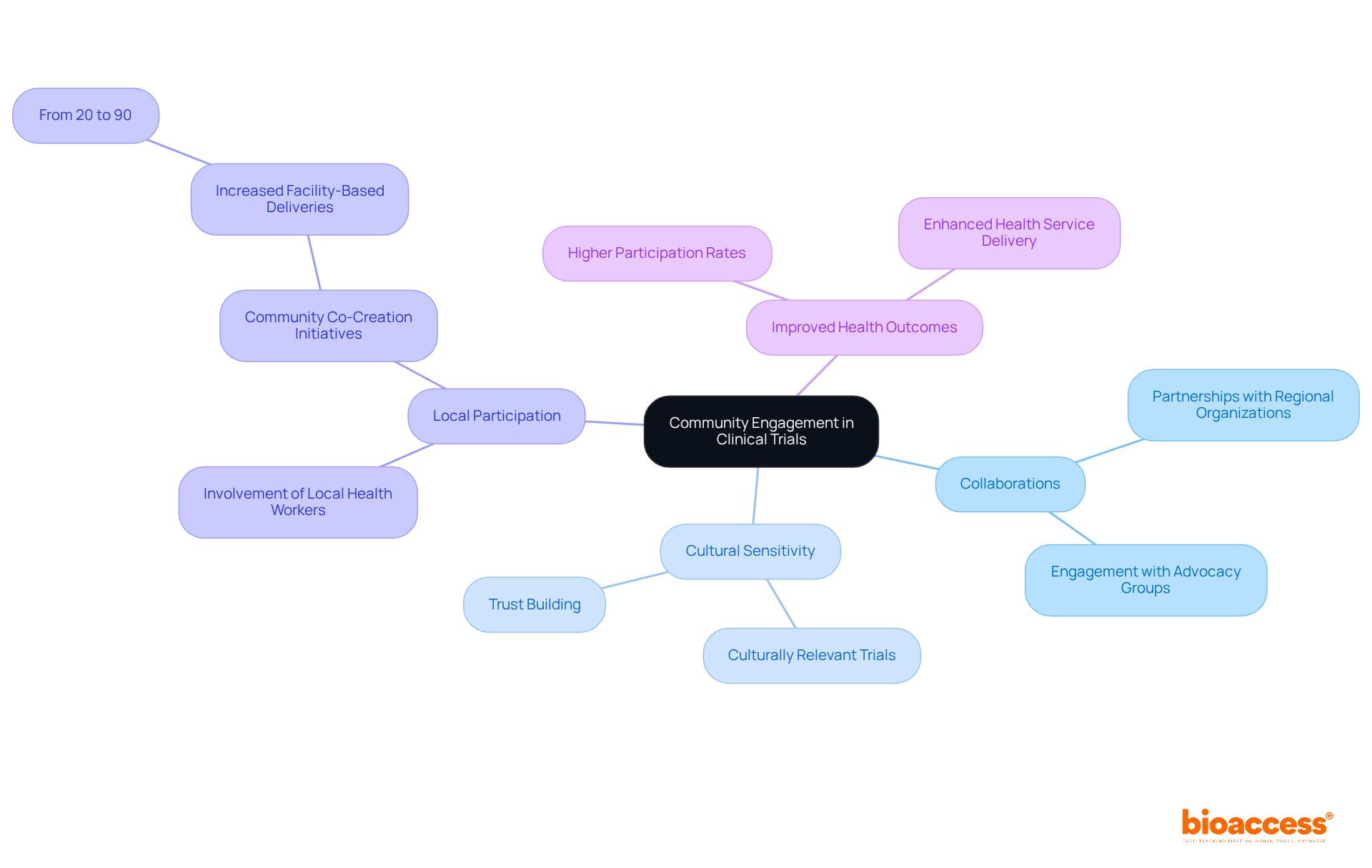
Community Engagement: Building Trust for Diverse Participation
Successful engagement approaches are vital for building confidence and encouraging in research studies. Collaborating with regional organizations, leaders, and advocacy groups significantly enhances the understanding of the importance of clinical studies while addressing potential challenges.
For instance, initiatives that have effectively partnered with health workers demonstrate improved and increased participation rates. By actively involving local participants throughout the research process—from study design to implementation—researchers can develop trials that are . This strategy not only fosters trust but also leads to among diverse populations, as evidenced by initiatives that have elevated facility-based deliveries from 20% to 90% through collaborative efforts.
Ultimately, such are essential for ensuring that incorporates , reflecting the needs and perspectives of all groups and thereby enhancing the overall effectiveness and impact of medical advancements.
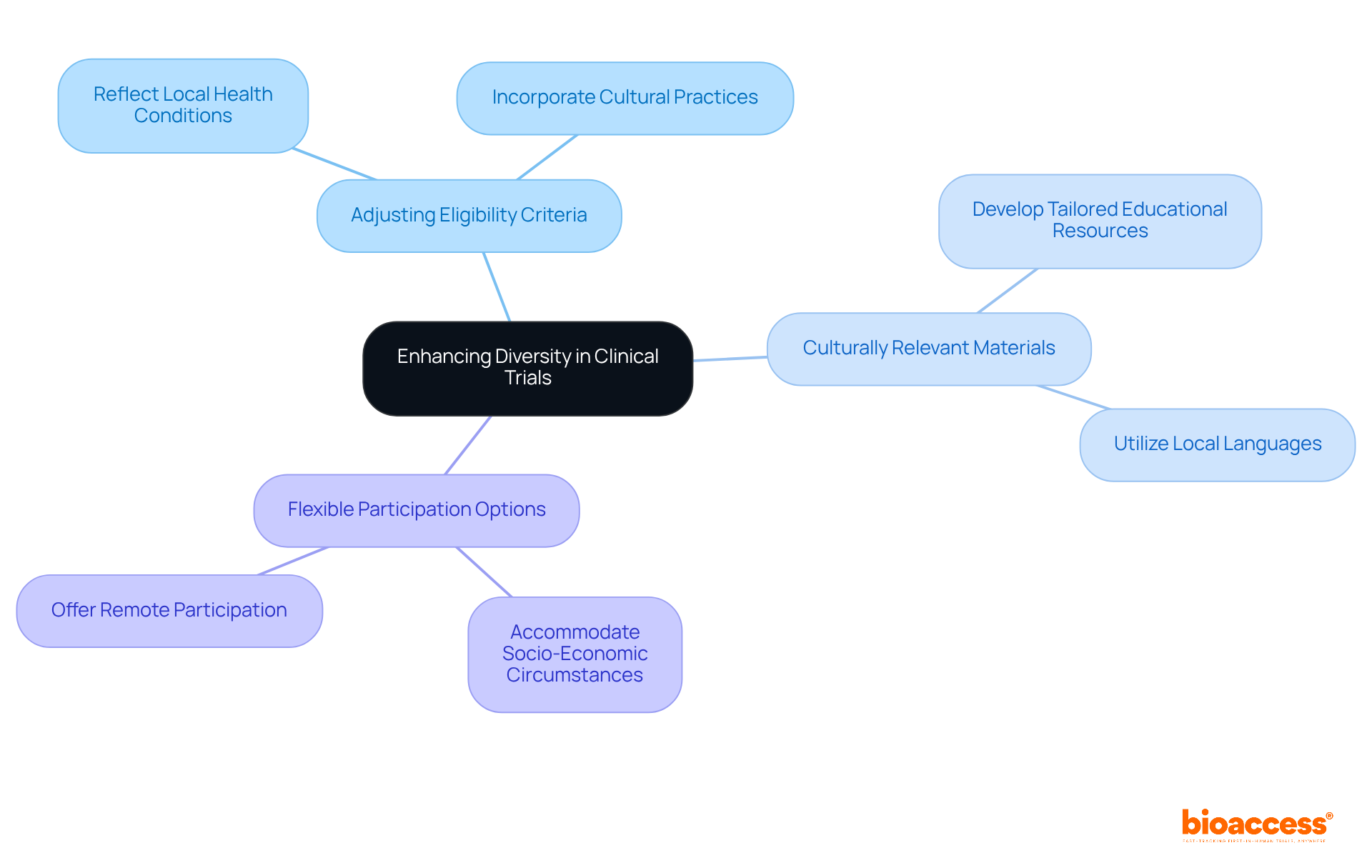
Tailored Strategies: Enhancing Diversity in Clinical Trial Design
To enhance , it is essential to implement tailored strategies that consider the specific needs and preferences of various populations, particularly in the . This includes:
- Adjusting eligibility criteria to reflect local health conditions and cultural practices.
- Providing that resonate with diverse communities.
- Offering that accommodate varying socio-economic circumstances.
As industry leaders have highlighted, understanding the and regulatory landscapes in Latin America is crucial for effective market access. By creating , researchers can promote , attracting a wider variety of participants and resulting in more thorough and relevant outcomes. Involving local specialists and utilizing their knowledge can further improve the significance and efficacy of medical studies in this varied area.
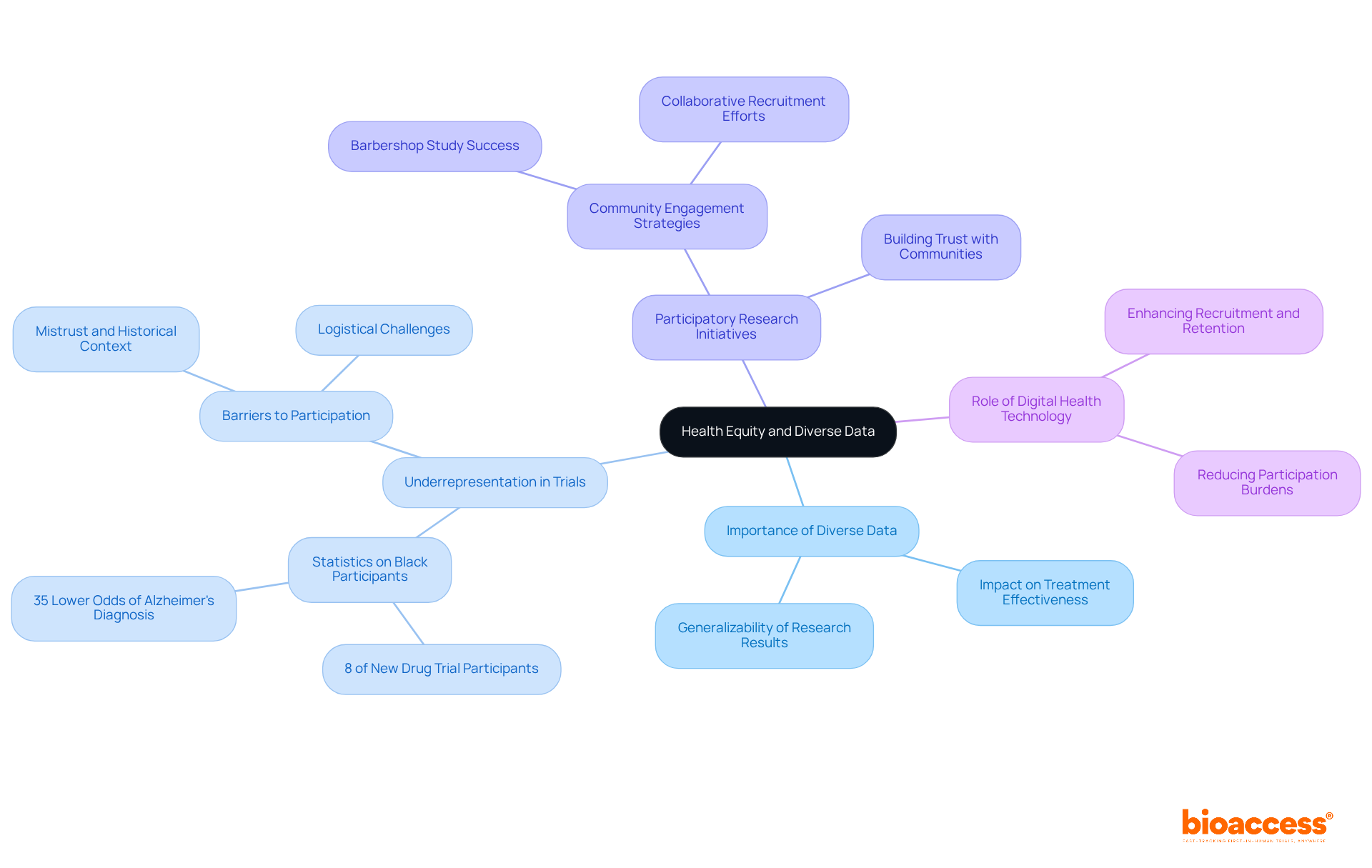
Health Equity: The Impact of Diverse Data on Public Health
Varied information gathered from research studies plays an essential role in influencing public health policies aimed at . By including a broad spectrum of participants, medical research highlights the importance of to understand how diverse populations respond to treatments, which is crucial for developing .
For instance, studies have shown that Black participants are often underrepresented in , with only 8% of new drug trial participants being Black in 2020. The lack of can result in treatments that are less effective for these populations, perpetuating health inequities.
Furthermore, initiatives such as participatory research emphasize to enhance the recruitment and retention of , ultimately leading to more . A significant instance is the randomized medical study that successfully enrolled over 300 non-Hispanic Black men through collaborations with local barbershops, achieving a remarkable retention rate of 95%.
Such efforts not only enhance the generalizability of research results but also promote , ensuring that advancements in medicine benefit all communities and lead to improved wellness outcomes across the board. As public wellness policies increasingly prioritize equity in well-being, the incorporation of varied research data becomes indispensable for developing interventions that genuinely address the needs of all communities.
Additionally, leveraging digital health technology can further improve recruitment and retention of by alleviating participation burdens.
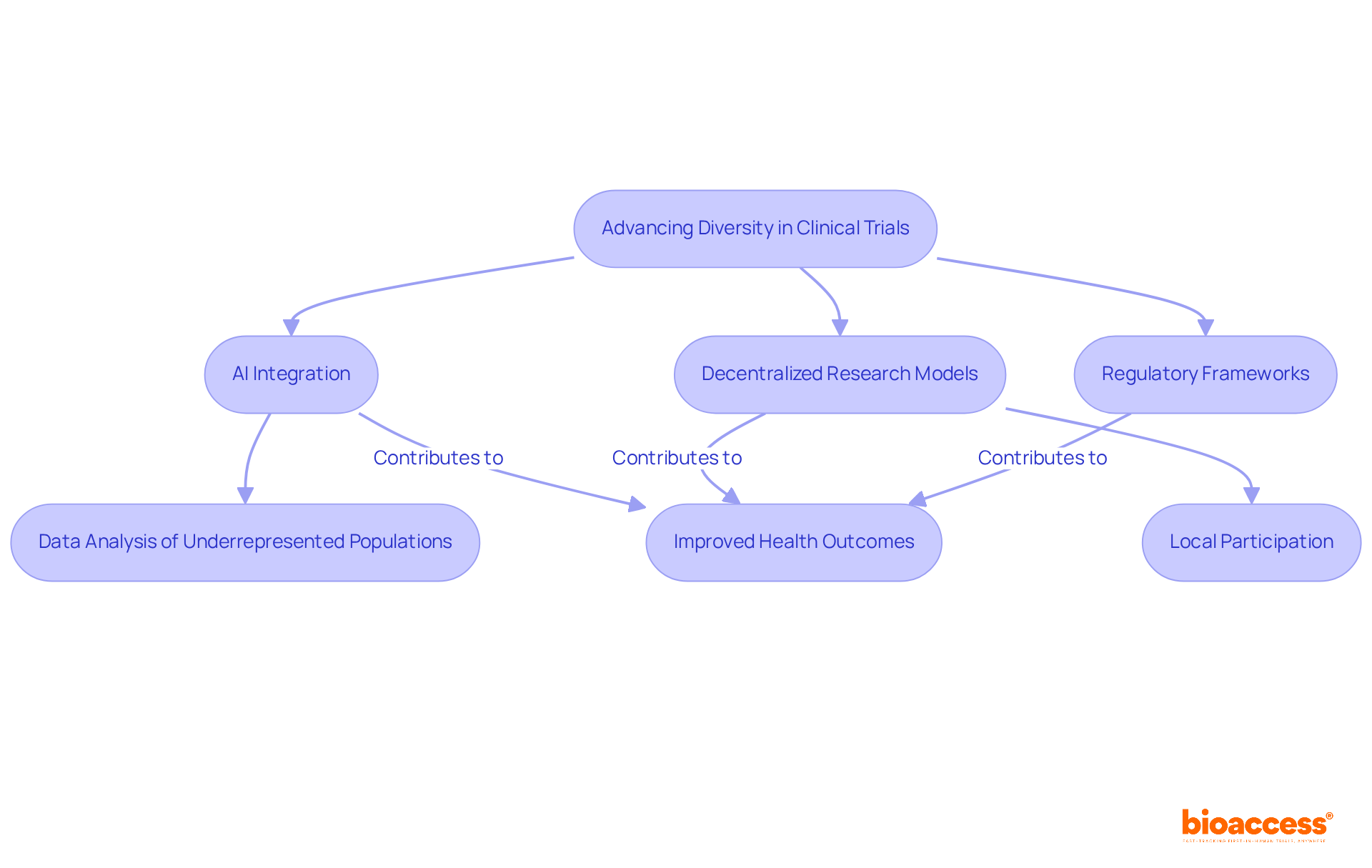
Future Directions: Advancing Diversity in Clinical Trials
The progression of clinical studies necessitates a steadfast commitment to . A pivotal advancement is the integration of , which can significantly streamline the identification and recruitment of . Dr. Sergio Alvarado, Clinical Study Manager at bioaccess®, emphasizes the potential of AI algorithms to analyze extensive datasets, revealing underrepresented populations and ensuring that studies reflect the demographics of the wider community. His work in Latin America, particularly on projects addressing degenerative disc disease and vascular access technologies, showcases how innovative medical solutions can improve by increasing health literacy and participation among diverse populations.
Furthermore, are emerging as a powerful tool to enhance accessibility. By permitting participants to engage in tests from their local areas or even from home, these models dismantle geographical obstacles that frequently impede varied involvement. The success of locally-driven initiatives, such as the in Vanuatu, which involved collective efforts and significantly improved service delivery, underscores the potential of decentralized models in promoting inclusivity. Coupled with , these innovations foster a more equitable research environment.
As the medical research field embraces these advancements, the potential for more inclusive and representative studies increases, ultimately leading to . The dedication to diversity in transcends mere regulatory necessity; it is essential for the progression of medical science and the enhancement of healthcare for varied communities, a vision that bioaccess® actively champions under the guidance of experts like Dr. Alvarado.
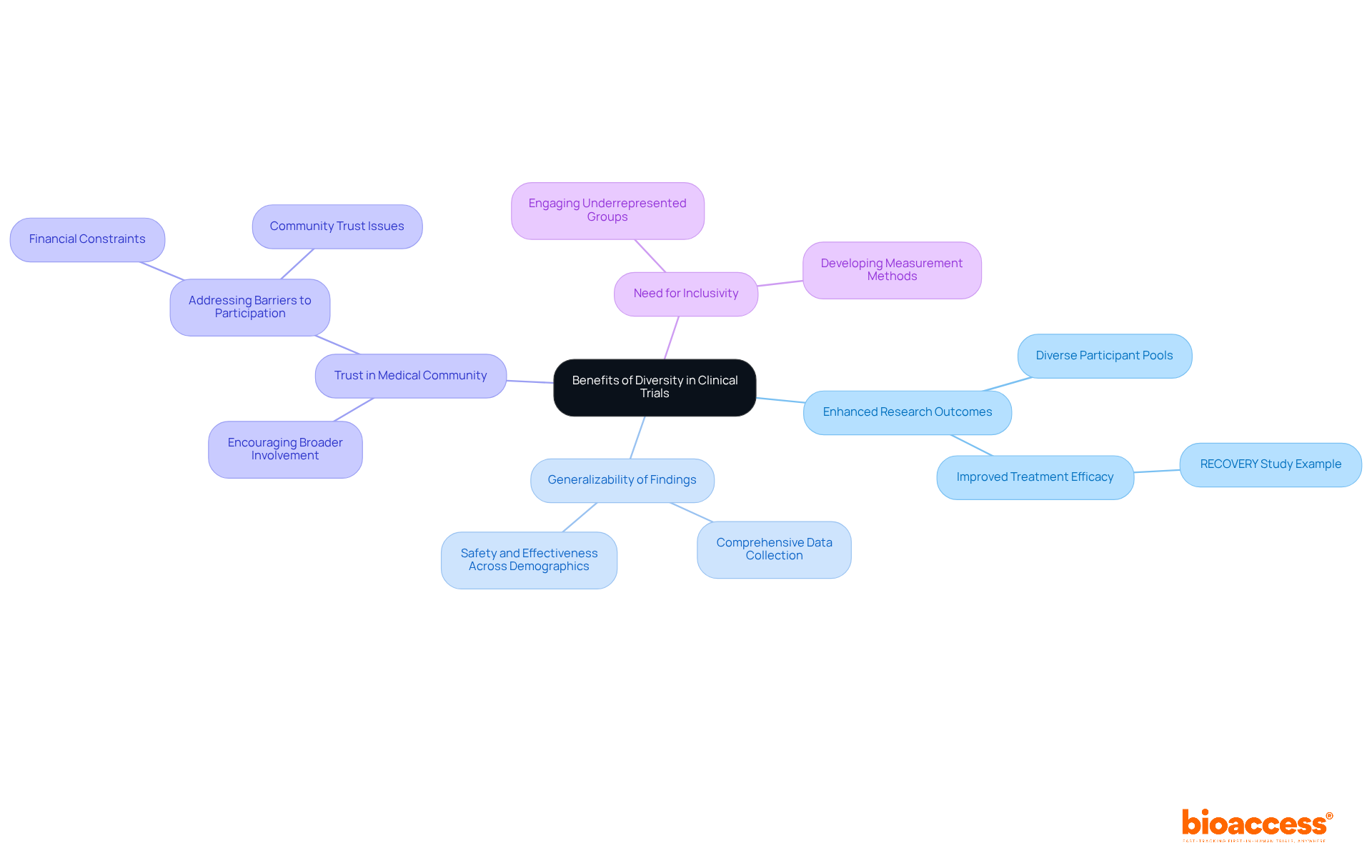
The Benefits of Diversity: Why It Matters in Clinical Trials
Variety in presents numerous benefits that significantly enhance research outcomes. By integrating , studies contribute to , generating more comprehensive data that improves the generalizability of findings and ensures that treatments are safe and effective across various demographics. For instance, research demonstrates that the inclusion of allows for a more accurate assessment of treatment efficacy, ultimately leading to . This was notably observed during the , where diverse participation was crucial in evaluating the effectiveness of dexamethasone for COVID-19 patients, ultimately saving lives.
Moreover, prioritizing diversity fosters trust within the medical community, encouraging broader involvement in research initiatives. A 2024 Ipsos study revealed that while 58% of UK adults expressed willingness to participate in research trials, only 41% of ethnic minority adults felt the same. This disparity highlights the urgent need for that not only engage underrepresented groups but also tackle barriers such as financial constraints and lack of community trust.
By recognizing the importance of diversity, stakeholders can drive , leading to innovations that cater to the needs of all populations. This dedication to inclusivity not only elevates the quality of clinical trials but also aids in the development of that genuinely reflect the of the patient population.
Conclusion
Diversity in clinical trials transcends regulatory requirements; it is a fundamental aspect that enhances the validity and applicability of medical research. By prioritizing inclusive practices, the clinical research community ensures that studies reflect the varied demographics of the population, ultimately leading to more effective and equitable healthcare solutions.
The article delineates several key strategies to enhance diversity in clinical trials:
- Leveraging global agility
- Addressing barriers to participation
- Employing technology
- Engaging with communities
Implementing tailored approaches that consider the unique needs of diverse populations enables stakeholders to significantly improve recruitment and retention rates. Notable examples, such as successful partnerships and innovative use of telemedicine, underscore the potential for achieving meaningful progress in this area.
The significance of diversity in clinical trials extends beyond individual studies; it has profound implications for public health and health equity. As the medical field evolves, embracing diversity is essential for developing treatments that are safe and effective for all groups. A concerted effort to enhance representation in research not only fosters trust within communities but also drives advancements that can lead to healthier outcomes across populations. The call to action is unequivocal: prioritize diversity to ensure that medical research serves everyone equitably, paving the way for a healthier future.
Frequently Asked Questions
What is bioaccess® and what is its main goal?
bioaccess® is an organization that leverages the regulatory speed of Latin America, diverse patient populations in the Balkans, and Australia’s efficient pathways to expedite ethical approvals and enhance patient enrollment in clinical trials. Its main goal is to accelerate medical studies while enhancing diversity in clinical trials.
Why is diversity in clinical trials important?
Diversity in clinical trials is essential for ensuring that results are relevant to the wider population. It helps uncover variations in drug effectiveness and safety among different racial, ethnic, and socioeconomic groups, enhancing the scientific validity of research and fostering trust in the medical community.
How does bioaccess® improve representation in clinical trials?
bioaccess® improves representation by judiciously selecting test sites and implementing tailored recruitment strategies that focus on marginalized groups. This approach addresses critical gaps in research and enhances the diversity of participant demographics.
Can you provide an example of bioaccess®’s successful recruitment strategy?
A notable example is a pilot study on an electronic patient-reported outcome (ePRO) intervention for breast cancer patients in Mexico, which achieved a 100% retention rate, demonstrating the effectiveness of tailored recruitment strategies.
What are some barriers to inclusion in clinical trials?
Barriers to inclusion often stem from distrust in the healthcare system, lack of awareness regarding research opportunities, and logistical challenges such as transportation and financial constraints.
How can these barriers be overcome?
To overcome these barriers, it is crucial to engage directly with communities, provide comprehensive information about the benefits of participation, and ensure that research sites are easily accessible.
What partnerships has bioaccess® formed to enhance diversity in clinical trials?
bioaccess® has partnered with Caribbean Health Group to establish Barranquilla as a premier location for medical studies in Latin America, supported by Colombia’s Minister of Health. Additionally, its collaboration with GlobalCare Clinical Trials has resulted in over a 50% reduction in recruitment time and a 95% retention rate.
What is the participation rate of eligible individuals in clinical trials globally?
Fewer than 5% of eligible individuals worldwide participate in clinical trials, with rural, Indigenous, and low-income communities often being overlooked.
List of Sources
- bioaccess®: Accelerating Diversity in Clinical Trials Through Global Agility
- bioaccessla.com (https://bioaccessla.com/blog/patient-recruitment-strategies-in-chile-for-clinical-trials-success)
- es-us.ups.com (https://es-us.ups.com/us/en/healthcare/learning-center/blog/rethinking-clinical-trials)
- Latin America’s Landscape For Medtech Clinical Trials (https://clinicalleader.com/doc/latin-america-s-landscape-for-medtech-clinical-trials-0001)
- Diverse Representation: Ensuring Accurate Clinical Trial Outcomes
- The Importance of Diversity in Clinical Trials – Journal of Oncology Navigation & Survivorship (https://jons-online.com/issues/2024/july-2024-vol-15-no-7/the-importance-of-diversity-in-clinical-trials)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC8643184)
- walgreensbootsalliance.com (https://walgreensbootsalliance.com/news-media/our-stories/women-clinical-trials-walgreens-inspire-action)
- Why Diverse Representation in Clinical Research Matters and the Current State of Representation within the Clinical Research Ecosystem – Improving Representation in Clinical Trials and Research – NCBI Bookshelf (https://ncbi.nlm.nih.gov/books/NBK584396)
- Leveraging Technology: Expanding Access to Diverse Populations in Trials
- New Data Links Decentralized Clinical Trial Approaches to Improved Diversity in Patient Populations | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/new-data-links-decentralized-clinical-trial-approaches-improved-diversity-patient-populations)
- fiercebiotech.com (https://fiercebiotech.com/cro/clinical-trials-industry-set-uncertain-interconnected-2025-according-velocity-clinical-research)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC9133187)
- nih.gov (https://nih.gov/news-events/news-releases/nih-developed-ai-algorithm-matches-potential-volunteers-clinical-trials)
- globalforum.diaglobal.org (https://globalforum.diaglobal.org/issue/march-2025/top-10-challenges-and-solutions-for-decentralized-clinical-trials-technology-diversity-and-regulations-in-2025-and-beyond)
- Community Engagement: Building Trust for Diverse Participation
- The critical role of communications (https://recoverytrial.net/case_studies/the-critical-role-of-communications)
- pubmed.ncbi.nlm.nih.gov (https://pubmed.ncbi.nlm.nih.gov/34014324)
- tandfonline.com (https://tandfonline.com/doi/full/10.1080/23288604.2018.1541497)
- Community-Based Participatory Research: Insights, Challenges, and Successes From the Perspectives of Frontline Recruiters and Investigators | Published in Journal of Participatory Research Methods (https://jprm.scholasticahq.com/article/77399-community-based-participatory-research-insights-challenges-and-successes-from-the-perspectives-of-frontline-recruiters-and-investigators)
- narrativestrategies.com (https://narrativestrategies.com/media-and-public-relations)
- Health Equity: The Impact of Diverse Data on Public Health
- Embracing Diversity: The Imperative for Inclusive Clinical Trials | Harvard Medical School Professional, Corporate, and Continuing Education (https://learn.hms.harvard.edu/insights/all-insights/embracing-diversity-imperative-inclusive-clinical-trials)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC9133187)
- kff.org (https://kff.org/racial-equity-and-health-policy/elimination-of-federal-diversity-initiatives-implications-for-racial-health-equity)
- yaledailynews.com (https://yaledailynews.com/blog/2025/02/14/analysis-loss-of-cdc-data-is-impacting-public-health)
- nia.nih.gov (https://nia.nih.gov/news/data-shows-racial-disparities-alzheimers-disease-diagnosis-between-black-and-white-research)
- Future Directions: Advancing Diversity in Clinical Trials
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC11044643)
- tandfonline.com (https://tandfonline.com/doi/full/10.1080/23288604.2018.1541497)
- The Benefits of Diversity: Why It Matters in Clinical Trials
- The critical role of communications (https://recoverytrial.net/case_studies/the-critical-role-of-communications)
- clinicaltrialsarena.com (https://clinicaltrialsarena.com/news/diversity-commitment-persists-in-clinical-trials-amidst-policy-shifts)
- abpi.org.uk (https://abpi.org.uk/media/news/2025/june/coalition-calls-for-roadmap-to-improve-inclusivity-in-clinical-research)

Leave a Reply