Introduction
Navigating the complexities of ANVISA-regulated trials is a critical endeavor for clinical research organizations aiming to ensure compliance and efficiency. With the Brazilian research market poised for significant growth, understanding the regulatory framework—including the latest updates like Resolution No. 945/2024—is essential for successful trial management. However, the challenge remains: how can organizations effectively implement best practices to not only meet compliance requirements but also enhance the overall quality and success of their studies?
This article delves into proven strategies for managing ANVISA-regulated trials, offering insights that could transform the landscape of clinical research in Brazil.
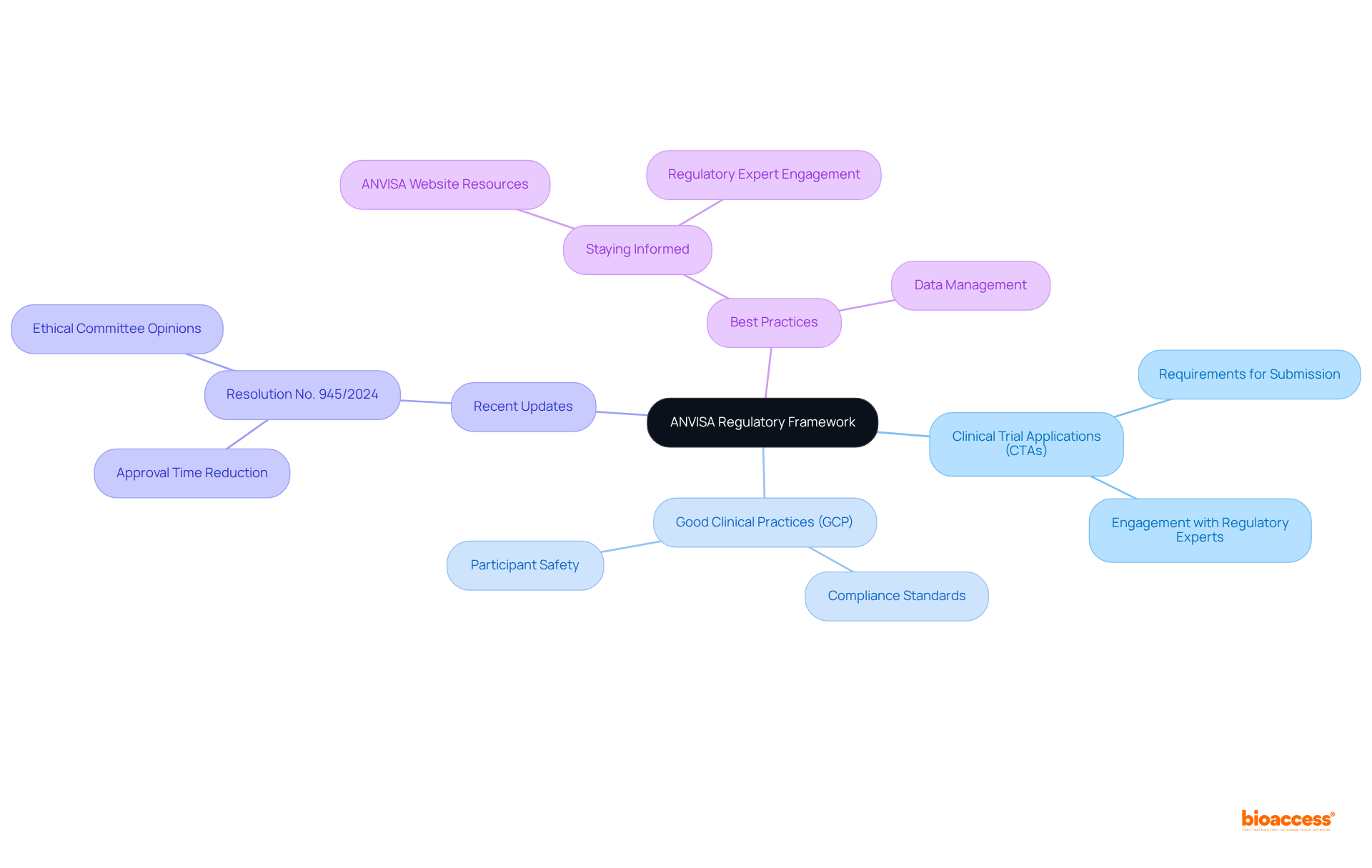
Understand ANVISA Regulatory Framework
Effectively navigating ANVISA-regulated studies requires a comprehensive understanding of the regulations and guidelines, which is part of the . Understanding the requirements for , , and specific protocols for various medical products is essential to apply the .
Notably, recent updates, particularly , have streamlined the processes for conducting research studies. This resolution mandates that ethical committees provide prompt opinions within 30 business days, a significant improvement in efficiency. This change is part of a broader initiative aimed at enhancing the transparency of the approval process, which has seen the average review time for .
Staying informed through resources such as the ANVISA website and engaging with regulatory experts are considered , as they can significantly aid in ensuring compliance and expediting the approval process.
With the Brazilian research market projected to expand from USD 316.6 million in 2024 to USD 443.5 million by 2032, understanding these is increasingly crucial for successful research applications.
Implement Compliance Protocols for Trials
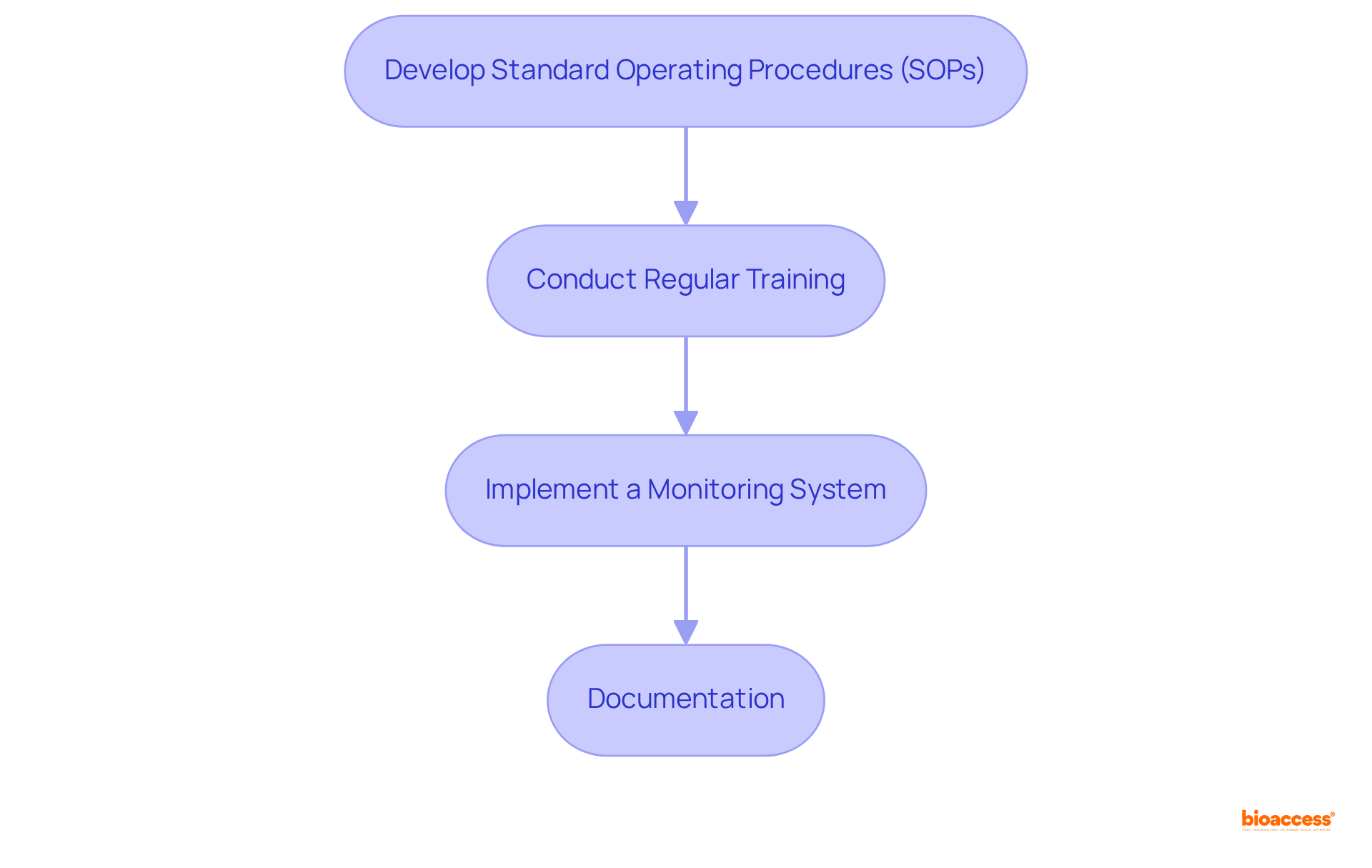
Establishing is essential for upholding the integrity and success of overseen by ANVISA. A comprehensive framework must include regular audits, , and continuous training for all team members. Key steps to consider are:
- Develop : Establish SOPs that clearly delineate processes for study conduct, data management, and . In Brazil, approximately 70% of have implemented SOPs, underscoring their vital role in ensuring compliance.
- Conduct Regular Training: Guarantee that all team members undergo thorough training on and , complemented by periodic refresher courses to keep everyone abreast of evolving guidelines.
- Implement a : Deploy a robust to track adherence to established protocols and pinpoint areas for enhancement. This may involve regular site visits and audits to ensure compliance with SOPs.
- Documentation: Keep meticulous records of all experimental activities, including consent forms, protocol amendments, and communications with regulatory bodies. Proper documentation is crucial for during inspections and safeguarding the integrity of study data.
By adhering to these best practices, clinical research organizations can enhance their operational efficiency and successfully navigate ANVISA-regulated studies.
Adopt Efficient Project Management Strategies
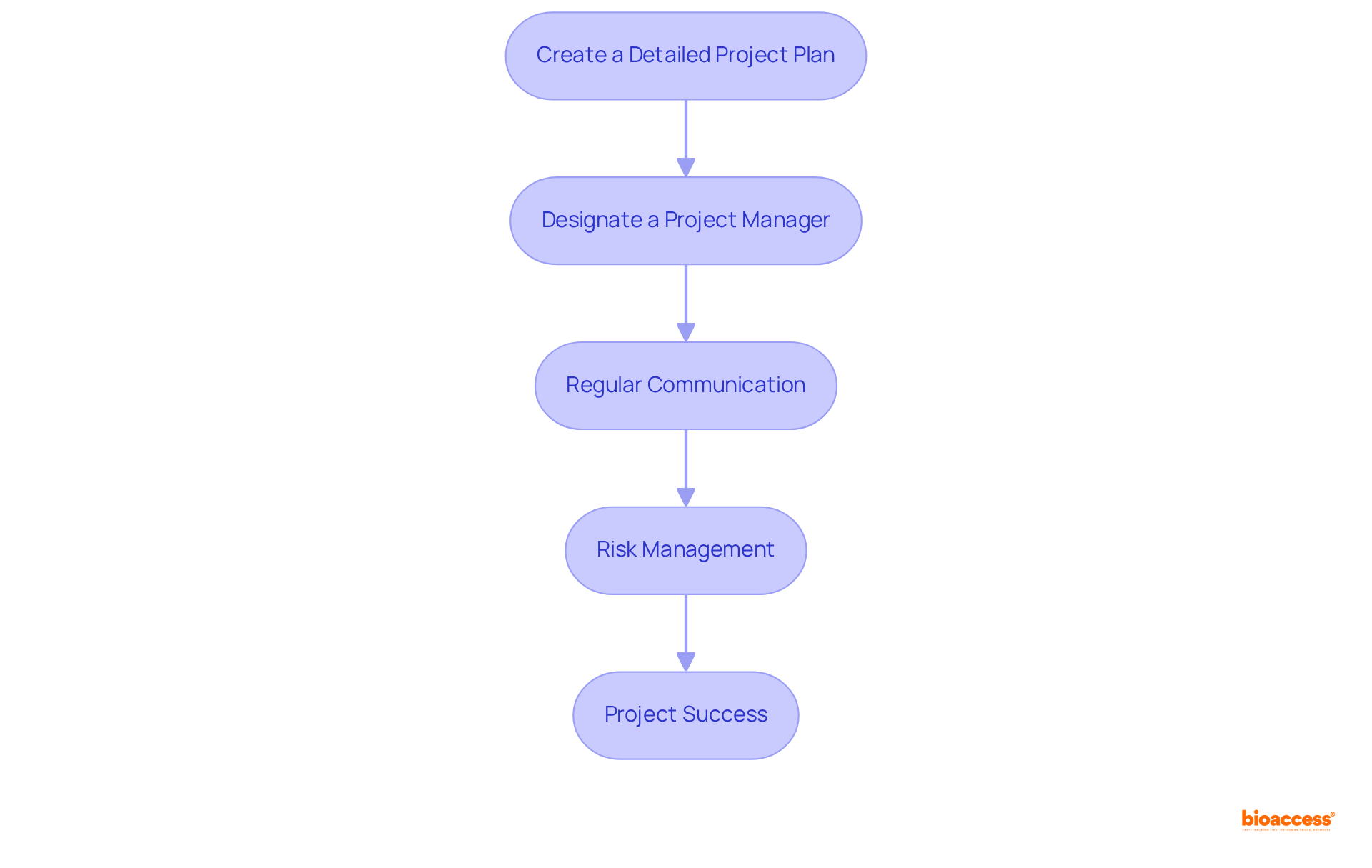
To enhance in ANVISA-regulated trials, it is imperative to implement several key strategies:
- Create a : Develop a comprehensive outline that encompasses all phases of the study, including timelines, milestones, and resource allocation. By utilizing software, organizations can significantly improve tracking and accountability; in fact, those employing such tools complete .
- Designate a Project Manager: Appoint an experienced project manager to oversee the experiment. This individual plays a crucial role in ensuring that all team members remain aligned and informed, which is essential for maintaining momentum and meeting deadlines.
- Regular Communication: Establish a routine for meetings to discuss progress, address challenges, and adjust timelines as necessary. Effective communication is vital for fostering collaboration and problem-solving, with .
- : Identify potential risks early in the project and develop mitigation strategies. This proactive approach not only prevents delays but also follows the , ultimately enhancing the likelihood of project success. Statistics reveal that projects with structured management practices are , underscoring the importance of thorough planning and .
Invest in Ongoing Training for Research Teams
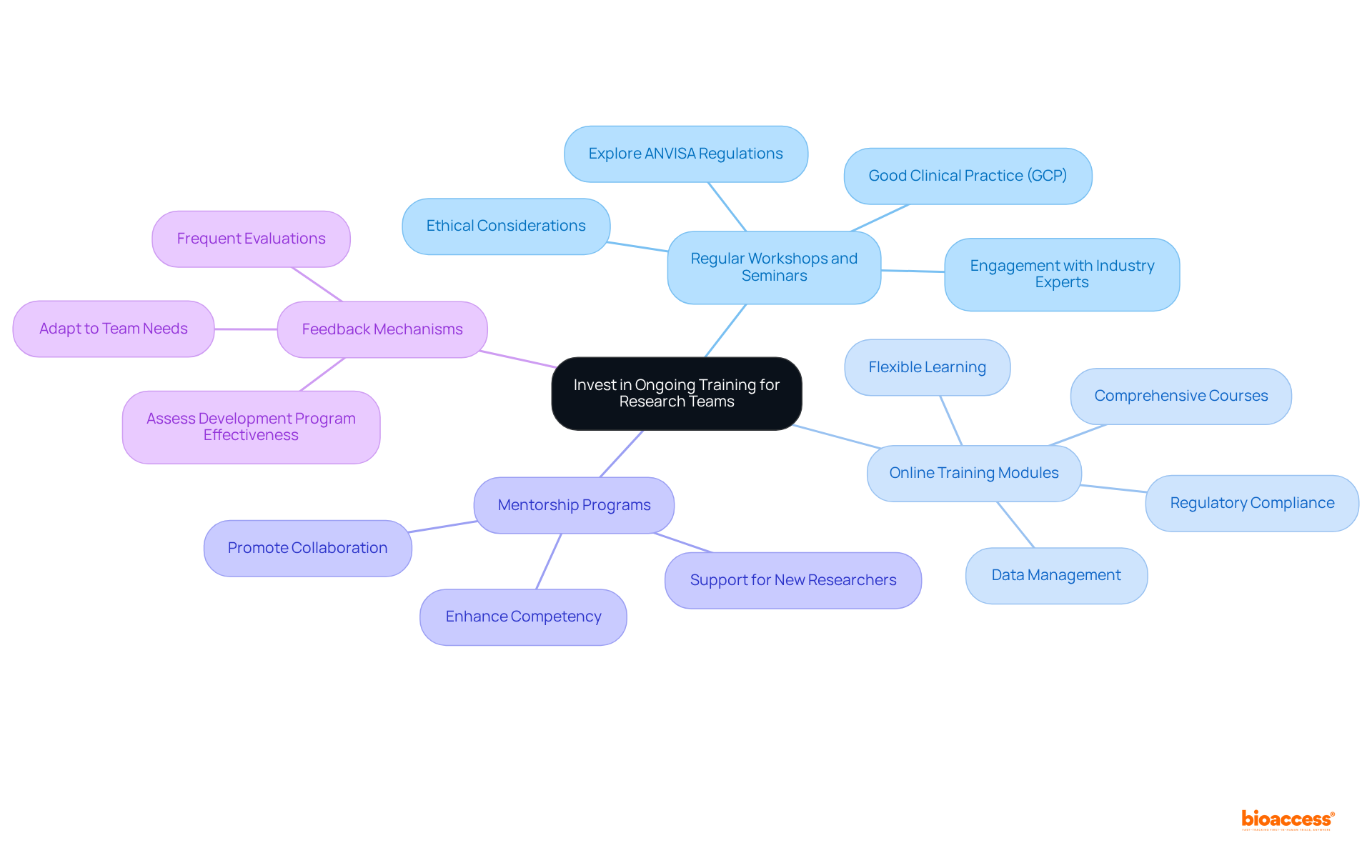
Investing in continuous education for research teams is essential for improving and ensuring compliance with . Key initiatives include:
- Regular Workshops and Seminars: Conduct workshops that explore , , and ethical considerations in research. Engaging industry experts to share insights and updates can significantly enrich the learning experience.
- Online Training Modules: Offer access to online training platforms that provide comprehensive courses on various aspects of , including and data management. This flexibility enables group members to learn at their own pace while remaining informed on essential subjects.
- Mentorship Programs: Establish . This promotes a culture of learning and collaboration, enhancing the overall competency of the research group.
- Feedback Mechanisms: Implement robust to assess the effectiveness of development programs. Frequent evaluations facilitate essential modifications according to team needs and changing regulatory standards, ensuring that development remains pertinent and effective.
These initiatives not only enhance adherence to but also represent the best practices for managing ANVISA-regulated trials, ultimately . Significantly, nurses and data managers account for more than 30% of the overall time and effort needed to carry out a medical study, highlighting the significance of customized education for these positions. With more than 20 years of experience in research services, bioaccess® highlights that for upholding high standards in studies. By merging educational programs with our extensive offerings, such as feasibility studies, site selection, and project management, we guarantee that our groups are well-prepared to handle the intricacies of research in healthcare. This underscores the critical need for comprehensive training initiatives that cater to all team members involved in clinical trials.
Conclusion
Effectively managing ANVISA-regulated trials requires a profound understanding of the regulatory landscape, strict adherence to compliance protocols, and the execution of robust project management strategies. By comprehending the complexities of ANVISA regulations and recent updates, including streamlined approval processes, organizations can significantly boost their operational efficiency and secure successful research outcomes.
Key practices include:
- The development of standard operating procedures
- Regular training sessions
- Meticulous documentation
The significance of a structured project management approach is paramount, as it not only ensures the timely completion of trials but also mitigates risks associated with research activities. Furthermore, investing in ongoing training for research teams solidifies compliance and fosters a culture of continuous improvement, which is essential for navigating the complexities of clinical research.
Ultimately, a commitment to understanding ANVISA’s regulatory framework, implementing rigorous compliance measures, and cultivating a well-trained research team is vital for the success of clinical trials in Brazil. As the research market continues to expand, embracing these best practices will not only enhance the quality of trials but also contribute to the advancement of innovative medical solutions, significantly impacting healthcare outcomes.
Frequently Asked Questions
What is ANVISA and why is it important in clinical trials?
ANVISA (Agência Nacional de Vigilância Sanitária) is the Brazilian Health Regulatory Agency responsible for overseeing the regulation of medical products and clinical trials. Understanding ANVISA’s regulatory framework is essential for effectively managing ANVISA-regulated studies.
What are the key components of managing ANVISA-regulated trials?
Key components include understanding the requirements for Clinical Trial Applications (CTAs), adhering to Good Clinical Practices (GCP), and following specific protocols for various medical products.
What recent changes have been made to streamline the research approval process in Brazil?
Recent updates, particularly Resolution No. 945/2024, have mandated that ethical committees provide opinions within 30 business days, improving the efficiency of the approval process and reducing the average review time for ethical approvals from 45 days to approximately 30 days.
How can researchers stay informed about ANVISA regulations?
Researchers can stay informed by utilizing resources such as the ANVISA website and engaging with regulatory experts, which are considered best practices for ensuring compliance and expediting the approval process.
What is the projected growth of the Brazilian research market?
The Brazilian research market is projected to expand from USD 316.6 million in 2024 to USD 443.5 million by 2032, highlighting the importance of understanding regulatory frameworks for successful research applications.
List of Sources
- Understand ANVISA Regulatory Framework
- 10 Essential Clinical Trial Requirements Under Anvisa | bioaccess® (https://bioaccessla.com/blog/10-essential-clinical-trial-requirements-under-anvisa)
- statista.com (https://statista.com/statistics/1067438/brazil-clinical-trials-share-worldwide)
- bdlaw.com (https://bdlaw.com/news/chemical-watch-quotes-miriam-mazza-on-brazils-chemical-framework-management-bill)
- Brazil Clinical Trials Market Size, Share | Analysis Report [2032] (https://fortunebusinessinsights.com/brazil-clinical-trials-market-112310)
- linkedin.com (https://linkedin.com/posts/countryregulatoryaffairs_regulatoryaffairs-freyrbrazil-anvisa-activity-7256572319433699329-uDgz)
- Implement Compliance Protocols for Trials
- bmcmedresmethodol.biomedcentral.com (https://bmcmedresmethodol.biomedcentral.com/articles/10.1186/s12874-020-01109-5)
- gcpblog.org (https://gcpblog.org/establishing-gcp-compliance-in-clinical-research-organizations)
- Clinical Research Compliance: 5 Critical Factors for 2024 Success (https://infonetica.net/articles/clinical-research-compliance)
- relias.com (https://relias.com/blog/10-inspiring-patient-experience-quotes)
- qualtrics.com (https://qualtrics.com/blog/research-quotes)
- Adopt Efficient Project Management Strategies
- qualtrics.com (https://qualtrics.com/blog/research-quotes)
- 110+ project management statistics and trends for 2026 (https://monday.com/blog/project-management/project-management-statistics)
- 31 Must-Know Project Management Statistics in 2026 (https://proofhub.com/articles/project-management-statistics)
- Project Management Statistics By Team Size, Remote Work, Software And Features (2026) (https://electroiq.com/stats/project-management-statistics)
- 153 Project Management Quotes To Inspire You In 2026 (https://thedigitalprojectmanager.com/project-management/project-management-quotes-inspiration)
- Invest in Ongoing Training for Research Teams
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC3092661)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC4430465)
- 18 of Our Favorite Quotes About the Power of Training & Development – Abilitie (https://abilitie.com/blog/2018-7-6-18-of-our-favorite-quotes-about-the-power-of-training-development)
- 65 Quotes About Workplace Learning & Development (https://growthengineering.co.uk/65-quotes-about-workplace-learning)

Leave a Reply