Introduction
Clinical trials play a vital role in the development of medical treatments, requiring meticulous monitoring to ensure their integrity and success. In this article, we will explore the importance of monitoring in clinical trials, the different types of monitoring methods, and the benefits of remote monitoring. We will also discuss the challenges involved in clinical trial monitoring and best practices for effective monitoring.
Additionally, we will delve into the technological advancements in monitoring, such as AI and automation, and the regulatory guidelines that govern clinical trial monitoring. Through case studies, we will highlight successful monitoring approaches and emphasize the critical nature of patient-centric monitoring and rigorous data management. Join us as we delve into the world of clinical trial monitoring and its impact on patient safety and medical advancements.
Importance of Monitoring in Clinical Trials
are indispensable in the development of , requiring rigorous monitoring to safeguard their integrity and outcomes. These trials are structured into , each with a specific focus—Phase 1 ensures safety, Phase 2 evaluates both safety and efficacy, and Phase 3 confirms these attributes on a larger scale, ultimately leading to a potential .
Monitoring activities encompass a thorough examination of collected data and to promptly identify and rectify any problems, thereby guaranteeing that data is both precise and consistent with the trial’s goals. An illustrative case involves a patient from rural Pennsylvania with a rare disease, who faces the daunting challenge of participating in a clinical trial abroad. The complexities of international travel, language barriers, and document management underscore the that can arise during , further emphasizing the need for meticulous oversight to maintain and trial validity.
Types of Monitoring: On-Site vs. Remote
are pivotal in the journey of bringing a medical device to market, ensuring the safety and effectiveness of new treatments. To facilitate this, two predominant are employed: on-site and .
is the traditional approach, where monitors physically visit the study site to scrutinize study documents, confirm data accuracy, and verify adherence to protocols. , in contrast, harnesses technology to oversee study data from afar, providing a nimble and efficient alternative.
This modern approach not only enhances and lowers costs but also surmounts geographical hurdles. For example, consider a patient from rural Pennsylvania with a rare disease, who is given the chance to join a clinical trial in Turkey. could play a crucial role in such cases, allowing the trial to extend its reach without imposing the burden of travel logistics on participants. Ultimately, the integration of into is revolutionizing the process, making trials more accessible and manageable for patients globally.
Benefits of Remote Monitoring in Clinical Trials
The inclusion of in presents a transformative approach to patient participation and . Consider the case of a patient in rural Pennsylvania with an ultra-rare disease, who is offered a chance to join a clinical trial in Turkey.
The logistical challenges of cross-border travel, from securing visas to handling unfamiliar paperwork, can be daunting. circumvents these barriers, providing the patient with the means to engage in the trial without the stress of travel.
It facilitates , allowing for swift identification and resolution of issues, and reduces the need for travel, conserving time and resources. Furthermore, it upholds data quality and integrity through a centralized review platform. Significantly, it prioritizes participant safety by enabling detailed monitoring of adverse events for timely intervention. In essence, refines the monitoring process, heightening efficiency and expanding the reach of to patients regardless of their geographic location.
Challenges in Clinical Trial Monitoring
Monitoring is a complex endeavor, particularly when it comes to during remote monitoring. To safeguard sensitive participant information, it’s crucial to .
De-identification plays a key role in protecting individual privacy by stripping away personal identifiers from patient data, rendering it anonymous. This enables researchers to utilize the data for in-depth analysis, identifying trends and patterns that could lead to advances in and more effective disease prevention strategies.
Additionally, establishing clear communication channels between monitors and study sites is essential to swiftly address any issues that may arise. The intricate and abundant data collected in necessitates the use of sophisticated . These tools are not just essential for handling the data but are also vital for extracting meaningful insights from it, thereby ensuring the safety of trial participants and the integrity of the study findings. The importance of meticulous data management is underscored by the Who’s estimate that with two million different medical devices on the market, virtually everyone will engage with such devices, emphasizing the impact that have on and medical device efficacy.
 Flowchart: Data Management and Analysis in Clinical Trials](https://vectorly-files.s3.eu-central-1.amazonaws.com/22176642-2663-4edb-8623-4e19845b85a1.jpg)
Best Practices for Effective Monitoring
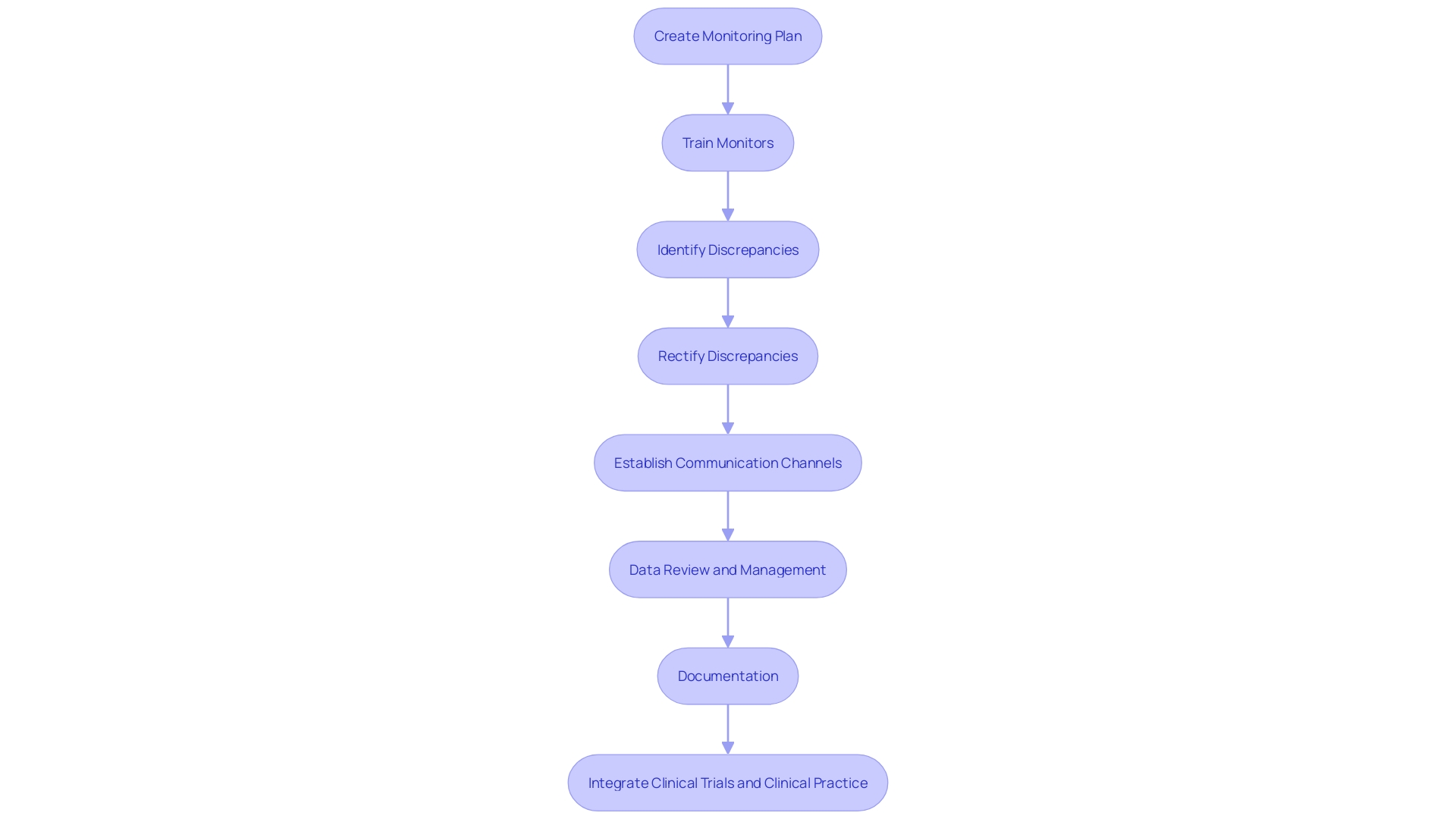
In the realm of , robust monitoring practices are pivotal to safeguarding and participant safety. The inception of any study necessitates a meticulously crafted , outlining the extent and regularity of surveillance measures. Monitors, the custodians of compliance, must be equipped with comprehensive training to adeptly identify and rectify any discrepancies in data or deviations from the study protocol.
A noteworthy illustration of the complexities involved in is the case of a patient from rural Pennsylvania facing an ultra-rare disease, who must navigate international travel to join a trial in Turkey. Here, the underscore the necessity for clear and proactive communication channels among monitors, investigators, and study sites to promptly address such issues. Furthermore, the importance of rigorous data review and management cannot be overstated, as highlighted by the ‘s estimate of two million medical devices in circulation globally.
The European Union Medical Device Regulation (EU MDR) Article 62 emphasizes the critical nature of this process. To encapsulate the monitoring process, is indispensable, serving as a testament to the trial’s adherence to stringent standards, thereby ensuring transparency and accountability. This approach not only upholds the integrity of the trial but also bridges the gap between and practical application, as discussed at the JAMA Summit, where the need for integration between and clinical practice was a focal point of discourse.
Technological Advancements in Monitoring: AI and Automation
The landscape of is undergoing a transformative shift with the integration of Artificial Intelligence (AI) and automation tools. Experts like Devine and Mark Leimbeck, who bring decades of experience in , , and program management, underscore the impact of AI in enhancing the monitoring process. By leveraging AI algorithms, vast datasets can be analyzed more effectively, uncovering patterns and anomalies that might elude human monitors.
This technology also aids in , such as , thereby allocating more time for monitors to concentrate on critical activities. The utilization of digital workflows and data orchestration contributes to the personalization of patient treatments, and the acceleration of . Moreover, Ai’s role in simulating clinical and operational outcomes from the onset of drug development illustrates its capacity to identify potential drug candidates and optimize study designs.
This preemptive modeling can pinpoint potential challenges, diminish risks of failure, and enhance study efficiency, ultimately benefiting both patients and pharmaceutical companies. The advent of a self-driving clinical trial, powered by the increasing volume of health data, looms on the horizon, representing a significant leap forward in pharmacology and patient care. With Ai’s aid, the daunting task of is also being revolutionized, expediting enrollment rates and driving the industry towards unprecedented accuracy and efficiency.
Regulatory Guidelines for Clinical Trial Monitoring
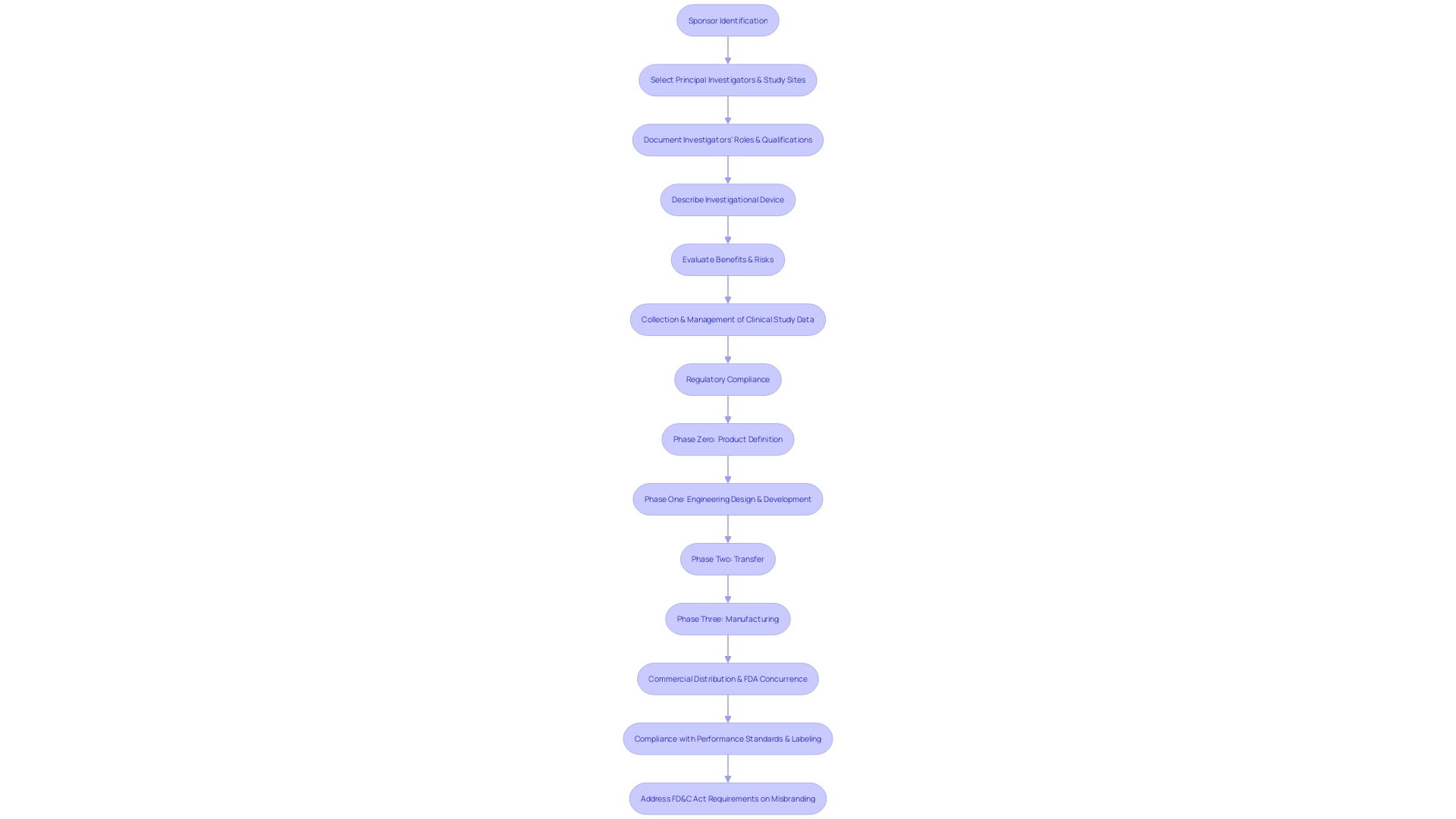
In the realm of medical device , the importance of precise and comprehensive monitoring is underscored by guidelines such as those detailed in MDCG 2024-3. For instance, Section 3.1 mandates the inclusion of fundamental information like sponsor identity, principal investigators, and study sites, along with a delineation of the investigators’ roles and qualifications.
This ensures accountability and clarity in the conduct of the trial. Section 3.2 delves into the specifics of the , requiring a detailed description that encompasses its purpose, intended patient populations, and indications for use.
The thorough understanding of the device is critical for assessing its applicability and safety in clinical settings. Moreover, Section 3.3 demands a comprehensive evaluation of the benefits and risks associated with the device, necessitating a calculated with a well-founded rationale.
Such stringent guidelines are in place to safeguard patient safety and validate the integrity of the clinical investigation outcomes. The gravity of rigorous is highlighted by the World Health Organization’s estimate that over two million types of are in circulation globally.
This makes the meticulous collection and management of clinical study data a vital component of participant safety and the accuracy of study results. As Article 62 of the EU MDR articulates, the stakes are high when human subjects are involved, and the potential for to affect millions underscores the necessity of . The FDA’s evolving regulatory landscape for pharmaceuticals further illustrates the complexity of maintaining compliance. Amendments to regulations not only influence the cost of drug development but also carry the risk of significant delays and reputational damage. A case in point is the recent progress in Alzheimer’s treatment, emphasizing the impact of regulatory adherence on the advancement of medical therapies. Adhering to such comprehensive guidelines is not only a regulatory requirement but also a moral imperative to ensure the safety and effectiveness of medical interventions.
Case Studies: Successful Monitoring in Clinical Trials
The complexities of extend beyond the confines of data collection and adherence to protocols. Consider the poignant scenario of a patient from rural Pennsylvania diagnosed with an ultra-rare disease, for which there is no FDA-approved treatment.
Presented with the chance to join a clinical trial in Turkey that might save their life, the patient faces not only the disease but also the daunting task of managing international travel logistics. This patient’s story underscores the critical nature of , addressing the .
Meanwhile, the significance of meticulous cannot be overstated. With the World Health Organization reporting around two million different on the global market, the impact of is ubiquitous. Ensuring participant safety and the integrity of study conclusions demands rigorous data management, as emphasized by Article 62 of the European Union Medical Device Regulation. This regulation underlines the non-negotiable importance of in clinical research, which in turn informs the safety and efficacy of used worldwide.
Conclusion
In conclusion, clinical trial monitoring is vital for the development of medical treatments. It ensures data integrity and participant safety throughout the trial phases.
On-site and remote monitoring methods offer distinct benefits, with remote monitoring overcoming geographical barriers and prioritizing patient convenience. Challenges such as data privacy and communication must be addressed with robust systems and protocols.
Advanced data management tools are necessary to handle the abundance of trial data. Best practices include comprehensive monitoring plans, thorough training for monitors, proactive communication channels, and rigorous data review.
Compliance with regulatory guidelines is crucial to uphold participant safety and validate study outcomes. Technological advancements like AI and automation are transforming the monitoring process.
AI algorithms enhance data analysis capabilities and automate tasks, contributing to personalized patient treatments and accelerating trial efficiency. Successful case studies highlight patient-centric approaches that address real-world challenges. Meticulous data management is essential for patient safety and device efficacy due to the widespread use of medical devices. In summary, clinical trial monitoring plays a critical role in medical advancements. By embracing technology, implementing best practices, and complying with regulations, we can continue to improve patient outcomes through effective monitoring processes.
Frequently Asked Questions
Why is monitoring important in clinical trials?
Monitoring is crucial in clinical trials to ensure the integrity and outcomes of medical treatments. It involves thorough examination of collected data and trial procedures to identify and rectify issues, guaranteeing that the data aligns with the trial’s goals.
What are the distinct phases of clinical trials?
Clinical trials are structured into three phases: Phase 1 ensures safety, Phase 2 evaluates both safety and efficacy, and Phase 3 confirms safety and efficacy on a larger scale, leading to potential FDA approval.
What are the two predominant methods of monitoring in clinical trials?
The two main methods of monitoring are On-Site Monitoring, where monitors physically visit study sites, and Remote Monitoring, which utilizes technology to oversee study data from a distance.
What are the benefits of remote monitoring?
Remote monitoring eliminates travel logistics for participants, facilitates real-time data access for timely issue resolution, maintains data quality and integrity, and prioritizes participant safety by closely monitoring adverse events.
What challenges are associated with clinical trial monitoring?
Key challenges include data privacy and security, the need for robust systems to protect sensitive information, establishing clear communication channels, and handling complex data management and analysis.
What best practices should be followed for effective monitoring?
Effective monitoring practices include developing a comprehensive monitoring plan, providing training for monitors, ensuring proactive communication among stakeholders, and maintaining rigorous data review and management.
How are technological advancements impacting clinical trial monitoring?
Technological advancements, particularly AI and automation, enhance monitoring by analyzing vast datasets, automating mundane tasks, optimizing study designs, and expediting patient recruitment.
What regulatory guidelines govern clinical trial monitoring?
Regulatory guidelines like MDCG 2024-3 emphasize comprehensive documentation of study details, detailed descriptions of investigational devices, and rigorous evaluation of benefits and risks to ensure patient safety.
Can you provide an example highlighting the importance of monitoring?
A notable example involves a patient from rural Pennsylvania with an ultra-rare disease facing logistical challenges in participating in a clinical trial in Turkey, illustrating the necessity of patient-centric monitoring approaches.
What impact do clinical trials have on medical devices and patient safety?
Clinical trials significantly impact the safety and efficacy of medical devices, making rigorous monitoring and data management vital to ensure these devices are safe and effective for public use.
List of Sources
- Importance of Monitoring in Clinical Trials
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- nytimes.com (https://www.nytimes.com/2024/06/28/health/wang-cassava-alzheimers-fraud.html)
- healthywomen.org (https://www.healthywomen.org/condition/clinical-trials-for-lupus)
- Types of Monitoring: On-Site vs. Remote
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- greenlight.guru (https://www.greenlight.guru/blog/clinical-data-reporting-launch)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- greenlight.guru (https://www.greenlight.guru/blog/guide-clinical-data-management-medtech)
- Benefits of Remote Monitoring in Clinical Trials
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- Challenges in Clinical Trial Monitoring
- greenlight.guru (https://www.greenlight.guru/blog/guide-clinical-data-management-medtech)
- imerit.net (https://imerit.net/blog/challenges-and-benefits-of-data-de-identification-in-healthcare-analytics/)
- infomeddnews.com (https://infomeddnews.com/hipaa-compliance-checklist-7-key-steps-to-ensure-data-security-introduction/)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- Best Practices for Effective Monitoring
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- raps.org (https://www.raps.org/News-and-Articles/News-Articles/2024/6/Experts,-FDA-officials-discuss-future-of-clinical?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- greenlight.guru (https://www.greenlight.guru/blog/guide-clinical-data-management-medtech)
- fda.gov (https://www.fda.gov/medical-devices/human-factors-and-medical-devices/human-factors-postmarket-information-device-surveillance-and-reporting-processes)
- Technological Advancements in Monitoring: AI and Automation
- medtechintelligence.com (https://medtechintelligence.com/column/the-power-of-ai-to-enhance-clinical-trials/)
- hitconsultant.net (https://hitconsultant.net/2023/09/01/self-driving-clinical-trial-ai-ml-optimization/)
- forbes.com (https://www.forbes.com/sites/forbesbusinessdevelopmentcouncil/2023/09/14/the-next-generation-of-clinical-trials-ai-and-new-ways-to-use-data/)
- vimeo.com (https://vimeo.com/891707034/c2b8ebb0fd?share=copy|)
- Regulatory Guidelines for Clinical Trial Monitoring
- greenlight.guru (https://www.greenlight.guru/blog/mdcg-2024-3-mdr-clinical-investigation-plan)
- greenlight.guru (https://www.greenlight.guru/blog/guide-clinical-data-management-medtech)
- dev.to (https://dev.to/nidhi_acharya_427558b1130/understanding-the-regulatory-framework-of-fda-for-drug-approval-3ebf)
- starfishmedical.com (https://starfishmedical.com/blog/medical-device-phase-one-product-development-pro-tips/)
- fda.gov (https://www.fda.gov/medical-devices/regulatory-controls/general-controls-medical-devices)
- Case Studies: Successful Monitoring in Clinical Trials
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- greenlight.guru (https://www.greenlight.guru/blog/guide-clinical-data-management-medtech)

Leave a Reply