Introduction
The landscape of clinical research is undergoing rapid evolution, with regulatory standards such as ISO 10993-18 playing a pivotal role in ensuring the safety and efficacy of medical devices. This standard underscores the importance of chemical characterization of materials, a process that not only protects patient health but also facilitates compliance for manufacturers. As the demand for innovative medical solutions escalates, organizations must consider: how can they effectively navigate the complexities of ISO 10993-18 to achieve success in clinical research? This article delves into ten key insights that illuminate the path to compliance, highlighting best practices and strategies for overcoming the inherent challenges within this critical regulatory framework.
bioaccess: Accelerating Clinical Research for ISO 10993-18 Compliance
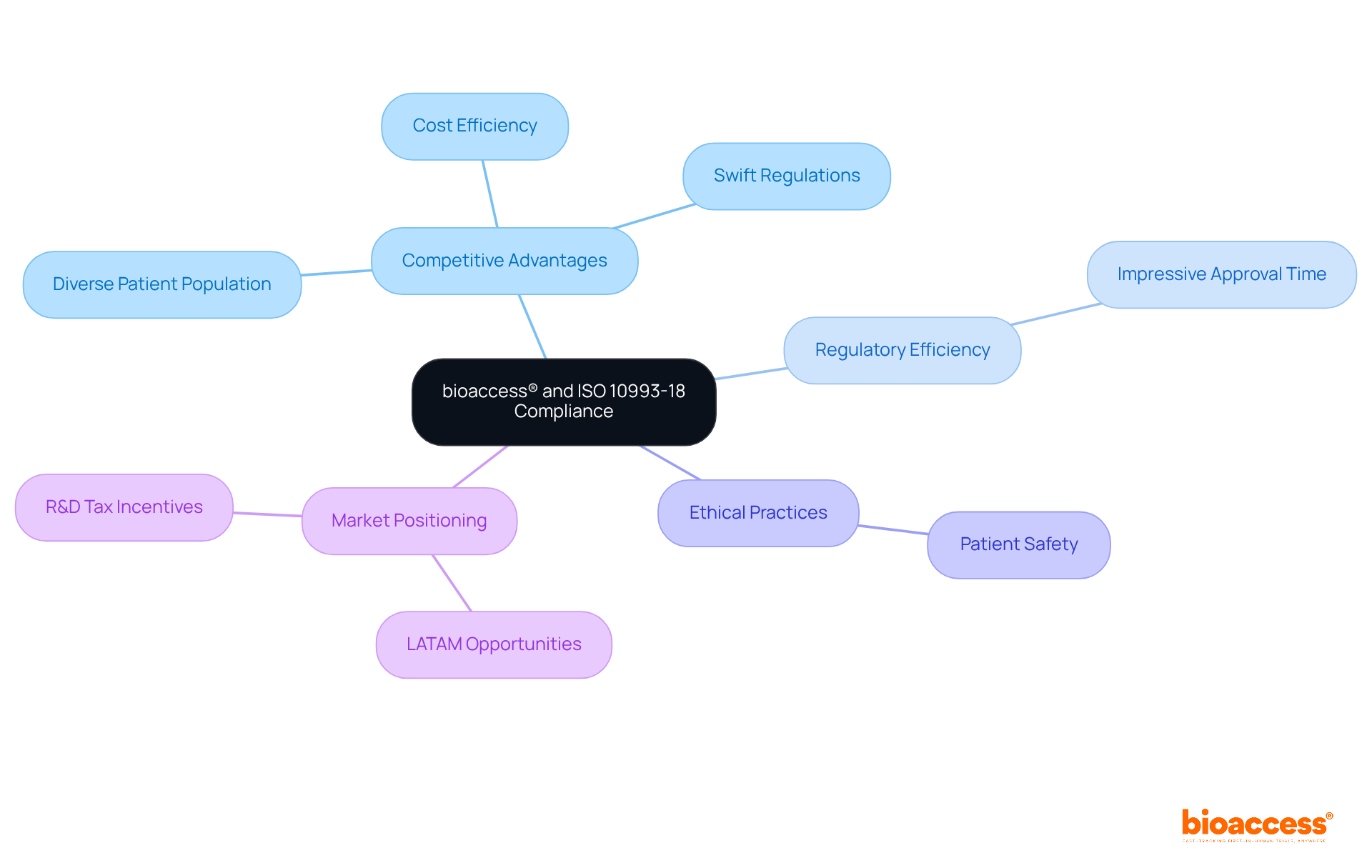
bioaccess® leverages its extensive expertise in early-phase to ensure , thereby capturing the attention of innovators in Medtech, Biopharma, and Radiopharma. By harnessing Colombia’s competitive advantages—swift regulations, compared to North America, and a diverse patient population—bioaccess® secures within an impressive 90-120 days. This rapid turnaround is essential for innovators striving to meet the rigorous standards set by , which focuses on the chemical characterization of materials used in medical devices.
Dedicated to ethical practices and patient safety, bioaccess® empowers clients to navigate the complexities of compliance effectively, thereby supporting successful projects that align with . In the context of LATAM, where are increasingly relevant, bioaccess® emerges as a leader in accelerating while ensuring adherence to evolving standards. This is further bolstered by Colombia’s cost-effective, and R&D tax incentives, which enhance the appeal of conducting trials in the region.
The collaboration with bioaccess® not only streamlines the compliance process but also positions clients to capitalize on the unique opportunities within the . As the landscape of continues to evolve, engaging with a partner that prioritizes both speed and ethical standards is crucial for success.
Chemical Characterization: A Cornerstone of ISO 10993-18
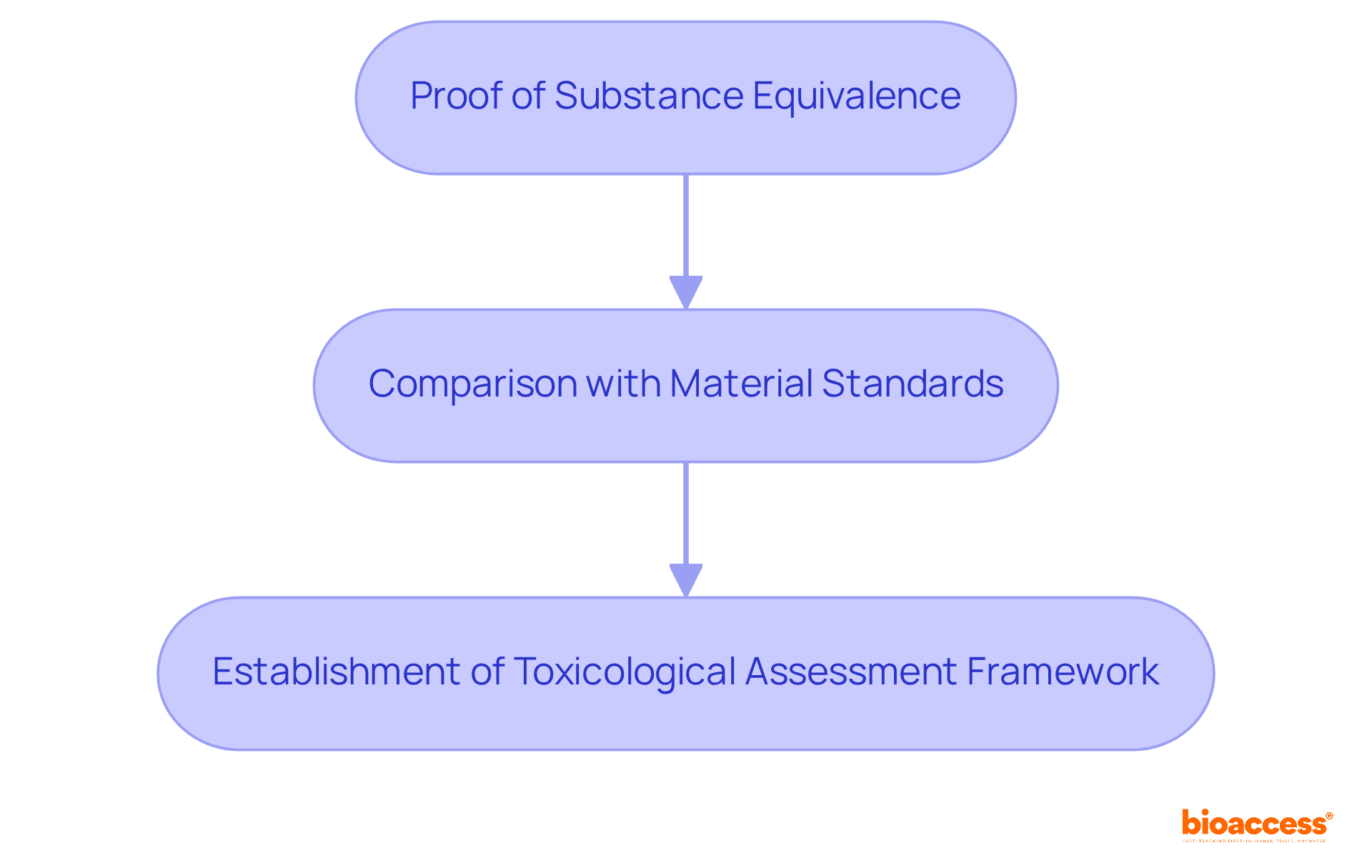
is a cornerstone of , focusing on the identification and quantification of material constituents in medical apparatus. This process is vital for assessing potential associated with the materials used in production. In 2025, the significance of thorough is paramount, as it supports manufacturers in ensuring their products do not release harmful materials that could jeopardize patient health. Notably, research indicates that approximately 30% of medical instruments require further testing due to issues identified during , underscoring the critical nature of this phase in the development process.
Successful examples of illustrate its effectiveness in enhancing and improving the safety profile of medical devices. Key stages in this process include:
- Comparison with
- Establishment of a as outlined in
Experts emphasize that a well-organized strategy can mitigate hazards and streamline the path to market approval, ultimately benefiting both producers and patients. As Eric M. Sussman observes, ” can generate information for and is an alternative approach for addressing some biocompatibility endpoints that can reduce the time and cost of testing and the need for animal testing.” As the landscape of medical apparatus development evolves, integrating robust practices remains essential for safeguarding public health.
Extractables and Leachables Testing: Ensuring Biocompatibility Under ISO 10993-18
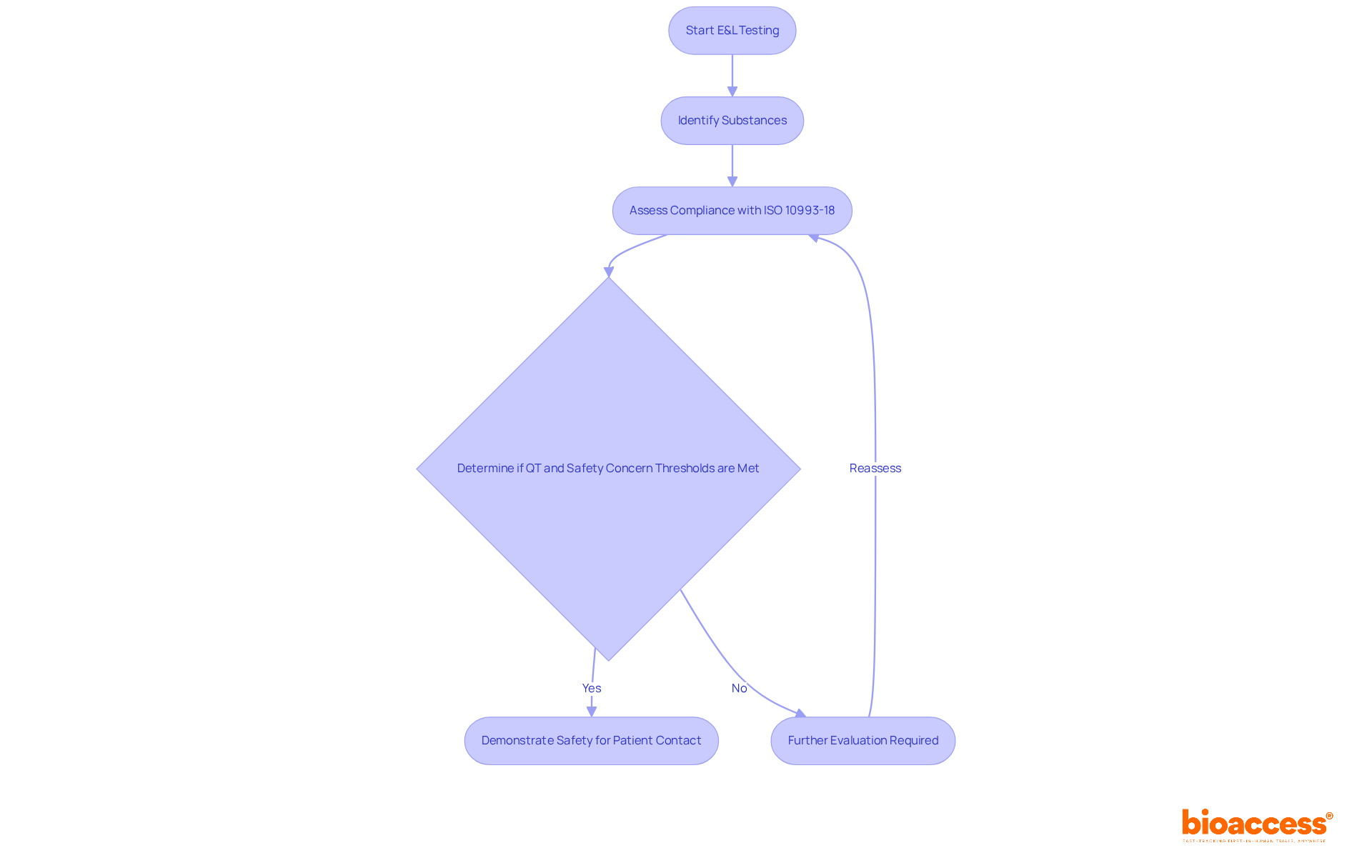
Extractables and leachables (E&L) testing is a critical component of , which is essential for assessing the safety of medical instruments. This testing identifies from devices into the body during use, potentially posing health risks. By conducting , manufacturers can demonstrate compliance with , ensuring their products are safe for patient contact. This process not only satisfies regulatory requirements but also builds trust with healthcare providers and patients alike.
Recent updates indicate that by 2025, the percentage of passing following has notably improved, reflecting advancements in testing methodologies and regulatory compliance. The integration of into the development process has become increasingly vital, as manufacturers recognize its role in mitigating risks associated with chemical migration. The qualification threshold (QT) for is established at 5 µg/day, while the is set at 0.15 µg/day, emphasizing the stringent standards that must be adhered to.
As Chris Allen, CEO of Broughton, underscores, ” are an essential part of product development to ensure safety and manage risk.” This statement reinforces the fundamental significance of within the medical equipment sector, particularly as regulatory bodies continue to enforce rigorous guidelines to safeguard patient health. Furthermore, the is anticipated to reach a valuation of $1 billion by 2028, underscoring the increasing importance of in the industry.
Understanding the Analytical Evaluation Threshold (AET) in ISO 10993-18
Understanding the
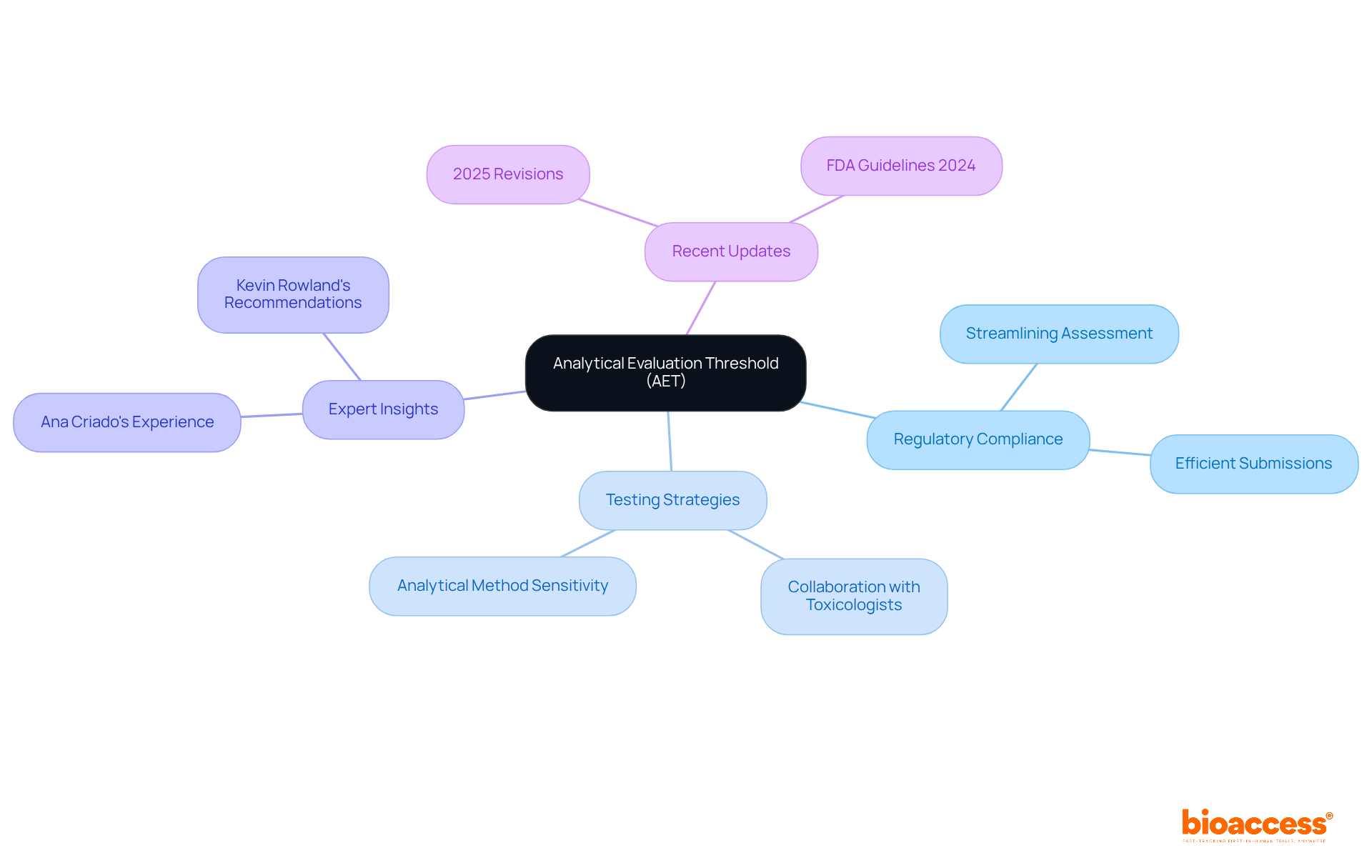
The Analytical Evaluation Threshold (AET) is a pivotal element in ISO 10993-18, establishing the minimum concentration of a substance that must be reliably quantified during . Substances detected below this threshold typically do not necessitate further , thereby streamlining the assessment process. By grasping the AET, manufacturers can strategically allocate resources to identify and assess potentially harmful substances, ensuring compliance while optimizing testing efforts. This focused strategy not only improves the effectiveness of the oversight process but also enables prompt market entry for .
Ana Criado, our Director of Regulatory Affairs and an expert in biomedical engineering and health economics, emphasizes the significance of AET in guiding manufacturers toward that align with . For instance, in her experience consulting for global companies, she has observed that a clear understanding of AET can lead to more efficient submissions and reduce the risk of costly delays. Recent updates in 2025 further emphasize the significance of the AET in , promoting a cooperative method between toxicologists and chemists to guarantee precise application of the AET, ultimately aiding in .
Safety Data Sheets (SDS): Key to Regulatory Compliance in ISO 10993-18
are essential documents that provide comprehensive information regarding the properties, hazards, and safe handling of substances utilized in . In alignment with , manufacturers must reference SDS to ensure thorough . These documents not only assist in meeting but also play a pivotal role in managing uncertainties and safety evaluations throughout the product lifecycle. By keeping SDS current, manufacturers can showcase their .
The structured format of SDS, divided into 16 sections, ensures that , facilitating effective . Expert insights indicate that can considerably diminish potential risks, ensuring that organizations prioritize safety in their operations.
Linking ISO 10993-18 and ISO 14971: A Risk Management Perspective
and ISO 14971 are pivotal standards that collectively create a robust framework for addressing hazards associated with . While provides comprehensive management guidelines throughout the device lifecycle, . By integrating standards, manufacturers can systematically identify, assess, and mitigate risks related to chemical constituents, ensuring a thorough evaluation of potential hazards.
This comprehensive approach not only enhances product safety but also ensures , ultimately leading to improved . Industry leaders emphasize that ; as one expert stated, ‘ firms MUST have established threat management processes that adhere to ISO 14971.’ Moreover, recent statistics reveal that over 70% of manufacturers are now embracing both ISO standards, reflecting a growing acknowledgment of their significance in promoting safer medical innovations.
Examples of include:
- Conducting that consider both substance safety and overall equipment performance.
This ensures that every aspect of a device’s safety profile is assessed, reinforcing the commitment to patient protection and compliance.
Reporting Requirements: Transparency in ISO 10993-18 Compliance
Transparency in reporting is vital for adherence to . Manufacturers must meticulously document and communicate their , findings, and associated risks. This requirement encompasses comprehensive reports on , detailing the rationale behind the selected analytical methods. By adhering to strict , including , manufacturers not only demonstrate their but also cultivate trust with oversight authorities and stakeholders. Effective reporting practices significantly enhance interactions during audits and reviews. Studies indicate that organizations with transparent documentation experience improved . Furthermore, insights from compliance specialists underscore that can , reducing the likelihood of delays and fostering a culture of accountability within the organization.
Challenges in Demonstrating Biological Equivalence Under ISO 10993-18
presents significant challenges for producers, particularly amid the evolving compliance landscape in Colombia. A major hurdle is the , which leads to discrepancies in the interpretation and application of the standard across various regions. Reports indicate that approximately 60% of producers face inconsistencies in guidance, complicating their compliance efforts.
Moreover, manufacturers must navigate . Experts such as Ana Criado, Director of Compliance and a professor in biomedical engineering, and Katherine Ruiz, a specialist in compliance for medical tools and in vitro diagnostics, underscore the importance of a . This includes:
- Meticulous planning
- Engaging with
- Employing collaborative strategies to enhance adherence and facilitate successful market entry for
As industry leaders emphasize, effectively navigating these is vital for sustaining competitiveness in a rapidly changing environment.
Adopting a Risk-Based Approach in Chemical Characterization for ISO 10993-18
Adopting a in is essential for compliance with . This approach enables producers to prioritize testing activities based on the potential hazards associated with specific materials and their intended uses. By focusing on high-risk components, organizations can allocate resources more efficiently, streamlining the compliance process. This method not only enhances the but also strengthens the overall safety and efficacy of , ensuring while minimizing unnecessary testing.
Industry leaders emphasize that focusing on the evaluation of challenges can lead to improved . For instance, 31% of executives view as the greatest challenge to company growth, underscoring the importance of prioritizing testing based on potential issues. Furthermore, early planning is crucial to avoid missing critical data, which can significantly impact compliance timelines.
Establishing a framework for is also vital, as it allows organizations to adapt to evolving conditions and maintain compliance. By employing a formula like Likelihood x Impact, manufacturers can effectively prioritize their , ensuring that they address the most significant hazards in their processes.
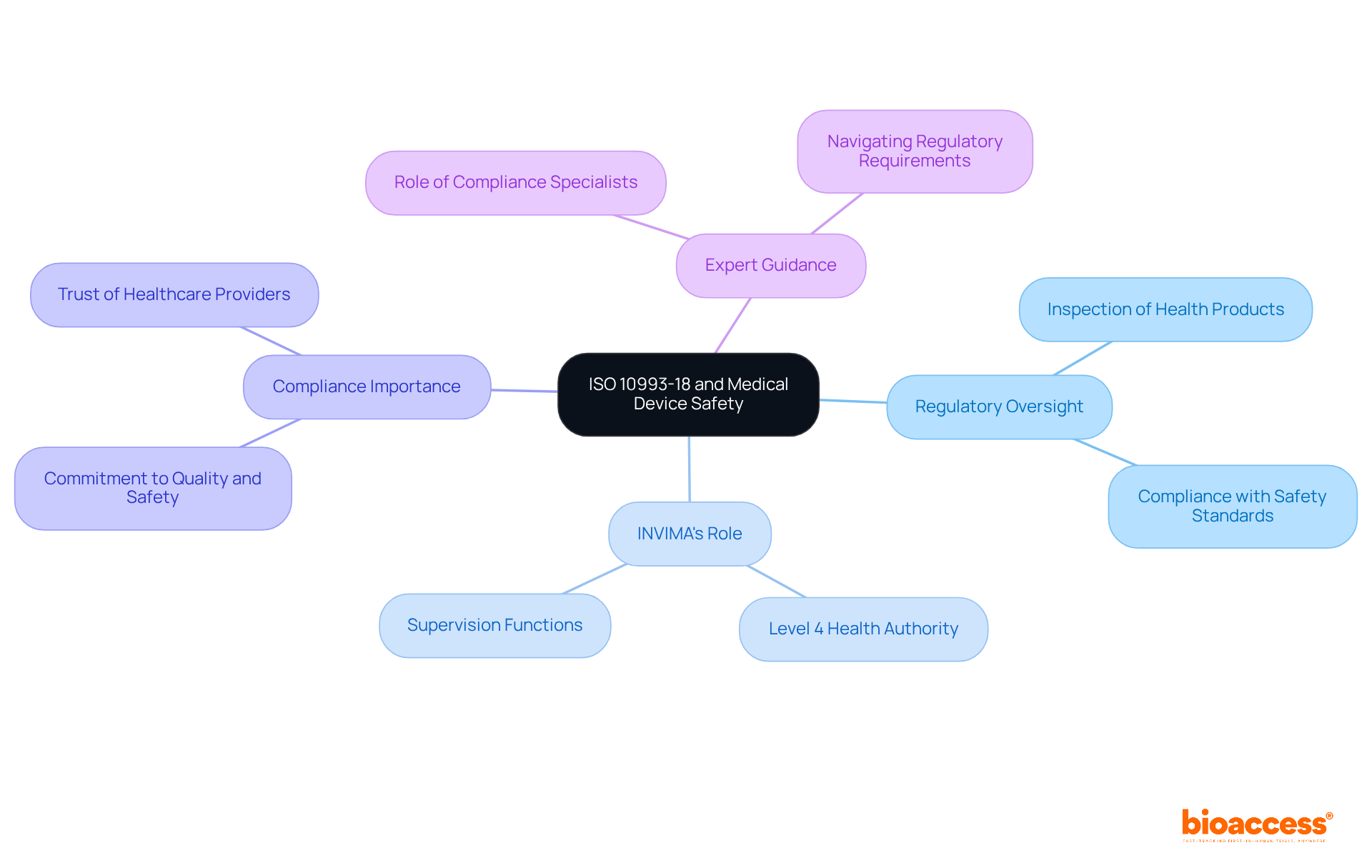
The Significance of ISO 10993-18 in Ensuring Medical Device Safety
The framework for the established by is pivotal in safeguarding the safety of medical instruments. This standard is instrumental in identifying potential linked to component elements, thereby protecting patient health.
In Colombia, the oversight of is significantly shaped by INVIMA (), recognized as a Level 4 health authority by PAHO/WHO. INVIMA’s oversight functions include:
- Inspection and supervision of health products
- Ensuring compliance with safety and efficacy standards
By adhering to the standards set by , manufacturers can affirm their , ultimately bolstering the alike.
Specialists such as Ana Criado, Director of Compliance at Bioaccess, leverage their extensive experience in compliance and biomedical engineering to guide companies through the complexities of meeting .
As the medical device landscape evolves, compliance with , supported by robust regulatory frameworks, remains a critical factor for the of safe and effective medical technologies.
Conclusion
The insights presented on ISO 10993-18 underscore its critical role in ensuring the safety and compliance of medical devices. By focusing on rigorous chemical characterization and biocompatibility testing, manufacturers can effectively navigate the complexities of regulatory requirements while prioritizing patient safety. Collaboration with bioaccess® further enhances the ability to meet these standards efficiently, showcasing the importance of strategic partnerships in clinical research.
Key arguments throughout the article highlight the significance of:
- Substance characterization
- Extractables and leachables testing
- The Analytical Evaluation Threshold (AET) in streamlining compliance processes
The integration of Safety Data Sheets (SDS) and a risk-based approach to chemical characterization also emerge as essential components for maintaining transparency and enhancing the safety profile of medical devices. As the landscape of regulatory compliance continues to evolve, the proactive adoption of these strategies is paramount for success.
Ultimately, embracing the principles outlined in ISO 10993-18 not only fosters regulatory compliance but also contributes to the advancement of safer medical technologies. Manufacturers are encouraged to prioritize these practices, as they safeguard patient health and build trust with healthcare providers and stakeholders. Engaging with experts and leveraging available resources can drive innovation and ensure that medical devices meet the highest standards of safety and efficacy.
Frequently Asked Questions
What is bioaccess® and what role does it play in clinical research?
bioaccess® is a company that leverages its expertise in early-phase clinical research to ensure compliance with ISO 10993-18, focusing on the chemical characterization of materials used in medical devices. It helps innovators in Medtech, Biopharma, and Radiopharma navigate compliance effectively and supports successful clinical research projects.
How does bioaccess® ensure compliance with ISO 10993-18?
bioaccess® secures ethical approvals within 90-120 days by utilizing Colombia’s competitive advantages such as swift regulations, cost efficiency, and a diverse patient population. This rapid turnaround is essential for meeting the rigorous standards set by ISO 10993-18.
What are the benefits of conducting clinical trials in Colombia?
Conducting clinical trials in Colombia offers cost savings exceeding 30% compared to North America, access to a high-quality healthcare system, and R&D tax incentives, making it an appealing location for clinical research.
What is the significance of substance characterization in ISO 10993-18?
Substance characterization is crucial for identifying and quantifying material constituents in medical devices, which helps assess potential biological risks. It is essential for ensuring that products do not release harmful materials that could jeopardize patient health.
What are the key stages in the substance characterization process?
Key stages include proof of substance equivalence, comparison with material standards, and the establishment of a toxicological assessment framework as outlined in ISO 10993-18.
Why is extractables and leachables (E&L) testing important?
E&L testing is critical for assessing the safety of medical instruments by identifying substances that may leach into the body during use. It ensures compliance with biocompatibility standards and helps build trust with healthcare providers and patients.
What are the established thresholds for E&L testing?
The qualification threshold (QT) for E&L testing is set at 5 µg/day, while the safety concern threshold is established at 0.15 µg/day, highlighting the stringent standards that must be adhered to.
What is the projected market value for extractable and leachable testing by 2028?
The extractable and leachable testing market is anticipated to reach a valuation of $1 billion by 2028, indicating its increasing importance in the medical equipment sector.
List of Sources
- bioaccess: Accelerating Clinical Research for ISO 10993-18 Compliance
- campoly.com (https://campoly.com/blog/seismic-shifts-iso-10993-182020)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- Comparison of the Latin America Regulation Landscape and International Reference Health Authorities to Hasten Drug Registration and Clinical Research Applications – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10579156)
- measurlabs.com (https://measurlabs.com/blog/biocompatibility-testing)
- oatext.com (https://oatext.com/Current-barriers-for-developing-clinical-research-in-Latin-America-A-cross-sectional-survey-of-medical-oncologists.php/1000)
- Chemical Characterization: A Cornerstone of ISO 10993-18
- gradientcorp.com (https://gradientcorp.com/trend_articles/new-developments-in-medical-device-chemical-characterization)
- researchgate.net (https://researchgate.net/publication/358650455_Chemical_Characterization_and_Non-targeted_Analysis_of_Medical_Device_Extracts_A_Review_of_Current_Approaches_Gaps_and_Emerging_Practices)
- acdlabs.com (https://acdlabs.com/blog/iso1099318-2020-simplifying-the-chemical-characterization-of-medical-device-materials)
- blog.johner-institute.com (https://blog.johner-institute.com/regulatory-affairs/iso-10993-18-chemical-characterization)
- Extractables and Leachables Testing: Ensuring Biocompatibility Under ISO 10993-18
- towardshealthcare.com (https://towardshealthcare.com/insights/extractable-and-leachable-testing-services-market-sizing)
- rootsanalysis.com (https://rootsanalysis.com/reports/extractable-and-leachable-testing-market-2018-2028/220.html)
- measurlabs.com (https://measurlabs.com/blog/medical-device-extractables-and-leachables-testing)
- basciences.com (https://basciences.com/services/services-by-category/extractables-leachables/extractables-leachables-testing/iso-10993-18)
- Understanding the Analytical Evaluation Threshold (AET) in ISO 10993-18
- blog.johner-institute.com (https://blog.johner-institute.com/regulatory-affairs/iso-10993-18-chemical-characterization)
- azom.com (https://azom.com/article.aspx?ArticleID=19230)
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S0273230024001661)
- namsa.com (https://namsa.com/resources/blog/a-guide-to-iso-10993-18s-analytical-evaluation-threshold)
- medicaldesignbriefs.com (https://medicaldesignbriefs.com/component/content/article/36027-preparing-for-mdr-don-t-overlook-the-analytical-evaluation-threshold)
- Safety Data Sheets (SDS): Key to Regulatory Compliance in ISO 10993-18
- The Role of Safety Data Sheets in Compliance Readiness (https://evotix.com/resources/blog/the-role-of-safety-data-sheets-in-compliance-readiness)
- stericycle.ca (https://stericycle.ca/en-ca/resource-centre/blog/importance-of-safety-data-sheets)
- researchgate.net (https://researchgate.net/publication/381844214_Enhancing_Chemical_Safety_and_Regulatory_Compliance_through_Safety_Data_Sheets_SDS_Challenges_Innovations_and_Future_Directions)
- verifiedmarketreports.com (https://verifiedmarketreports.com/product/safety-data-sheet-sds-management-market)
- totalmedicalcompliance.com (https://totalmedicalcompliance.com/how-often-does-a-safety-data-sheet-sds-need-to-be-updated-and-who-is-responsible?srsltid=AfmBOoqajeZzJXwK1CBlUWOjhOAfPJIwQHddc6Bq9pI2XpQa-by49yqy)
- Linking ISO 10993-18 and ISO 14971: A Risk Management Perspective
- 50 Risk Management Quotes: Wisdom for Smart Decision-making | ITD World (https://itdworld.com/blog/leadership/risk-management-quotes)
- greenlight.guru (https://greenlight.guru/blog/iso-14971-risk-management)
- researchgate.net (https://researchgate.net/publication/357843718_Risk_Management_Standards)
- nectarpd.com (https://nectarpd.com/iso10993-1-and-iso14971)
- ketryx.com (https://ketryx.com/blog/iso-14971-a-comprehensive-guide-to-risk-management-in-medical-devices)
- Reporting Requirements: Transparency in ISO 10993-18 Compliance
- acdlabs.com (https://acdlabs.com/blog/iso1099318-2020-simplifying-the-chemical-characterization-of-medical-device-materials)
- blog.johner-institute.com (https://blog.johner-institute.com/regulatory-affairs/iso-10993-18-chemical-characterization)
- intertek.com (https://intertek.com/medical/iso10993part18-medical-device-chemical-characterisation-extractables)
- Compliance Quotes (83 quotes) (https://goodreads.com/quotes/tag/compliance)
- Challenges in Demonstrating Biological Equivalence Under ISO 10993-18
- medinstitute.com (https://medinstitute.com/blog/challenges-of-demonstrating-biological-equivalence-per-iso-10993-18-and-eu-mdr)
- linkedin.com (https://linkedin.com/pulse/unresolved-statistical-challenges-bioequivalence-chandramouli-r-gkj5c)
- Statistical and regulatory considerations in assessments of interchangeability of biological drug products – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC4046084)
- journals.sagepub.com (https://journals.sagepub.com/doi/10.1177/01926233251316283?int.sj-abstract.similar-articles.2)
- testlabsuk.com (https://testlabsuk.com/blog/biocompatibility-in-medical-devices-ensuring-safety-compliance-and-market-success)
- Adopting a Risk-Based Approach in Chemical Characterization for ISO 10993-18
- sgs.com (https://sgs.com/en/webinars/2024/04/recent-trends-and-regulatory-expectations-in-chemical-characterization-of-medical-devices)
- 50+ Risk Management Statistics to Know in 2026 (https://secureframe.com/blog/risk-management-statistics)
- blog.johner-institute.com (https://blog.johner-institute.com/regulatory-affairs/iso-10993-18-chemical-characterization)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC8641414)
- The Significance of ISO 10993-18 in Ensuring Medical Device Safety
- iso.org (https://iso.org/standard/41106.html)
- blog.johner-institute.com (https://blog.johner-institute.com/regulatory-affairs/iso-10993-18-chemical-characterization)
- intertek.com (https://intertek.com/medical/iso10993part18-medical-device-chemical-characterisation-extractables)
- namsa.com (https://namsa.com/resources/blog/iso-10993-18-primer-medical-device-chemical-characterization)

Leave a Reply